098e60d8871d25529461e7a7ca69fb1a.ppt
- Количество слайдов: 58

BTG 424 Food Biotechnology 11 Biosafety Issues in Modern Biotechnology Dr. Friday Nwalo (FUNAI)

BIOSAFETY ‘Biosafety’ means the need to protect human and animal health and environment from the possible adverse effects of the products of modern biotechnology
![Convention of Biodiversity (CBD) [1992] n n n Focus: conservation and sustainable use of Convention of Biodiversity (CBD) [1992] n n n Focus: conservation and sustainable use of](https://present5.com/presentation/098e60d8871d25529461e7a7ca69fb1a/image-3.jpg)
Convention of Biodiversity (CBD) [1992] n n n Focus: conservation and sustainable use of biodiversity Recognized the potential of modern biotechnology for human well being Took cognizance that modern biotechnology could have serious effects on environment and health Article 8(g) emphasized the need to regulate the risks associated with the use of LMOS. Article 19(3) set the stage for a legally binding international instrument about biosafety.

The Cartagena Protocol on Biosafety (CPB) n n n Entered into force on 29 th December 1993 Focus on transboundary movement of the LMOS. Seeks to lay down an internationally acceptable framework to provide for an adequate level of protection against the possible adverse affects of LMOS on biodiversity and human health.

Basic Premises of CPB n n n “Advance Informed Agreement” between Parties (AIA) Decision on the basis of scientific risk assessment Precautionary Principle

How is Genetic Engineering (GE) different from conventional breeding (CB)? n n Combining DNA in new combinations and introducing it into a new organism are the GE tools. Main differences between CB and GE Ø Ability to move across sexual barriers Ø Amount of change: a specific gene embodying a particular trait or thousands of genes embodying desirable and undesirable traits Ø Occurrence of change in one or several generations.
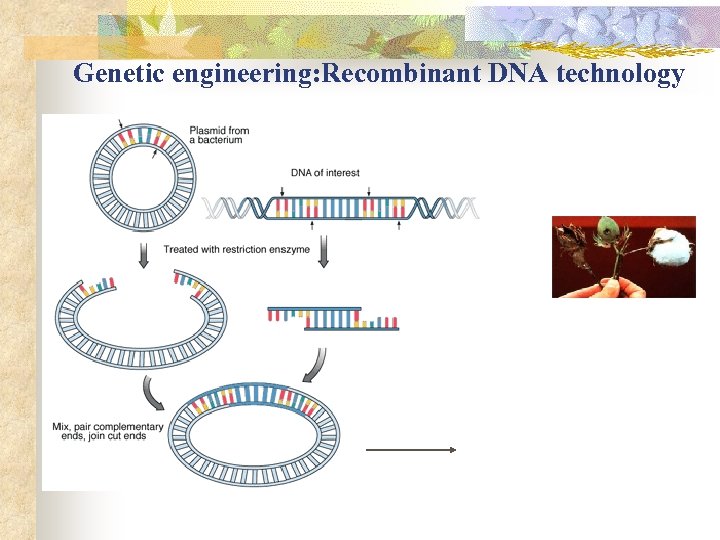
Genetic engineering: Recombinant DNA technology

Is GE inherently unsafe? n n Two diametrically opposite trends of thought US-Canada n No new risks associated with GM crops n New regulations not considered necessary n Safety assessments Ø ‘Product’ rather than ‘process’ based Ø In comparison and contrast to their ‘familiarity’ and ‘substantial’ equivalence to conventional crops

…Is GE inherently unsafe? n EU n n n Safety assessment n n n GE crops considered new and special Existing legislation not considered sufficient Process based Principle of ‘substantial equivalence’ beginning rather than the end Adoption of ‘Precautionary Principle’ as guide

…. Is GE inherently unsafe? n n GE technology carries certain inherent unpredictability Some facts n Isolation of a gene from its natural environment and integration into entirely different organism n Possible transgenic instability due to triggering of the inbuilt defense mechanisms of the host organism leading to inactivation or silencing of foreign genes.

