761f718dd170b160900e0a6fa6896789.ppt
- Количество слайдов: 68

Bisphonates vs RANK ligand inhibition in bone metastases New York, November 15, 2013 Vera Hirsh, MD, FRCP(C) Mc. Gill University Health Centre Montreal, QC, CANADA
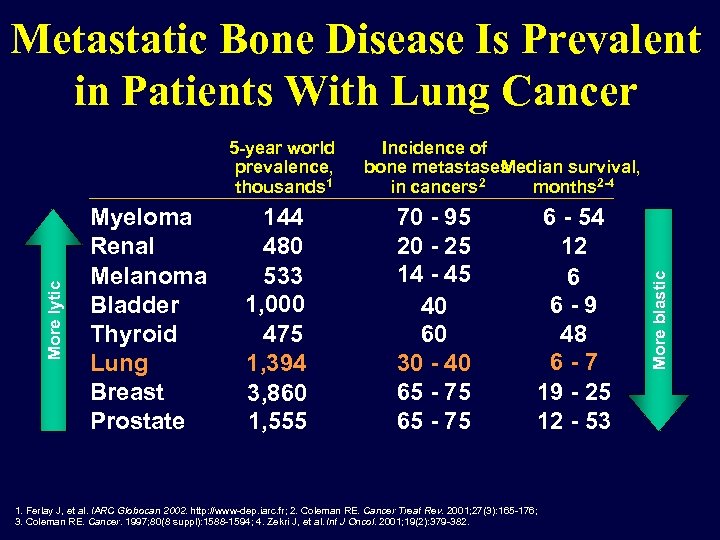
Metastatic Bone Disease Is Prevalent in Patients With Lung Cancer Myeloma Renal Melanoma Bladder Thyroid Lung Breast Prostate 144 480 533 1, 000 475 1, 394 3, 860 1, 555 Incidence of bone metastases Median survival, 2 in cancers months 2 -4 70 - 95 20 - 25 14 - 45 40 60 30 - 40 65 - 75 6 - 54 12 6 6 - 9 48 6 - 7 19 - 25 12 - 53 1. Ferlay J, et al. IARC Globocan 2002. http: //www-dep. iarc. fr; 2. Coleman RE. Cancer Treat Rev. 2001; 27(3): 165 -176; 3. Coleman RE. Cancer. 1997; 80(8 suppl): 1588 -1594; 4. Zekri J, et al. Int J Oncol. 2001; 19(2): 379 -382. More blastic More lytic 5 -year world prevalence, thousands 1

Bone Metastases Have Debilitating Consequences Disease Skeletal-related events Ultimate consequence Loss of autonomy Fracture Radiation to bone Bone metastases Consequences Spinal cord compression Hypercalcaemia Surgery to bone (incl. cementoplasty) Significant morbidity Reduced quality of life 2 Bone pain Increased healthcare costs and resources SREs = Skeletal-related events Kinnane N. Eur J Oncol Nurs 2007; 11(Suppl. ): S 28 -S 31; 2. Weinfurt KP, et al. Ann Oncol 2005; 16: 579 -584. Decreased survival
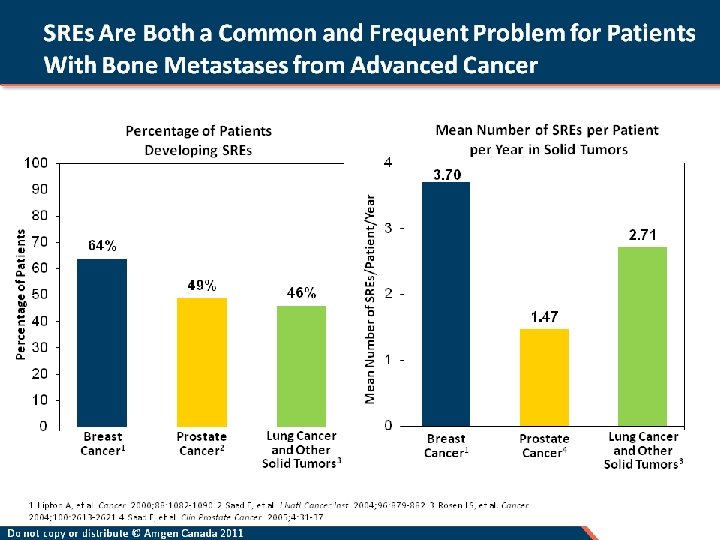
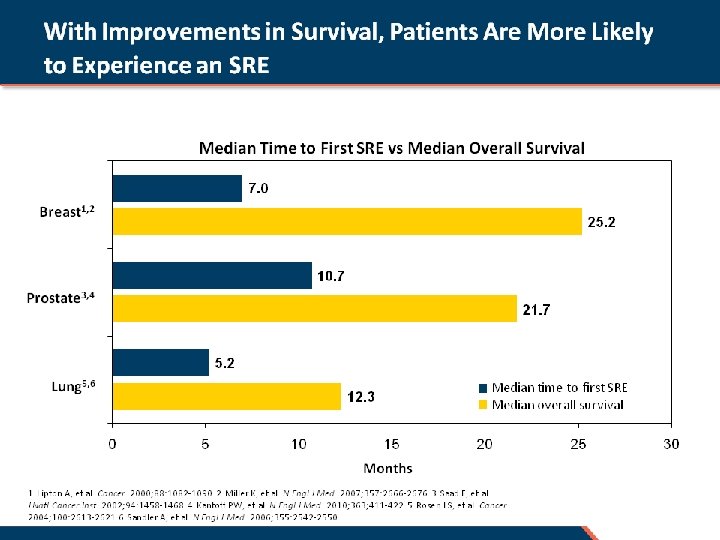
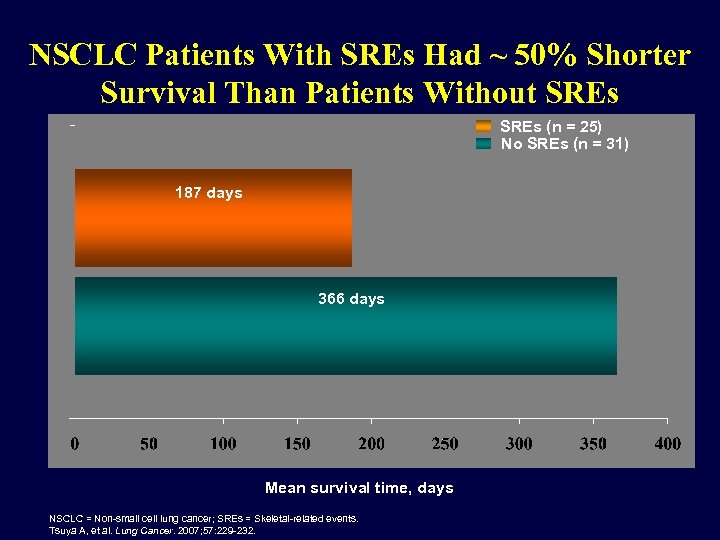
NSCLC Patients With SREs Had ~ 50% Shorter Survival Than Patients Without SREs (n = 25) No SREs (n = 31) 187 days 366 days Mean survival time, days NSCLC = Non-small cell lung cancer; SREs = Skeletal-related events. Tsuya A, et al. Lung Cancer. 2007; 57: 229 -232.
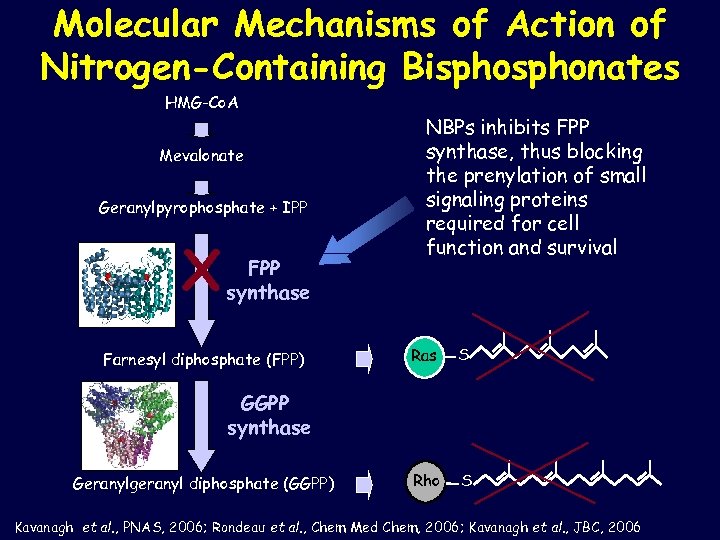
Molecular Mechanisms of Action of Nitrogen-Containing Bisphonates HMG-Co. A Mevalonate Geranylpyrophosphate + IPP X FPP synthase Farnesyl diphosphate (FPP) NBPs inhibits FPP synthase, thus blocking the prenylation of small signaling proteins required for cell function and survival Ras S Rho S GGPP synthase Geranylgeranyl diphosphate (GGPP) Kavanagh et al. , PNAS, 2006; Rondeau et al. , Chem Med Chem, 2006; Kavanagh et al. , JBC, 2006
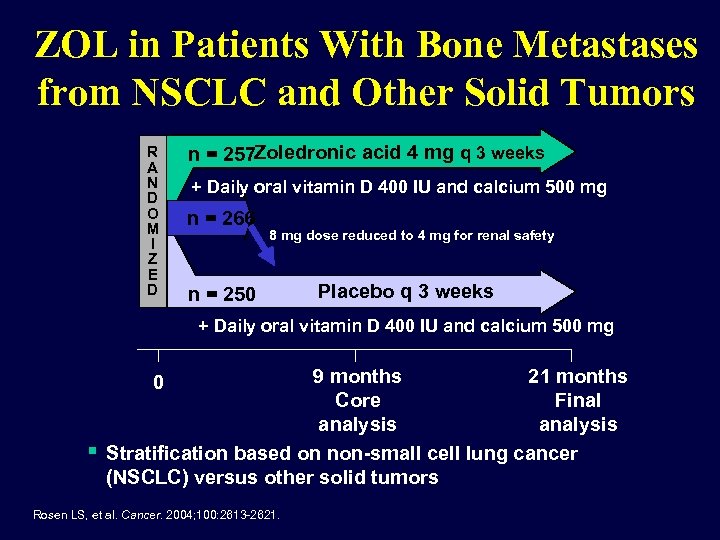
ZOL in Patients With Bone Metastases from NSCLC and Other Solid Tumors R A N D O M I Z E D n = 257 Zoledronic acid 4 mg q 3 weeks + Daily oral vitamin D 400 IU and calcium 500 mg n = 266 8 mg dose reduced to 4 mg for renal safety n = 250 Placebo q 3 weeks + Daily oral vitamin D 400 IU and calcium 500 mg 9 months 21 months Core Final analysis Stratification based on non-small cell lung cancer (NSCLC) versus other solid tumors 0 Rosen LS, et al. Cancer. 2004; 100: 2613 -2621.

