206904a70912ebbd8899db19564fe4b5.ppt
- Количество слайдов: 40

Benefits and Risks of ART Dr Paula Munderi WHO Training Course for Introducing Pharmacovigilance of HIV Medicines 23 - 28 November 2009, Dar Es Salaam

Summary Progress in ART coverage - WHO Progress Special Populations : Women & Children Epidemiology of Paediatric HIV Report Pregnancy and ART Benefits of ART Survival Decreased Morbidity Decreased Transmission Vertical & Horizontal Principles of ART Measurement of efficacy Risks of ART linked to AEs and Toxicities Adherence : determinant of ART efficacy Co-morbidities of specific concern : TB, Hep. B, Hep C, (Malaria) Programme implications Selection of ARVs in the PH approach ART principles & recommendations Current ART practice in LMIC What this means for countries

Acknowledgements Sources of Slide Material Published data WHO progress report on access 2009 Jens Lundgren Lynne Moffenson WHO HQ - HIV Dept (ATC team) DART study group

I Benefits of ART • Improved Survival • Reduced morbidity • Reduced vertical transmission of HIV • Possibly – reduced horizontal transmission of HIV
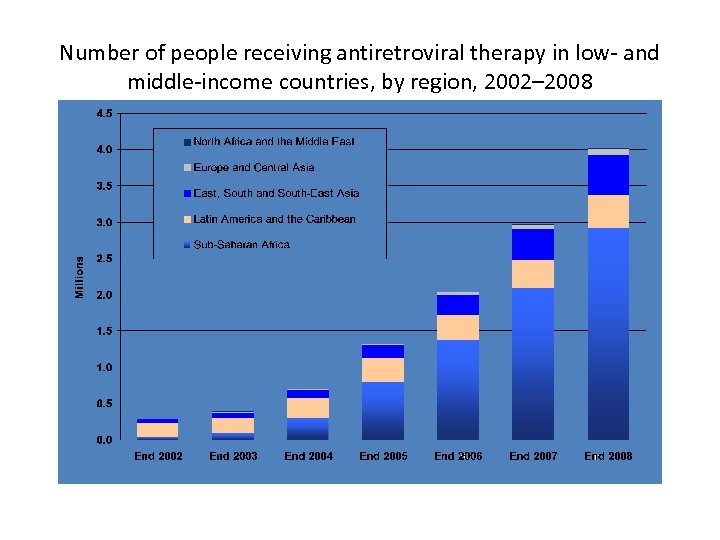
Number of people receiving antiretroviral therapy in low- and middle-income countries, by region, 2002– 2008
