ae8f5b96eb042ec71e741911c6a3a28e.ppt
- Количество слайдов: 52

Best Practice for Platelet and Plasma Transfusion Nicole Draper, MD

Platelets

Platelet Storage and QC • Whole-blood derived or apheresis • 5 days at 20 -24 o. C – Temp needs to be maintained in transport, while held in OR or ICU etc. • • Gently agitated Stored in plasma or additive solution Must test for bacterial contamination Must have >3. 0 x 10 11 platelets per apheresis unit
Case 1 • 30 -year-old woman with h/o tetralogy of Fallot with cadaveric pulmonic valve, ASD closure device. • Admitted with right heart failure found to have pulmonic valve vegetations complicated by severe pulmonic regurgitation. • OR tomorrow for redo pulmonary valve replacement • Cardiac bypass pump

Platelet Transfusion Indications • Prophylaxis – Non-bleeding patients – Platelet count <10 x 109/L • Treatment Hgb 6. 8 (11 -16 g/d. L) Plt 146 (150 -400 x 109/L) ACT 353 (74 -137 sec) PT 23. 8 (11. 4 -14. 4 sec) Fibrinogen 129 (150400 mg/d. L) Actively bleeding – Bleeding/surgical patient – Platelet count <50 x 109/L typically – Neurological often <100 x 109/L – Platelet dysfunction (aspirin, clopidogrel, uremia, plastic, pumps, congenital)
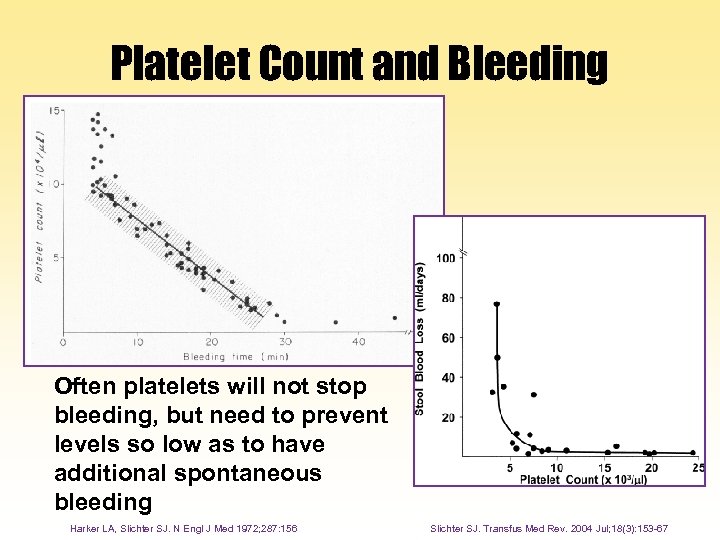
Platelet Count and Bleeding Often platelets will not stop bleeding, but need to prevent levels so low as to have additional spontaneous bleeding Harker LA, Slichter SJ. N Engl J Med 1972; 287: 156 Slichter SJ. Transfus Med Rev. 2004 Jul; 18(3): 153 -67

Platelet Count and Bleeding http: //imaging. ubmmedica. com/cancernetwork/journals/oncology/images/o 0009 sup 8 cf 2. gif

Platlet Count and Procedures Mc. Vay PA, Toy PT. Transfusion 1991; 31(2): 164 -71.

Platelet Count and Procedures • PTs and PTTs 1. 1 -1. 5 times midrange normal levels and platelet counts 50 -99 x 10(9)/L. • Percutaneous liver biopsy 177 inpatient procedures (155 standard, 22 fine needle). • Bleeding complications in patients with platelet counts greater than or equal to 50 x 10(9)/L was 3. 4% (6 of 175), with no significant difference from patients with normal parameters. • Highly associated with bleeding complications: a patient diagnosis of malignancy, 14% (7 of 50) compared with 0. 8% (1 of 127) among other patients (P less than 0. 001). Mc. Vay PA, Toy PT. Am J Clin Pathol 1990; 94(6): 747 -53.

