be22133ed6d93afea7cb3d0d09474cc4.ppt
- Количество слайдов: 65

 AVIAN INFLUENZA Prof. Dr. SALAH M. HASSAN College Vet. Medicine Mosul University
AVIAN INFLUENZA Prof. Dr. SALAH M. HASSAN College Vet. Medicine Mosul University
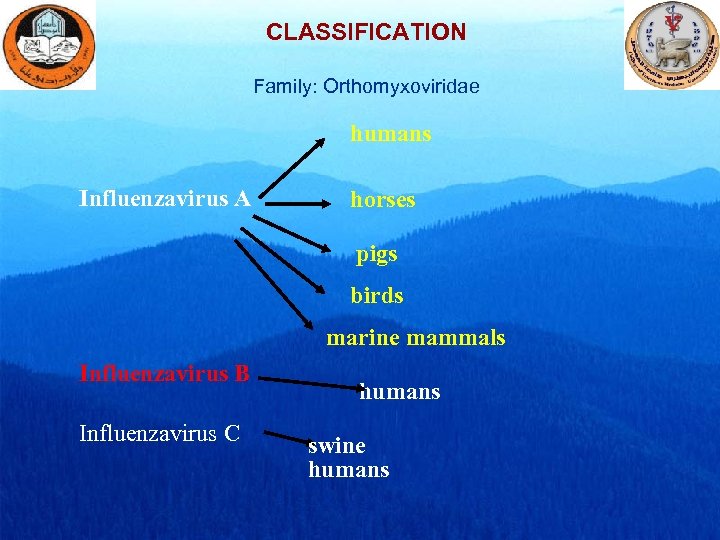 CLASSIFICATION Family: Orthomyxoviridae humans Influenzavirus A horses pigs birds marine mammals Influenzavirus B Influenzavirus C humans swine humans
CLASSIFICATION Family: Orthomyxoviridae humans Influenzavirus A horses pigs birds marine mammals Influenzavirus B Influenzavirus C humans swine humans
 Avian influenza (AI) • • asymptomatic infection to respiratory disease and drops in egg production to severe, systemic disease with near 100% mortality
Avian influenza (AI) • • asymptomatic infection to respiratory disease and drops in egg production to severe, systemic disease with near 100% mortality
 Etiology • Orthomyxoviridae, genus Influenzavirus A • single-stranded RNA • surface is covered by two types glycoprotein projections (HA), (NA).
Etiology • Orthomyxoviridae, genus Influenzavirus A • single-stranded RNA • surface is covered by two types glycoprotein projections (HA), (NA).
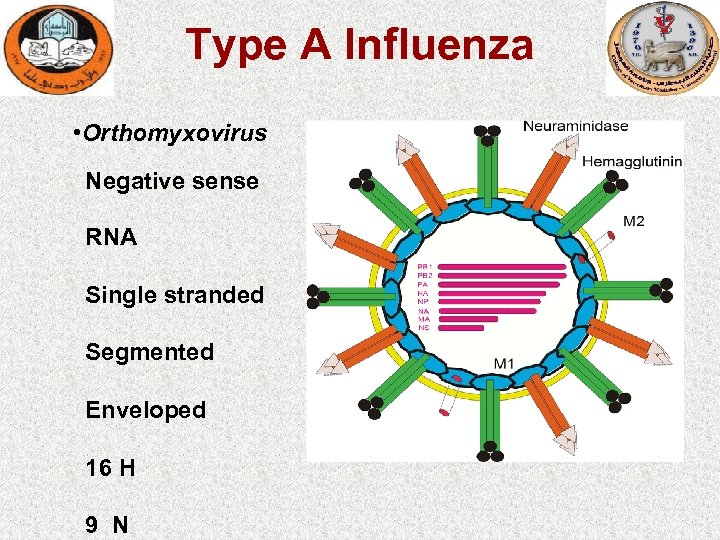 Type A Influenza • Orthomyxovirus Negative sense RNA Single stranded Segmented Enveloped 16 H 9 N
Type A Influenza • Orthomyxovirus Negative sense RNA Single stranded Segmented Enveloped 16 H 9 N
 Influenza Subtypes 16 Hemagglutinin subtypes 9 Neuraminidase subtypes 144 possible antigenic subtypes
Influenza Subtypes 16 Hemagglutinin subtypes 9 Neuraminidase subtypes 144 possible antigenic subtypes
 Avian Influenza Only viruses of H 5 and H 7 subtypes have been shown to cause HPAI in susceptible species
Avian Influenza Only viruses of H 5 and H 7 subtypes have been shown to cause HPAI in susceptible species
 Avian Influenza HPAI emerges from LPAI
Avian Influenza HPAI emerges from LPAI
 Avian Influenza Not all H 5 and H 7 viruses are HPAI
Avian Influenza Not all H 5 and H 7 viruses are HPAI
 Susceptibility to Chemical and Physical Agents • • • Unstable in the environment. Physical factors such as heat extremes of p. H dryness
Susceptibility to Chemical and Physical Agents • • • Unstable in the environment. Physical factors such as heat extremes of p. H dryness
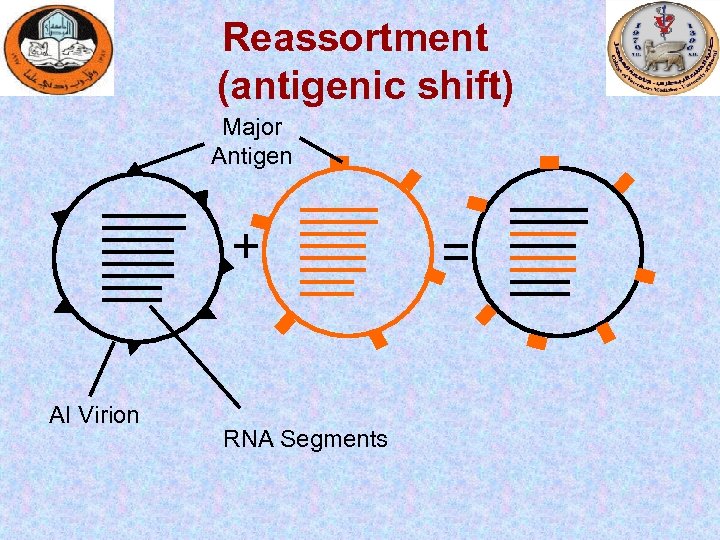 Reassortment (antigenic shift) Major Antigen AI Virion RNA Segments
Reassortment (antigenic shift) Major Antigen AI Virion RNA Segments
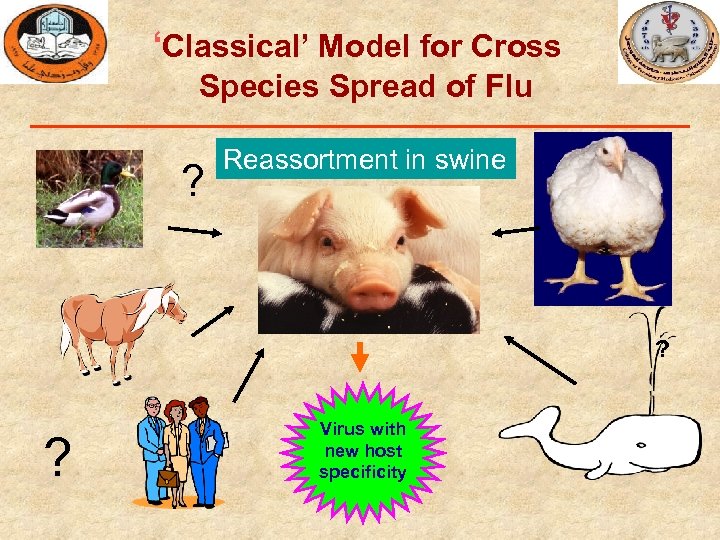 ‘Classical’ Model for Cross Species Spread of Flu ? Reassortment in swine ? ? Virus with new host specificity
‘Classical’ Model for Cross Species Spread of Flu ? Reassortment in swine ? ? Virus with new host specificity
 In Waterfowl, Influenza Viruses Replicate preferentially in the Intestinal tract. Virus transmission Among aquatic birds is via Contaminated water-oral Route.
