fc828985184129b212c2157e440761c4.ppt
- Количество слайдов: 63

Biopharmaceuticals and agrobioproducts: a North – South conflict? November 16 -17, 2006, Warsaw

The lecture: înew îinventive îcommercial

IPR in BIOTECHNOLOGY means: b CONNECTION of science, technology and industry; b Globalisation; b Innovations, property and profit; b Highly skilled researchers and workers in industry; b Productivity, growth and structural changes; b Information and communication policy.

SECTORS in ECONOMY b Agriculture b Health = = = green biotech white biotech red biotech b violet = legislation (IPR) b Industry b In summary: Contribution of biotechnology to the entire BIOeconomy

VAC b value added chain b= b science & technology + b law & IPR b+ b society & perception b
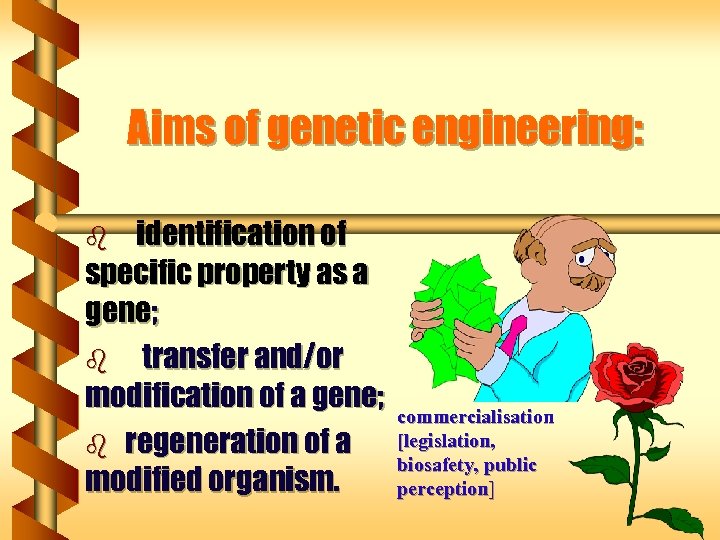
Aims of genetic engineering: identification of specific property as a gene; b transfer and/or modification of a gene; b regeneration of a modified organism. b commercialisation [legislation, biosafety, public perception]

Bioproducts in national and global economy: 1. Biomedicine – – Biopharmaceuticals, Molecular markers, Targeted therapies, Diagnosis. 2. Food & feed – Functional food – Nutraceutics 3. Plants for industry – biomaterials – bioenergy – bioreactors

Biotechnology is green: l l Essential for sustainable future of our society: Renewable resources (“green” bio-resources: biomass, bioenergy, biomaterials) l Clean production [recycling is possible] l The transition from Chemistry Bio-based chemistry one of the essential leads for the transition to renewable bio-resources

opportunities l Genomics and modern (Bio)Technology offers excellent tools for major improvements and innovations for the transition of our strong petro-based chemistry to bio-based chemistry l The use of bio-waste (“biomass”) offers a way of turning environmental burden into environmental advantage

b A report by the USDA national statistics service states that the U. S. has increased the acreage of soybean, corn, and cotton this year. Biotech soybean was the most popular choice amongst farmers, accounting for 89% of the total of all soybeans grown in the U. S. The statistics released include: * 66. 6 million acres of biotech soy were planted in ’ 06, up from 62. 76 million in 2005. * 12. 6 million acres of biotech cotton were planted, accounting for 83% of all cotton grown. * 48. 4 million acres of biotech corn were planted, accounting for 61% of all corn planted, and 6 million acres more than in 2005. Additional information is available at: http: //usda. mannlib. cornell. edu/usda/nass/Acre//2000 s/2006/Acre-06 -30 -2006. pdf

These can be converted to any desired product(s)

Shopping List • • • • Fruit pie Biscuits Bread rolls Chicken soup (tinned) (for vegetarians: vegetable soup) Chocolate bar Cornflakes Crisps Margarine Mayonnaise Pasta Ready meal Asian food Readily prepared schnitzel coated with breadcrumbs (for vegetarians: corn burger) Spaghetti sauce (instant) Sandwich spread Vanilla ice cream