Platelet Dysfunction: Aspirin Figure 3. Before and after transfusion platelet function assay results with change in platelet function. Figure 2. Before and after transfusion platelet function assay results without change in platelet function. Joseph B, Pandit V, Sadoun M, Larkins CG, Kulvatunyou N, Tang A, et al. J Trauma Acute Care Surg. 2013; 75(6): 990 -4. No difference in the progression of ICH (37. 5% vs. 30%, p = 0. 7), neurosurgical intervention (12. 5% vs. 15%, p = 0. 8), and platelet count (240. 9 vs. 252. 1 p = 0. 32)
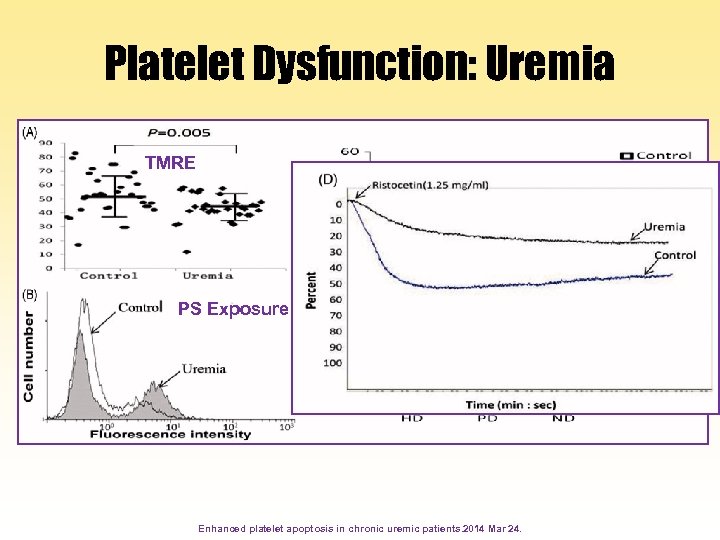
Platelet Dysfunction: Uremia TMRE PS Exposure Enhanced platelet apoptosis in chronic uremic patients. 2014 Mar 24.

Platelet Dysfunction: CPB • Several studies have found that laboratory predictors of platelet dysfunction do not significantly correlate with bleeding after CPB. • There is a clear correlation between the duration of CPB and the BMI with blood loss. Perioperative monitoring of primary and secondary hemostasis in coronary artery bypass grafting. Semin Thromb Hemost. 2005; 31(4): 426 -40.

Platelet Transfusion Contraindications • Nonbleeding patients on antiplatelet medications or with platelet dysfunction extrinsic to the platelet (uremia, von Willebrand disease) • Activation or autoimmune destruction of endogenous platelets (HIT, TTP, ITP) unless there is life-threatening hemorrhage

Blood Samples • Two unique patient identifiers • Zero-tolerance for clerical errors – Most common cause of fatal hemolytic transfusion reactions • New sample every 3 days required if – Pregnant or transfused RBCs in the past 3 months – Usually a universally applied criteria for RBCs • Platelets and plasma often transfused on historical blood type

Platelet Compatibility • Weak ABO antigens on platelets • 20 -40% reduction in count increase if incompatible – Care more about the ABO antibodies in the plasma that can hemolyze red cells – Soluble A or B antigenic substance in pt plasma • Type-A donors recruited to apheresis platelets • Type-O donors recruited to RBC donation

Question 1 An Rh+ platelet is transfused to an Rh- patient. Which of the following does the patient need? A. Rh. Ig regardless of age and sex B. Rh. Ig if female with childbearing potential C. No administration of Rh. Ig
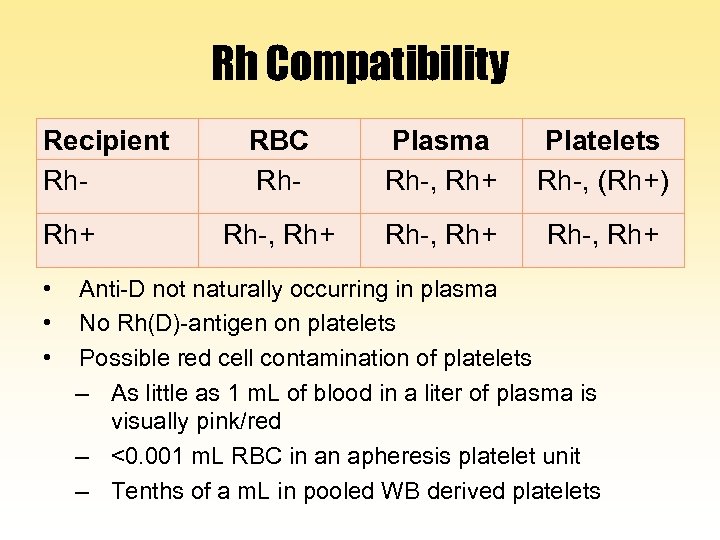
Rh Compatibility Recipient Rh. Rh+ • • • RBC Rh- Plasma Rh-, Rh+ Platelets Rh-, (Rh+) Rh-, Rh+ Anti-D not naturally occurring in plasma No Rh(D)-antigen on platelets Possible red cell contamination of platelets – As little as 1 m. L of blood in a liter of plasma is visually pink/red – <0. 001 m. L RBC in an apheresis platelet unit – Tenths of a m. L in pooled WB derived platelets

