1fd7be72a96f89757f91bf11ced15f7a.ppt
- Количество слайдов: 15

Astrocyte-mediated control of cerebral blood flow Nature Neuroscience 9, 260 -267. 2006. Takahiro Takano, Guo-Feng Tian, Weiguo Peng, Nanhong Lou, Witold Libionka, Xiaoning Han & Maiken Nedergaard. Center for Aging and Developmental Biology, Department of Neurosurgery, University of Rochester Medical School. Neurovascular Regulation of Astrocytes in the Biomedical and Clinical Context Matthew Engel December 13, 2006

Role of Astrocytes in Normal/Pathological Brain Function Central Nervous System Neurons Functional Hyperemia Glia Astrocytes New Drug Target Neurodegenerative Diseases Sensory Processing Epilepsy, Alzheimer's, Parkinson’s Diseases Tian, G. F. , Takano, T. , Lin, J. H. , Wang, X. , Bekar, L. , and Nedergaard, M. (2006) Imaging of cortical astrocytes using 2 photon laser scanning microscopy in the intact mouse brain, Advanced drug delivery reviews 58, 773 -787.
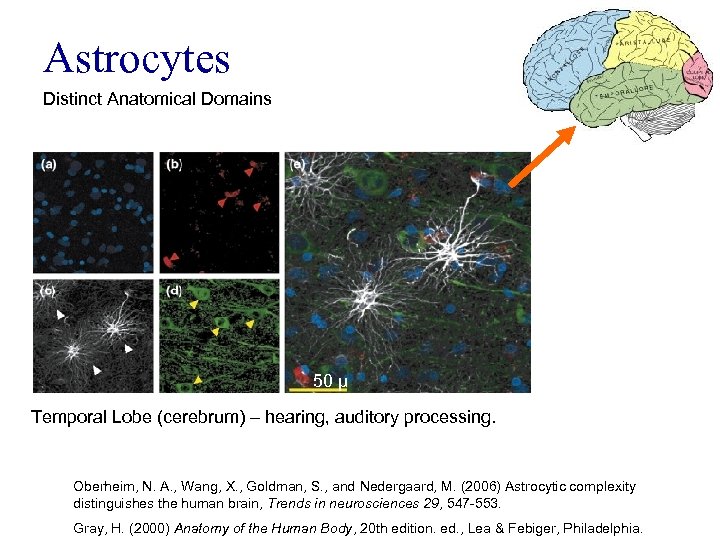
Astrocytes Distinct Anatomical Domains 50 μ Temporal Lobe (cerebrum) – hearing, auditory processing. Oberheim, N. A. , Wang, X. , Goldman, S. , and Nedergaard, M. (2006) Astrocytic complexity distinguishes the human brain, Trends in neurosciences 29, 547 -553. Gray, H. (2000) Anatomy of the Human Body, 20 th edition. ed. , Lea & Febiger, Philadelphia.
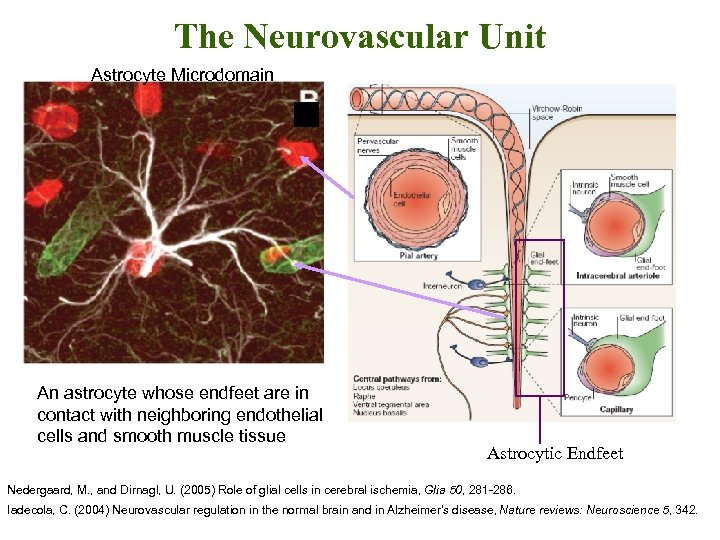
The Neurovascular Unit Astrocyte Microdomain An astrocyte whose endfeet are in contact with neighboring endothelial cells and smooth muscle tissue Astrocytic Endfeet Nedergaard, M. , and Dirnagl, U. (2005) Role of glial cells in cerebral ischemia, Glia 50, 281 -286. Iadecola, C. (2004) Neurovascular regulation in the normal brain and in Alzheimer's disease, Nature reviews: Neuroscience 5, 342.

