a2c92003ea9cd0c3a7d680aff43010db.ppt
- Количество слайдов: 45
 ASTM International C. Peter Waegemann Immediate Past Chair Committee E 31 on Health Informatics CEO, Medical Records Institute Chair, Mobile Healthcare Alliance 1
ASTM International C. Peter Waegemann Immediate Past Chair Committee E 31 on Health Informatics CEO, Medical Records Institute Chair, Mobile Healthcare Alliance 1
 My Background in Health Informatics Standards 2006 Immediate past Chair of ASTM E 31 1996 -2001 Chair, ANSI Healthcare Informatics Standards Board 2002 -2006 Chair of ASTM E 31 2000 -2004 Chair of US TAG to ISO TC 215 1992 -1999 Member of ASTM, AMIA, AHIMA, HL 7, IEEE, ABA (HEALTHCARE), MCC, NCPDP, WEDI, ASC X 12, etc. 1998 Founding Father of ISO TC 215 1997 Chair and Founding Father of Corba. Med OMG 1994 Chair: New ASTM Subcommittee on Authenticity of Computer-based Patient Records Chair : Committee for Improving International Relations at the ANSI HISPP International and Regional Subcommittee Appointment by CEN TC 251 as Expert for Project Team 13 Member of the ASTM Executive Committee 1993 Chair: Task Force on Ownership of Health Information at ASTM Subcommittee 31. 17 Privacy, Confidentiality and Access And others such as Board Member of SNOMED International, etc. 2
My Background in Health Informatics Standards 2006 Immediate past Chair of ASTM E 31 1996 -2001 Chair, ANSI Healthcare Informatics Standards Board 2002 -2006 Chair of ASTM E 31 2000 -2004 Chair of US TAG to ISO TC 215 1992 -1999 Member of ASTM, AMIA, AHIMA, HL 7, IEEE, ABA (HEALTHCARE), MCC, NCPDP, WEDI, ASC X 12, etc. 1998 Founding Father of ISO TC 215 1997 Chair and Founding Father of Corba. Med OMG 1994 Chair: New ASTM Subcommittee on Authenticity of Computer-based Patient Records Chair : Committee for Improving International Relations at the ANSI HISPP International and Regional Subcommittee Appointment by CEN TC 251 as Expert for Project Team 13 Member of the ASTM Executive Committee 1993 Chair: Task Force on Ownership of Health Information at ASTM Subcommittee 31. 17 Privacy, Confidentiality and Access And others such as Board Member of SNOMED International, etc. 2
 About ASTM International ASTM Organized in 1898 l Independent, private sector, not-for-profit organization l Provides a management system and administrative framework for development of voluntary, consensus standards and promotion of related knowledge, which demonstrate a high degree of technical quality and global market relevance. l 3
About ASTM International ASTM Organized in 1898 l Independent, private sector, not-for-profit organization l Provides a management system and administrative framework for development of voluntary, consensus standards and promotion of related knowledge, which demonstrate a high degree of technical quality and global market relevance. l 3
 ASTM Serves Multiple Sectors Health Informatics l Metals l Petroleum and Lubricants l Environment l l l – Air l – Soil l – Water Construction Materials l Consumer Products l l Mechanical Testing Plastics Road and Paving Medical and Surgical Materials and Devices Textiles Corrosion of Metals Pharmaceutical Process Analytical Technology 4
ASTM Serves Multiple Sectors Health Informatics l Metals l Petroleum and Lubricants l Environment l l l – Air l – Soil l – Water Construction Materials l Consumer Products l l Mechanical Testing Plastics Road and Paving Medical and Surgical Materials and Devices Textiles Corrosion of Metals Pharmaceutical Process Analytical Technology 4