biopharmaceuticals & biomarket trends b Market: 2005 - 602 bln USD (+7, 7% from 2004) – 2010 - 842 bln USD Participation in the market: North America + EU + Japan = 82, 3% Generics: USA 40% Germany 60% Poland 90% New biopharmaceuticals: nervous system + cardiovascular + metabolism therapy HORMONES The leader in biotechnology: AMGEN (12, 5 bln USD 2005)
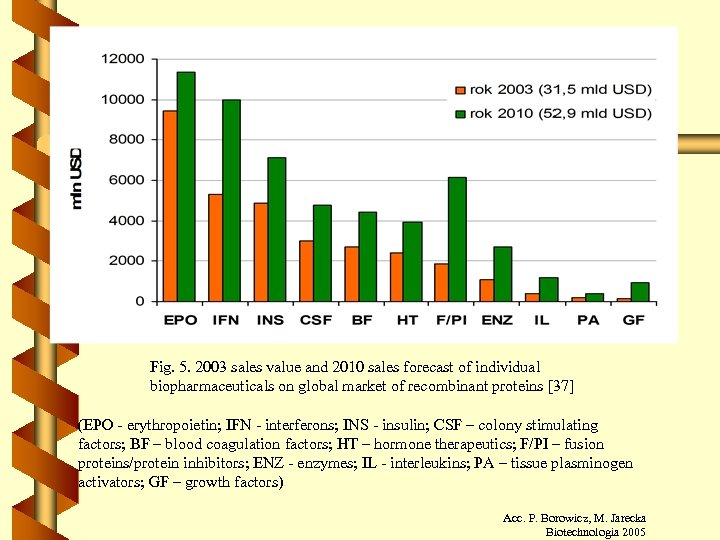
Fig. 5. 2003 sales value and 2010 sales forecast of individual biopharmaceuticals on global market of recombinant proteins [37] (EPO - erythropoietin; IFN - interferons; INS - insulin; CSF – colony stimulating factors; BF – blood coagulation factors; HT – hormone therapeutics; F/PI – fusion proteins/protein inhibitors; ENZ - enzymes; IL - interleukins; PA – tissue plasminogen activators; GF – growth factors) Acc. P. Borowicz, M. Jarecka Biotechnologia 2005

Bioeconomy - how to describe it? b The question: What are the basic indicators of biotech contribution to productivity and overall economy? b The answer: Patents; but: internationally comparable indicators are not available