Tumor Types Enrolled: Randomized, Placebo-Controlled Trial of ZOL in Patients With Solid Tumors Tumor type NSCLC Small-cell lung cancer (SCLC) Renal cell carcinoma Colon/rectum/intestinal Cancer unknown primary Bladder Esophagus/gastroesophageal Head and neck Melanoma Thyroid Other tumor types (n = 11) Rosen LS, et al. Cancer. 2004; 100: 2613 -2621. n % 244 36 46 37 35 26 12 10 6 39 49 7 7 5 2 2 2 1 8
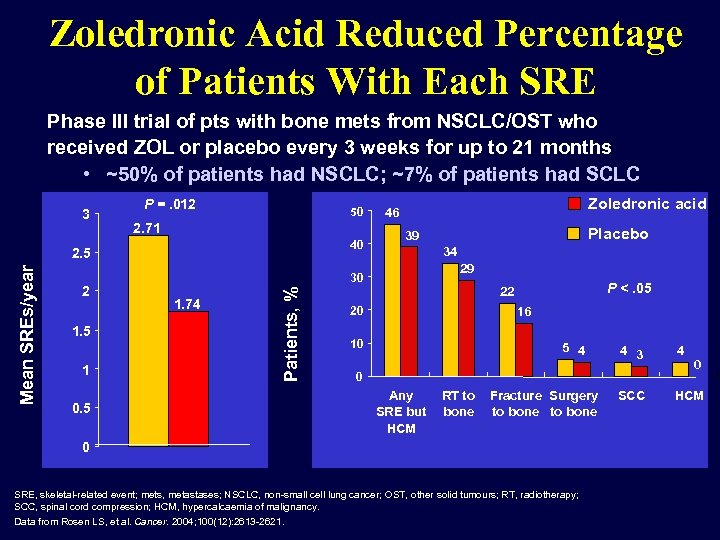
Zoledronic Acid Reduced Percentage of Patients With Each SRE Phase III trial of pts with bone mets from NSCLC/OST who received ZOL or placebo every 3 weeks for up to 21 months • ~50% of patients had NSCLC; ~7% of patients had SCLC 3 P =. 012 50 2. 71 40 2 1. 5 1 0. 5 1. 74 Placebo 39 34 29 30 Patients, % Mean SREs/year 2. 5 Zoledronic acid 46 P <. 05 22 20 16 10 5 4 4 3 4 SCC HCM 0 Any SRE but HCM RT to bone Fracture Surgery to bone 0 SRE, skeletal-related event; mets, metastases; NSCLC, non-small cell lung cancer; OST, other solid tumours; RT, radiotherapy; SCC, spinal cord compression; HCM, hypercalcaemia of malignancy. Data from Rosen LS, et al. Cancer. 2004; 100(12): 2613 -2621. 0
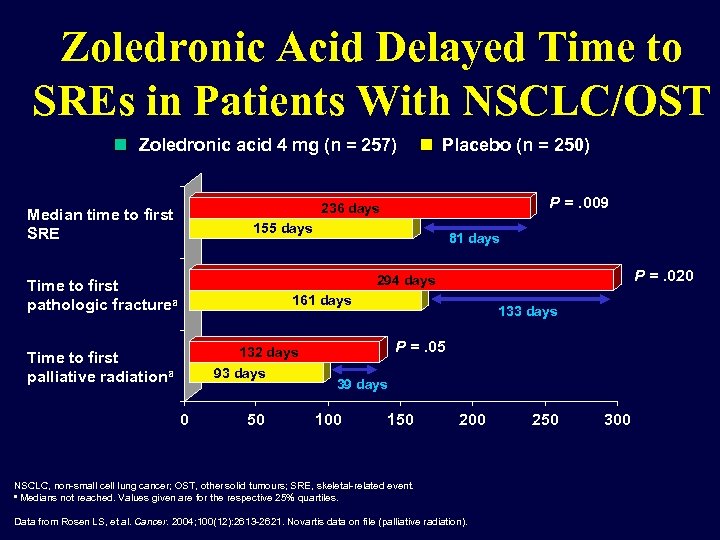
Zoledronic Acid Delayed Time to SREs in Patients With NSCLC/OST Zoledronic acid 4 mg (n = 257) Placebo (n = 250) P =. 009 236 days Median time to first SRE 155 days 81 days P =. 020 294 days Time to first pathologic fracturea 161 days 133 days P =. 05 132 days Time to first palliative radiationa 93 days 0 50 39 days 100 150 200 NSCLC, non-small cell lung cancer; OST, other solid tumours; SRE, skeletal-related event. a Medians not reached. Values given are for the respective 25% quartiles. Data from Rosen LS, et al. Cancer. 2004; 100(12): 2613 -2621. Novartis data on file (palliative radiation). 250 300