In Waterfowl, Influenza Viruses Replicate preferentially in the Intestinal tract. Virus transmission Among aquatic birds is via Contaminated water-oral Route.
 Transmission and Carriers All 16 HA and 9 NA Subtypes Of Influenza A Virus are Maintained in aquatic Bird populations (Ducks, Shorebirds and gulls)
Transmission and Carriers All 16 HA and 9 NA Subtypes Of Influenza A Virus are Maintained in aquatic Bird populations (Ducks, Shorebirds and gulls)
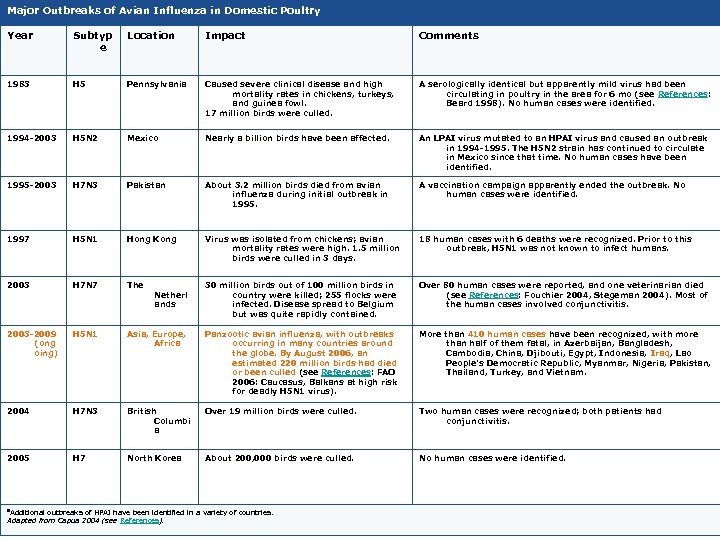 Major Outbreaks of Avian Influenza in Domestic Poultry Year Subtyp e Location Impact Comments 1983 H 5 Pennsylvania Caused severe clinical disease and high mortality rates in chickens, turkeys, and guinea fowl. 17 million birds were culled. A serologically identical but apparently mild virus had been circulating in poultry in the area for 6 mo (see References: Beard 1998). No human cases were identified. 1994 -2003 H 5 N 2 Mexico Nearly a billion birds have been affected. An LPAI virus mutated to an HPAI virus and caused an outbreak in 1994 -1995. The H 5 N 2 strain has continued to circulate in Mexico since that time. No human cases have been identified. 1995 -2003 H 7 N 3 Pakistan About 3. 2 million birds died from avian influenza during initial outbreak in 1995. A vaccination campaign apparently ended the outbreak. No human cases were identified. 1997 H 5 N 1 Hong Kong Virus was isolated from chickens; avian mortality rates were high. 1. 5 million birds were culled in 3 days. 18 human cases with 6 deaths were recognized. Prior to this outbreak, H 5 N 1 was not known to infect humans. 2003 H 7 N 7 The 30 million birds out of 100 million birds in country were killed; 255 flocks were infected. Disease spread to Belgium but was quite rapidly contained. Over 80 human cases were reported, and one veterinarian died (see References: Fouchier 2004, Stegeman 2004). Most of the human cases involved conjunctivitis. 2003 -2009 (ong oing) H 5 N 1 Asia, Europe, Africa Panzootic avian influenza, with outbreaks occurring in many countries around the globe. By August 2006, an estimated 220 million birds had died or been culled (see References: FAO 2006: Caucasus, Balkans at high risk for deadly H 5 N 1 virus). More than 410 human cases have been recognized, with more than half of them fatal, in Azerbaijan, Bangladesh, Cambodia, China, Djibouti, Egypt, Indonesia, Iraq, Lao People's Democratic Republic, Myanmar, Nigeria, Pakistan, Thailand, Turkey, and Vietnam. 2004 H 7 N 3 British Columbi a Over 19 million birds were culled. Two human cases were recognized; both patients had conjunctivitis. 2005 H 7 North Korea About 200, 000 birds were culled. No human cases were identified. a. Additional Netherl ands outbreaks of HPAI have been identified in a variety of countries. Adapted from Capua 2004 (see References).