…. Is GE inherently unsafe? n Possibilities of integration of foreign gene at a site predisposed to silencing of genes (position effect). Ø Variance in the levels of expression of the transgene in different environmental conditions (heat, humidity, light…. . ) Ø Possibilities of silencing of genes arising in subsequent generations Case by case sound scientific assessment is of utmost significance
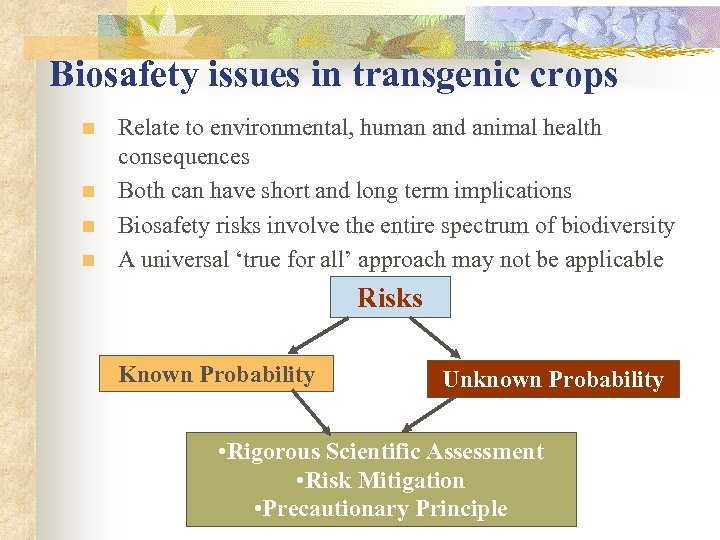
Biosafety issues in transgenic crops n n Relate to environmental, human and animal health consequences Both can have short and long term implications Biosafety risks involve the entire spectrum of biodiversity A universal ‘true for all’ approach may not be applicable Risks Known Probability Unknown Probability • Rigorous Scientific Assessment • Risk Mitigation • Precautionary Principle

. . Biosafety issues in transgenic crops Biosafety concerns arise from: n n n Horizontal gene transfer Genetic contamination Transfer of allergens and toxins from one life form to another and creation of new toxins and allergenic compounds

. . Biosafety issues in transgenic crops Main - Concerns Ø Development of aggressive weeds/ wild relatives by transfer of transgenic traits Ø Erosion of land races/wild relatives by genetic pollution in centres of origin/ diversity Ø Harm to the non-target organisms Ø Development of pest resistance by prolonged use Ø Monoculture and limitations to farmers’ choice in crop management Ø Hazard to human and animal health by transfer of toxins and allergens and by creation of new toxins and allergenic compounds

. . Biosafety issues in transgenic crops Assessment n n n GE venturing into an unknown biological territory ASILOMAR Conference (1975): No research till safety guidelines in place Initially, focus on laboratory safety procedures Wider definition of biosafety with possibilities of commercialization of GM products The broad format of biosafety parametres essentially the same in all regulations

. . Biosafety issues in transgenic crops Two main stages: 1. 2. Laboratory/green house stage Confined Trial Stage IMPORTANT Prevention of the spread of genetically engineered material outside lab/field

Laboratory/green house stage n. Different biosafety levels as per the degree of risk involved n. Two methods of containment v. Physical v. Biological

Confined Trial Stage A confined trial is a small scale release of a transgenic plant species for research purposes conducted under conditions that prevent spread of the organism and mitigate its impact on the surrounding environment Objective is to collect data to evaluate the crops’ performance

Focus on Risk Mitigation Risk mitigation – the terms and conditions that are necessary to conduct the trial safely. n n n Prevent Gene Flow Prevent entry of GMOs into food chain Prevent Persistence of GMOs in the field

Bio-pharmaceutical therapeutics Biosafety risk n Survival, multiplication and dissemination of GMOs in contained/ open environment n Interaction of GMOs with biological systems n Routes of dissemination: physical; biological Risk depends upon n Nature of organism invovled n Extent of use of LMOs n End product LMO or not?

…Bio-pharmaceutical therapeutics Risk categorization of micro organisms: determining factors n Capability to cause disease n Hazard to laboratory workers n Risk of spread to community n Availability of effective treatment Health risks n Toxigenicity Pathogenicity n Allergenicity Antibiotic resistance

. . Bio-pharmaceutical therapeutics Environmental risks n Outcrossing between GMOs and pathogens n Negative effects on populations of non target organisms Risk assessment n Access n Expression n Damage Risk management and communication n Physical n Biological

GM foods: need for safety assessment n n n Expressed proteins generally not a part of regular food supply Food complex mixtures e. g. nutrients, antinutrients and natural toxins Directly enter human system Assume different forms Involve storage, processing, transportation

. . Safety assessment of GM foods comprise Guidelines by Codex Alimentarius Commission n Assessment of possible allergenicity n Assessment of possible toxicity n Compositional analysis of key components n Food processing n Nutritional modification

…. GM foods: Allergenicity; Toxicity Allergy It is a hypersensitive reaction initiated by immunologic mechanisms caused by specific substances called allergens. Assessment n Is the gene source allergenic? n Expression level of introduced gene n Unintended effect n Digestibility and heat stability Toxicity n n n New proteins as a result of intended modification Unintended new proteins as a result of the modification Natural constituents beyond their level of normal variation