Track to receive the biotechnological invention

Track to receive the biotechnological invention discovery
Track to receive the biotechnological invention discovery aim
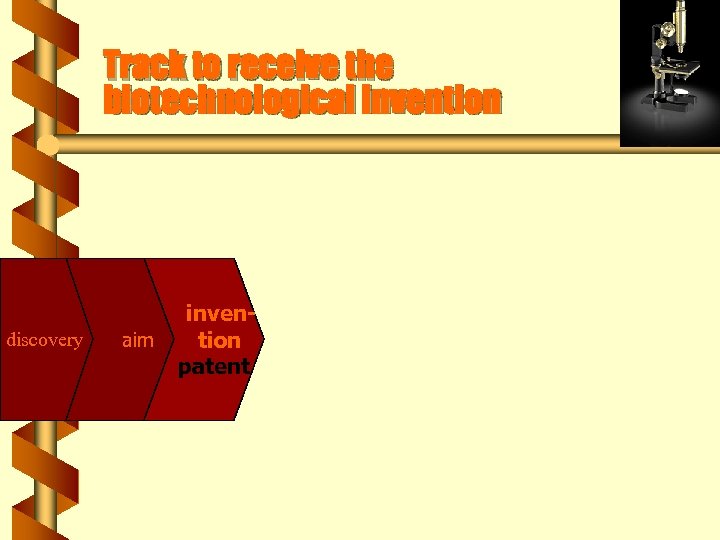
Track to receive the biotechnological invention discovery aim invention patent
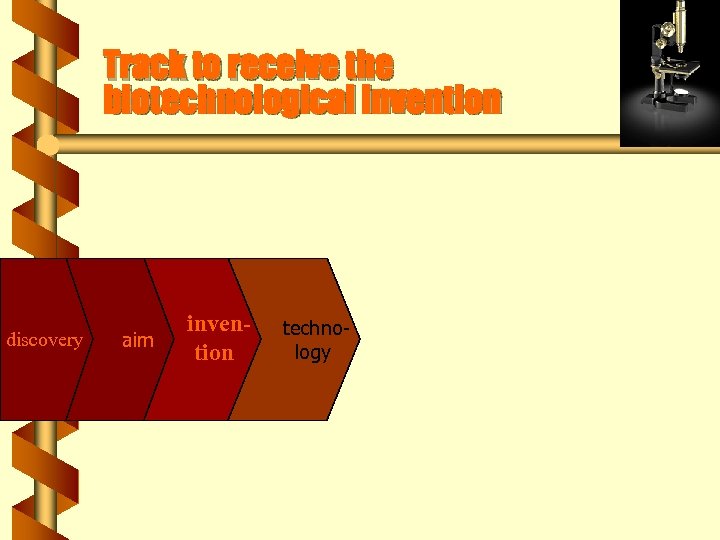
Track to receive the biotechnological invention discovery aim invention technology
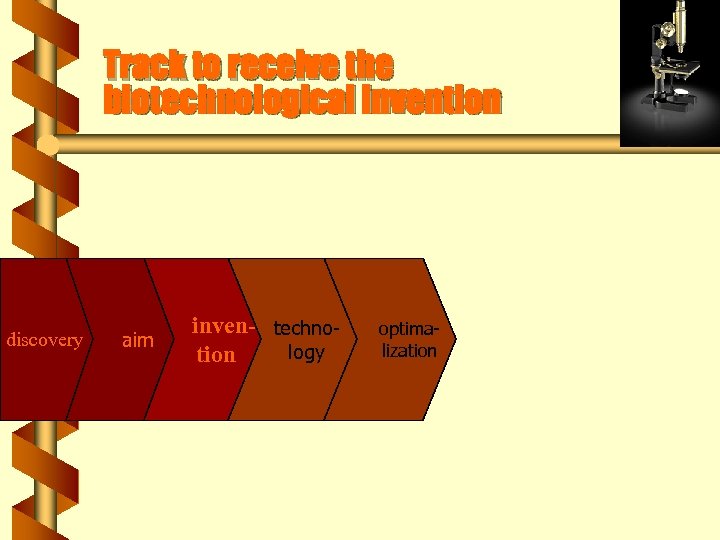
Track to receive the biotechnological invention discovery aim inven- technology tion optimalization
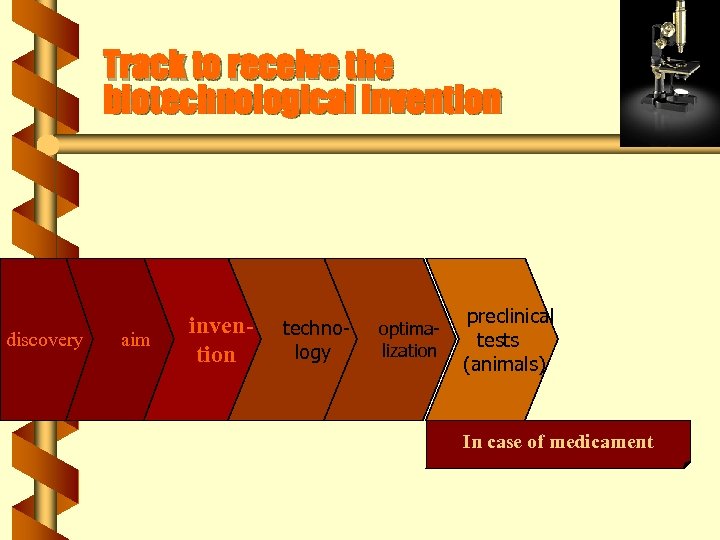
Track to receive the biotechnological invention discovery aim invention technology optimalization preclinical tests (animals) In case of medicament
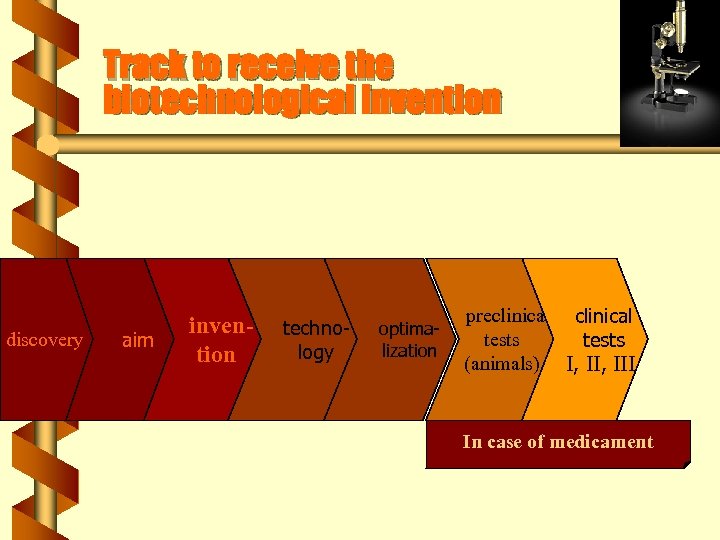
Track to receive the biotechnological invention discovery aim invention technology optimalization preclinical tests (animals) I, III In case of medicament
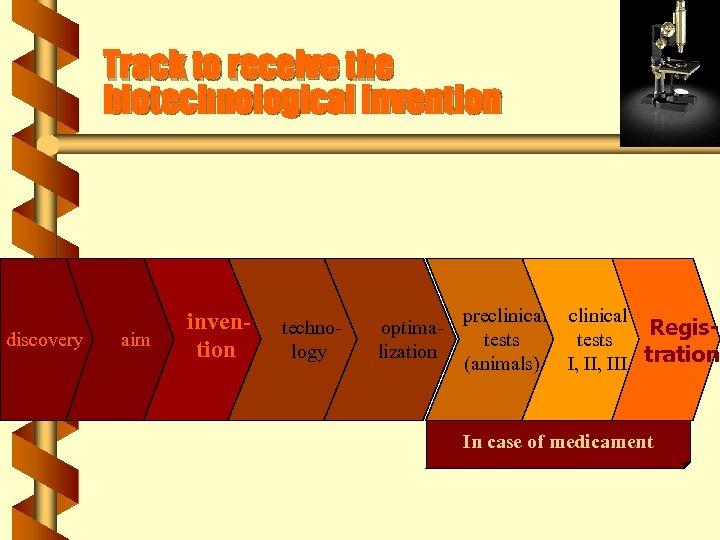
Track to receive the biotechnological invention discovery aim invention technology optimalization preclinical tests (animals) clinical Registests I, III tration In case of medicament
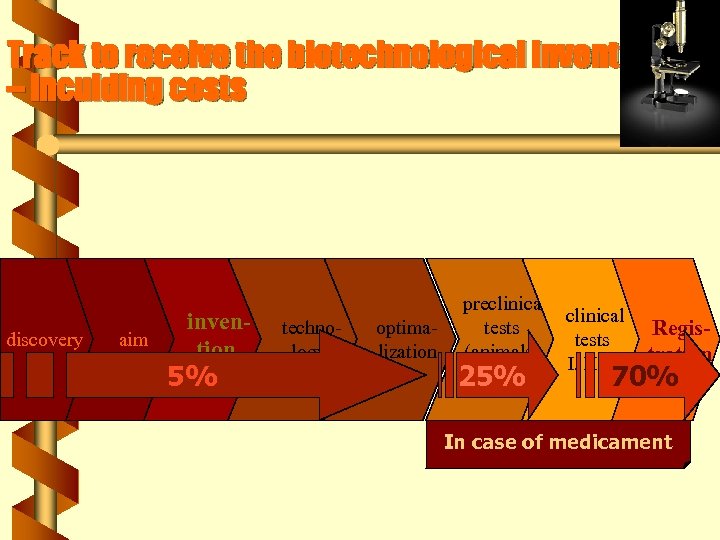
Track to receive the biotechnological invention – inculding costs discovery aim invention 5% technology optimalization preclinical tests Registests (animals) I, III tration 25% 70% In case of medicament