Major Outbreaks of Avian Influenza in Domestic Poultry Year Subtyp e Location Impact Comments 1983 H 5 Pennsylvania Caused severe clinical disease and high mortality rates in chickens, turkeys, and guinea fowl. 17 million birds were culled. A serologically identical but apparently mild virus had been circulating in poultry in the area for 6 mo (see References: Beard 1998). No human cases were identified. 1994 -2003 H 5 N 2 Mexico Nearly a billion birds have been affected. An LPAI virus mutated to an HPAI virus and caused an outbreak in 1994 -1995. The H 5 N 2 strain has continued to circulate in Mexico since that time. No human cases have been identified. 1995 -2003 H 7 N 3 Pakistan About 3. 2 million birds died from avian influenza during initial outbreak in 1995. A vaccination campaign apparently ended the outbreak. No human cases were identified. 1997 H 5 N 1 Hong Kong Virus was isolated from chickens; avian mortality rates were high. 1. 5 million birds were culled in 3 days. 18 human cases with 6 deaths were recognized. Prior to this outbreak, H 5 N 1 was not known to infect humans. 2003 H 7 N 7 The 30 million birds out of 100 million birds in country were killed; 255 flocks were infected. Disease spread to Belgium but was quite rapidly contained. Over 80 human cases were reported, and one veterinarian died (see References: Fouchier 2004, Stegeman 2004). Most of the human cases involved conjunctivitis. 