50dab138d0643c249f6d95deb8962fcd.ppt
- Количество слайдов: 28

Brachy Mesh Implants for Lung Cancer Cindy Thomason Aurora Health. Care – Metro
Brachy Mesh Implants • I-125 seeds sewn in mesh in a grid layout • Indications • Stage I nonsmall cell lung cancer • Metastases
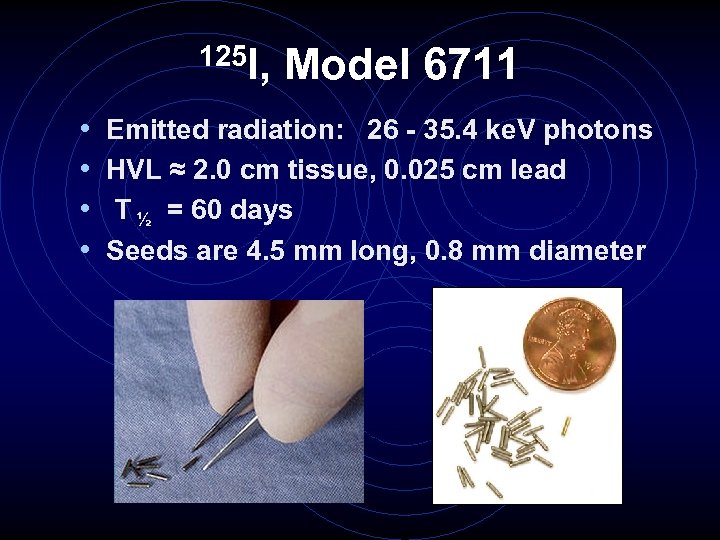
125 I, • • Model 6711 Emitted radiation: 26 - 35. 4 ke. V photons HVL ≈ 2. 0 cm tissue, 0. 025 cm lead T ½ = 60 days Seeds are 4. 5 mm long, 0. 8 mm diameter

125 I Brachy Mesh • Basis is a Vicryl mesh which is a flexible, absorbable mesh • Tensile strength of mesh is 10% is 6 weeks • 125 I seeds in suture are embedded in mesh in grid array • Size and spacing of array decided by Radiation Oncologist and Thoracic Surgeon

125 I Brachy Mesh • Typical size / spacing • 4 rows x 10 seeds/row, 1 cm spacing between seeds and rows • Ave activity / seed = 0. 41 m. Ci • Ave total activity = 16. 5 m. Ci
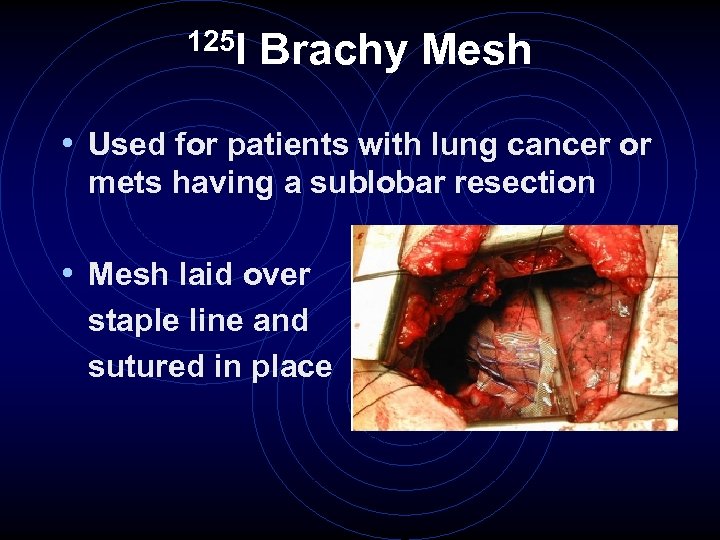
125 I Brachy Mesh • Used for patients with lung cancer or mets having a sublobar resection • Mesh laid over staple line and sutured in place
125 I Brachy Mesh Most procedures can be done via minimally invasive surgery
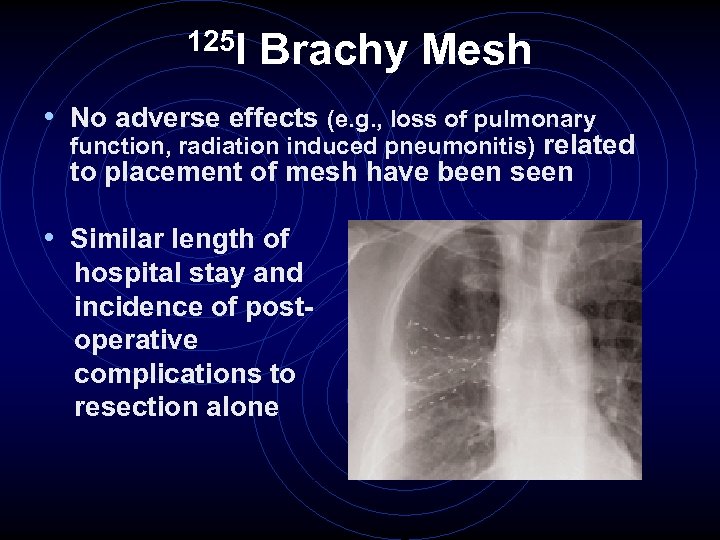
125 I Brachy Mesh • No adverse effects (e. g. , loss of pulmonary function, radiation induced pneumonitis) related to placement of mesh have been seen • Similar length of hospital stay and incidence of postoperative complications to resection alone