Scientific Risk of Research Activities Discovery and Development of a Successful Medicine YEARS 15 14 13 12 11 10 9 8 7 6 5 4 3 2 1 0 PHASES INTRODUCTION/ REGISTRATION DEVELOPMENT 1 2 2 -5 POST-MARKETING SURVEILLANCE III CLINICAL TEST (HUMANS) BASIC RESEARCH II I 5 - 10 10 - 20 IV PRECLINICAL TEST (ANIMALS) 3, 000 - 10, 000 QUANTITY OF SUBSTANCES Source: Based on Ph. RMA analysis, updated for data per Tufts Center for the Study of Drug Development (CSDD) database SYNTHESIS, EXAMINATION & SCREENING
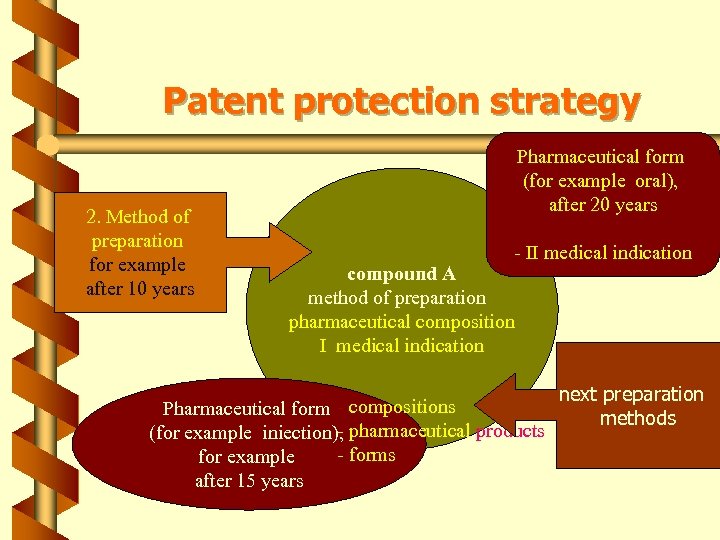
Patent protection strategy 2. Method of preparation for example after 10 years Pharmaceutical form (for example oral), after 20 years - II medical indication compound A method of preparation pharmaceutical composition I medical indication Pharmaceutical form - compositions (for example iniection), pharmaceutical products - forms for example after 15 years next preparation methods
![New pharmaceuticals and generics Pharmaceutical industry NEW [BIO] PHARMACEUTICALS GENERICS Patents SPC Waiting for New pharmaceuticals and generics Pharmaceutical industry NEW [BIO] PHARMACEUTICALS GENERICS Patents SPC Waiting for](https://present5.com/presentation/fc828985184129b212c2157e440761c4/image-29.jpg)
New pharmaceuticals and generics Pharmaceutical industry NEW [BIO] PHARMACEUTICALS GENERICS Patents SPC Waiting for patent expiration EXCLUSIVITY COMPETITION