Zoledronic Acid Reduced the Risk of Developing an SRE Risk reduction P value 31% . 003 32% . 016 0. 693 NSCLC + OST 1 0. 675 NSCLC 2 0 0. 2 0. 4 0. 6 0. 8 1 1. 2 1. 4 1. 6 1. 8 2 Risk ratio (zoledronic acid 4 mg versus placebo) In favour of zoledronic acid In favour of placebo NSCLC, non-small cell lung cancer; OST, other solid tumours; SRE, skeletal-related event. 1. Rosen LS, et al. Cancer. 2004; 100(12): 2613 -2621; 2. Belch A, et al. Proc Am Soc Clin Oncol. 2003; 22: 761. Abstract 3058.
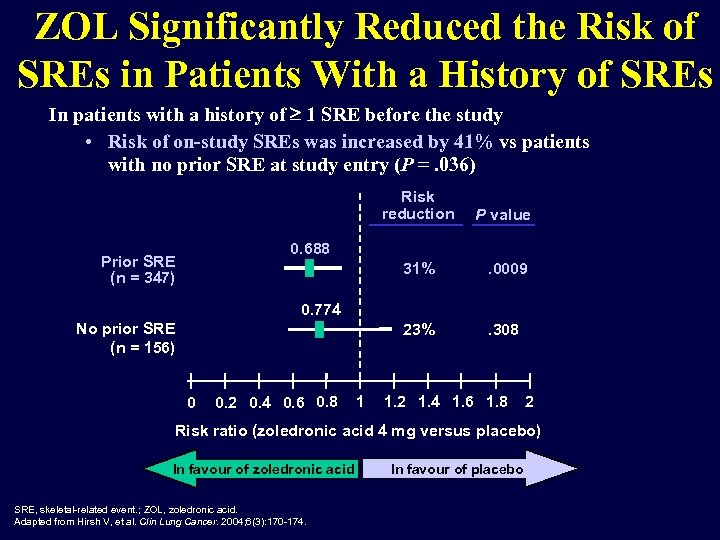
ZOL Significantly Reduced the Risk of SREs in Patients With a History of SREs In patients with a history of 1 SRE before the study • Risk of on-study SREs was increased by 41% vs patients with no prior SRE at study entry (P =. 036) Risk reduction P value 0. 688 Prior SRE (n = 347) 31% . 0009 23% . 308 0. 774 No prior SRE (n = 156) 0 0. 2 0. 4 0. 6 0. 8 1 1. 2 1. 4 1. 6 1. 8 2 Risk ratio (zoledronic acid 4 mg versus placebo) In favour of zoledronic acid SRE, skeletal-related event. ; ZOL, zoledronic acid. Adapted from Hirsh V, et al. Clin Lung Cancer. 2004; 6(3): 170 -174. In favour of placebo
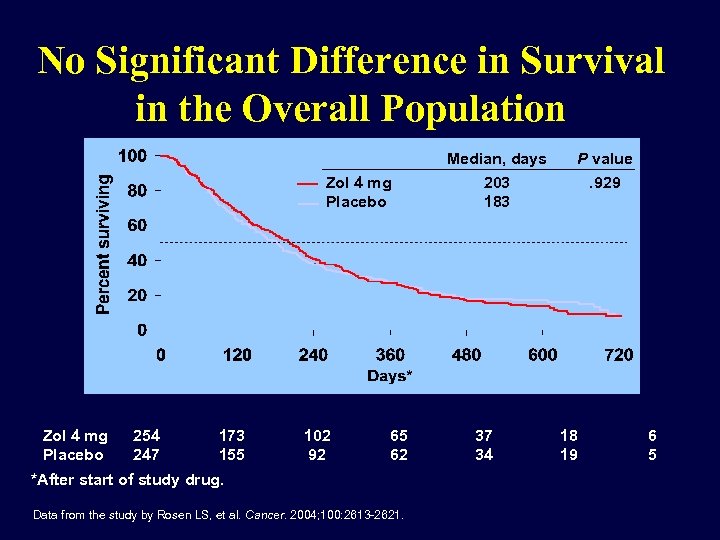
No Significant Difference in Survival in the Overall Population Median, days Zol 4 mg Placebo 254 247 173 155 102 92 65 62 *After start of study drug. Data from the study by Rosen LS, et al. Cancer. 2004; 100: 2613 -2621. P value 203 183 . 929 37 34 18 19 6 5
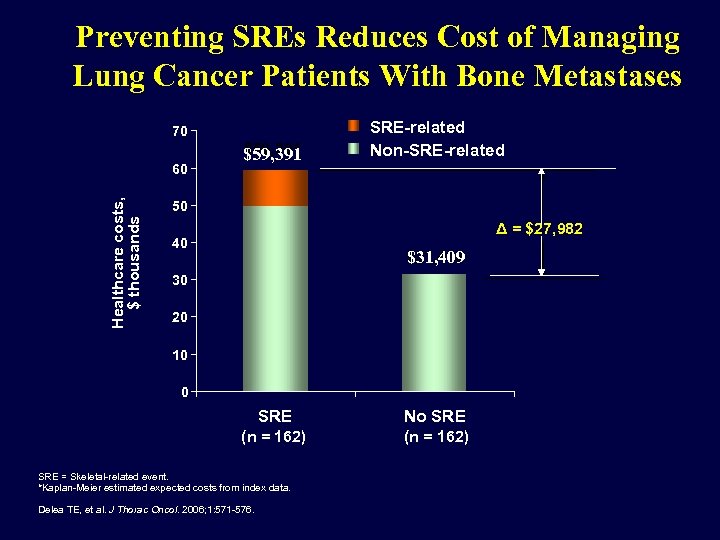
Preventing SREs Reduces Cost of Managing Lung Cancer Patients With Bone Metastases 70 Healthcare costs, $ thousands 60 $59, 391 SRE-related Non-SRE-related 50 Δ = $27, 982 40 $31, 409 30 20 10 0 SRE No SRE (n = 162) SRE = Skeletal-related event. *Kaplan-Meier estimated expected costs from index data. Delea TE, et al. J Thorac Oncol. 2006; 1: 571 -576.
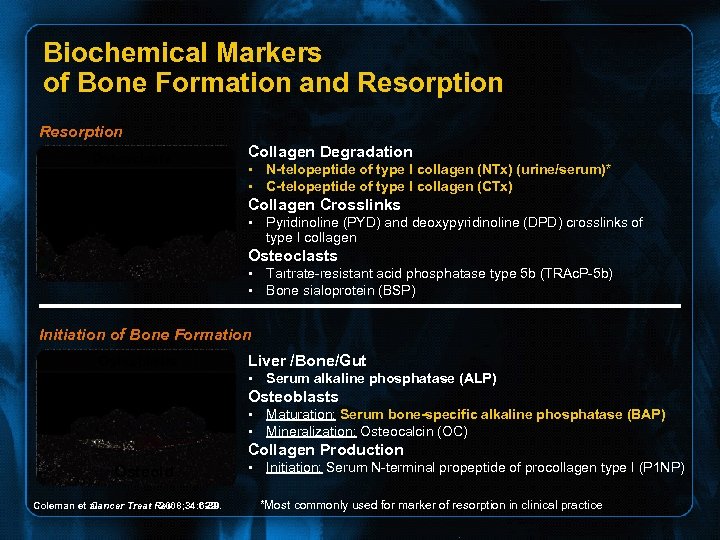
Biochemical Markers of Bone Formation and Resorption Osteoclasts Collagen Degradation • N-telopeptide of type I collagen (NTx) (urine/serum)* • C-telopeptide of type I collagen (CTx) Collagen Crosslinks • Pyridinoline (PYD) and deoxypyridinoline (DPD) crosslinks of type I collagen Osteoclasts • Tartrate-resistant acid phosphatase type 5 b (TRAc. P-5 b) • Bone sialoprotein (BSP) Initiation of Bone Formation Osteoblasts Liver /Bone/Gut • Serum alkaline phosphatase (ALP) Osteoblasts • Maturation: Serum bone-specific alkaline phosphatase (BAP) • Mineralization: Osteocalcin (OC) Collagen Production Osteoid Coleman et al. Cancer Treat Rev 2008; 34: 629 -39. • Initiation: Serum N-terminal propeptide of procollagen type I (P 1 NP) *Most commonly used for marker of resorption in clinical practice.
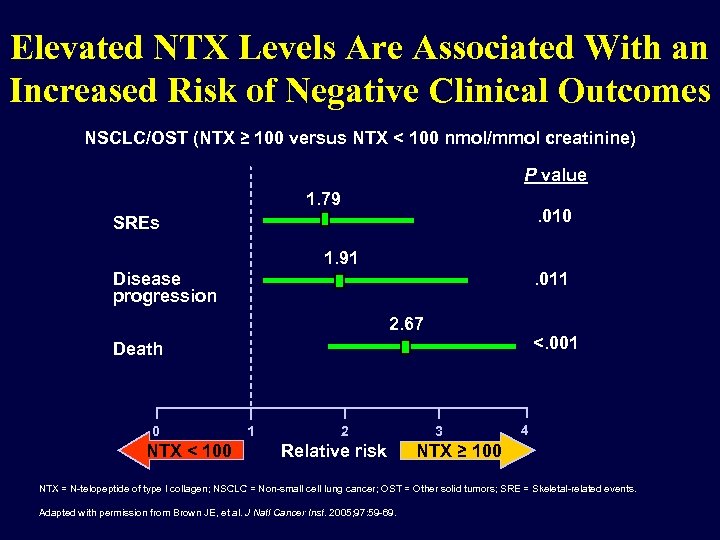
Elevated NTX Levels Are Associated With an Increased Risk of Negative Clinical Outcomes NSCLC/OST (NTX ≥ 100 versus NTX < 100 nmol/mmol creatinine) P value 1. 79 . 010 SREs 1. 91 . 011 Disease progression 2. 67 <. 001 Death 0 NTX < 100 1 2 Relative risk 3 4 NTX ≥ 100 NTX = N-telopeptide of type I collagen; NSCLC = Non-small cell lung cancer; OST = Other solid tumors; SRE = Skeletal-related events. Adapted with permission from Brown JE, et al. J Natl Cancer Inst. 2005; 97: 59 -69.
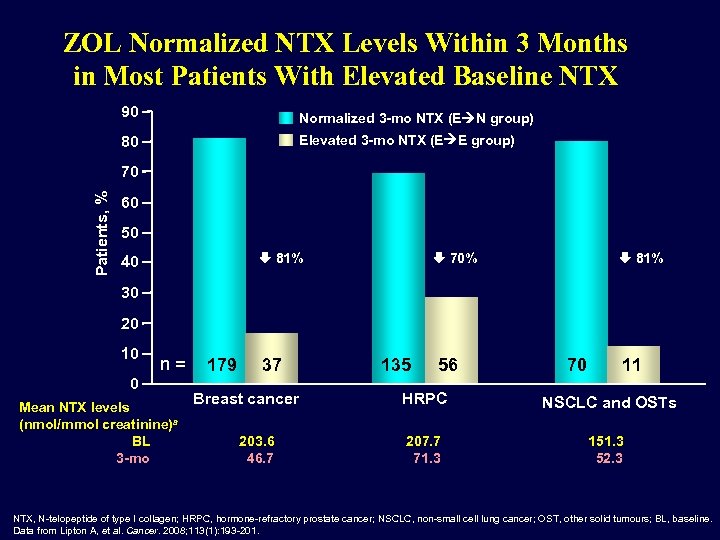
ZOL Normalized NTX Levels Within 3 Months in Most Patients With Elevated Baseline NTX 90 Normalized 3 -mo NTX (E N group) 80 Elevated 3 -mo NTX (E E group) Patients, % 70 60 50 81% 40 70% 81% 30 20 10 0 n = 179 37 Breast cancer 135 56 70 11 HRPC NSCLC and OSTs Mean NTX levels (nmol/mmol creatinine)a BL 203. 6 207. 7 151. 3 3 -mo 46. 7 71. 3 52. 3 NTX, N-telopeptide of type I collagen; HRPC, hormone-refractory prostate cancer; NSCLC, non-small cell lung cancer; OST, other solid tumours; BL, baseline. Data from Lipton A, et al. Cancer. 2008; 113(1): 193 -201.
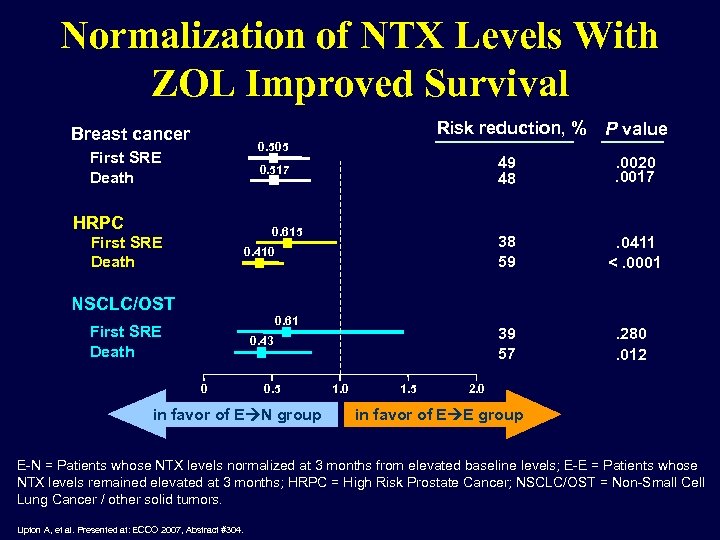
Normalization of NTX Levels With ZOL Improved Survival Risk reduction, % P value Breast cancer 0. 505 First SRE Death 49 48 HRPC First SRE Death 0. 410 . 0411 <. 0001 39 57 0. 615 . 0020. 0017 38 59 0. 517 . 280. 012 NSCLC/OST 0. 61 First SRE Death 0. 43 0 0. 5 in favor of E N group 1. 0 1. 5 2. 0 in favor of E E group E-N = Patients whose NTX levels normalized at 3 months from elevated baseline levels; E-E = Patients whose NTX levels remained elevated at 3 months; HRPC = High Risk Prostate Cancer; NSCLC/OST = Non-Small Cell Lung Cancer / other solid tumors. Lipton A, et al. Presented at: ECCO 2007, Abstract #304.
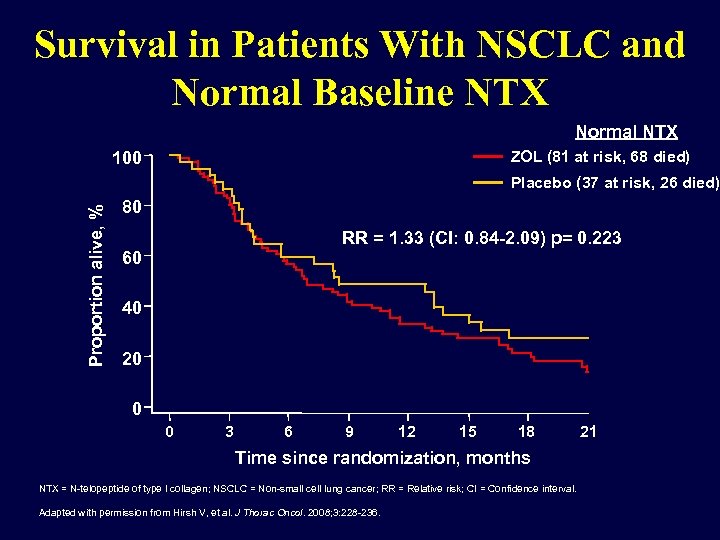
Survival in Patients With NSCLC and Normal Baseline NTX Normal NTX 100 ZOL (81 at risk, 68 died) Proportion alive, % Placebo (37 at risk, 26 died) 80 RR = 1. 33 (CI: 0. 84 -2. 09) p= 0. 223 60 40 20 0 0 3 6 9 12 15 18 Time since randomization, months NTX = N-telopeptide of type I collagen; NSCLC = Non-small cell lung cancer; RR = Relative risk; CI = Confidence interval. Adapted with permission from Hirsh V, et al. J Thorac Oncol. 2008; 3: 228 -236. 21
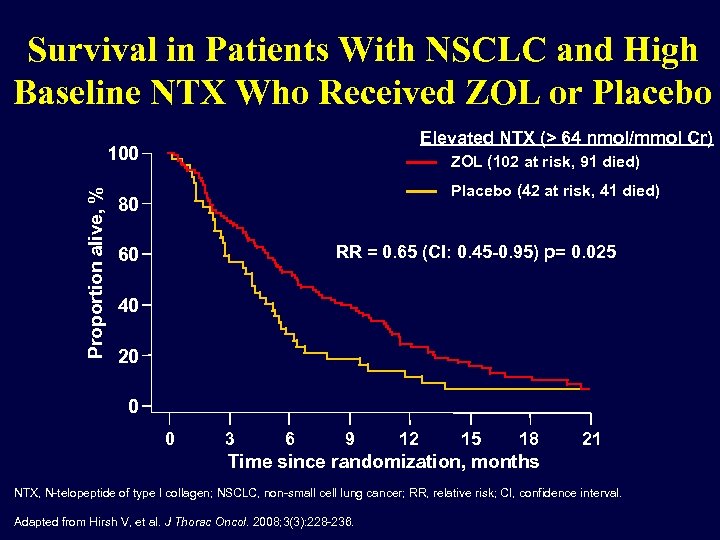
Survival in Patients With NSCLC and High Baseline NTX Who Received ZOL or Placebo Elevated NTX (> 64 nmol/mmol Cr) Proportion alive, % 100 ZOL (102 at risk, 91 died) Placebo (42 at risk, 41 died) 80 RR = 0. 65 (CI: 0. 45 -0. 95) p= 0. 025 60 40 20 0 0 3 6 9 12 15 18 21 Time since randomization, months NTX, N-telopeptide of type I collagen; NSCLC, non-small cell lung cancer; RR, relative risk; CI, confidence interval. Adapted from Hirsh V, et al. J Thorac Oncol. 2008; 3(3): 228 -236.
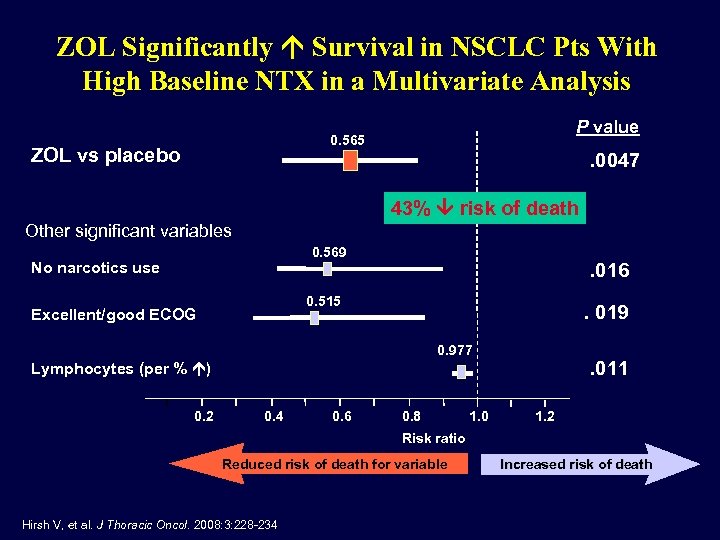
ZOL Significantly Survival in NSCLC Pts With High Baseline NTX in a Multivariate Analysis P value 0. 565 ZOL vs placebo . 0047 43% risk of death Other significant variables 0. 569 No narcotics use . 016 0. 515 Excellent/good ECOG . 019 0. 977 . 011 Lymphocytes (per % ) 0. 2 0. 4 0. 6 0. 8 1. 0 1. 2 Risk ratio Reduced risk of death for variable Hirsh V, et al. J Thoracic Oncol. 2008: 3: 228 -234. Increased risk of death