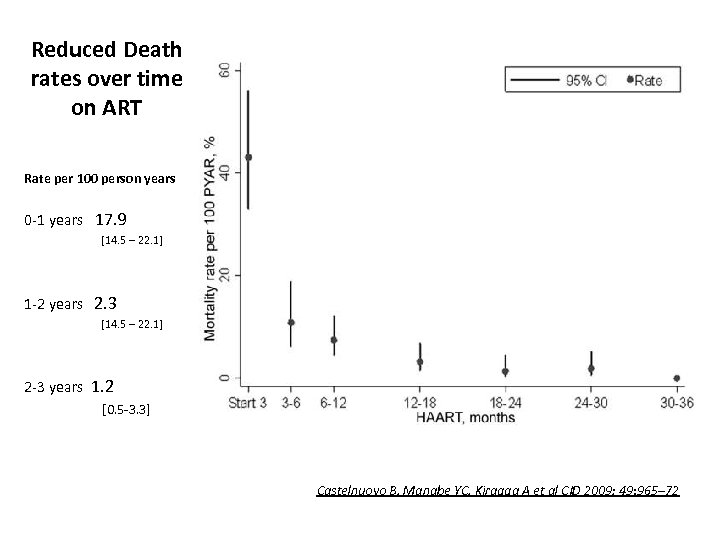
Reduced Death rates over time on ART Rate per 100 person years 0 -1 years 17. 9 [14. 5 – 22. 1] 1 -2 years 2. 3 [14. 5 – 22. 1] 2 -3 years 1. 2 [0. 5 -3. 3] Castelnuovo B, Manabe YC, Kiragga A et al CID 2009; 49: 965– 72
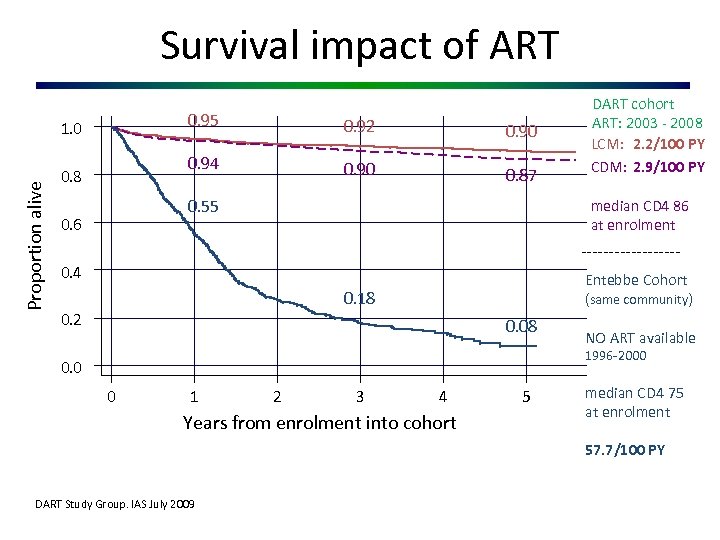
Survival impact of ART 0. 95 Proportion alive 0. 8 0. 92 0. 90 0. 94 1. 0 0. 90 0. 87 0. 55 0. 6 DART cohort ART: 2003 - 2008 LCM: 2. 2/100 PY CDM: 2. 9/100 PY median CD 4 86 at enrolment --------- Entebbe Cohort (same community) 0. 4 0. 18 0. 2 0. 08 NO ART available 1996 -2000 0. 0 0 1 2 3 4 Years from enrolment into cohort 5 median CD 4 75 at enrolment 57. 7/100 PY DART Study Group. IAS July 2009
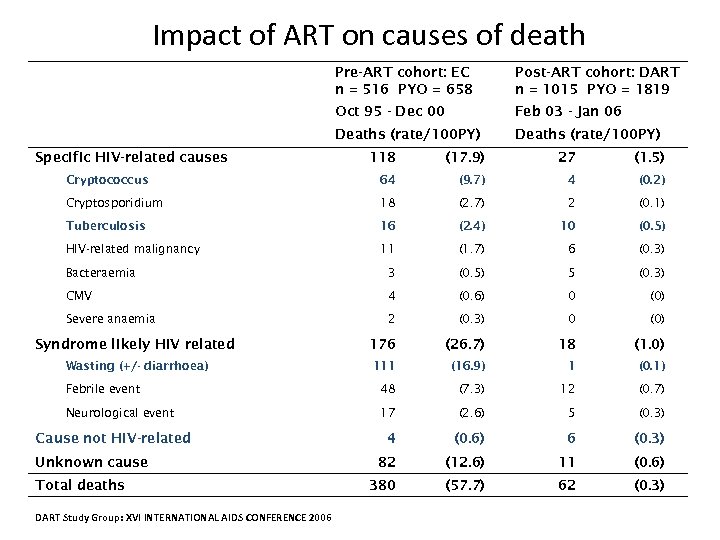
Impact of ART on causes of death Pre-ART cohort: EC n = 516 PYO = 658 Oct 95 - Dec 00 Feb 03 - Jan 06 Deaths (rate/100 PY) Specific HIV-related causes Post-ART cohort: DART n = 1015 PYO = 1819 Deaths (rate/100 PY) 118 (17. 9) 27 (1. 5) Cryptococcus 64 (9. 7) 4 (0. 2) Cryptosporidium 18 (2. 7) 2 (0. 1) Tuberculosis 16 (2. 4) 10 (0. 5) HIV-related malignancy 11 (1. 7) 6 (0. 3) Bacteraemia 3 (0. 5) 5 (0. 