Rh. Ig and Platelets • Anti-D alloimmunization after D-incompatible platelet transfusions: a 14 -year single-institution retrospective review at Beth Israel Deaconess Medical Center. • Of 130 eligible D− patients, 48% women and 57% immunocompetent, who received a total of 565 apheresis PLTs, none formed anti-D. 28% Transfusion. 2014 Mar; 54(3): 650 -4.

Platelet Dosage and Effect • Whole-blood-derived platelets and apheresis platelets have equivalent efficacy • Dose – 1 apheresis platelet – 6 -pack of whole blood platelets – 5 -10 m. L/kg in pediatric patients • Increase by 30 -60 x 109/L in 70 kg adult • Typical life-span of 3 -4 days post transfusion
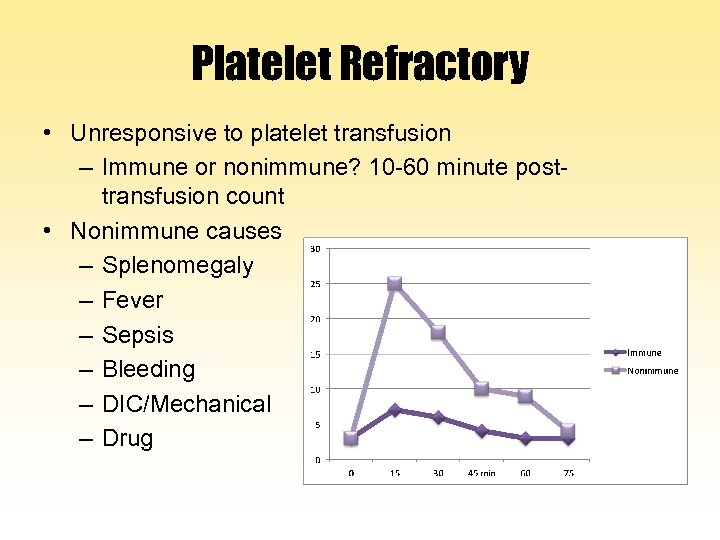
Platelet Refractory • Unresponsive to platelet transfusion – Immune or nonimmune? 10 -60 minute posttransfusion count • Nonimmune causes – Splenomegaly – Fever – Sepsis – Bleeding – DIC/Mechanical – Drug

Platelet Refractory • Platelet alloantibodies: Anti-HLA class I or platelet-specific antibodies – Previous transfusion or transplantation – Pregnancy – Recipient dependent, not dose • Treatment: HLA-matched or crossmatched platelets • Prevention: Leukocyte reduction

Case 2 50 -year-old woman with suspected aplastic anemia Pre Plt Count 2/21 2/22 1830 0030 1030 5 3 4 Post Plt Count 2030 0130 1400 3 4 4

Platelet Refractory: PRA

HLA-Matched Platelets Apheresis Platelet Unit
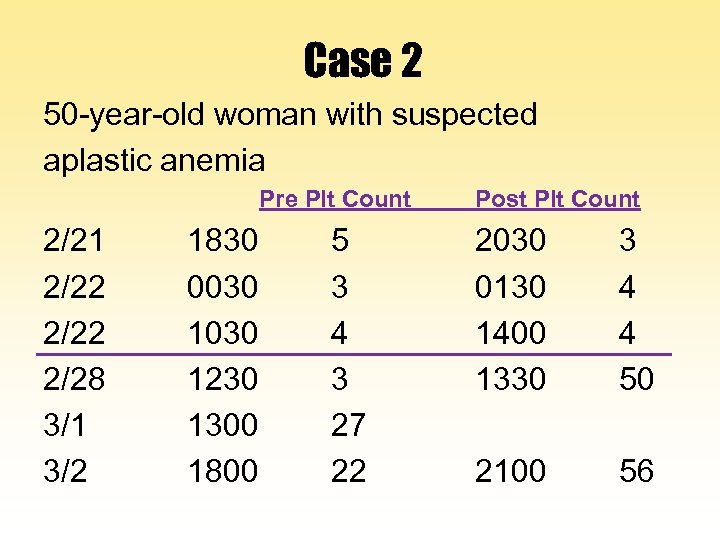
Case 2 50 -year-old woman with suspected aplastic anemia Pre Plt Count 2/21 2/22 2/28 3/1 3/2 1830 0030 1230 1300 1800 5 3 4 3 27 22 Post Plt Count 2030 0130 1400 1330 3 4 4 50 2100 56