RANK Ligand Plays a Destructive Role in Patients With Bone Metastases From Solid Tumours
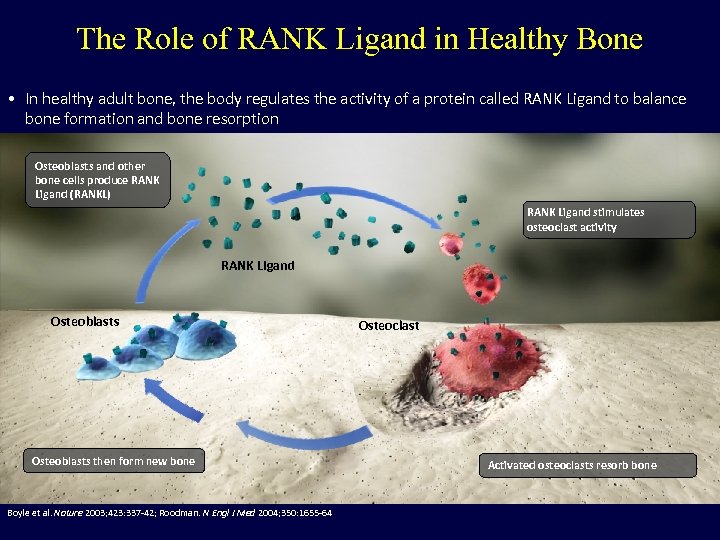
The Role of RANK Ligand in Healthy Bone • In healthy adult bone, the body regulates the activity of a protein called RANK Ligand to balance bone formation and bone resorption Osteoblasts and other bone cells produce RANK Ligand (RANKL) RANK Ligand stimulates osteoclast activity RANK Ligand Osteoblasts then form new bone Boyle et al. Nature 2003; 423: 337 -42; Roodman. N Engl J Med 2004; 350: 1655 -64 Osteoclast Activated osteoclasts resorb bone
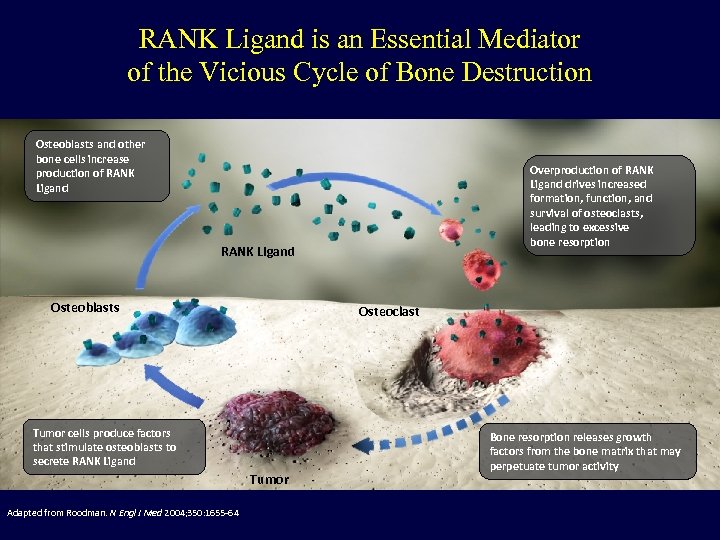
RANK Ligand is an Essential Mediator of the Vicious Cycle of Bone Destruction Osteoblasts and other bone cells increase production of RANK Ligand Overproduction of RANK Ligand drives increased formation, function, and survival of osteoclasts, leading to excessive bone resorption RANK Ligand Osteoblasts Osteoclast Tumor cells produce factors that stimulate osteoblasts to secrete RANK Ligand Tumor Adapted from Roodman. N Engl J Med 2004; 350: 1655 -64 Bone resorption releases growth factors from the bone matrix that may perpetuate tumor activity

Denosumab: A RANK Ligand Inhibitor • RANK Ligand is essential for osteoclast formation, function, and survival • Fully human monoclonal antibody to RANK Ligand • Ig. G 2 immunoglobulin isotype • High affinity and specificity for human Rank Ligand • No detectable binding to other key TNF family members • Administered via subcutaneous (SC) injection TNF = Tumor Necrosis Factor Bekker et al J Bone Miner Res 2004; 19: 1059 -66, Boyle et al Nature 2003; 423: 337 -42, Peterson et al J Bone Miner Res 2003; 18: S 166. Abstract SA 393.
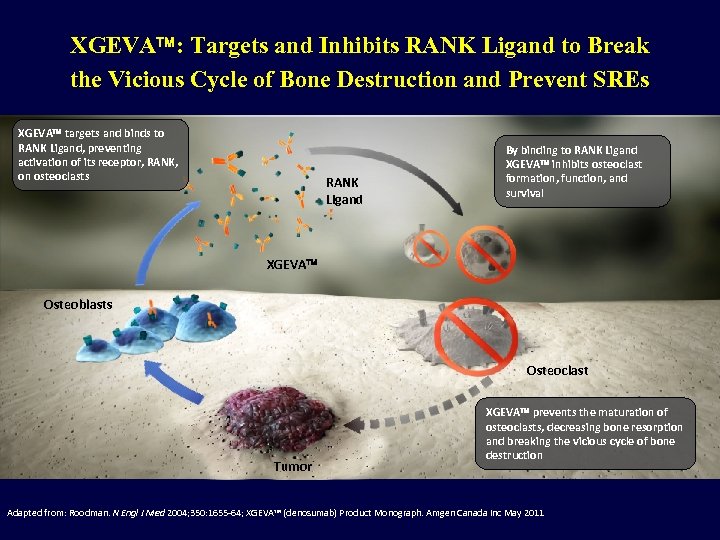
XGEVA : Targets and Inhibits RANK Ligand to Break the Vicious Cycle of Bone Destruction and Prevent SREs XGEVA targets and binds to RANK Ligand, preventing activation of its receptor, RANK, on osteoclasts RANK Ligand By binding to RANK Ligand XGEVA inhibits osteoclast formation, function, and survival XGEVA Osteoblasts Osteoclast Tumor XGEVA prevents the maturation of osteoclasts, decreasing bone resorption and breaking the vicious cycle of bone destruction Adapted from: Roodman. N Engl J Med 2004; 350: 1655 -64; XGEVA™ (denosumab) Product Monograph. Amgen Canada Inc May 2011

Randomized, Double-Blind, Study of Denosumab vs Zoledronic Acid in the Treatment of Bone Metastases in Patients With Advanced Cancer (Excluding Breast and Prostate Cancer) or Multiple Myeloma Henry DH, Costa L, Goldwasser F, et al. J Clin Oncol. 2011; 29: 1125 -1132