2003 -2009 (ong oing) H 5 N 1 Asia, Europe, Africa Panzootic avian influenza, with outbreaks occurring in many countries around the globe. By August 2006, an estimated 220 million birds had died or been culled (see References: FAO 2006: Caucasus, Balkans at high risk for deadly H 5 N 1 virus). More than 410 human cases have been recognized, with more than half of them fatal, in Azerbaijan, Bangladesh, Cambodia, China, Djibouti, Egypt, Indonesia, Iraq, Lao People's Democratic Republic, Myanmar, Nigeria, Pakistan, Thailand, Turkey, and Vietnam. 2004 H 7 N 3 British Columbi a Over 19 million birds were culled. Two human cases were recognized; both patients had conjunctivitis. 2005 H 7 North Korea About 200, 000 birds were culled. No human cases were identified. a. Additional Netherl ands outbreaks of HPAI have been identified in a variety of countries. Adapted from Capua 2004 (see References).
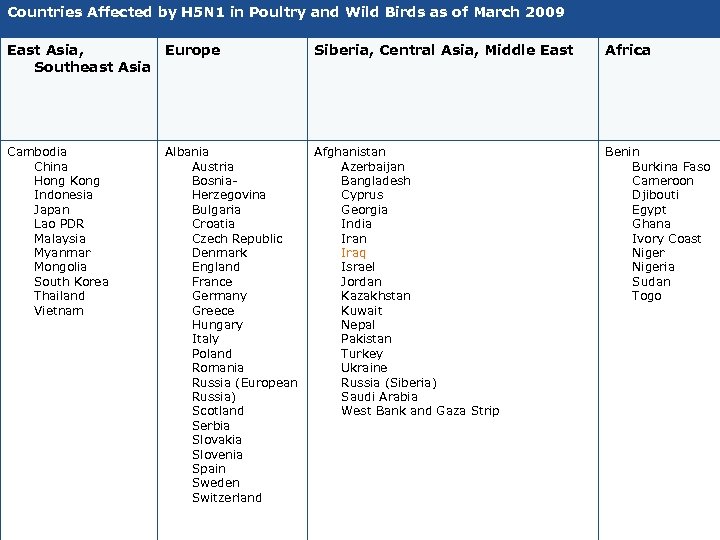 Countries Affected by H 5 N 1 in Poultry and Wild Birds as of March 2009 East Asia, Europe Southeast Asia Siberia, Central Asia, Middle East Africa Cambodia China Hong Kong Indonesia Japan Lao PDR Malaysia Myanmar Mongolia South Korea Thailand Vietnam Afghanistan Azerbaijan Bangladesh Cyprus Georgia India Iran Iraq Israel Jordan Kazakhstan Kuwait Nepal Pakistan Turkey Ukraine Russia (Siberia) Saudi Arabia West Bank and Gaza Strip Benin Burkina Faso Cameroon Djibouti Egypt Ghana Ivory Coast Nigeria Sudan Togo Albania Austria Bosnia. Herzegovina Bulgaria Croatia Czech Republic Denmark England France Germany Greece Hungary Italy Poland Romania Russia (European Russia) Scotland Serbia Slovakia Slovenia Spain Sweden Switzerland
Countries Affected by H 5 N 1 in Poultry and Wild Birds as of March 2009 East Asia, Europe Southeast Asia Siberia, Central Asia, Middle East Africa Cambodia China Hong Kong Indonesia Japan Lao PDR Malaysia Myanmar Mongolia South Korea Thailand Vietnam Afghanistan Azerbaijan Bangladesh Cyprus Georgia India Iran Iraq Israel Jordan Kazakhstan Kuwait Nepal Pakistan Turkey Ukraine Russia (Siberia) Saudi Arabia West Bank and Gaza Strip Benin Burkina Faso Cameroon Djibouti Egypt Ghana Ivory Coast Nigeria Sudan Togo Albania Austria Bosnia. Herzegovina Bulgaria Croatia Czech Republic Denmark England France Germany Greece Hungary Italy Poland Romania Russia (European Russia) Scotland Serbia Slovakia Slovenia Spain Sweden Switzerland
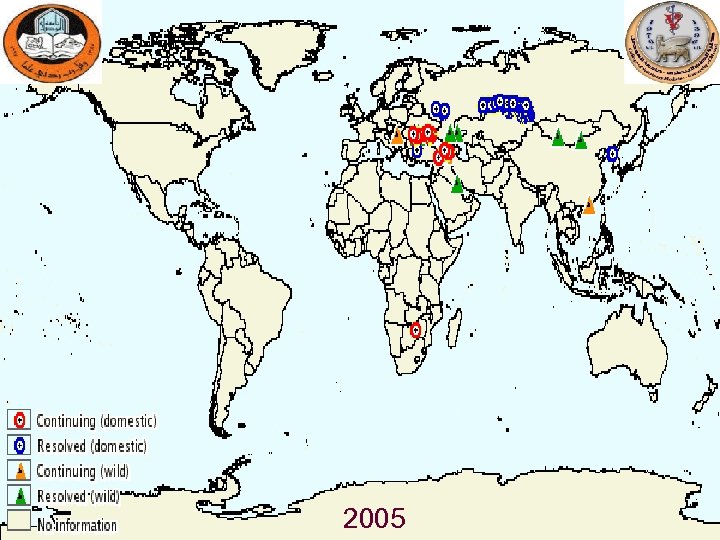 2005
2005
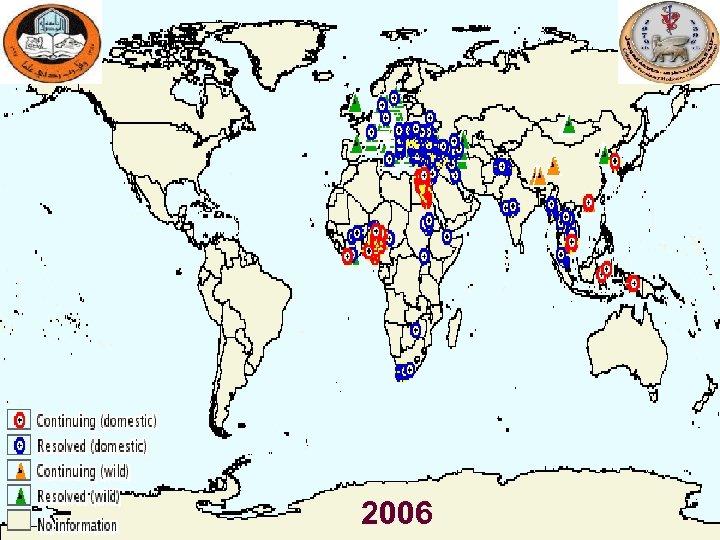 2006
2006
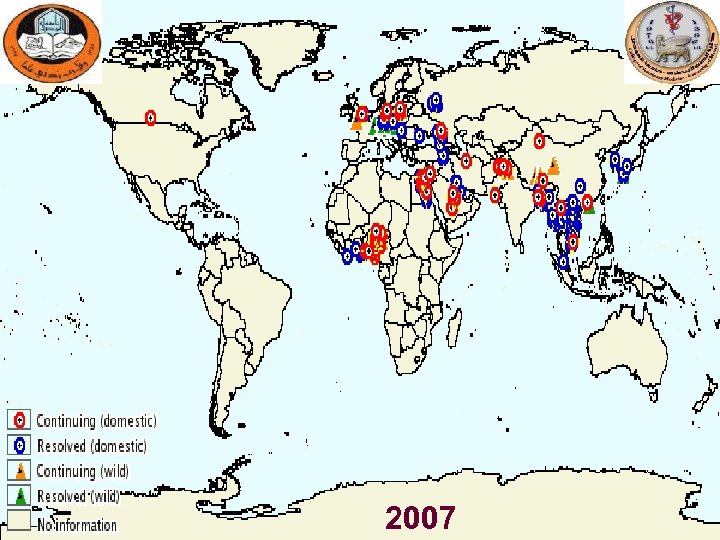 2007
2007
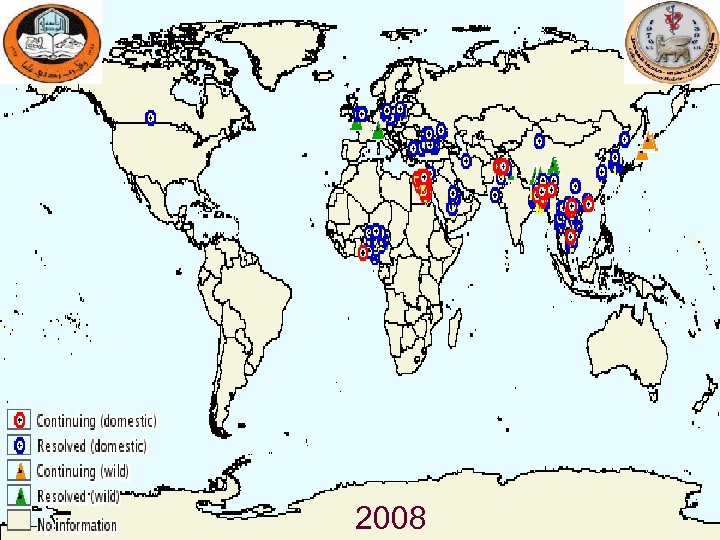 2008
2008
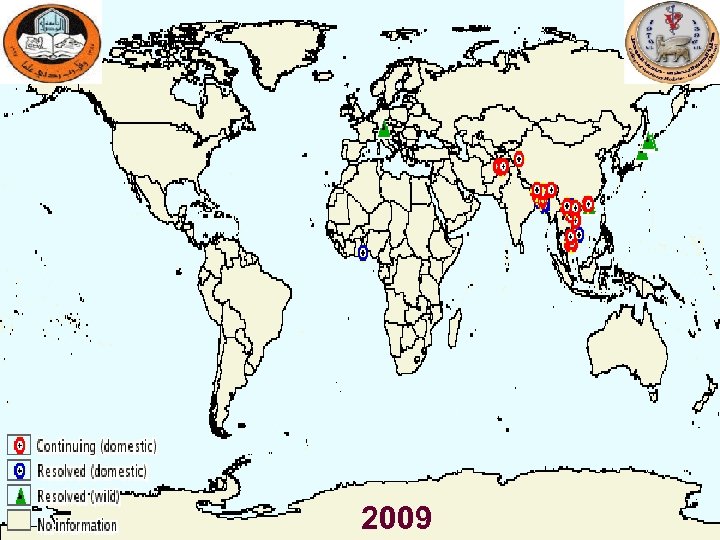 2009
2009