Plasma

Plasma Volume: 200 – 600 m. L Content: Plasma Anticoagulant 200 m. L 250 m. L 300 m. L 500 m. L 600 m. L PLASMA = INR = 1. 3

Plasma Types • Fresh Frozen Plasma (FFP): frozen within 8 hours of collection • Plasma Frozen within 24 Hours (PF 24): frozen within 24 hours of collection • Thawed Plasma (TP): derived from FFP or FP 24 and maintained for a maximum of 5 days after the day of thaw • Plasma Cryoprecipitate Reduced: low levels of fibrinogen, FVIII, v. WF, FXIII, fibronectin

Handling Options for FFP Stored frozen < -18°C Thawed at 30 -37ºC FFP, Thawed Store at 1 -6ºC >24 h Transfuse Thawed Plasma (up to 5 days after thawing)

Coagulation Factor Activity of Thawed Plasma Day 1 Day 2 Day 3 Day 4 Day 5 % change Day 1 to 5 p Fibr 225 224 224 225 0 NS II 81 81 81 80 80 1 NS V 79 75 71 68 66 16 NS VII 90 81 76 72 72 20 NS VIII 107 76 66 65 65 41 <. 02 X 85 84 84 82 80 6 NS Tabular entries as % activity NS = not statistically significant Downes K et al. Transfusion 2001; 41: 570

Question 2 All of the following are preferred uses of fresh frozen plasma except? A. Massive transfusion B. Reversal of warfarin anticoagulation C. Treatment of hemophilia A D. Treatment of TTP

Plasma Transfusion Indications • Bleeding or preoperative patients – Deficiency of multiple coagulation factors • • liver disease warfarin therapy massive transfusion disseminated intravascular coagulation – Specific factor deficiency, no concentrate • Thrombotic thrombocytopenic purpura • Rare specific plasma protein deficiency

Contraindications • When a coagulopathy can be corrected more effectively with a specific therapy – Vitamin K – Cryoprecipitated AHF – Prothrombin complex concentrates • When blood volume can be safely and adequately replaced with other volume expanders

Plasma Dosage and Effect • The volume transfused depends on the clinical situation and patient size • May be guided by laboratory assays of coagulation function • No QC for plasma products

Plasma Dosage and Effect Determinants PLASMA Rx: 2 units? ? Patient size Bleeding site Factor activity: Initial, target Factor concentration in plasma Factor half-life in vivo Unit volume USUAL DOSE FOR CONTROL OF BLEEDING: 10 -20 m. L/kg 70 kg x 15 m. L/kg x 1 unit/250 ml = 4. 2 4 units Hgb 6. 8 (11 -16) Plt 146 (150 -400 L) ACT 353 (74 -137) PT 23. 8 (11. 4 -14. 4) Fibrinogen 129 (150 -400) Actively bleeding

Abnormalities in Coagulation Testing do not Necessarily Indicate a Clinical Coagulopathy Fibrinogen Factor VIII Normal 200 -400 mg/d. L 1 U/m. L Hemostatic 50 -100 mg/d. L 5 -25% Normal concentration: 1 U/m. L = 100% activity Edmunds LH. Hemostasis and thrombosis: basic principles and clinical practice. 4 th ed. 2001 p 1031 -43

Using Screening Tests to Predict Plasma Need Mild elevations of PT, INR, a. PTT overestimate clinical benefit of transfusing plasma for patients in most clinical situations. Generally recommended transfusion trigger points in appropriate situations: 1. 3 x upper limit of reference range (in seconds) - or – 1. 5 x midpoint of reference range (in seconds) -Mc. Vay PA et al. AJCP 1990; 94: 737 -53. -Mc. Vay PA et al. Transfusion 1991; 31: 164 -71. -Counts RB et al. Ann Surg 1979; 190: 91 -9. -Ciavarella D et al. Br J Haematol 1987; 67: 365 -8. -Auble T et al. Acad Emerg Med 2002; 567 -574 -Stanworth SJ, Hematology Am Soc Hematol Educ Program 2007: 179 -86