Study Design: International, Randomized, Double-Blind, Active-Controlled, Phase 3 Trial Key Inclusion • Adults with solid tumors and bone metastases (excluding breast and prostate) or multiple myeloma Key Exclusion • Current or prior intravenous bisphonate administration 1° Endpoint 2° Endpoints N = 886 Denosumab 120 mg SC and Placebo IV* Q 4 W Supplemental Calcium and Vitamin D N = 890 Zoledronic Acid 4 mg IV* and Placebo SC Q 4 W • Time to first on-study SRE (non inferiority) • Time to first on-study SRE (superiority) • Time to first-and-subsequent on-study SRE (superiority) *IV product dose adjusted for baseline creatinine clearance and subsequent dose intervals determined by serum creatinine (per Zometa label) Henry D et al. European Journal of Cancer Supplements, Vol. 7, No 3, September 2009, Page 11. Abstract 20 LBA and Oral Presentation.
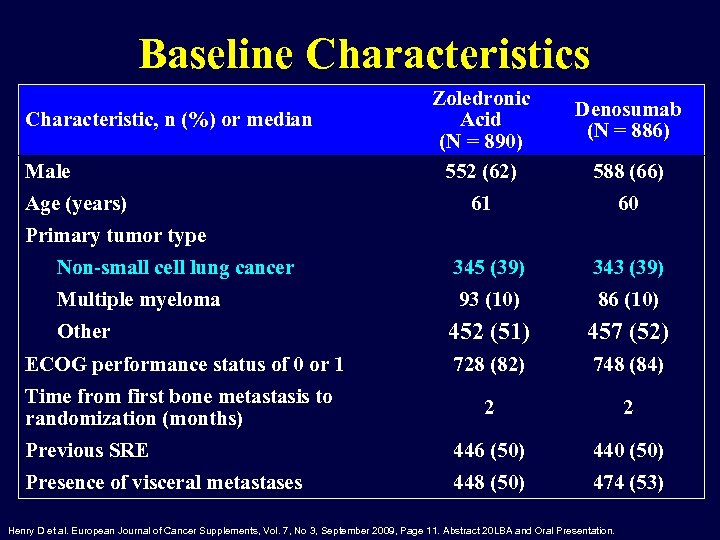
Baseline Characteristics Characteristic, n (%) or median Male Age (years) Zoledronic Acid (N = 890) 552 (62) Denosumab (N = 886) 588 (66) 61 60 Primary tumor type Non-small cell lung cancer 345 (39) 343 (39) Multiple myeloma 93 (10) 86 (10) 452 (51) 457 (52) ECOG performance status of 0 or 1 728 (82) 748 (84) Time from first bone metastasis to randomization (months) 2 2 Previous SRE 446 (50) 440 (50) Presence of visceral metastases 448 (50) 474 (53) Other Henry D et al. European Journal of Cancer Supplements, Vol. 7, No 3, September 2009, Page 11. Abstract 20 LBA and Oral Presentation.
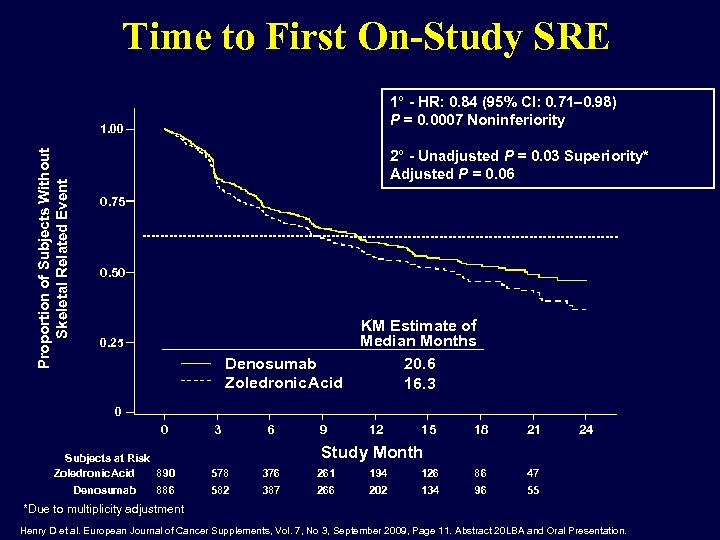
Time to First On-Study SRE 1° - HR: 0. 84 (95% CI: 0. 71– 0. 98) P = 0. 0007 Noninferiority Proportion of Subjects Without Skeletal Related Event 1. 00 2° - Unadjusted P = 0. 03 Superiority* Adjusted P = 0. 06 0. 75 0. 50 0. 25 Denosumab Zoledronic. Acid KM Estimate of Median Months 20. 6 16. 3 0 0 3 6 9 12 15 18 21 24 Study Month Subjects at Risk 890 Zoledronic Acid 578 376 261 194 126 86 47 886 582 387 266 202 134 96 55 Denosumab *Due to multiplicity adjustment Henry D et al. European Journal of Cancer Supplements, Vol. 7, No 3, September 2009, Page 11. Abstract 20 LBA and Oral Presentation.
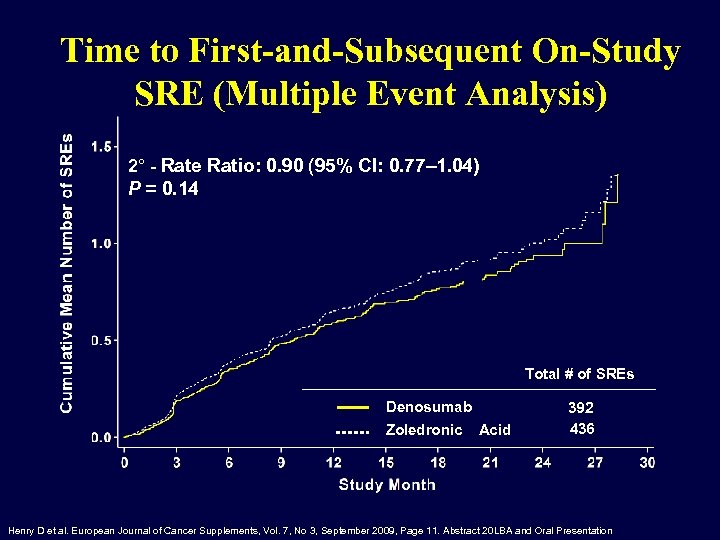
Time to First-and-Subsequent On-Study SRE (Multiple Event Analysis) 2° - Rate Ratio: 0. 90 (95% CI: 0. 77– 1. 04) P = 0. 14 Total # of SREs Denosumab Zoledronic Acid 392 436 Henry D et al. European Journal of Cancer Supplements, Vol. 7, No 3, September 2009, Page 11. Abstract 20 LBA and Oral Presentation.
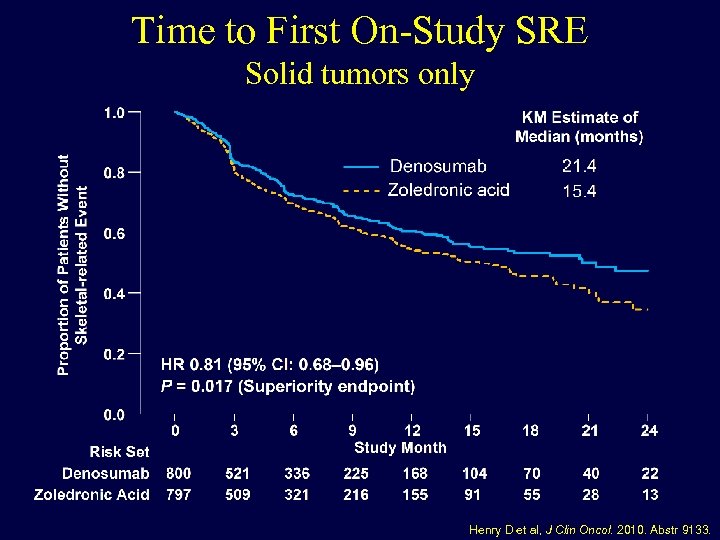
Time to First On-Study SRE Solid tumors only Henry D et al, J Clin Oncol. 2010. Abstr 9133.
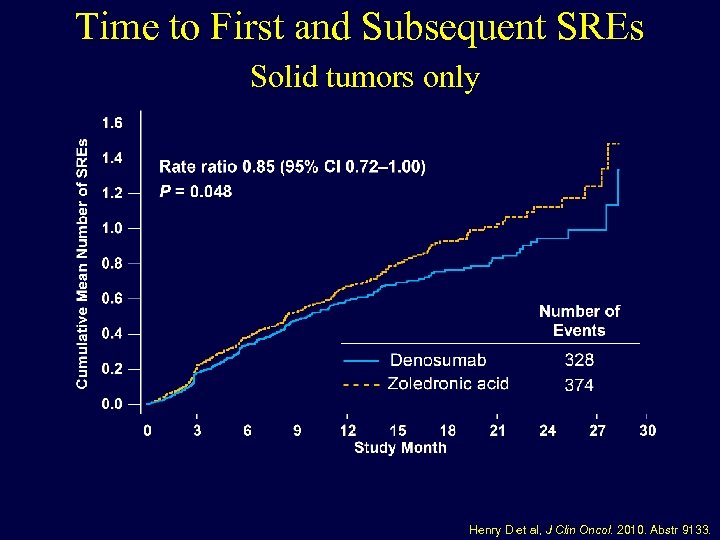
Time to First and Subsequent SREs Solid tumors only Henry D et al, J Clin Oncol. 2010. Abstr 9133.
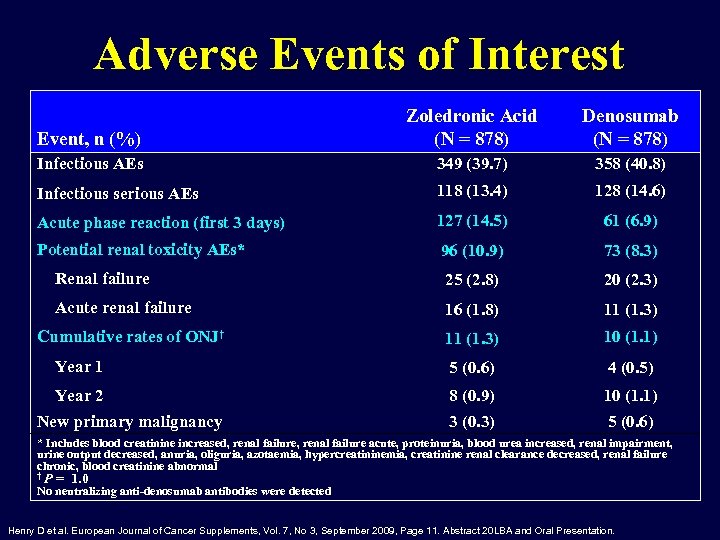
Adverse Events of Interest Event, n (%) Zoledronic Acid (N = 878) Denosumab (N = 878) Infectious AEs 349 (39. 7) 358 (40. 8) Infectious serious AEs 118 (13. 4) 128 (14. 6) Acute phase reaction (first 3 days) 127 (14. 5) 61 (6. 9) Potential renal toxicity AEs* 96 (10. 9) 73 (8. 3) Renal failure 25 (2. 8) 20 (2. 3) Acute renal failure 16 (1. 8) 11 (1. 3) 10 (1. 1) Year 1 5 (0. 6) 4 (0. 5) Year 2 8 (0. 9) 10 (1. 1) 3 (0. 3) 5 (0. 6) Cumulative rates of ONJ† New primary malignancy * Includes blood creatinine increased, renal failure acute, proteinuria, blood urea increased, renal impairment, urine output decreased, anuria, oliguria, azotaemia, hypercreatininemia, creatinine renal clearance decreased, renal failure chronic, blood creatinine abnormal † P = 1. 0 No neutralizing anti-denosumab antibodies were detected Henry D et al. European Journal of Cancer Supplements, Vol. 7, No 3, September 2009, Page 11. Abstract 20 LBA and Oral Presentation.
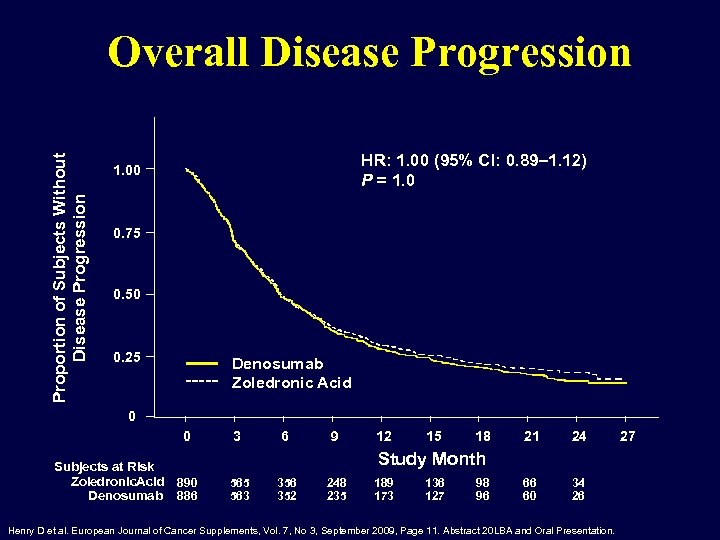
Proportion of Subjects Without Disease Progression Overall Disease Progression HR: 1. 00 (95% CI: 0. 89– 1. 12) P = 1. 00 0. 75 0. 50 0. 25 Denosumab Zoledronic Acid 0 0 Subjects at Risk Zoledronic. Acid 890 Denosumab 886 3 6 9 12 15 18 21 24 66 60 34 26 Study Month 565 563 356 352 248 235 189 173 136 127 98 96 Henry D et al. European Journal of Cancer Supplements, Vol. 7, No 3, September 2009, Page 11. Abstract 20 LBA and Oral Presentation. 27
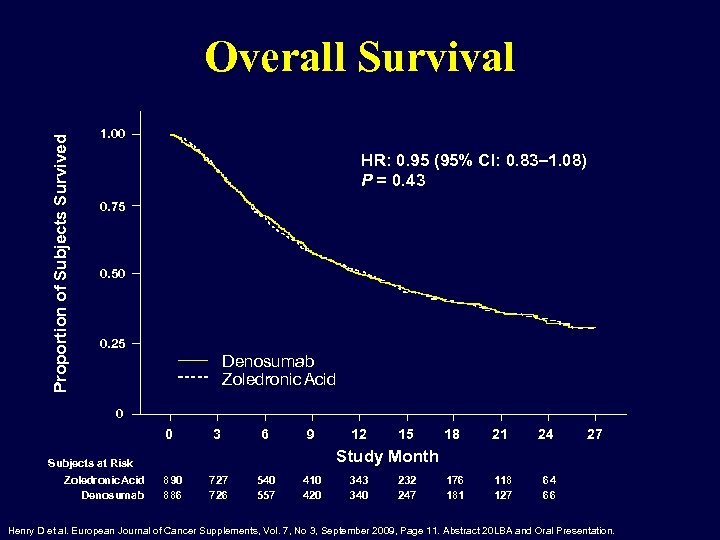
Proportion of Subjects Survived Overall Survival 1. 00 HR: 0. 95 (95% CI: 0. 83– 1. 08) P = 0. 43 0. 75 0. 50 0. 25 Denosumab Zoledronic Acid 0 0 3 6 9 15 18 21 24 176 181 118 127 27 64 66 Study Month Subjects at Risk Zoledronic. Acid Denosumab 12 890 886 727 726 540 557 410 420 343 340 232 247 Henry D et al. European Journal of Cancer Supplements, Vol. 7, No 3, September 2009, Page 11. Abstract 20 LBA and Oral Presentation.