Suplementary Protection Certificate (SPC) b for pharmaceuticals and plant protection products • these regulations allow the owners of patents involving innovative drugs and plant protection products to extend the patent protection for even up to five years.

Bolar Amendment in CE b The Bolar Amendment permits generic firms to make use of originating firms’ otherwise protected clinical data before the relevant patent have expired. b In contrast, regulatory regimes and enabling legislations in many other developed nations are complex and tend to inhibit the rapid marketing of generic drugs.

Consequence: unfair commercial use b Generic drug registration is used/relied on data of original drug before the relevant patent has expired. PHARMACEUTICALS originals generics Submit undisclosed (safety and efficacy data) Submit bioequivalent studies
![Aspects: b b b b b the balance between [costs – losses – profits]; Aspects: b b b b b the balance between [costs – losses – profits];](https://present5.com/presentation/fc828985184129b212c2157e440761c4/image-33.jpg)
Aspects: b b b b b the balance between [costs – losses – profits]; transfer of knowledge [North to South? ? ? , one-way direction] fostering innovation; preservation of national heritage and biodiversity; regional specificity; local needs for highly qualified staff; functioning of current licensing practice; cost of licensing vs price of patenting; availability of licensing; blocking [false] patents.

INNOVATION -specific parameters vs. value of innovation b Priority, b Affordability, b Quality, b Sustainability, b Uncertainty, b Monopoly rights, b Stimulation of research, b Avoiding duplication.
![IPR actors: b b b b b [1] Private sector Public sector Finance and IPR actors: b b b b b [1] Private sector Public sector Finance and](https://present5.com/presentation/fc828985184129b212c2157e440761c4/image-35.jpg)
IPR actors: b b b b b [1] Private sector Public sector Finance and banking Busines & economy Science & research & development Research at academia & industry Industry at agriculture, pharmacy, food and feed, etc. Management Legislation Non-governmental organisation
![IPR actors [2] b Scientists b Managers b Business people b Greens b Lay IPR actors [2] b Scientists b Managers b Business people b Greens b Lay](https://present5.com/presentation/fc828985184129b212c2157e440761c4/image-36.jpg)
IPR actors [2] b Scientists b Managers b Business people b Greens b Lay people

INVENTION Patents: î new î inventive î commercial Art. 10 Art. 24 p. w. p (Poland) Art. 52. 1 EPC in biotech: a) product of biology, b) containing biologics, c) biological method.