Calcium Propagation Glutamate, GABA release at synapse In Astrocytes: Synaptic activity • Oscillatory Ca+2 increase • Intracellular Ca+2 waves -Spatially restricted • Relaxation of smooth muscle • Significant Vasodilation • Ca+2 waves propagated to neighboring cells. • Release gliotransmitters. Vwave = 8 -10 μ/s Dwave = 150 -200 μm pre/post-synaptic ADP receptors Inhibitor of excitatory transmission Astrocytes release: ATP hydrolysis → ADP Wang, X. , Lou, N. , Xu, Q. , Tian, G. F. , Peng, W. G. , Han, X. , Kang, J. , Takano, T. , and Nedergaard, M. (2006) Astrocytic Ca 2+ signaling evoked by sensory stimulation in vivo, Nature neuroscience 9, 816 -823.

Q: How is this measured? A: Real time imaging of Ca+2 in vivo. §Image of ‘caged’ dye, serves as fluorescent tracer – photolyzable chelator. §DMNP-EDTA: Upon photostimulation, affinity for calcium decreases 600, 000 fold and kd increases from 3 n. M to 5 m. M. Photolysis results in pulse of free calcium. Red: NP-EGTA 2 Photon emission Blue: DMNP-EDTA Single photon emission has insufficient depth. DMNP-EDTA resolution of conventional caging is inadequate. Cage excitation Ca+2 release Molecular Probes. Caged Probes for a Variety of Purposes. Section 17. 2. pp 711 -715.

Preliminary Results Penetrating artery 30 μ Rhod-2 and GFP (under GFAP-promoter) colocalized. Indicates that rhod-2 calcium indicator (dye) preferentially taken up by astrocytes. Notes: • Choice of (transgenic) animal and anesthetics are imperative. • Animal is intubated and artificially ventilated. Measure blood-gas content from femoral catheter. • Use of cranial window requires removal of skull and dura matter. Takano, T. , Tian, G. F. , Peng, W. , Lou, N. , Libionka, W. , Han, X. , and Nedergaard, M. (2006) Astrocyte-mediated control of cerebral blood flow, Nature neuroscience 9, 260 -267. Supplementary material.

Results b. Cross section over time of penetrating artery targeted by DMNP-EDTA photolysis. Displays rapid increase in endfoot calcium and vasodilation. c. Time course of calcium uncaging caused by photolysis. Targeted photolysis of DMNP-EDTA in astrocytic endfeet stimulated vasodilation. Takano, T. , Tian, G. F. , Peng, W. , Lou, N. , Libionka, W. , Han, X. , and Nedergaard, M. (2006) Astrocyte-mediated control of cerebral blood flow, Nature neuroscience 9, 260 -267.