3) CMV 4 (0. 6) 0 (0) Severe anaemia 2 (0. 3) 0 (0) Syndrome likely HIV related 176 (26. 7) 18 (1. 0) Wasting (+/- diarrhoea) 111 (16. 9) 1 (0. 1) Febrile event 48 (7. 3) 12 (0. 7) Neurological event 17 (2. 6) 5 (0. 3) 4 (0. 6) 6 (0. 3) 82 (12. 6) 11 (0. 6) 380 (57. 7) 62 (0. 3) Cause not HIV-related Unknown cause Total deaths DART Study Group: XVI INTERNATIONAL AIDS CONFERENCE 2006
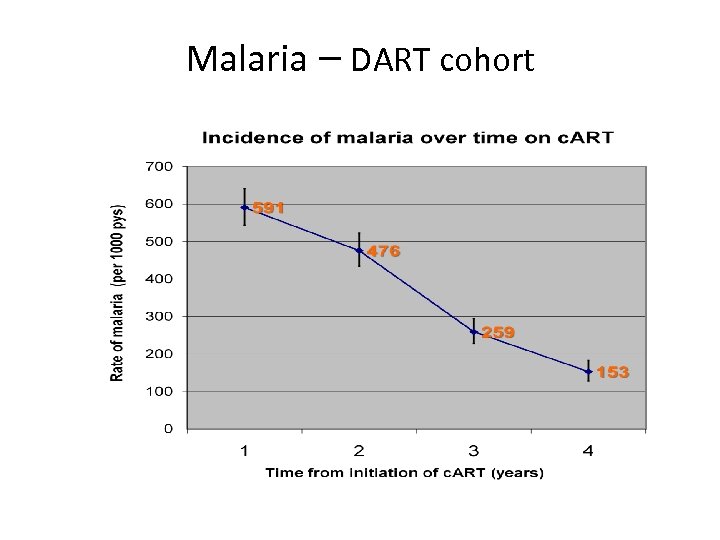
Malaria – DART cohort
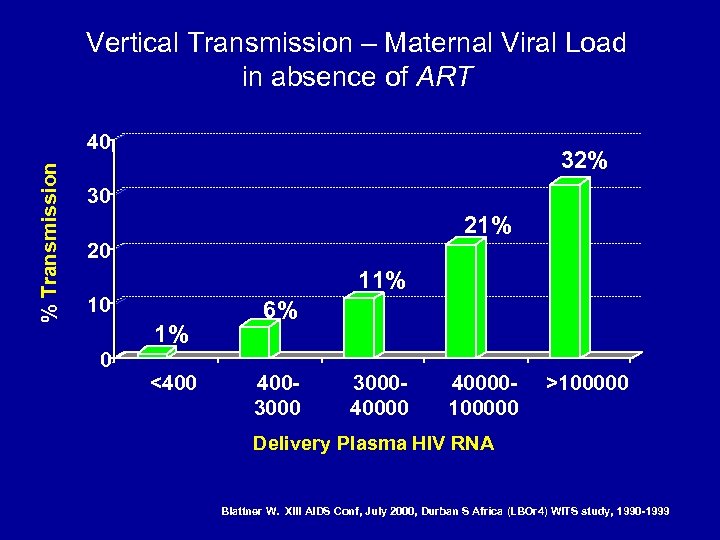
Vertical Transmission – Maternal Viral Load in absence of ART % Transmission 40 32% 30 21% 20 11% 10 1% 0 <400 6% 400300040000100000 >100000 Delivery Plasma HIV RNA Blattner W. XIII AIDS Conf, July 2000, Durban S Africa (LBOr 4) WITS study, 1990 -1999
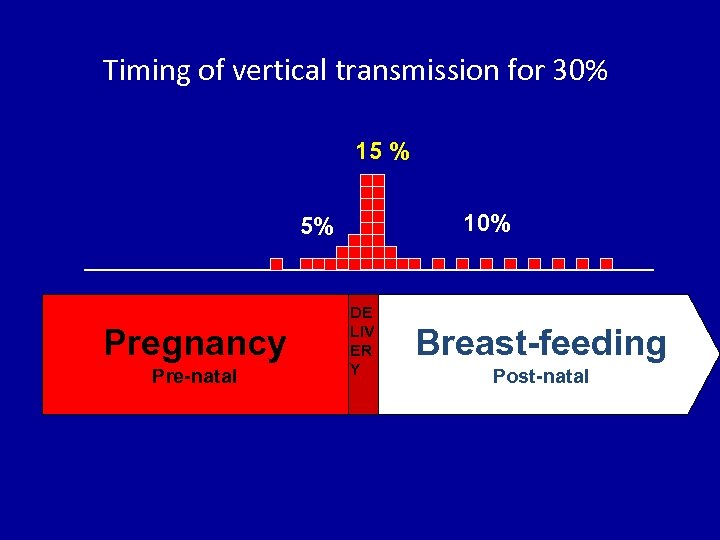
Timing of vertical transmission for 30% 15 % 10% 5% Pregnancy Pre-natal DE LIV ER Y Breast-feeding Post-natal
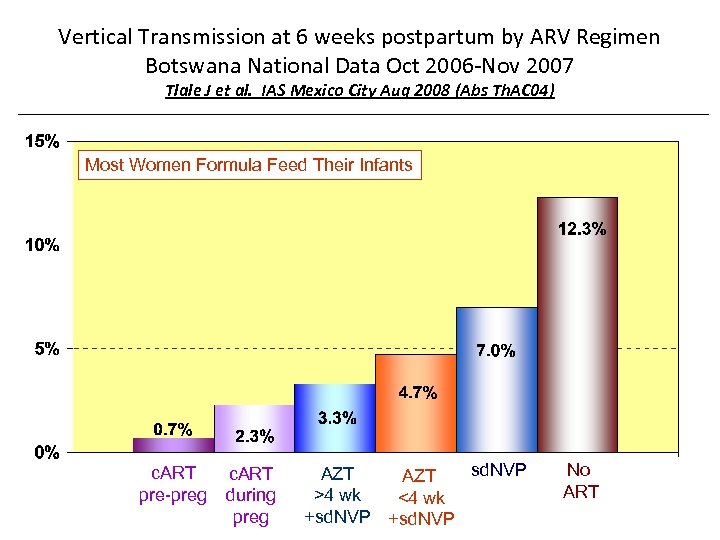
Vertical Transmission at 6 weeks postpartum by ARV Regimen Botswana National Data Oct 2006 -Nov 2007 Tlale J et al. IAS Mexico City Aug 2008 (Abs Th. AC 04) Most Women Formula Feed Their Infants c. ART pre-preg c. ART during preg sd. NVP AZT >4 wk <4 wk +sd. NVP No ART