Generics b Essential similarity b Biosimilar (but not identical) the same chemical formula (? ) the same bioavailability (? ) the same bioeffect (? ) b simplified registration procedure b low cost

Different Levels of Added Value Research-based Industry Employment Trade balance Research Development costs Development time Development risk 560, 000 (incl. 88, 000 R&D) in 2002 - 114, 800 within German Market € 28, 000 million in 2001 (up from € 5, 200 million in 1985) - 35 NCE (New Chemical Entites) global launched in 2000 (5 global annual „blockbusters”) - 36 NCE (New Chemical Entites) global launched in 2001 Generics manufacturer 85, 000 (mostly CEE) ? (negative) no discovery or advanced € 895 millions and growing less than € 1 million 10 -13 years 2 -3 years significant: one in 5 -10, 000 zero risk, pick most successful
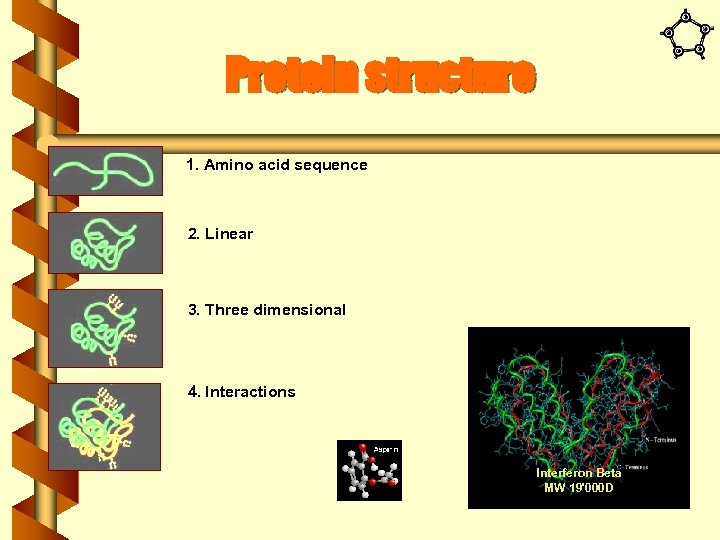
Protein structure 1. Amino acid sequence 2. Linear 3. Three dimensional 4. Interactions Interferon Beta MW 19'000 D
Denaturation and Breakdown
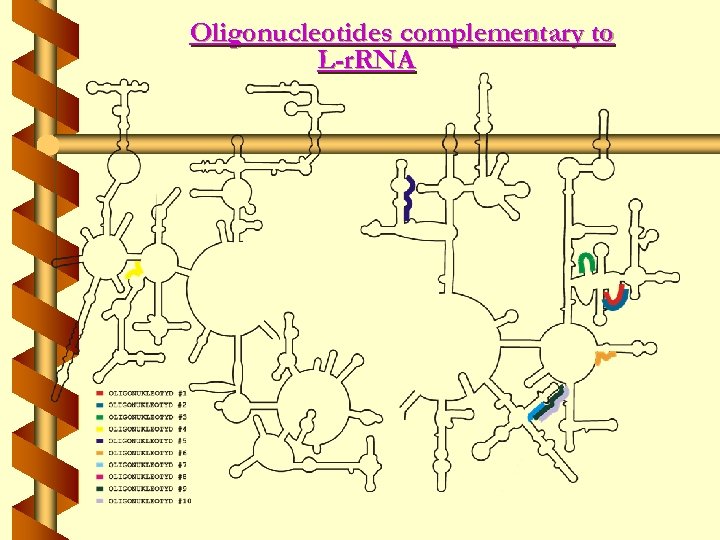
Oligonucleotides complementary to L-r. RNA

To differentiate Biosimilars and Generics • Manufacturing Process; • Molecular Properties; • Safety; • Efficacy

Safety Considerations For Biotech Medicines, each product has a unique safety profile dependent on its: • Mechanism of action • Unique manufacturing process • Composition (by-products and impurities)
![Aspects of novelty [1]: b Research b Commercialization b Simplicity covered by “omics and Aspects of novelty [1]: b Research b Commercialization b Simplicity covered by “omics and](https://present5.com/presentation/fc828985184129b212c2157e440761c4/image-45.jpg)
Aspects of novelty [1]: b Research b Commercialization b Simplicity covered by “omics and logics” b Monopoly vs public goods b Biodiversity vs monoculture of agriculture
![Aspects of novelty [2]; solutions: b Patent pool [collaborative mechanism]; b Clearing-house mechanism; b Aspects of novelty [2]; solutions: b Patent pool [collaborative mechanism]; b Clearing-house mechanism; b](https://present5.com/presentation/fc828985184129b212c2157e440761c4/image-46.jpg)
Aspects of novelty [2]; solutions: b Patent pool [collaborative mechanism]; b Clearing-house mechanism; b Fostering collaboration. b The necessary factors to be included: technical, business, financial, legal, public perception, anti-trust concerns.
![Patent pool [collaborative mechanism] b The term patent pool is commonly used to refer Patent pool [collaborative mechanism] b The term patent pool is commonly used to refer](https://present5.com/presentation/fc828985184129b212c2157e440761c4/image-47.jpg)
Patent pool [collaborative mechanism] b The term patent pool is commonly used to refer to a joint patent licensing program that consists of a number of companies who have decided to offer a joint license for their patents that are essential to the practice of a defined specification. b An Essential Patent is a patent that contains one or more claims that are unavoidably and necessarily infringed by the implementation of a specification for which it is essential. b Competition authorities have indicated that the pro-competitive benefits of patent pools-of allowing a broader group of companies to have access to Essential Patents efficiently through a joint licensing program-may be diluted by including patents that are not Essential Patents. www. oecd. org