Pain Outcomes in a Randomized Phase 3 Clinical Trial of Denosumab vs Zoledronic Acid (ZA) in Patients with Solid Tumors and Bone Metastases L Fallowfield, 1 R von Moos, 2 D Patrick, 3 CS Cleeland, 4 DH Henry, 5 V Hirsh, 6 K Zarogoulidis, 7 W Ying, 8 K Chung, 9 Z Cong, 9 H Yeh 10 1 University of Sussex, Cancer Research UK, Brighton, United Kingdom; 2 Kantonsspital Graubünden, Oncology, Chur, Switzerland; 3 University of Washington, Health Services, Seattle, USA; 4 University of Texas, Symptom Research Division of Internal Medicine, Texas, USA; 5 Pennsylvania Hospital, Joan Karnell Cancer Center, Philadelphia, USA; 6 Mc. Gill University Health Centre, Oncology, Montreal, Canada; 7 Aristotle University of Thessaloniki G. Papanikolaou Hospital, Pulmonary Department, Thessaloniki, Greece; 8 Amgen Inc. , Biostatistics, Thousand Oaks, USA; 9 Amgen Inc. , Health Economics, Thousand Oaks, USA; 10 Amgen Inc. , Clinical Research, Thousand Oaks, USA
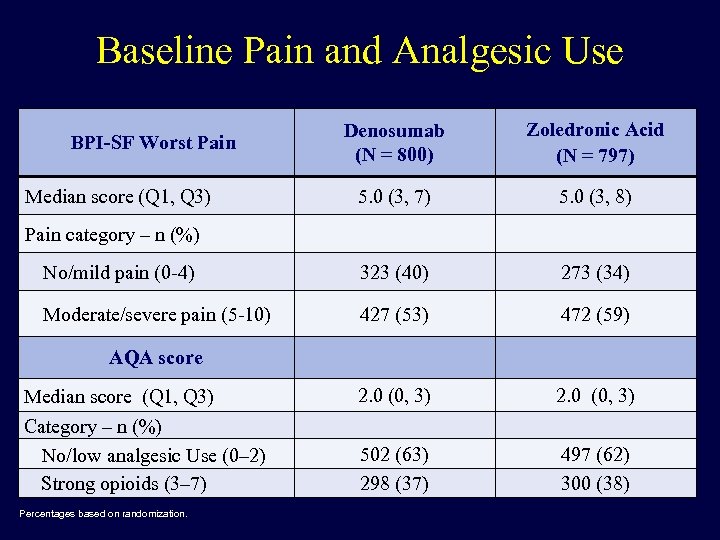
Baseline Pain and Analgesic Use Denosumab (N = 800) Zoledronic Acid (N = 797) 5. 0 (3, 8) No/mild pain (0 -4) 323 (40) 273 (34) Moderate/severe pain (5 -10) 427 (53) 472 (59) Median score (Q 1, Q 3) 2. 0 (0, 3) Category – n (%) No/low analgesic Use (0– 2) Strong opioids (3– 7) 502 (63) 298 (37) 497 (62) 300 (38) BPI-SF Worst Pain Median score (Q 1, Q 3) Pain category – n (%) AQA score Percentages based on randomization.
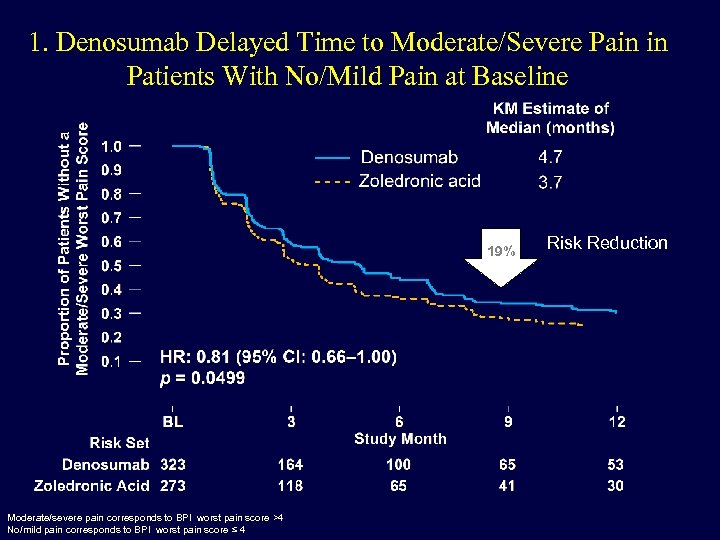
1. Denosumab Delayed Time to Moderate/Severe Pain in Patients With No/Mild Pain at Baseline 19% Moderate/severe pain corresponds to BPI worst pain score >4 No/mild pain corresponds to BPI worst pain score ≤ 4 Risk Reduction
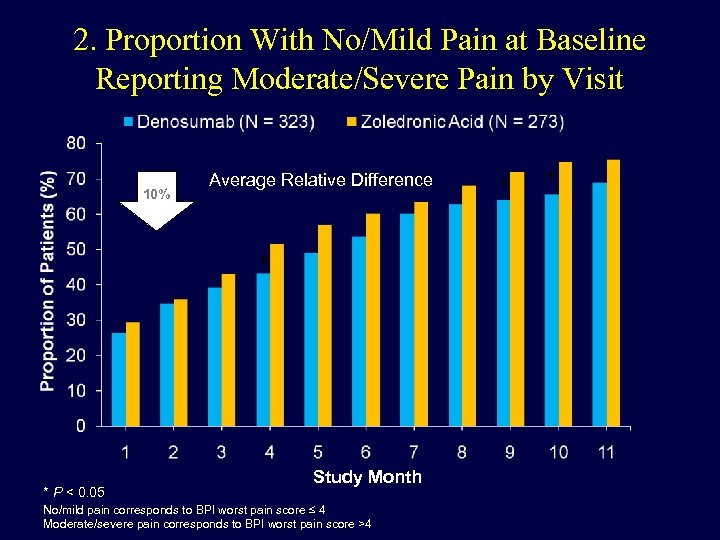
2. Proportion With No/Mild Pain at Baseline Reporting Moderate/Severe Pain by Visit 10% Average Relative Difference * * P < 0. 05 Study Month No/mild pain corresponds to BPI worst pain score ≤ 4 Moderate/severe pain corresponds to BPI worst pain score >4 * *
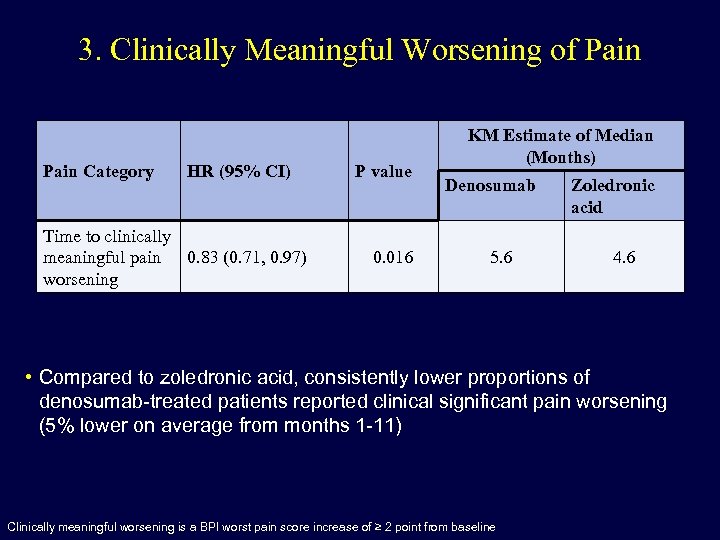
3. Clinically Meaningful Worsening of Pain Category HR (95% CI) Time to clinically meaningful pain 0. 83 (0. 71, 0. 97) worsening P value 0. 016 KM Estimate of Median (Months) Denosumab 5. 6 Zoledronic acid 4. 6 • Compared to zoledronic acid, consistently lower proportions of denosumab-treated patients reported clinical significant pain worsening (5% lower on average from months 1 -11) Clinically meaningful worsening is a BPI worst pain score increase of ≥ 2 point from baseline
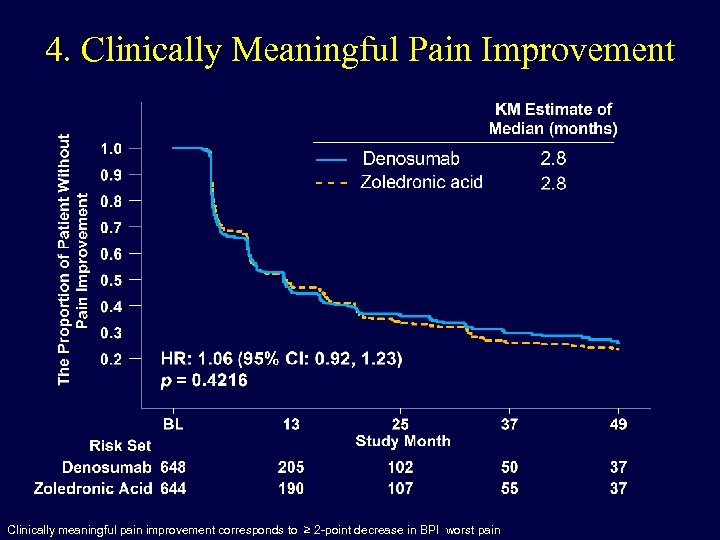
4. Clinically Meaningful Pain Improvement Clinically meaningful pain improvement corresponds to ≥ 2 -point decrease in BPI worst pain
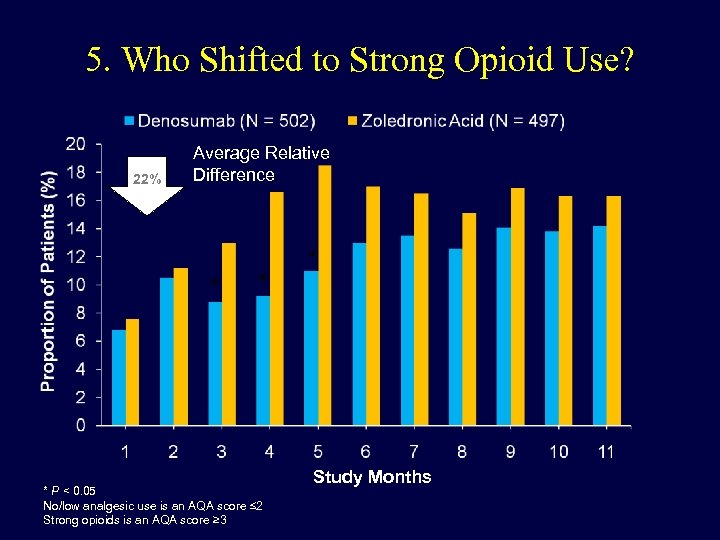
5. Who Shifted to Strong Opioid Use? 22% Average Relative Difference * * * P < 0. 05 No/low analgesic use is an AQA score ≤ 2 Strong opioids is an AQA score ≥ 3 * Study Months