Prophylactic Plasma Transfusion Almost no effect with an INR <1. 85 Patients receiving FFP and having pretransfusion and posttransfusion PT/INR. Patients with acute trauma, in the operating room, with excessive factor consumption (ie, DIC), or given PCC were excluded. Holland LL, Brooks JP Am J Clin Pathol. 2006 Jul; 126(1): 133 -9. Abdel-Wahab OI, Healy B, Dzik WH Transfusion. 2006 Aug; 46(8): 1279 -85
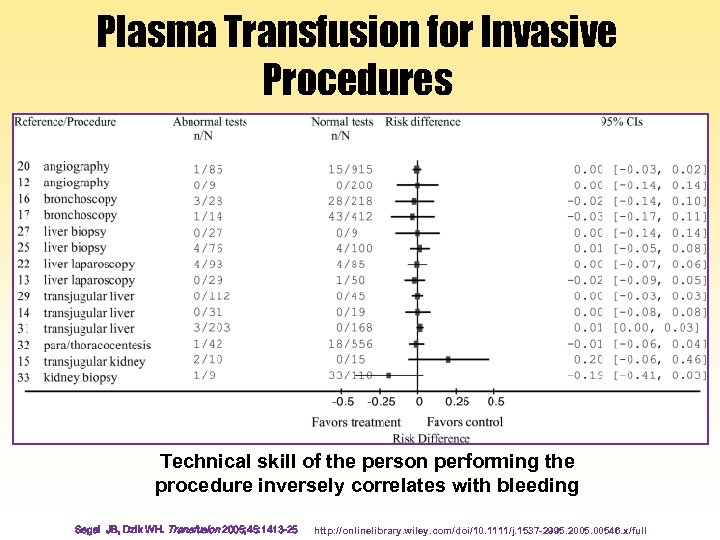
Plasma Transfusion for Invasive Procedures Technical skill of the person performing the procedure inversely correlates with bleeding Segal JB, Dzik WH. Transfusion 2005; 45: 1413 -25 http: //onlinelibrary. wiley. com/doi/10. 1111/j. 1537 -2995. 2005. 00546. x/full

Thrombelastography (TEG) • In 76 patients, routine coagulation tests (i. e. prothrombin time, fibrinogen level, d -dimer, and platelet count), thrombelastography, and whole blood aggregometry were obtained perioperatively and on days 1 and 3 after OPCAB. • Intra- and postoperative blood loss was determined Poston R et al. Eur J Cardiothorac Surg 2005; 27: 584 -591
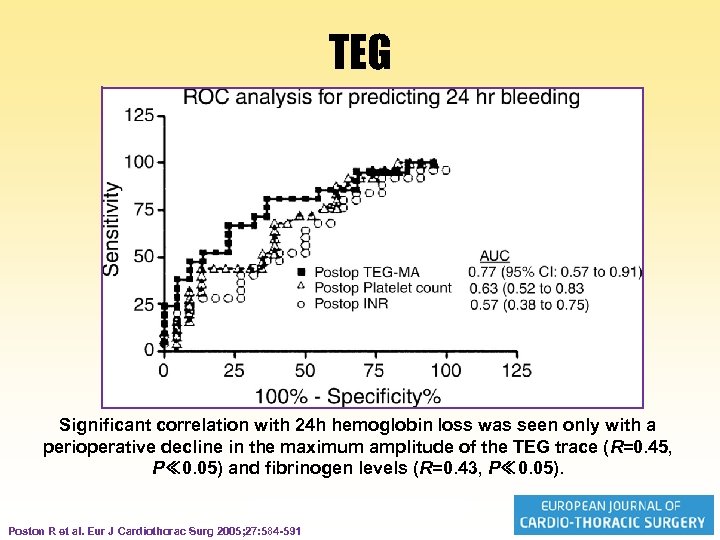
TEG Significant correlation with 24 h hemoglobin loss was seen only with a perioperative decline in the maximum amplitude of the TEG trace (R=0. 45, P≪ 0. 05) and fibrinogen levels (R=0. 43, P≪ 0. 05). Poston R et al. Eur J Cardiothorac Surg 2005; 27: 584 -591
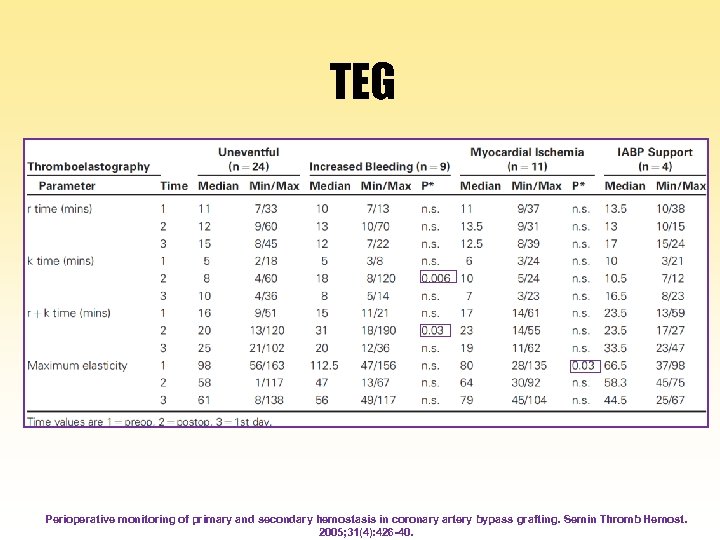
TEG Perioperative monitoring of primary and secondary hemostasis in coronary artery bypass grafting. Semin Thromb Hemost. 2005; 31(4): 426 -40.