 How is the disease transmitted and spread? Several factors can contribute to the spread of AI viruses including globalization and international trade (legally and illegally), marketing practices (live bird markets), farming practices and the presence of the viruses in wild birds.
How is the disease transmitted and spread? Several factors can contribute to the spread of AI viruses including globalization and international trade (legally and illegally), marketing practices (live bird markets), farming practices and the presence of the viruses in wild birds.
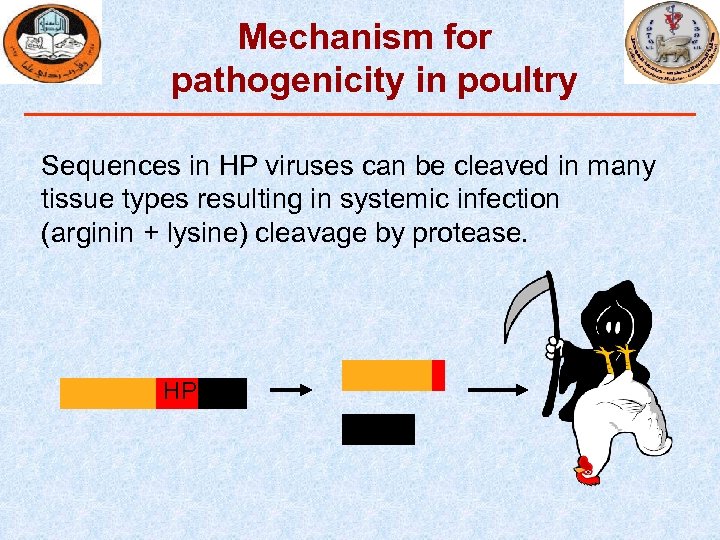 Mechanism for pathogenicity in poultry Sequences in HP viruses can be cleaved in many tissue types resulting in systemic infection (arginin + lysine) cleavage by protease. HP
Mechanism for pathogenicity in poultry Sequences in HP viruses can be cleaved in many tissue types resulting in systemic infection (arginin + lysine) cleavage by protease. HP
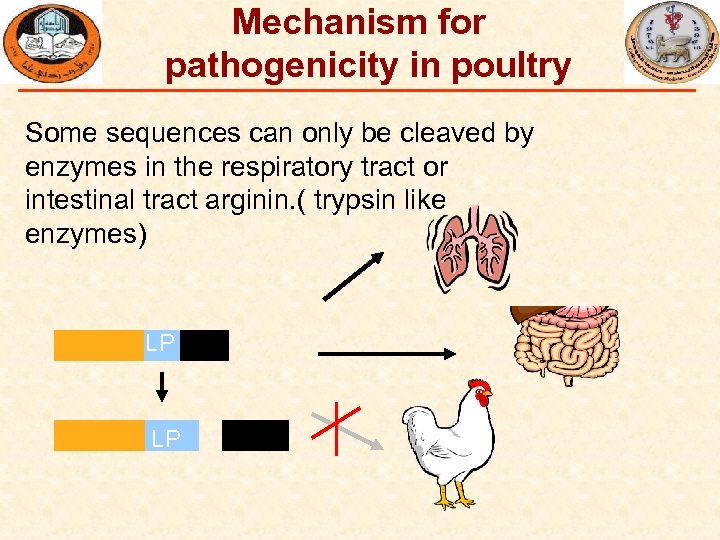 Mechanism for pathogenicity in poultry Some sequences can only be cleaved by enzymes in the respiratory tract or intestinal tract arginin. ( trypsin like enzymes) LP LP
Mechanism for pathogenicity in poultry Some sequences can only be cleaved by enzymes in the respiratory tract or intestinal tract arginin. ( trypsin like enzymes) LP LP
 Incubation Period • • • 3 up to 14 days dose of virus route of exposure species exposed ability to detect clinical signs