Vertical transmission of HIV 15 -35% <1% Antenatal screen Maternal ART Neonatal ART C. section No breastfeeding
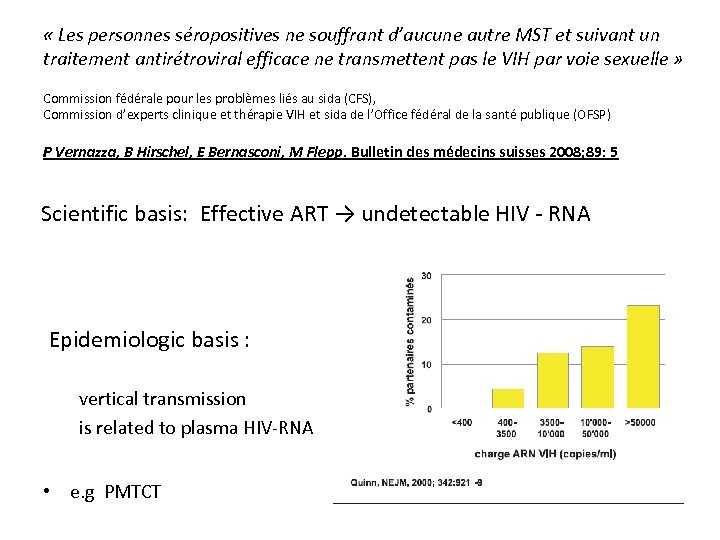
« Les personnes séropositives ne souffrant d’aucune autre MST et suivant un traitement antirétroviral efficace ne transmettent pas le VIH par voie sexuelle » Commission fédérale pour les problèmes liés au sida (CFS), Commission d’experts clinique et thérapie VIH et sida de l’Office fédéral de la santé publique (OFSP) P Vernazza, B Hirschel, E Bernasconi, M Flepp. Bulletin des médecins suisses 2008; 89: 5 Scientific basis: Effective ART → undetectable HIV - RNA Epidemiologic basis : vertical transmission is related to plasma HIV-RNA • e. g PMTCT