Seconds Effect of Body Temperature on Coagulant Activity o. C Rohrer MJ, Natale AM. Crit Care Med 1992; 20: 1402 -5

Effect of Acid/Base Balance on Coagulant Activity Meng ZH et al. J Trauma 2003; 55: 886 -91 © 2003 Lippincott Williams & Wilkins, Inc. Published by Lippincott Williams & Wilkins, Inc.

Question 3 56 -year-old woman with ESLD secondary to hepatitis C is reported to have sudden onset respiratory distress at approximately 10: 30 am. Intubated at 11 am. She was scheduled for a procedure in IR and received 6 units FFP from 5 am to 10 am. Time Hb INR T Bili Haptoglobin 0400 8. 6 2. 5 3. 7 1130 6. 9 1. 9 4. 1 17. 0 (41– 165) A. Hemolysis B. Fluid overload C. TRALI D. Bacterial contamination

At the Bedside • Clerical check • Visual check • 170 -260µ filter removes fibrin clots, aggregates • 22 -14 gauge needle/catheter – 24 for pediatric if necessary • • 0. 9% (normal) saline Appropriate blood warmers Transfusion must be completed within 4 hrs Stop transfusion if suspect reaction

Blood is a Drug • The blood bank is the only part of the laboratory that is regulated by the FDA – Blood products are biologic drugs – Lab + pharmacy • Include transfusion history as part of a drug history

Possible Side Effects • More likely with massive transfusion – Hypothermia – Hyperkalemia – Metabolic acidosis (citric acid) – Hypocalcemia, hypomagnesemia

Infectious Disease Transmission • Infectious Disease Testing – HIV: anti-HIV-1/2, HIV RNA (1: 1. 5 million) – HCV: anti-HCV, HCV RNA (1: 1. 2 million) – HBV: HBs. Ag, anti-HBc, (1: 280, 000) – HTLV: anti-HTLV-I/II – WNV: WNV RNA – Syphilis: anti-Treponema pallidum – Chagas: based on history – CMV: optional

Types of Transfusion Reactions • Fever – Febrile – Hemolytic (delayed vs. acute) – Bacterial sepsis • Respiratory distress – Transfusion related acute lung injury (TRALI) – Transfusion associated circulatory overload (TACO) – Allergic (anaphylaxis) • Rash – Allergic – TA-GVHD ● Thrombocytopenia – Posttransfusion purpura – Platelet refractory

References • Transfusion therapy: clinical principles and practice / editor, Paul D Mintz. 3 rd ed. AABB 2011. • Technical manual / editor John D. Roback. 17 th ed. AABB 2011. • Circular of information for the use of human blood and blood components. http: //www. fda. gov/biologicsbloodvaccines/gui dancecomplianceregulatoryinformation/guida nces/blood/ucm 364565. htm

Questions
ae8f5b96eb042ec71e741911c6a3a28e.ppt