Incubation Period • • • 3 up to 14 days dose of virus route of exposure species exposed ability to detect clinical signs
 Morbidity and Mortality • LP AI less than 5% unless • HP AI 50— 89% and can reach 100%
Morbidity and Mortality • LP AI less than 5% unless • HP AI 50— 89% and can reach 100%
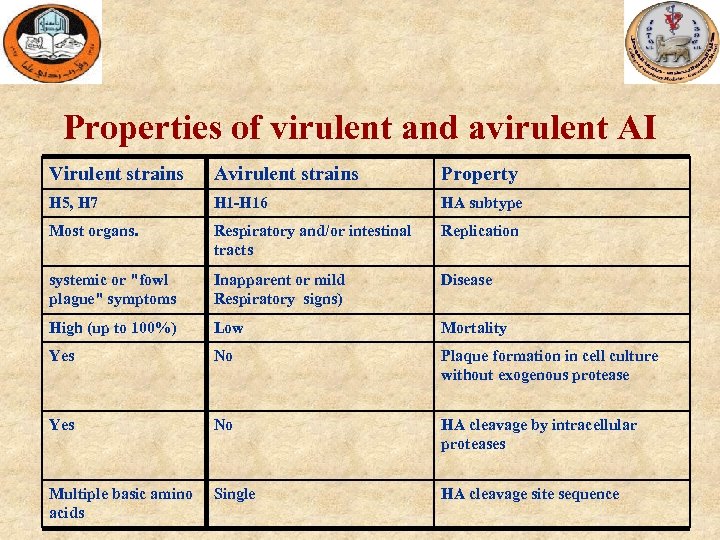 Properties of virulent and avirulent AI Virulent strains Avirulent strains Property H 5, H 7 H 1 -H 16 HA subtype Most organs. Respiratory and/or intestinal tracts Replication systemic or "fowl plague" symptoms Inapparent or mild Respiratory signs) Disease High (up to 100%) Low Mortality Yes No Plaque formation in cell culture without exogenous protease Yes No HA cleavage by intracellular proteases Multiple basic amino acids Single HA cleavage site sequence
Properties of virulent and avirulent AI Virulent strains Avirulent strains Property H 5, H 7 H 1 -H 16 HA subtype Most organs. Respiratory and/or intestinal tracts Replication systemic or "fowl plague" symptoms Inapparent or mild Respiratory signs) Disease High (up to 100%) Low Mortality Yes No Plaque formation in cell culture without exogenous protease Yes No HA cleavage by intracellular proteases Multiple basic amino acids Single HA cleavage site sequence


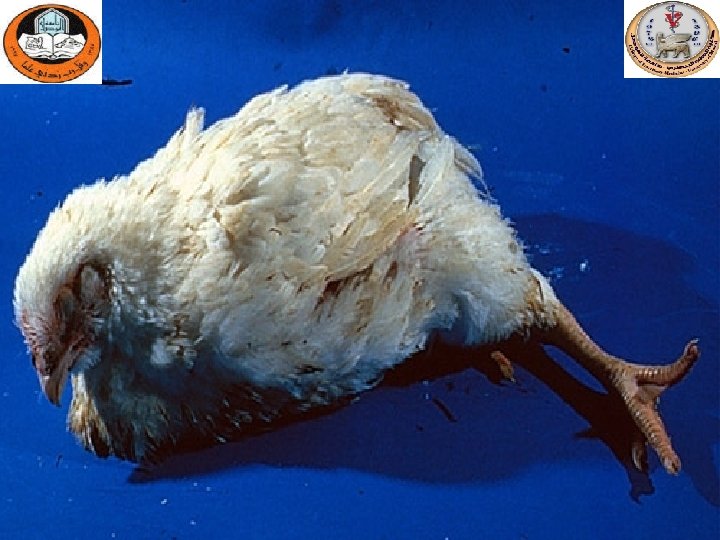

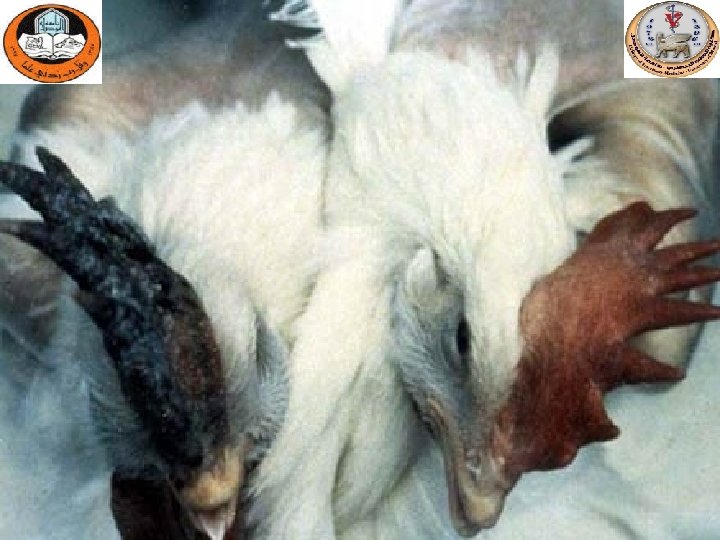
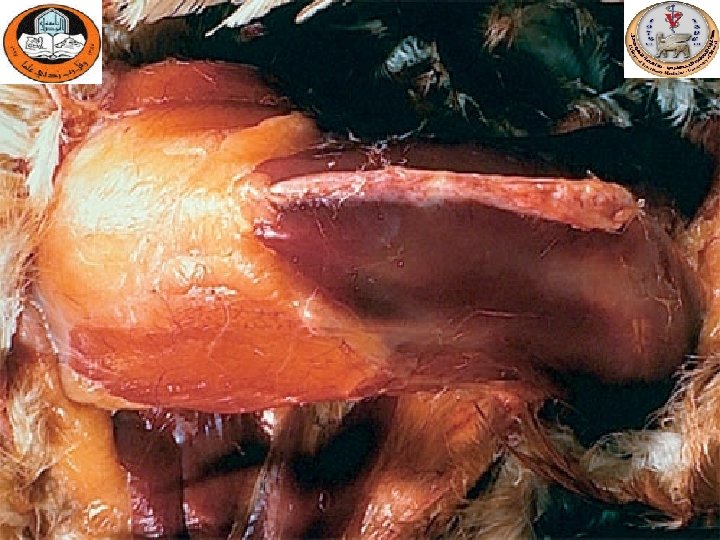