125 I Brachy Mesh 110 patients median 11 month follow-up (range of 1 -68 months) significant improvement in local recurrence 19% with sublobar alone vs 1% with sublobar resection + brachytherapy Voynov, et al. , Brachytherapy 4: 278 -285, 2005 Stantos, et al. , Surgery 134: 691 -697, 2003

Dose Prescription • 120 / 100 Gy to a point 0. 5 cm above / below plane of implant with margin of 1 -2 cm along each side of the resection staple line • 100 Gy used for patients with previous external beam treatment or tumor location near heart, aorta or spinal cord • We use 100 Gy because of difficulty in predicting location of staple line / mesh

Treatment planning • Johnson, et. al. (PMB 52: 1237 -1245, 2007) prescribed to point average of peak over row and trough between rows • We chose to also average in points between seeds Therefore, we created our own nomogram for standard configurations and spacing • Spacing between rows and seeds can be modified to adjust for seed activity availability

Treatment Planning • Brachy. Vision commissioned for I-125 Model 6711 • Compared to hand calculation and Johnson, et. al. • Agreement with Johnson, et. al. to within 5% reasonable due to differences in prescription point and uncertainty in parameters used in publication


• Pre implant dosimetry based on nomogram generated in house • Prescription based solely on preimplant geometry of mesh, not patient anatomy

Estimated Personnel Exposure from Brachy Mesh Procedure • Thoracic Surgeon will receive greatest exposure • Estimated hand dose = 2. 5 mrem • Estimate 5 mrem / procedure x 50 procedures / year 250 mrem / year • This is less than 10% of the dose limit, 5000 mrem / yr • Therefore, film or ring badges are not necessary for any personnel

Regulatory Requirements for Release of Brachy Mesh Patients • Brachy Mesh patients can be released to general public if the exposure rate at 1 m from patient is 1 mrem / hr or less (WI HS 157, Appendix U, Table 15) • Expected exposure rate from patient is 0. 6 mrem / hr


For 1 st 29 patients Exposure range = 0. 05 – 0. 95 m. R/hr Average exposure rate = 0. 42 m. R/hr ( 0. 13 m. R/hr (energy corrected)) Brachy Mesh patient could be released into general population as soon as procedure is completed

OR Procedures • Handled similarly to a prostate implant • Seed count performed prior to patient leaving OR • Personnel and surroundings surveyed

Radiation Safety Procedures for Dislodged Radioactive Source • Patients in-house 1 - 5 days • Since 125 I sources are embedded in Vicryl mesh, it is very unlikely that a source will be dislodged • However, all patients leave surgery with a chest tube and atrium only potential route for 125 I seed to exit body provided

• Surveys performed • When atrium replaced • When chest tube and atrium removed

Posting • Minimal since patient could be released immediately • Posting on front of chart and patient room door to save chest tube or atrium if changed and to call physics for survey • Physics posts release notice after survey completed

Brachy Mesh Patient Radiation Safety Instruction • For 3 months, avoid prolonged contact with pregnant women or children at distances closer than 3 feet; no limitations at distances greater than 3 feet • Patients are given wallet card with implant information and facility information and made aware of increased security and monitoring for radioactivity

Challenges • Some (3) patients have received two Brachy Mesh implants • Request a minimum of 1 month between implants • Exposure rates for second implant have been comparable or less than for first implant

Challenges (cont. ) • Patients with defibrillators or pacemakers • Move device • Perform worse case estimate of minimum allowed distance • Complicated by difficulty in estimating distance between mesh and device prior to implant

Challenges (cont. ) • Patients with malignant effusion • Treatment consists of placing a pleural catheter for sporadic drainage, potentially long term • First issue - exposure to personnel performing procedure • Second issue - seed migration into drainage fluid • As mesh dissolves, fibrosis forming around seeds helps to keep them in place • Treat drainage fluid similarly to urine from prostate implant patients

Post planning?

In conclusion • After start-up process, Brachy Mesh implants are a relatively simple procedure for the staff with essentially no side effects for the patient • Early results show improved survival which hopefully will be reinforced as more data becomes available
50dab138d0643c249f6d95deb8962fcd.ppt