Longitudinal studies in HIV sero-discordant couples Rwanda & Zambia 393 couples followed for 14 years no transmission from person on c. ART 8. 6 % transmission with no c. ART 1 -------------------------93 couples; in 41 +ve partner on c. ART 6 infections – all in non treated 2 -------------------------62 couples; Males +ve pregnancy desired no infection in female partner 3 • 2, 993 couples • followed from 2002 – 2008 • HIV testing every 3 months • 5’ 609 person years • 4 HIV infections from partners on c. ART • 171 infections partners not on c. ART Incidence density of HIV transmission 4 0. 7% on c. ART vs 3. 4% off c. ART [RR = 0. 21, CI: 0. 08, 0. 59] NB: HIV persists in semen and cervicovaginal fluid during effective c. ART

ARVs for Prevention • PEP & PMTCT • Universal Test & Treat – combination ART • Pre- Exposure prophylaxis (Pr. EP) – Tenofovir • ARVs as “ vaginal microbicides” – Tenofovir – Maraviroc

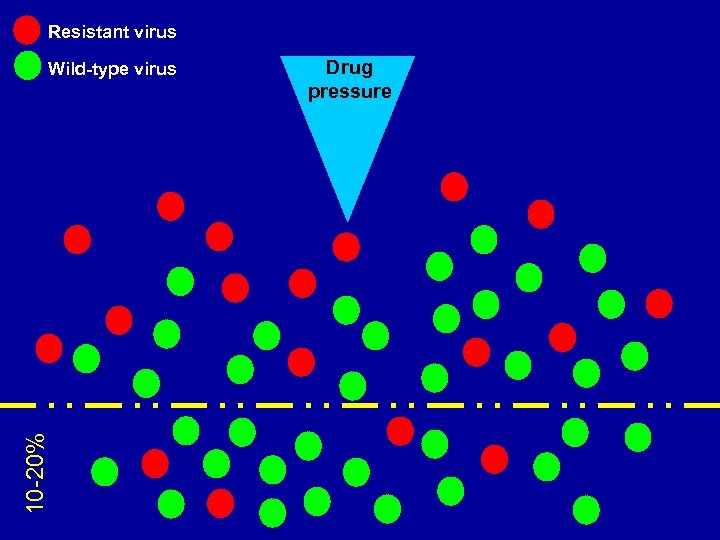
II Principles of ART • Combination therapy • Avoidance of resistance • Importance of adherence

For sustained efficacy of ART. . . • synergistic combinations of 3 active drugs usually from 2 different classes – single or dual drug therapy only in PMTCT; low risk PEP • the large number of possible combinations is only apparent – – cross resistance develops within classes Cross resistance may occur between NRTIs and NNRTIs • preserving future treatment options is critical – choice of initial regimen – rational sequencing of combinations thereafter

Measurement of efficacy of ART When is treatment working? • Absence of clinical disease – WHO clinical staging • Immune restoration – Rise in CD 4 count • Viral suppression – Undetectable VL When has treatment failed • Recurrence of clinical illness – WHO stage II/IV • Fall in CD 4 count – to below 100 cells/mm 3 • Detectable VL – to >5000 copies/ml
Resistant virus 10 -20% Wild-type virus
Resistant virus 10 -20% Wild-type virus Drug pressure
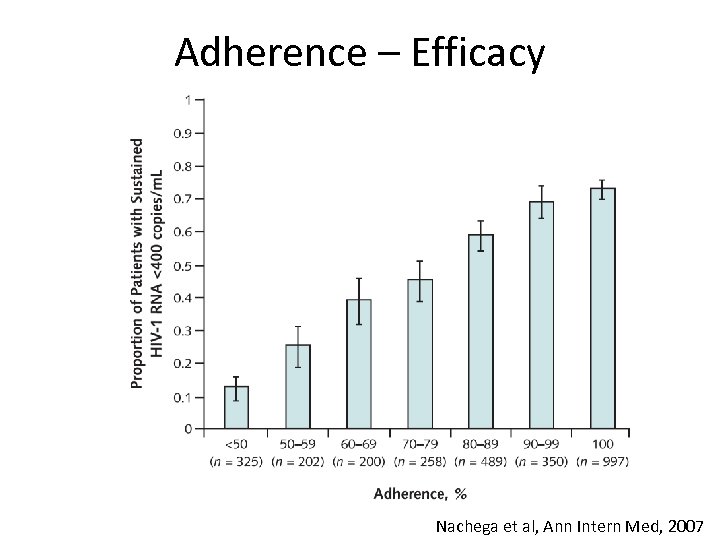
Adherence – Efficacy Nachega et al, Ann Intern Med, 2007

