85232a38065a7345921bd6b6d69fc98b.ppt
- Количество слайдов: 32

Automated Blood Cultures 韩向阳 Xiang-Yang Han, MD, Ph. D Department of Laboratory Medicine The University of Texas M. D. Anderson Cancer Center

Blood Cultures • Factors affecting bloodstream infections and culture yield • Parameters • Significance and interpretation • Systems and media • Fastidious organisms • Trends

Bloodstream Infection: Factors-1 • Bloodstream infection is the most severe form of infection and carries a high fatality (20% to 50%). • Microbial invasion into bloodstream reflects failure of initial host defense: loss of integrity of skin or mucosa, weakened innate or acquired immunity, or direct blood inoculation.

Bloodstream Infection: Factors-2 • Microbial factors: virulence mechanisms, such as toxins, intracellular survival, evasion or shielding from host defenses (leukocytes, complements, antibodies, etc). • The presence of antimicrobics in the circulation negatively affect culture positivity.

Blood Cultures: Parameters-1 • Timing – Blood should be drawn before antibiotic therapy, if all possible; – The presence of bacteria or fungi in the bloodstream is constant in case of endocarditis; – In other cases, microbes in circulation are not steady, and the best time to draw blood is during the rise of fever.

Blood Cultures: Parameters-2 • Volume – 20 -40 ml for each set of cultures (one aerobic bottle and one anaerobic bottle), e. g. , • 10 ml blood inoculated into 40 ml broth of BACTEC bottle to reach 1: 5 ratio; • 20 ml blood inoculated into 80 ml broth of ESP 80 A bottle (ratio 1: 5). – Pediatric cultures ranging from 1 to 10 ml, depending on the age.

Blood Cultures: Parameters-3 • Frequency – For each episode, 2 to 3 sets of culture should be obtained within first 24 hrs; – Data on 282 bacteremic episodes by Weinstein et al (RID 1983; 5: 35 -53): • First culture detected 257 (91%); • Two cultures together detected 281 (99%).

Blood Cultures: Parameters-4 • Incubation atomsphere – The proportion of anaerobic positive cultures is decreasing; – Thus, routine anaerobic cultures are not required now; – The situation of individual institution and patient population needs to be considered, such as surgical and OB/GYN patients.

Blood Cultures: Parameters-5 • How long to incubate? – Many studies have looked into this issue; – Five days are sufficient to detect nearly all (~99%) significant organisms; – Longer incubation mostly picks up contaminants; – A culture turned positive 6 -7 days later is unlikely to affect patient care

Blood Cultures: Interpretation • Almost always significant: – Staphylococcus aureus, Escherichia coli and other members of Enterobacteriaceae, Pseudomonas aeruginosa, Candida spp. • Common contaminant, but individual judgement needed: – – Coagulase-neg. staphylococci Corneform bacilli Alpha-hemolytic streptococci Propionibacterium acne

Blood Cultures: Methods • Manual method (1950 s-1970 s) – Incubation for 21 days, visual inspection of growth of organisms, and blind subcultures; – Isolator system (lysis centrifugation method) • Automated method (1980 s) – Automated detection of microbial CO 2 production, incubation shortened to 7 days, no blind subcultures; – Examples: BACTEC 460 and 660, later BACTEC NR 660. • Continuously monitoring blood culturing system (CMBCS)

Blood Cultures: CMBCS-1 • Bio. Merieux (Former Organon-Teknika, Durham, NC) – Bac. T/Alert series since 1990 • Colorimetric detection for CO 2 production; • Every 10 minutes to detect signal and go to algorithm for analysis to see if significant growth has occurred; • Newer system since 2001 Bac. T/Alert 3 D


Blood Cultures: CMBCS-2 • Becton-Dickinson (Sparks, MD) – BACTEC series • Fluorescent detection of CO 2 production • Every 10 minutes to detect signal • Newer system: BACTEC LX 2004– using laser to detect CO 2 production – Clinical evaluation in progress.