Anticancer Activity of Zoledronic Acid in NSCLC
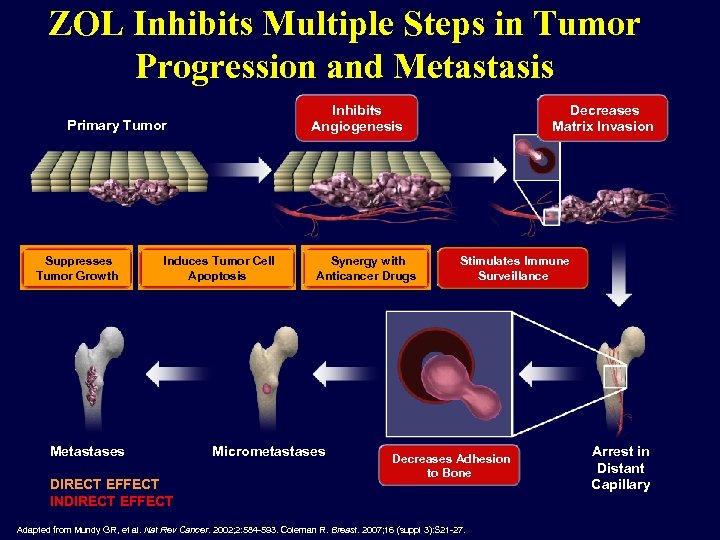
ZOL Inhibits Multiple Steps in Tumor Progression and Metastasis Inhibits Angiogenesis Primary Tumor Suppresses Tumor Growth Induces Tumor Cell Apoptosis Metastases DIRECT EFFECT INDIRECT EFFECT Synergy with Anticancer Drugs Micrometastases Decreases Matrix Invasion Stimulates Immune Surveillance Decreases Adhesion to Bone Adapted from Mundy GR, et al. Nat Rev Cancer. 2002; 2: 584 -593. Coleman R. Breast. 2007; 16 (suppl 3): S 21 -27. Arrest in Distant Capillary

Overall Survival Improvement in Patients With Lung Cancer Treated With Denosumab Versus Zoledronic Acid: Results From a Randomized Phase 3 Study G Scagliotti 1, V Hirsh 2, S Siena 3, D Henry 4, P Woll 5, C Manegold 6, P Solal-Celigny 7, G Rodriguez 8, M Krzakowski 9, ND Mehta 10, L Lipton 11, JA García-Sáenz 12, J Pereira 13, K Prabhash 14, C Tudor-Eliade 15, V Kanarev 16, A Feng 17, I Jacobs 17 1 University of Turino, Orbassano, Italy; 2 Mc. Gill University Health Centre, Montreal, Canada, 3 Ospedale Niguarda Cà Granda, Milan, Italy; 4 Joan Karnell Cancer Center, Philadelphia, PA, USA; 5 Weston Park Hospital, University of Sheffield, UK; 6 Klinikum Mannheim, Germany; 7 Clinique Victor Hugo, Le Mans, France; 8 South Texas Oncology and Hematology, San Antonio, TX, USA; 9 The Maria Sklodowska-Curie Institute of Oncology, Warsaw, Poland; 10 Oncology Hematology Associates of Northern Illinois, Gurnee, IL, USA; 11 Western Hospital Footscray, Vic, Australia; 12 Hospital Clínico San Carlos, Madrid, Spain; 13 Instituto do Cancer Arnaldo Vieira de Carvalho, Sao Paolo, Brazil; 14 Tata Memorial Hospital, Mumbai, India; 15 Institutul Oncologic I. Chiricuta, Cluj-Naooca, Romania; 16 Regional Oncology Dispensary with Inpatient Sector, Plovdiv, Bulgaria; 17 Amgen Inc. , Thousand Oaks, CA, USA
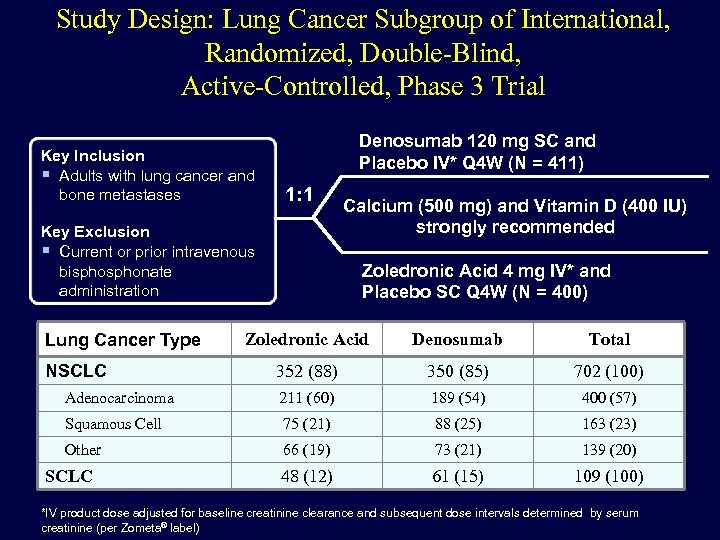
Study Design: Lung Cancer Subgroup of International, Randomized, Double-Blind, Active-Controlled, Phase 3 Trial Key Inclusion Adults with lung cancer and bone metastases Denosumab 120 mg SC and Placebo IV* Q 4 W (N = 411) 1: 1 Key Exclusion Current or prior intravenous bisphonate administration Lung Cancer Type Calcium (500 mg) and Vitamin D (400 IU) strongly recommended Zoledronic Acid 4 mg IV* and Placebo SC Q 4 W (N = 400) Zoledronic Acid Denosumab Total NSCLC 352 (88) 350 (85) 702 (100) Adenocarcinoma 211 (60) 189 (54) 400 (57) Squamous Cell 75 (21) 88 (25) 163 (23) Other 66 (19) 73 (21) 139 (20) SCLC 48 (12) 61 (15) 109 (100) *IV product dose adjusted for baseline creatinine clearance and subsequent dose intervals determined by serum creatinine (per Zometa® label)
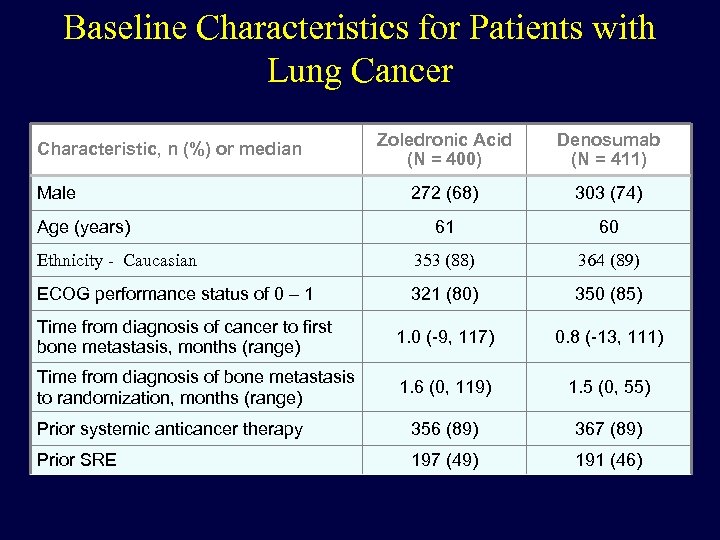
Baseline Characteristics for Patients with Lung Cancer Zoledronic Acid (N = 400) Denosumab (N = 411) 272 (68) 303 (74) 61 60 Ethnicity - Caucasian 353 (88) 364 (89) ECOG performance status of 0 – 1 321 (80) 350 (85) Time from diagnosis of cancer to first bone metastasis, months (range) 1. 0 (-9, 117) 0. 8 (-13, 111) Time from diagnosis of bone metastasis to randomization, months (range) 1. 6 (0, 119) 1. 5 (0, 55) Prior systemic anticancer therapy 356 (89) 367 (89) Prior SRE 197 (49) 191 (46) Characteristic, n (%) or median Male Age (years)
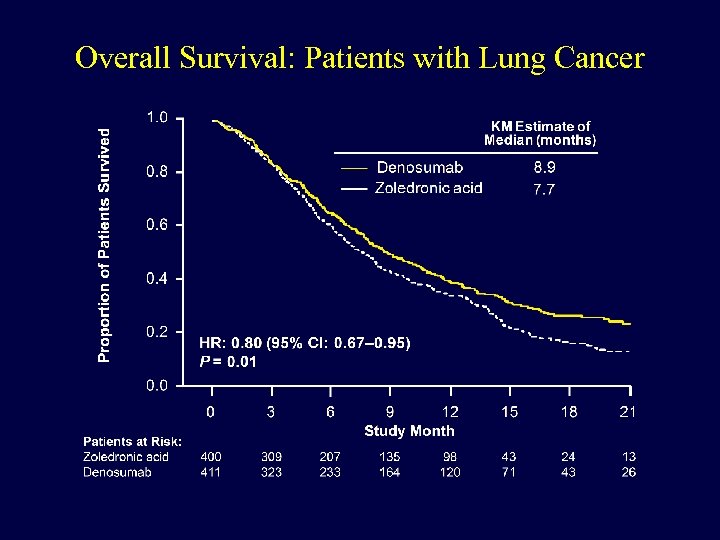
Overall Survival: Patients with Lung Cancer
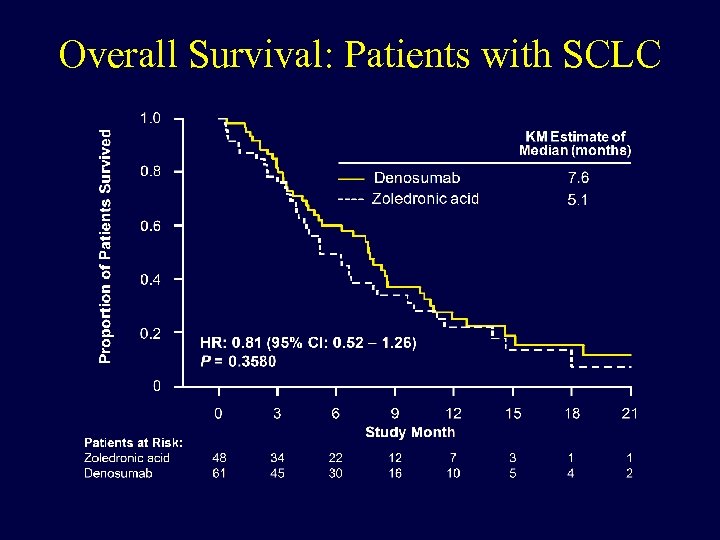
Overall Survival: Patients with SCLC
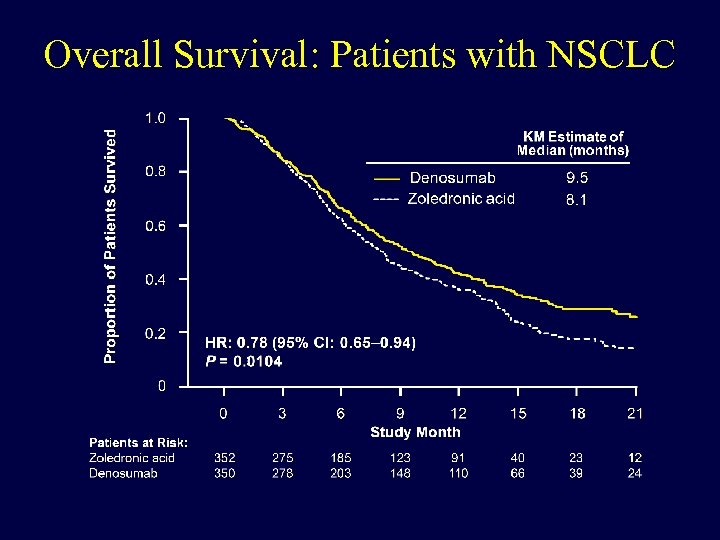
Overall Survival: Patients with NSCLC
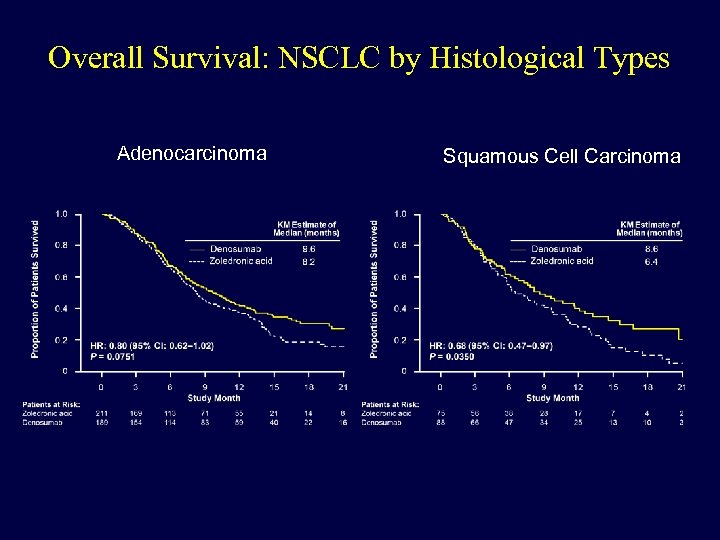
Overall Survival: NSCLC by Histological Types Adenocarcinoma Squamous Cell Carcinoma