III Risks of ART Mainly related to toxicities

NB: Most ART Toxicities are. . . - Predictable - Clinically detectable - Can be managed The key is patient and provider education !
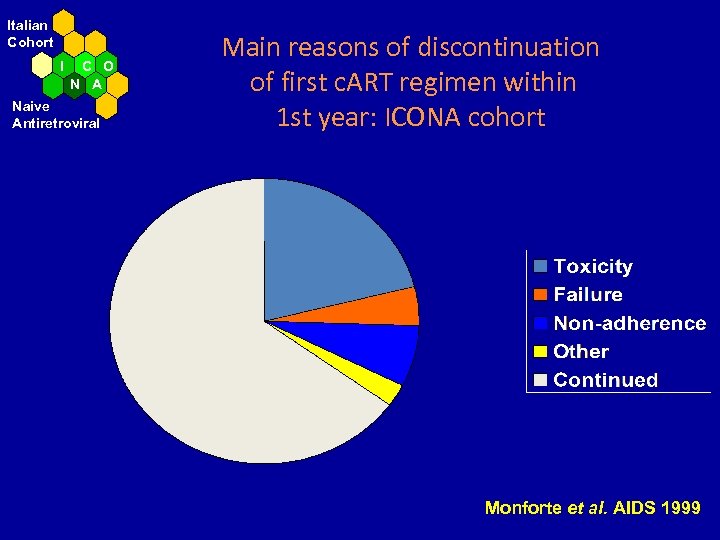
Italian Cohort I C O N A Naive Antiretroviral Main reasons of discontinuation of first c. ART regimen within 1 st year: ICONA cohort Monforte et al. AIDS 1999
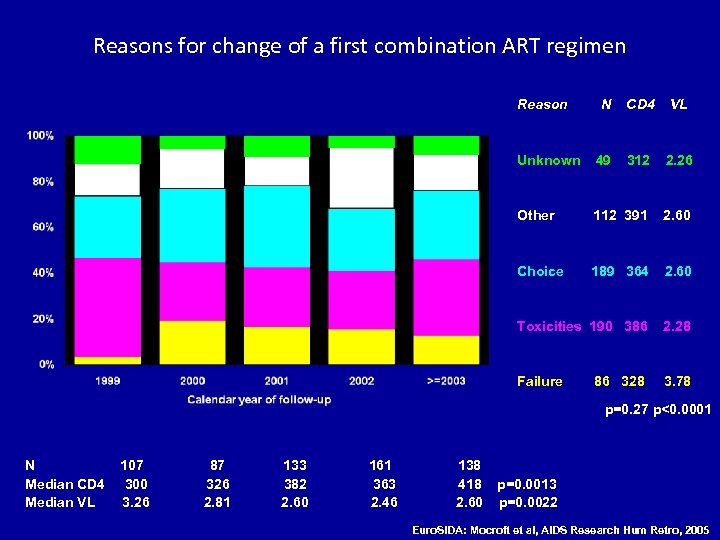
Reasons for change of a first combination ART regimen Reason N CD 4 VL Unknown 49 312 2. 26 Other 112 391 2. 60 Choice 189 364 2. 60 Toxicities 190 386 2. 28 Failure 3. 78 86 328 p=0. 27 p<0. 0001 N Median CD 4 Median VL 107 300 3. 26 87 326 2. 81 133 382 2. 60 161 363 2. 46 138 418 2. 60 p=0. 0013 p=0. 0022 Euro. SIDA: Mocroft et al, AIDS Research Hum Retro, 2005
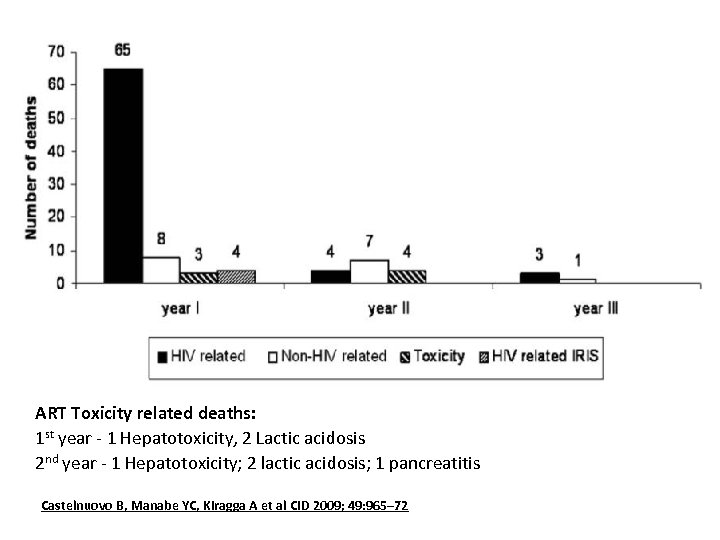
ART Toxicity related deaths: 1 st year - 1 Hepatotoxicity, 2 Lactic acidosis 2 nd year - 1 Hepatotoxicity; 2 lactic acidosis; 1 pancreatitis Castelnuovo B, Manabe YC, Kiragga A et al CID 2009; 49: 965– 72

NB: Most ART Toxicities are. . . - Predictable - Clinically detectable - Can be managed The key is patient and provider education !