Clearing-house mechanism b Expertise in managing information and technology varies enormously from country to country. For this reason, the Convention on Biological Diversity [in 1992] has established a "Clearing-House Mechanism" to ensure that all governments have access to the information and technologies they need for their work on biodiversity. The network of national focal points for the mechanism is being established to address matters relating to technical and scientific cooperation. The Parties have recently emphasized the need to strengthen the role of these focal points. Building a network of non-governmental organizations and other institutions working on biodiversity could contribute to this goal. Establishing National, Regional, Subregional and Thematic Clearing-House Focal Points for specific topics could also help. The clearing-house is based on the philosophy that broad participation and easy access must be a top priority. Its database can therefore be tapped through both traditional and electronic means of communication. Special efforts are made to ensure the participation of indigenous communities, whose unique knowledge and expertise are so important. The Clearing-House is coordinated by the Executive Secretary and overseen and guided by an Informal Advisory Committee (IAC) set up by the Parties to the Convention. www. oecd. org

Foster collaboration b Series of laws use patent ownership to foster collaboration between parties in the research and development enterprise. Patents protect the inventor’s investments in generating the knowledge that is the basis for innovation and serve as an incentive to the commercialization of new ideas. As R&D has become more expensive, ownership of title to inventions has been used as a means to encourage collaborative work among different players in the research enterprise. [Wendy H. Schacht]

Problems example: patent trolls b Patent Act: . . . The right to exclude others. . . from making, using, offering for sale, or selling the invention. . . b Patent trolls: patent holders who do not practice the invention but instead use patents primarily for obtaining license fees.

Quo vadis biotechnologiae? Differentiation: b North vs. b Rich vs. b Developed vs. b Members vs. b Technology reach b Science based b Modern drugs b Home cooking South poor undeveloped non-members vs. biodiversity reach vs. common knowledge vs. traditional medicine vs. nutraceutics
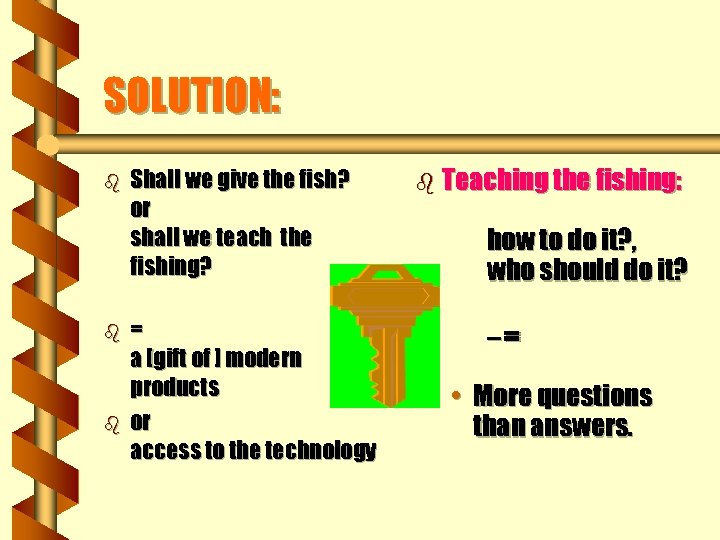
SOLUTION: b b b Shall we give the fish? or shall we teach the fishing? = a [gift of ] modern products or access to the technology b Teaching the fishing: how to do it? , who should do it? –= • More questions than answers.