Blood Cultures: CMBCS-3 • Trek (Former Difco, then ESP, Accumed, Cleveland, OH) – ESP series – Manometric detection of CO 2 production – Every 12 minutes detection – Newer system Versa. Trek, 2004

Blood Cultures: CMBCS-4 • Comparison of 3 systems – They are comparable overall – Bac. T/Alert FAN bottle (containing antimicrobic-removing substance) performs slightly better than standard bottles and other systems
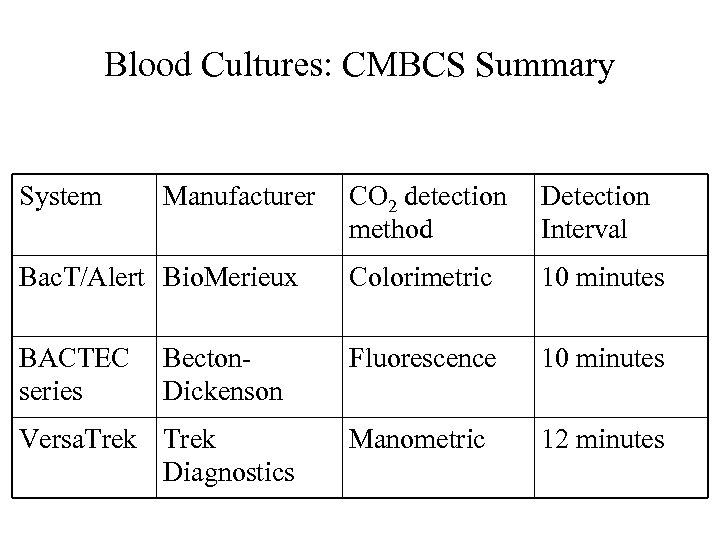
Blood Cultures: CMBCS Summary System Manufacturer CO 2 detection method Detection Interval Bac. T/Alert Bio. Merieux Colorimetric 10 minutes BACTEC series Fluorescence 10 minutes Manometric 12 minutes Becton. Dickenson Versa. Trek Diagnostics

Quantitative Blood Cultures: Lysis Centrifugation Method

Blood Cultures: Positive Rates • Overall positive rate ~10% • HACEK organisms, 0. 01% for all blood cultures or 1% of all blood isolates

Blood Cultures: Common Organisms • Data from Reimer et al. , 1997 1 2 3 4 5 6 7 8 9 10 1975 -77 E. coli S. aureus S. pneumoniae K. pneumoniae P. aeruginosa B. fragilis Enterococcus spp. S. pyogenes C. albicans P. mirabilis 1992 -93 S. aureus E. coli Coag-neg. Staph K. pneumoniae Enterococcus spp. P. aeruginosa S. pneumoniae Viridans streptococci C. albicans E. cloacae

Blood Cultures: Time of Detection • Data from BACTEC 9240 (Pat Murray, 1997) – – – – Streptococcus Enterobacteriaceae Enterococcus Staphylococcus aureus Pseudomonas Coag-neg. Staph Yeast 10. 3 hr 14. 0 hr 15. 1 hr 17. 8 hr 18. 5 hr 22. 9 hr 65. 1 hr

Blood culture for HACEK • Data from Septi-Chek system (Doern et al. , 1996) – – – – – Organism Days 1 -5 Haemophilus aphrophilus 19 A. actinomycetemcomitans 9 Cardiobacterium hominis 16 Eikenella corrodens 10 Kingella spp. 3 Brucella spp. 8 Francisella tularensis 5 Nutritionally v. strep 121 Total 191 6 -7 4 1 1 2 1 12 21 >=8 1 2 3 6 Mean 3. 3 3. 7 2. 9 3. 4 3. 8 5. 6 3. 6 4. 4