IV Women & Children • Pregnancy • In utero & Perinatal exposure to ARVs • c. ART in children

Incidence of Seroconversion in Pregnancy: Prevention of HIV in Pregnant Women is Critical Country Reference Incidence per 100 Pt-Yrs Pregnancy Uganda Gray R et al. Lancet 2005; 366: 1182 2. 3 Botswana Lu L et al. 2009 CROI Abs. 94 LB 1. 3 (0. 5 -3. 1) Zimbabwe, Uganda Morrison CS et al. AIDS 2007; 21: 1027 1. 6 South Africa Rehle T et al. S Afr Med J 2007; 97: 194 5. 2 (0 -12. 9) South Africa Moodley D et al. AIDS 2009; 23: 1255 10. 7 (8. 2 -13. 1)

43% of New Infant Infections in Botswana May be Due to Maternal Seroconversion in Pregnancy/PP Lu L et al. 16 th CROI, Montreal, Canada Feb 2009 Abs 94 LB HIV diagnosed before or during ANC # HIV+ women Estimated MTCT rate # infected infants New maternal infection late pregnancy New maternal infection 1 yr postpartum 13, 952 378 450 (incidence 1. 3%) (incidence 1. 8%) 73% 36% 276 186 4. 7% (with PMTCT ARV) 620 Of the estimated 1, 082 infant HIV infections in Botswana in 2007, 462 (43%) were due to incident cases of maternal HIV in pregnancy/PP

Prevention of HIV in Women, (Especially Young Women) Prevention of Unintended Pregnancies in HIV-Infected Women Prevention of Transmissio n from an HIV-Infected Woman to Her Infant Support for HIV-Infected Mother and Family Unintended Pregnancy Among HIV-Infected Women • 51% unintended pregnancies among women with HIV in Cote d’Ivoire. • 74% unintended pregnancies among women in HIV care in Rwanda. • 84% unintended pregnancies among PMTCT clients in South Africa. • 93% unintended pregnancies among women in HIV-ART Desgrees-du-Lou A et al. Int J STD AIDS 2002 care in Uganda. Bangendanye, 3 rd Ped CLS 2007 Rochet T et al. JAMA 2006 Homsy J et al. PLos. One 2009
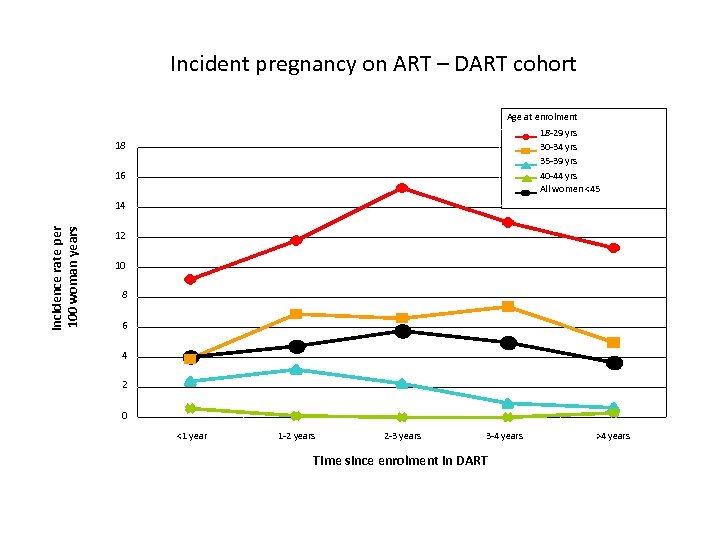
Incident pregnancy on ART – DART cohort Age at enrolment 18 -29 yrs 30 -34 yrs 35 -39 yrs 40 -44 yrs All women <45 18 16 Incidence rate per 100 woman years 14 12 10 8 6 4 2 0 <1 year 1 -2 years 2 -3 years 3 -4 years Time since enrolment in DART >4 years