…. GM foods: nutritional aspects; unintended effects n n Intended and unintended changes in nutrient levels Bioavailability of nutrients, stability and processing Presence and effect of anti-nutrients Impact of individual changes on overall nutritional profile Unintended effects Random integration of transgenes n Insertional mutagenesis n Disruption of gene functions n Production of new proteins n Changes in o Phenotype Metabolites o Enzymes Toxins o Genotype

Concluding Note…… n n n n Biosafety is integral to modern biotechnology The adoption of modern biotech products needs to be balanced with adequate biosafety safeguards Case by case scientific risk assessment and cost benefit analysis Greater acceptance of health care applications Need based adoption in GM crops and foods Participation of various stakeholders Dissemination of knowledge and information

What are GM’s? are a result of technology that has altered the DNA of living organisms (animals, plants or bacteria) Other terms that mean the same thing: n Genetically engineered n Transgenic n Recombinant DNA (r. DNA) technology n

How does this differ from Mendel and his peas? GM vs. Selective breading -slow -imprecise -modification of genes that naturally occur in the organism GM -very fast -precise -can introduce genes into an organism that would not occur naturally!

Why do it? n Rice- not high in essential nutrients Modification: n + daffodil genes and a bacterium = beta-carotene content drastically increased n + genes from a french bean = double the iron content. n Tomatoes- Introduce genes to increase shelf life.
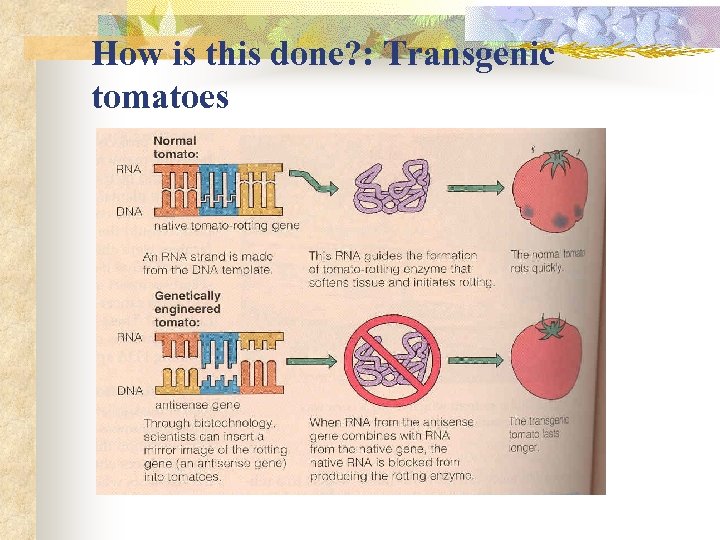
How is this done? : Transgenic tomatoes

Other applications n n Potato - modified to produce a beetle killing toxin Yellow squash – modified to contain to viral genes that resistant the most common viral diseases Develop foods that contain vaccines and antibodies that offer valuable protection against diseases such as cholera, hepatitis, and malaria Canola – modified to resist one type of herbicide or pesticide

Benefits of Genetic Engineering and Modifying 1. Higher yielding crops, more efficient use of land 2. Can save money and promote higher profits 3. Longer shelf life, less waste Example// Tomatoes from genetically modified seeds stay fresh longer. 4. Enhanced taste and quality 5. Reduced maturation time

Benefits of Genetic Engineering and Modifying 6. 7. 8. 9. Increased and improved nutrients and stress tolerance - A single genetically engineered into cauliflower can increase production of beta-carotene 100 times. - A gene can be implanted into a soybean upgrading the soy protein to a quality equal to that of milk. - Corn can be modified to contain its two limiting amino acids, lysine or tryptophan Improved resistance to disease or illness - Foods can be enhanced with phytochemicals that help maintain health and reduce the risks of chronic disease. Improved crop resistance to disease, pests, weeds and herbicides New products and growing techniques - “Individuals allergic to milk may be able to buy milk that has been treated with the lactase enzyme” (Whiney, 2002). - Creating decaffeinated coffee beans are in a process of research.

Benefits of Genetic Engineering and Modifying n Society n Increased food security for growing populations and growth challenges (Human Genome Project Information (2003), http: //www. ornl. gov/sci/techresources/Human _Genome/elsi/gmfood. shtml)

Risks associated with Genetic Modification 1. Safety n Potential human health implications. n Potential environmental impact. n Out-crossing n n n 2. Inevitable out-crossing of transgenic plants with naturally occurring ones. Creation of super-weeds Creation of biological weapons. Access and Intellectual Property n Domination of world food production by a few companies and developing countries.