An astrocytic basis of epilepsy Normal brain function is disrupted by burst neurological activity. Seizures premeditated by associated Ca+2 signaling. 2 Photon images of in Ca+2 hippocampal slices with 4 -AP. Paroxysmal depolarization shift (PDS) independent of action potential – no electrical stimulation required. Photolysis of caged Ca+2 causes PDS – hallmark depolarization discharge present in EEG. Tian, G. F. , Azmi, H. , Takano, T. , Xu, Q. , Peng, W. , Lin, J. , Oberheim, N. , Lou, N. , Wang, X. , Zielke, H. R. , Kang, J. , and Nedergaard, M. (2005) An astrocytic basis of epilepsy, Nature medicine 11, 973 -981.
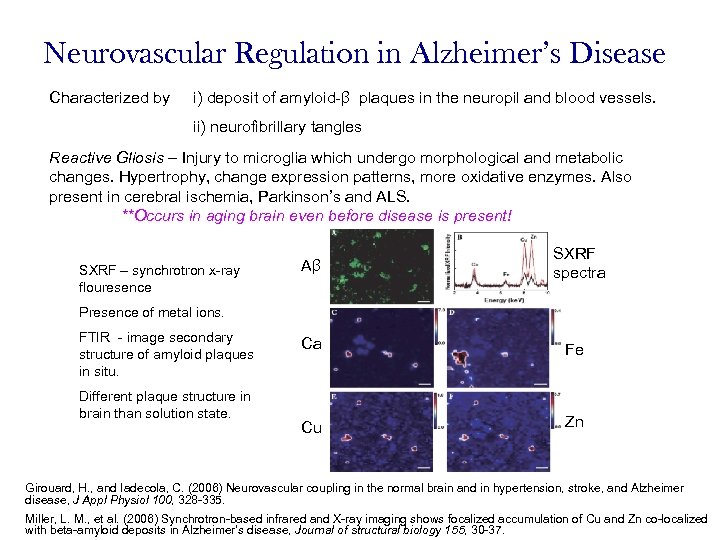
Neurovascular Regulation in Alzheimer’s Disease Characterized by i) deposit of amyloid-β plaques in the neuropil and blood vessels. ii) neurofibrillary tangles Reactive Gliosis – Injury to microglia which undergo morphological and metabolic changes. Hypertrophy, change expression patterns, more oxidative enzymes. Also present in cerebral ischemia, Parkinson’s and ALS. **Occurs in aging brain even before disease is present! SXRF – synchrotron x-ray flouresence Aβ SXRF spectra Presence of metal ions. FTIR - image secondary structure of amyloid plaques in situ. Different plaque structure in brain than solution state. Ca Fe Cu Zn Girouard, H. , and Iadecola, C. (2006) Neurovascular coupling in the normal brain and in hypertension, stroke, and Alzheimer disease, J Appl Physiol 100, 328 -335. Miller, L. M. , et al. (2006) Synchrotron-based infrared and X-ray imaging shows focalized accumulation of Cu and Zn co-localized with beta-amyloid deposits in Alzheimer's disease, Journal of structural biology 155, 30 -37.
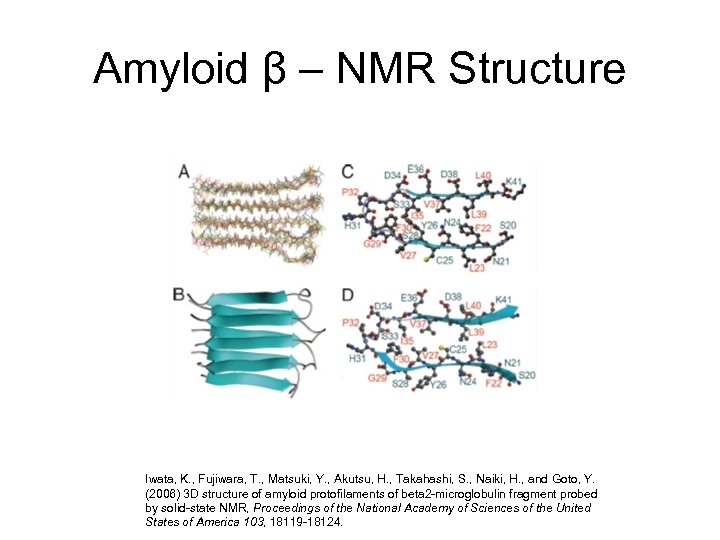
Amyloid β – NMR Structure Iwata, K. , Fujiwara, T. , Matsuki, Y. , Akutsu, H. , Takahashi, S. , Naiki, H. , and Goto, Y. (2006) 3 D structure of amyloid protofilaments of beta 2 -microglobulin fragment probed by solid-state NMR, Proceedings of the National Academy of Sciences of the United States of America 103, 18119 -18124.


The Neurovascular Unit §Neurons, glia and cerebral blood vessels form an integrated unit at the pial surface. §Glutamate release from neurons triggers Ca+2 wave propagation in astrocytic end feet. §Increases in astrocytic calcium result in coordinated vascular response: vasodilation. Endothelial sheath is surrounded by nervous tissue and smooth muscle. §Result: Calcium stimulation of perivascular layer mediates in local hemodynamic response. Intracerebral arterioles and capillaries. Iadecola, C. (2004) Neurovascular regulation in the normal brain and in Alzheimer's disease, Nature reviews: Neuroscience 5, 347 -360.

regulation Integration of vascular & neurological systems. Biological Perspective: Engineering Perspective: • Understanding cerebrovascular regulation in the context of cardiovascular and neurodegenerative pathologies. • Mapping interactions between traditionally distinct physiological systems will lead to improved de novo design of tissues for implantation. • Role of astrocytes in higher brain function and cognition is currently unknown, can we uncover this? • Drug and gene delivery to astrocytes presents challenges specific to this cell type and anatomical setting. • Regulation of functional hyperemia by astrocytes. • Design of drugs to promote astrocytic calcium release increasing cerebral perfusion. • Comprehend mechanics of anesthesia targeting specific regions of the brain inducing different affects. • Improved systems which mimic information transmission in CNS leads to neural engineerin.

How was the experiment performed? Multiphoton imaging – Study electrically non-excitable astrocytes. No electric transmission. 1. Glucose/lactose uptake 2. Uptake of neurotransmitters released during synaptic activity 3. Water/ion homeostasis 4. Maintain blood-brain barrier 5. Photolysis of caged calcium 6. CAGED CALCIUM : Photolyzable chelators (cages)
1fd7be72a96f89757f91bf11ced15f7a.ppt