Physiologic Changes During Pregnancy Can Affect Therapeutic Drug Administration • Increased plasma volume – dilution effect • Decreased in serum albumin – increase in free fraction of drug • Increased GFR 20 -60% starting 1 st trimester – change in drug clearance • Changes in hepatic enzyme activity – increase CYP 34 A, 2 D 6 = change in drug metabolism • Decreased gastric acid secretion, prolonged gastric emptying and intestinal transit time – decreased oral drug absorption
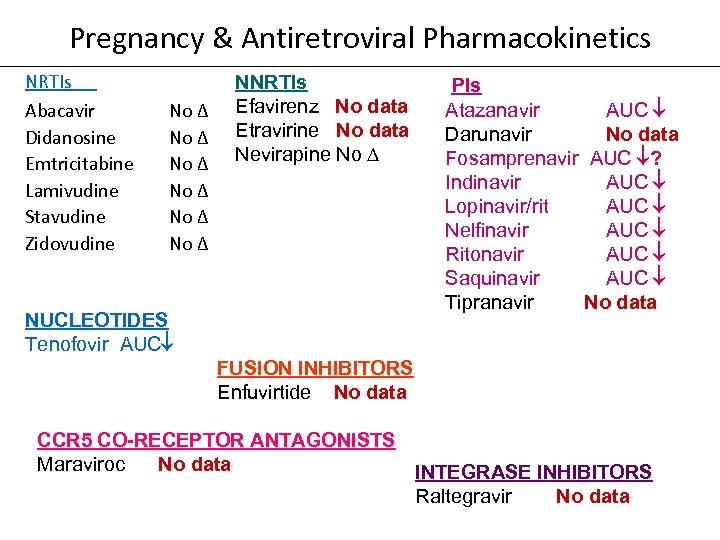
Pregnancy & Antiretroviral Pharmacokinetics NRTIs Abacavir Didanosine Emtricitabine Lamivudine Stavudine Zidovudine NNRTIs No ∆ Efavirenz No data No ∆ Etravirine No data No ∆ Nevirapine No ∆ NUCLEOTIDES Tenofovir AUC PIs Atazanavir AUC Darunavir No data Fosamprenavir AUC ? Indinavir AUC Lopinavir/rit AUC Nelfinavir AUC Ritonavir AUC Saquinavir AUC Tipranavir No data FUSION INHIBITORS Enfuvirtide No data CCR 5 CO-RECEPTOR ANTAGONISTS Maraviroc No data INTEGRASE INHIBITORS Raltegravir No data

Toxicity - associations in pregnancy Maternal Foetal • d 4 T / dd. I – lactic acidosis • EFAVIRENZ – neural tube defects • NEVIRAPINE – hepatic toxicity • TENOFOVIR – bone growth defects • ZIDOVUDINE – anaemia

Antiretroviral Pregnancy Registry 1/89 - 1/09 Prospective Cases (http: //www. APRegistry. com) % Birth Defect CDC general birth defect surveillance 1 st trimester any ARV exposure Atazanavir sulfate-containing (7/292) ABC-containing (18/608) AZT-containing (95/3108) 3 TC-containing (93/3226) d 4 T-containing (19/754) Indinavir-containing (6/276) Nelfinavir-containing (37/1074) Nevirapine-containing (18/817) Ritonavir-containing (20/883) Lopinavir-containing (8/470) Tenofovir-containing (16/678) dd. I-containing (16/365) 2. 7% (2. 7 -2. 8%) 2. 9% (2. 4 - 3. 4%) 2. 4% (1. 0 - 4. 9%) 3. 0% (1. 8 – 4. 6%) 3. 1% (2. 5 - 3. 7%) 2. 9% (2. 3 - 3. 5%) 2. 5% (1. 5 – 3. 9%) 2. 2% (0. 8 - 4. 7%) 3. 4% (2. 4 – 4. 7%) 2. 2% (1. 3 – 3. 5%) 2. 3% (1. 4 – 3. 5%) 1. 7% (0. 7 – 3. 3%) 2. 4% (1. 4 – 3. 8%) 4. 4% (2. 5 – 7. 0%)

HIV negative but exposed children following in utero exposure to ARVs Clinically symptomatic mitochondrial dysfunction – rare 0. 3% - 3 per 1, 000 – very rarely (0. 07% - 7 per 10, 000) can be fatal Mild, clinically asymptomatic, but persistent hematologic abnormalities – anaemia, neutropenia Transient elevations in lactic acid – common with in utero exposure – usually asymptomatic – resolve within months

ARV toxicities in treated children similar spectrum to adults Of particular importance in children: Metabolic abnormalities – hyperlipidaemia – glucose intolerance Fat redistribution Bone density & growth
206904a70912ebbd8899db19564fe4b5.ppt