Blood culture for Brucella • Bannatyne et al. JCM 1997; 35: 2673 -4 • BACTEC 9240 System – – – – Days 1 -3 4 -5 6 -7 8 -9 10 Total No. isolated 48 (49. 5%) 42 43. 4%) 4 (4. 1%) 3 (3. 1%) 0 97 (100%)

Blood Culture Protocol for Endocarditis? • Not necessary (Baron EJ et al. , CID 2005;

Blood Culture Media • Aerobic media – Standard aerobic (SA) bottles for BACTEC, Bac. T/Alert, and Trek • Anaerobic media – Standard anaerobic (SN) bottles for BACTEC, Bac. T/Alert, and Trek • Mycobacterial media: based on Middlebrook 7 H 9 broth • Additives to remove antimicrobics – Resins in BACTEC bottles – FAN in Bac. T/Alert bottles

Isolation and Pure Culture
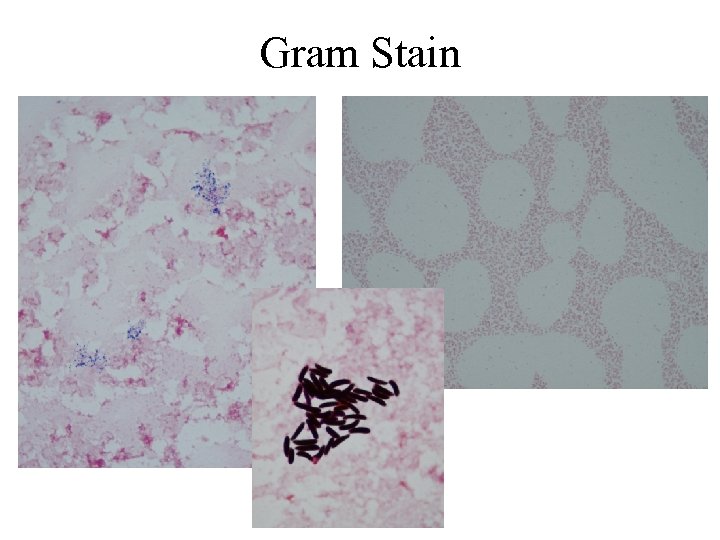
Gram Stain
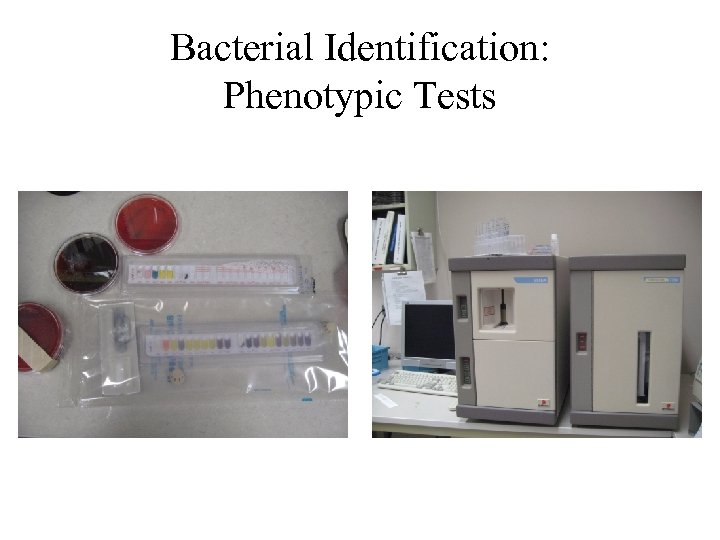
Bacterial Identification: Phenotypic Tests

Antimicrobial Susceptibility Tests

Bacterial Identification: Genotypic Tests • PCR sequencing of the 16 S r. RNA gene – DNA extraction from colony or positive liquid – PCR amplification – Sequencing – Matching with database – Correlating with culture features – Report final identification

85232a38065a7345921bd6b6d69fc98b.ppt