Risks associated with Genetic Modification – cont. 3. Ethics n n 4. Labeling n n 5. “Playing God” Tampering with nature by mixing genes among species. Not mandatory in some countries (e. g. , Canada and the United States). Mixing GM crops with non-GM confounds labeling attempts. Society n New advances may be skewed to the interests of rich countries. (Human Genome Project Information (2003), http: //www. ornl. gov/sci/techresources/Human_Genome/elsi/gmfood. shtml)

Risks with GM continued: Biodiversity n n n Addition of Bt gene into plants including corn, potatoes and cotton to increase resistance to plants Bt gene obtained from Bacillus thuringiensis (a soil bacterium that produces a natural insecticide) Problem: plants producing Bt toxin are releasing toxin in pollen Draper, D. (2002). Our Environment: A Canadian Perspective 2 nd Ed. Scarborough: Thompson Canada Lmt.

Pollen from a Bt plant was dusted on to milkweed: - only 56% of young monarch butterfly larvae lived - whereas pollen from organic plants dusted on the milkweed produced a survival rate of 100%. Approximately half of the monarch butterfly population live in the “corn belt” of the USA = this new gene could have serious repercussions for this organism

Socio-economic concerns of GMOs

Technology and society

Socio-economic? n n Socio-economic is the way in which economic activity affects and is shaped by social processes It has to do with societies progress, stagnation, or regression because of their local or regional economy, or the global economy

GMOs socio-economic considerations n There is not yet a clear and agreed definition on what socio-economic considerations entail in the context of biosafety regulations n Socio-economic considerations related to GMOs could be defined as the set of the intertwined social and economic consequences resulting from the changes arising from the introduction of GMOs into the environment, which need to be taken into account in the biosafety decision-making processes Sadler and Mc. Cabe (2002)

n Concerns with GMOs Scientific n Non-scientific n Environment n n n Is it natural, ‘crossing the species barrier Can the plants spread uncontrollably n n n Does the food contains bacterial genes Are the crops regulated; well scrutinized n n n Is it ethical Is it natural Socio-economic n Health safety n Ethical Whose interest does it represent Income security Rural labor Intellectual Property Rights (IPRs) Religious Political n Food security

International biosafety context n n n Initial scope of biosafety expanded to include food and feed safety Proposal to expand it further to include socio-economics, ethical, religious, philosophical and other considerations Socio-economics has little to do with the safety profile Two contrasting points of view Oppose: • • n Decisions should be left to end-users Open for blanket (no need to show real proof ) rejections of technology Favor • Critical to ensure that GM crops will not harm communities and producers IFPRI

in Cartagena Protocol n Article 26 SOCIO-ECONOMIC CONSIDERATIONS 1. The Parties, in reaching a decision on import under this Protocol or under its domestic measures implementing the Protocol, may take into account, consistent with their international obligations, socio-economic considerations arising from the impact of living modified organisms on the conservation and sustainable use of biological diversity, especially with regard to the value of biological diversity to indigenous and local communities. 2. The Parties are encouraged to cooperate on research and information exchange on any socio-economic impacts of living modified organisms, especially on indigenous and local communities.

ACT, 2015 n Anticipated changes in the existing social and economic patterns resulting from the introduction of the genetically modified organism or products n Possible treats to biological diversity, traditional crops or other products and in particular, farmers’ varieties and sustainable agriculture n impacts likely to be posed by the possibility of substituting traditional crops, products and indigenous technologies through modern biotechnology outside of their agro-climatic zones.

Socio-economic consideration NBMA ACT, 2015 n Anticipated social and economic costs due to loss of genetic diversity, employment, market opportunities and in general, means of livelihood of the communities likely to be affected by the introduction of the genetically modified organisms or products n Possible countries and communities to be affected in terms of disruptions to their social and economic welfare n Possible effects which are contrary to the social, cultural, ethical and religious values of communities arising from the use of release of the genetically modified organism or the product

Issues and roles for Socio Economic Considerations in Biosafety n Socio economic assessments for biosafety purposes are ex ante n Technical know how to do “feasible” Ex ante studies is available in the Nigeria n Methods to answer many of the questions posed by some stakeholder in an ex ante framework n Question of timing • • Not all the technologies that enter the biosafety regulatory process will be released into the environment The earlier assessments are required in the regulatory process, the more assumptions one has to make IFPRI