SUMMARY • In this exploratory analysis of a large subgroup of patients with NSCLC and SCLC cancer and bone metastases, denosumab treatment is associated with significantly improved overall survival versus zoledronic acid • Preclinical investigation underway to evaluate if RANKL inhibition has a direct anti-tumor effects on lung cancer cells (eg apoptosis, inhibition of cancer migration/invasion) • These findings warrant further clinical investigation

Summary • Results from a phase 3 RCT in patients with solid tumors (excluding breast and prostate cancer and multiple myeloma) and bone metastases showed denosumab was superior to zoledronic acid in: – delaying time to first on-study SRE 1 – delaying time to first and subsequent SREs 1 • The subgroup analysis demonstrated: – superior survival on denosumab vs zoledronic acid – better pain control 1. Henry D et al, J Clin Oncol. 2010. Abstr 9133.

THANK YOU!

MC GILL UNIVERSITY

Malignant Bone Disease Src and Osteoclast Function • • SRC activity regulates normal osteoclast function during bone resorption Metastases in bone activate SRC-dependent osteoclast bone resorption Bone Inhibition of SRC reduces bone resorption in preclinical models, healthy volunteers and patients Luo FR, et al. (2005). Evaluation of Anti-osteoclastic Activity of the Novel, Oral Multi-targeted Kinase Inhibitor Dasatinib (BMS-354825). AACR-NCI-EORTC International Conference. Molecular Targets and Cancer Therapeutics, Philadelphia, PA, p 173 (abstr. B 178)
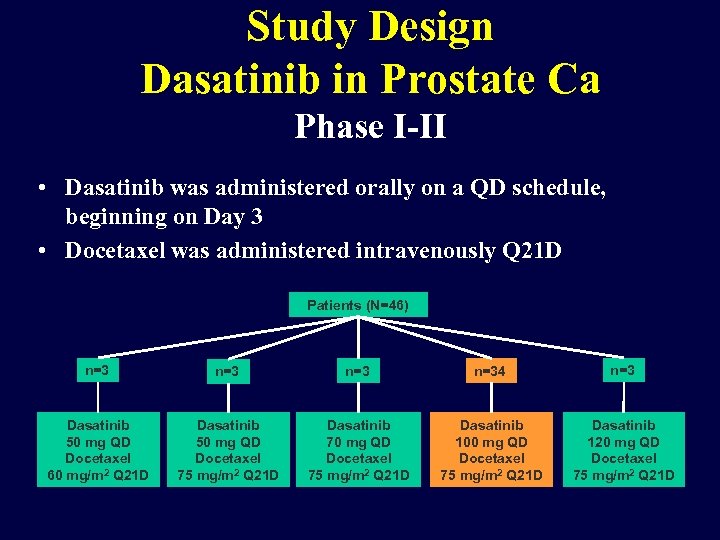
Study Design Dasatinib in Prostate Ca Phase I-II • Dasatinib was administered orally on a QD schedule, beginning on Day 3 • Docetaxel was administered intravenously Q 21 D Patients (N=46) n=3 n=34 n=3 Dasatinib 50 mg QD Docetaxel 60 mg/m 2 Q 21 D Dasatinib 50 mg QD Docetaxel 75 mg/m 2 Q 21 D Dasatinib 70 mg QD Docetaxel 75 mg/m 2 Q 21 D Dasatinib 100 mg QD Docetaxel 75 mg/m 2 Q 21 D Dasatinib 120 mg QD Docetaxel 75 mg/m 2 Q 21 D
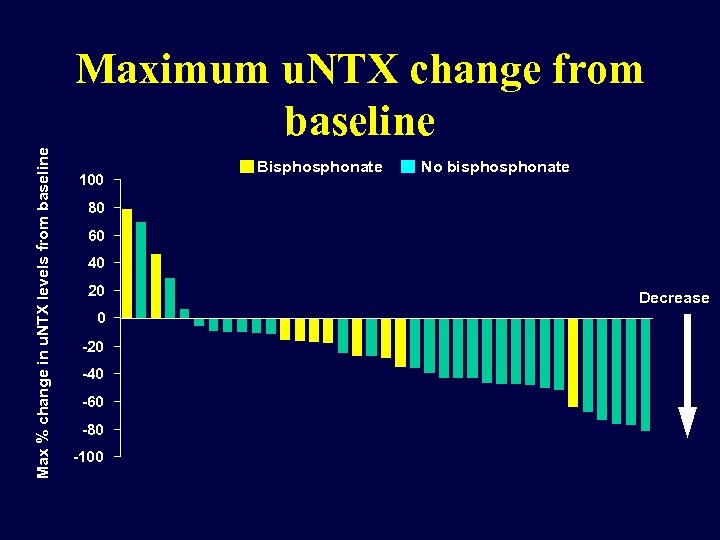
Max % change in u. NTX levels from baseline Maximum u. NTX change from baseline 100 Bisphonate No bisphonate 80 60 40 20 0 -20 -40 -60 -80 -100 Decrease
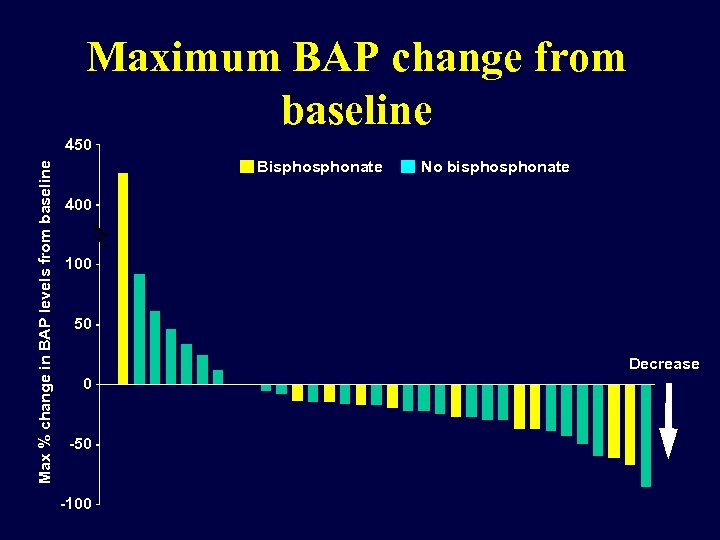
Maximum BAP change from baseline Max % change in BAP levels from baseline 450 Bisphonate No bisphonate 400 100 50 Decrease 0 -50 -100

MC GILL UNIVERSITY

Conclusions • Responses achieved demonstrate the antitumor activity of dasatinib in combination with docetaxel, including patients pretreated with chemotherapy. – 77% had clinical benefit or disease control • 57% had confirmed PR (RECIST), 1 patient had a u. CR – 59% had a PSA response – 42% had no new bone lesions • Dasatinib favorably modulates bone turnover as a single agent 5 and in combination with chemotherapy, supporting dual antitumor and antiosteoclast targeting 7, 8 – 86% had a decrease in u. NTX from baseline – 73% had a decrease in BAP from baseline • Dasatinib and docetaxel combination treatment was well tolerated – low frequency of grade 3/4 AEs – median treatment duration was 6. 2 months for dasatinib and 6 cycles for docetaxel • The results of this study serve as the basis for an ongoing phase 3 study of dasatinib 100 mg QD in combination with docetaxel 75 mg/m 2 Q 21 D

Sotatercept and Activin Confidential 65

THANK YOU!

MC GILL UNIVERSITY

Phase 1: Sotatercept Safety Profile • Hypertension in subjects with marked increase in Hgb (dose-limiting toxicity) at 1 mg/kg DL • Dose-dependent decrease in FSH in postmenopausal women • Mild AEs also reported: – Viral upper respiratory tract infection – Headache – Dizziness – Hot flash – Paresthesia – Muscle spasms – Fatigue and asthenia Confidential 73

MC GILL UNIVERSITY
761f718dd170b160900e0a6fa6896789.ppt