Intellectual Property Blacklist – „Watch LIST” of the United States Trade Representantive (USTR) b Intellectual property rights legislation is issued at the national level, but World Trade Organization member states must adhere to the requirements of the WTO Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS). b The 2005 ”Special 301 Report” examines in detail the adequacy and effectiveness of IPR protection in 90 countries.

Watch LIST Poland - some progress in improving IPR protection; - deficiencies in patent protection for pharmaceuticals, especially lack of coordination between the Health Ministry and the Polish Patent agency that would prevent the registration of unauthorised patent-infringing products. Hungary - has made improvements to its IPR regime in the past years; - does not provide any coordination between its health regulatory agency and its patent authority to prevent the marketing registrations of patent-infringing products. Slovakia -does not provide a coordination mechanism between its health regulatory agency and its patent authority to prevent the registration of unauthorised patent-infringing products - Slovakia continues to store sensitive registration data on the premises of a generic drug manufacture.

Europe and piracy • Product piracy is responsible for – – – 70. 000 working places less each year Damage to the German economy alone: € 20 – 25 bln each year € 500 bln yearly turnover in fakes (10% of world trade) • Role of Customs Authorities: – – – fight against counterfeiting performing the best possible controls and goods help prevent consumption of unsafe products Source: Wirtschafts Woche, Nr. 27, June 30. 2005

Prices of generics in 2003 b Prices for generics in EU 15 are 15 -50% higher than in the US b In Japan – prices of generics are more than double the U. S. prices b in Poland – prices are lower than in the US Source: U. S. Department of Commerce calculations based on data from IMS Health, IMS MIDAS ™, Q 4/2003 and the Center for Medicare and medicaid Services (CMS)]
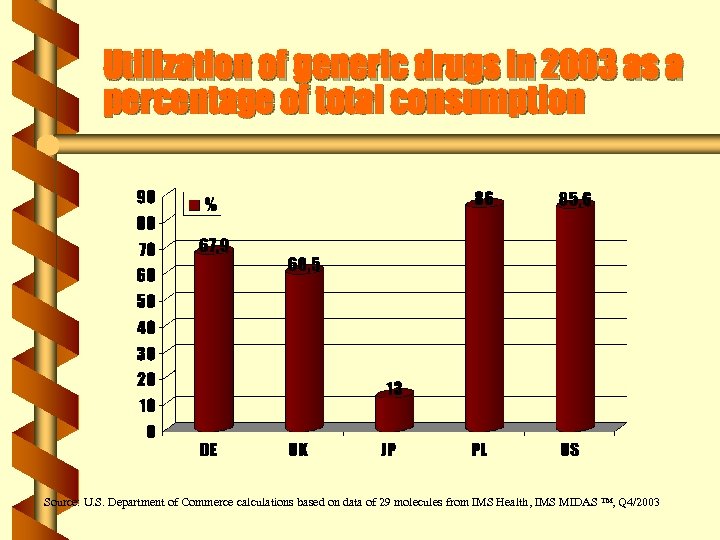
Utilization of generic drugs in 2003 as a percentage of total consumption Source: U. S. Department of Commerce calculations based on data of 29 molecules from IMS Health, IMS MIDAS ™, Q 4/2003

Public perception Patents and patenting = controversial issue. We need: Counter-balancing, in terms of information, various forms of information through international network.

SOLUTION: b Instead of recognising differentiation we should work on harmonisation and unification of IPR system on world arena. b In the today world of “global village” co-operation of all actors on international arena will give all of us the chance for synergy effect.

Why Resistance to IP Protection in Central Europe? • Tradition of weak protection of property in CE – good business with lack of IP • Resistance to change – survival of pre-existing alliances – distortion caused by conflict of interest not eliminated • IP not perceived as opportunity – fear – lack of positive experience with IP • Tradition of low economic freedom – low tradition of rewarding based on merits – low tradition of valuing human capital

Developing new products b The ability of innovative industries to continue to develope new products depends largely upon two factors: • a strong and effective intellectual property system • the capacity to market new products effectively during the period of time when the exclusive IPR exist. Without intellectual property: • no value created, • no investment, • no venture capital, • no trade.

Effective IP Enforcement through Cooperation b Effective IP enforcement requires sophisticated interaction b Cooperation between: – legal community, academia and scientists and creators – patent offices to promote IP and facilitate usage – venture capitalist, investors and scientists, creators of IP – courts, publication and discussion of reasoning – enforcement authorities – prosecutors, police, customs authorities

THANK YOU. . . I am ready to answer questions.
fc828985184129b212c2157e440761c4.ppt