Some countries approach to socioeconomic assessments n Large variation in terms of approaches n Argentina requires SEA, but limited to impacts on Argentinean exports n South Africa limits scope to impacts on surrounding communities n India not formally required in regulations yet regulatory authority required a baseline impact study n USA, Canada, and probably the EU, still voluntary information that may be included with dossiers IFPRI

Biotech and African Agriculture n Food security is still an issue in SSA n Millions of people are starving n Therefore, Africa needs all the appropriate tools in science & technology to increase productivity & enhance food security

Genetic Modification & Medicine n n n Europe is over producing food, thus enhancing agricultural productivity is no priority But Health and Longevity are very important to the West Hence, acceptance of GM medicine

Final comments n n Socio-economic impacts (positive or negative, predicted or unforeseen) are inherent part of technology introduction and adoption. This points out the need of including socio-economic considerations in the biosafety decision-making related to GMOs. In order to carry out these socio-economic assessments relevant to sustainability, precautionary or anticipatory (also called ex-ante) assessments are needed, complemented with regular monitoring (or ex-post). The Socio-economic consideration is already domesticated in the NBMA ACT, 2015

AATF
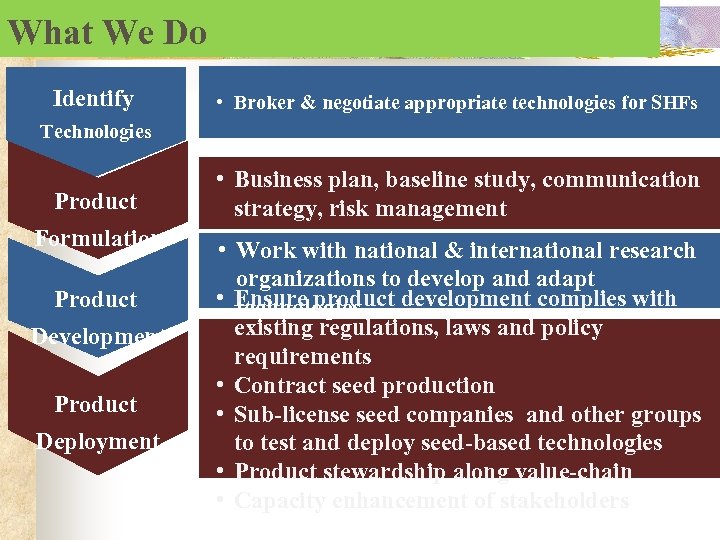
What We Do Identify • Broker & negotiate appropriate technologies for SHFs Technologies Product Formulation Product Development Product Deployment • Business plan, baseline study, communication strategy, risk management • Work with national & international research organizations to develop and adapt • Ensure product development complies with technologies existing regulations, laws and policy requirements • Contract seed production • Sub-license seed companies and other groups to test and deploy seed-based technologies • Product stewardship along value-chain • Capacity enhancement of stakeholders
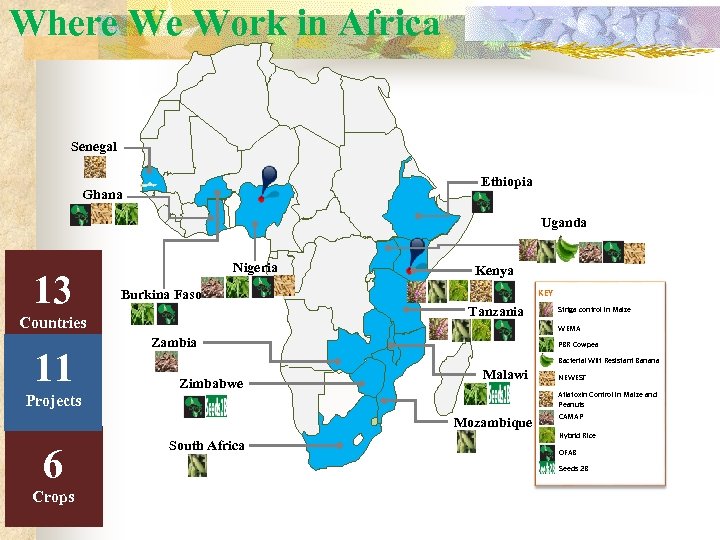
Where We Work in Africa Senegal Ethiopia Ghana Uganda 13 Nigeria KEY Burkina Faso Tanzania Countries 11 Kenya WEMA Zambia PBR Cowpea Bacterial Wilt Resistant Banana Zimbabwe Malawi Mozambique Crops South Africa NEWEST Aflatoxin Control in Maize and Peanuts Projects 6 Striga control in Maize CAMAP Hybrid Rice OFAB Seeds 2 B

Thank you
098e60d8871d25529461e7a7ca69fb1a.ppt