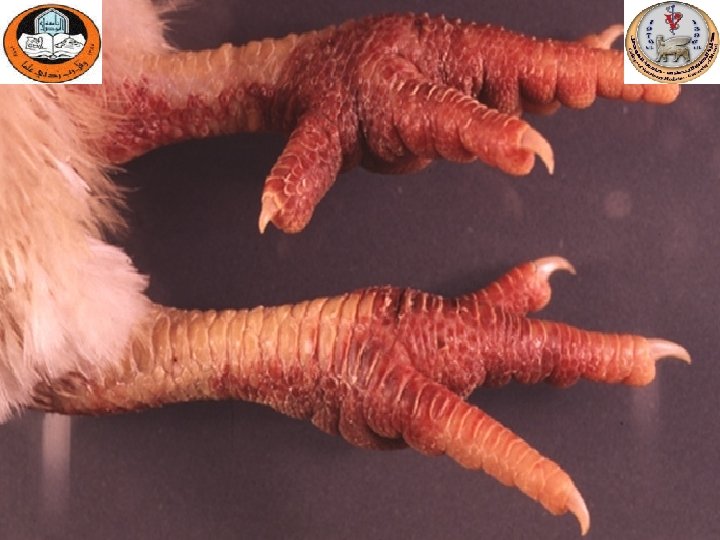
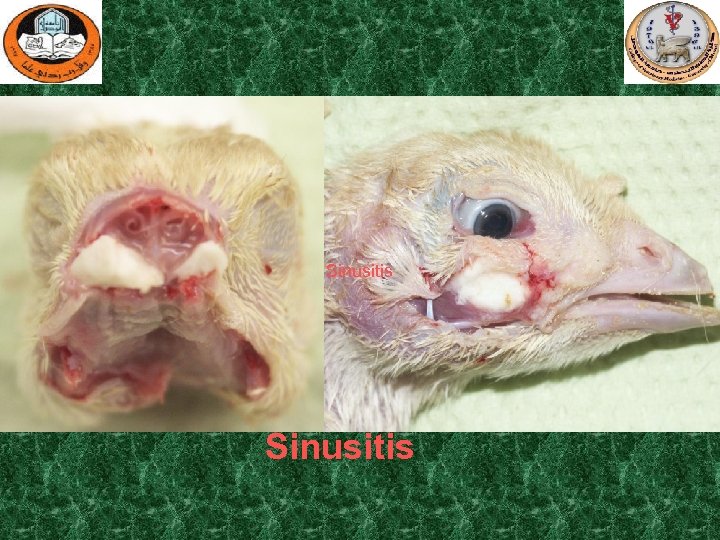 Sinusitis
Sinusitis
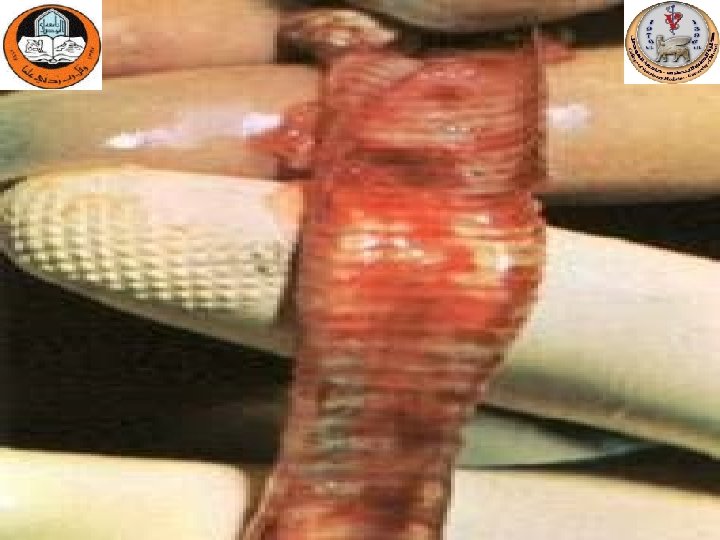
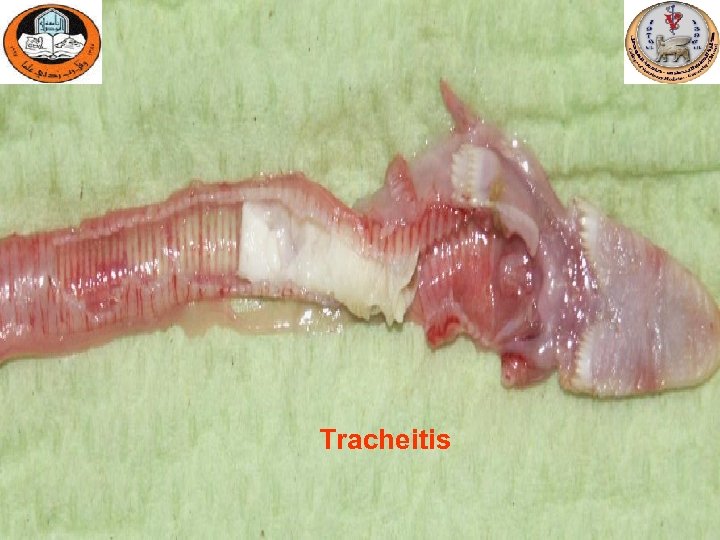 Tracheitis
Tracheitis
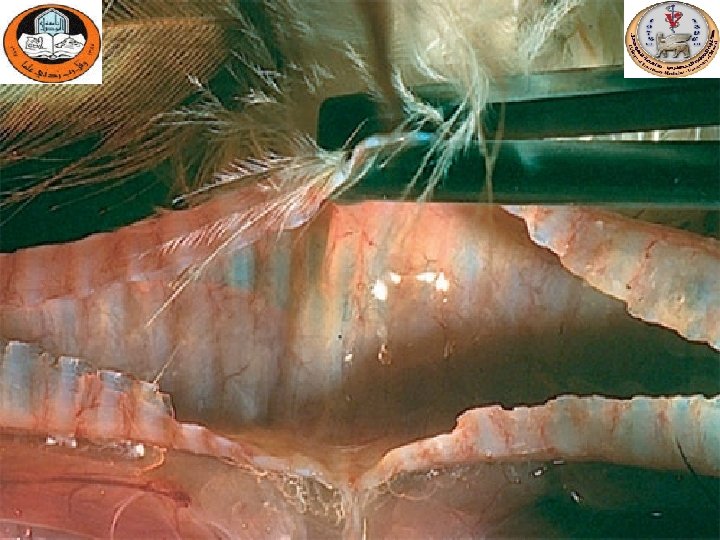
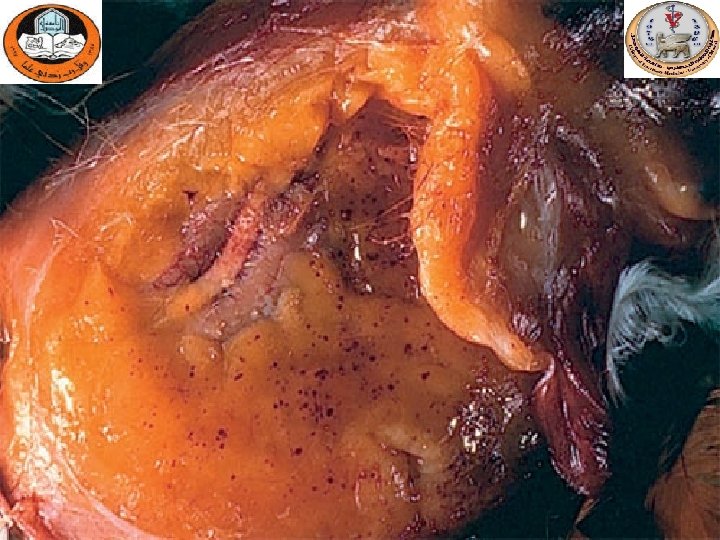
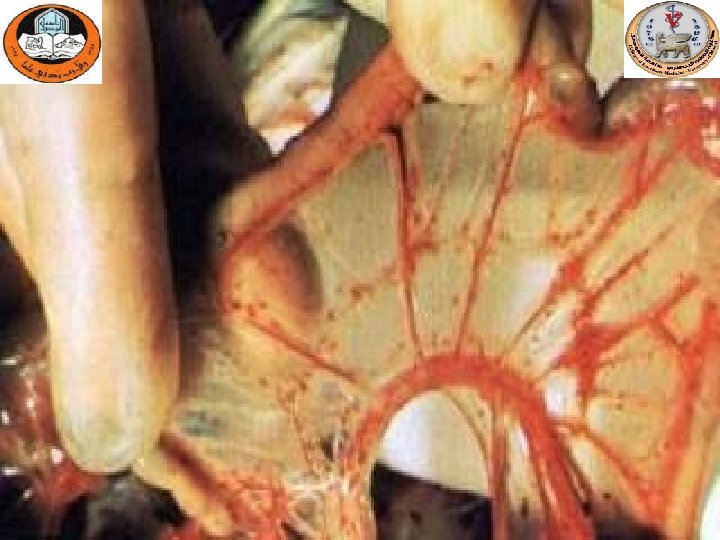

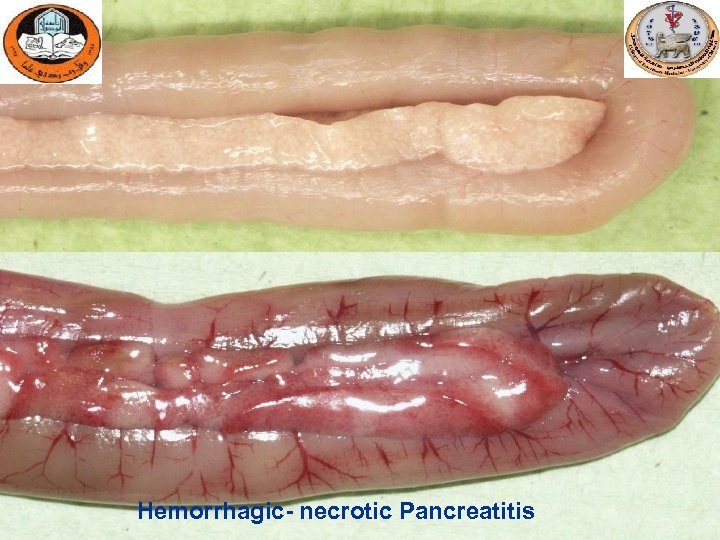 Hemorrhagic- necrotic Pancreatitis
Hemorrhagic- necrotic Pancreatitis

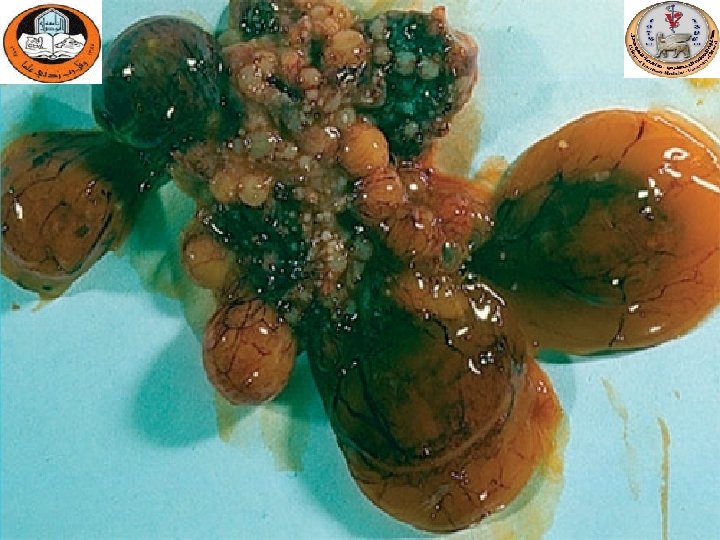
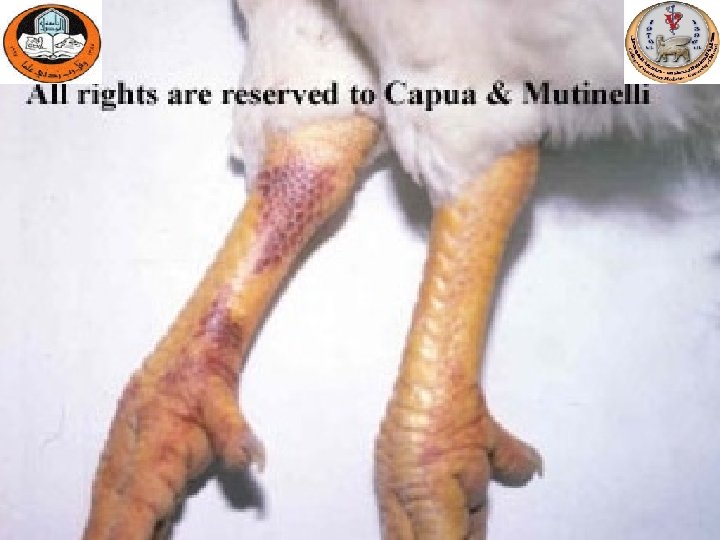
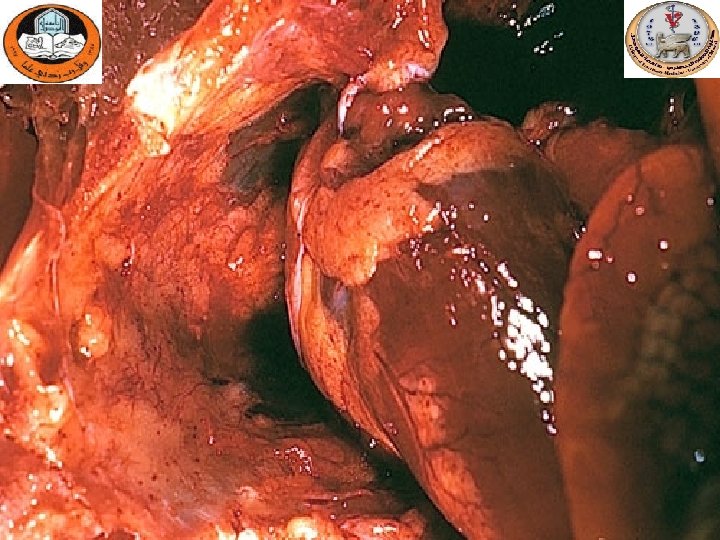
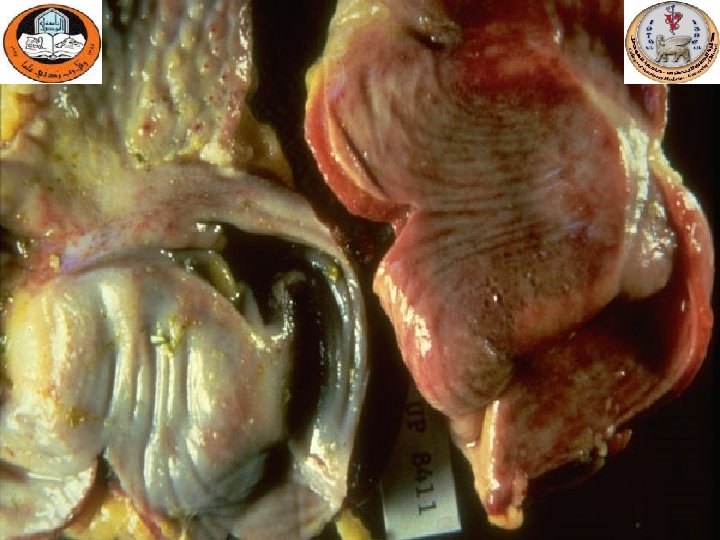
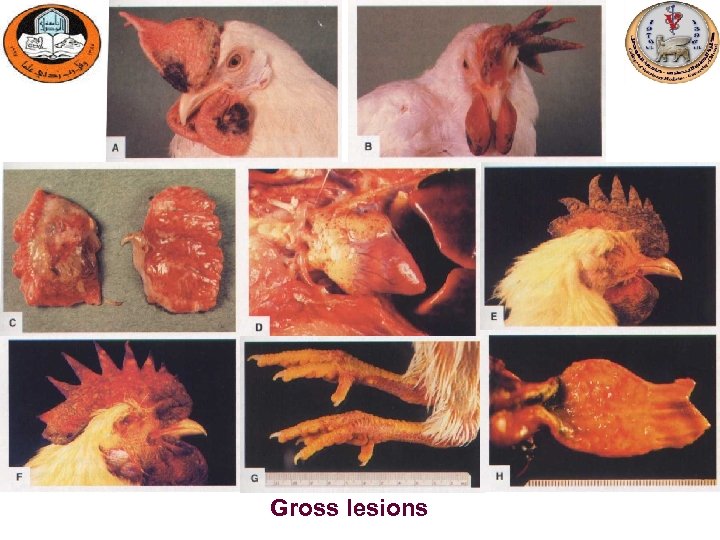 Gross lesions
Gross lesions
 In layers and breeders, reduce egg production
In layers and breeders, reduce egg production
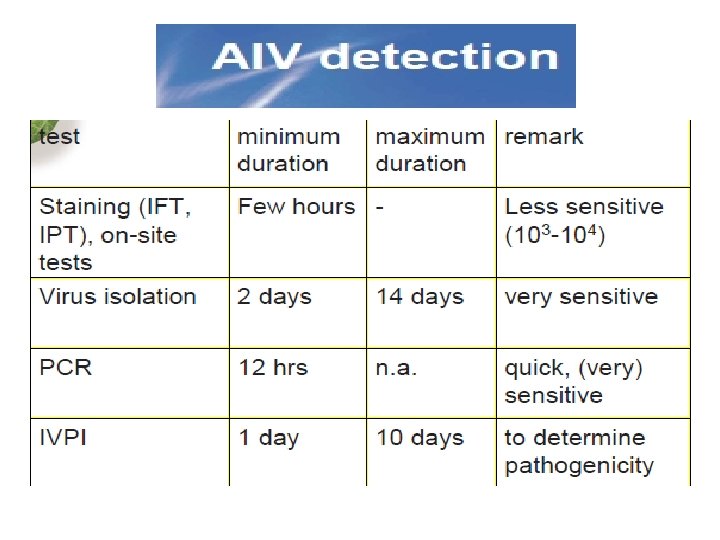
 (First) Diagnosis of AIV • • • - isolation of hemagglutinating agent in embryonated eggs; daily candling of eggs - determine H-subtype by HI-ab - determine virulence with IVPI - sequencing cleavage site hemagglutinin: (Note mixtures of LP and HP)
(First) Diagnosis of AIV • • • - isolation of hemagglutinating agent in embryonated eggs; daily candling of eggs - determine H-subtype by HI-ab - determine virulence with IVPI - sequencing cleavage site hemagglutinin: (Note mixtures of LP and HP)










 sm. hassan 04@gmail. com
sm. hassan 04@gmail. com