 ASTM Standards are Voluntary l They are: – Developed voluntarily – Used voluntarily l Mandatory only when: – Cited in a contractual agreement – Referenced by a government body 5
ASTM Standards are Voluntary l They are: – Developed voluntarily – Used voluntarily l Mandatory only when: – Cited in a contractual agreement – Referenced by a government body 5
 The ASTM Structure l 30, 000 Members – 104 countries represented – 90 industry sectors represented l 132 Technical committees l 2, 200 Subcommittees l Thousands of task groups 6
The ASTM Structure l 30, 000 Members – 104 countries represented – 90 industry sectors represented l 132 Technical committees l 2, 200 Subcommittees l Thousands of task groups 6
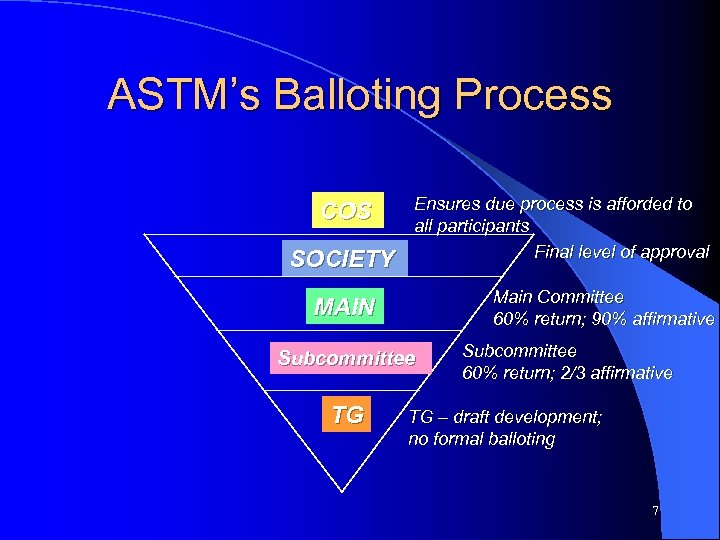 ASTM’s Balloting Process COS SOCIETY Ensures due process is afforded to all participants Final level of approval Main Committee 60% return; 90% affirmative MAIN Subcommittee TG Subcommittee 60% return; 2/3 affirmative TG – draft development; no formal balloting 7
ASTM’s Balloting Process COS SOCIETY Ensures due process is afforded to all participants Final level of approval Main Committee 60% return; 90% affirmative MAIN Subcommittee TG Subcommittee 60% return; 2/3 affirmative TG – draft development; no formal balloting 7
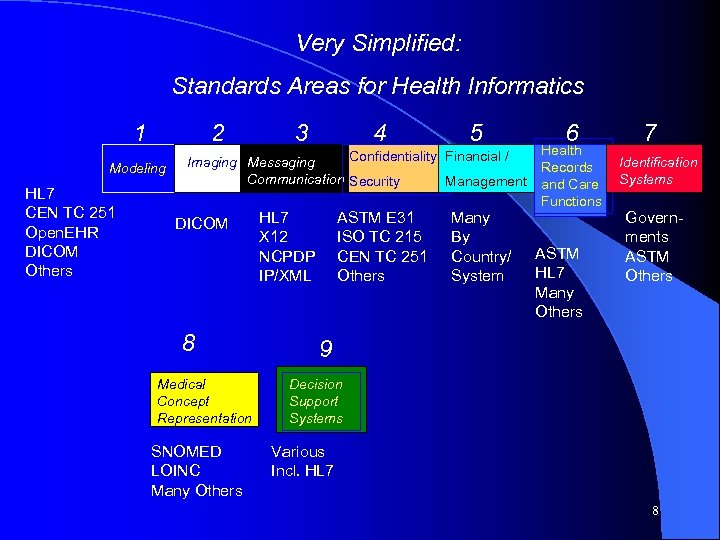 Very Simplified: Standards Areas for Health Informatics 1 2 Modeling HL 7 CEN TC 251 Open. EHR DICOM Others 3 4 5 Confidentiality Financial / Imaging Messaging Communication Security Management DICOM 8 Medical Concept Representation SNOMED LOINC Many Others HL 7 X 12 NCPDP IP/XML ASTM E 31 ISO TC 215 CEN TC 251 Others Many By Country/ System 6 Health Records and Care Functions ASTM HL 7 Many Others 7 Identification Systems Governments ASTM Others 9 Decision Support Systems Various Incl. HL 7 8
Very Simplified: Standards Areas for Health Informatics 1 2 Modeling HL 7 CEN TC 251 Open. EHR DICOM Others 3 4 5 Confidentiality Financial / Imaging Messaging Communication Security Management DICOM 8 Medical Concept Representation SNOMED LOINC Many Others HL 7 X 12 NCPDP IP/XML ASTM E 31 ISO TC 215 CEN TC 251 Others Many By Country/ System 6 Health Records and Care Functions ASTM HL 7 Many Others 7 Identification Systems Governments ASTM Others 9 Decision Support Systems Various Incl. HL 7 8
 Healthcare Informatics Standards Developers ANSI DICOM CEN TC 251 SNOMED ADA NCPDP HL 7 • JCAHO • NCQA • NCCLS • OMG • IHE • CDISC • Mo. HCA • OTHERS • (100+) ? WHO AHIMA HIMSS NLS NIST NAHIT LOINC PHIN Others ISO TC 215 ASTM E 31 ASC X 12 N/EDIFACT IEEE 9
Healthcare Informatics Standards Developers ANSI DICOM CEN TC 251 SNOMED ADA NCPDP HL 7 • JCAHO • NCQA • NCCLS • OMG • IHE • CDISC • Mo. HCA • OTHERS • (100+) ? WHO AHIMA HIMSS NLS NIST NAHIT LOINC PHIN Others ISO TC 215 ASTM E 31 ASC X 12 N/EDIFACT IEEE 9
 International Health Care Standards ISO Rep. of Korea Ecuador TC 215 Australian Standards Czech Republic Israel US Standards British Standards Austrian Standards Dutch Standards French Standards Swedish Standards German Standards South Africa Singapore Russia Switzerland Italian Standards Danish Standards Thailand Norwegian Standards New Zealand Finland Poland Japanese Standards Vietnam Yugoslavia Brazil Denmark Belgium CENT TC 251 Spain 10
International Health Care Standards ISO Rep. of Korea Ecuador TC 215 Australian Standards Czech Republic Israel US Standards British Standards Austrian Standards Dutch Standards French Standards Swedish Standards German Standards South Africa Singapore Russia Switzerland Italian Standards Danish Standards Thailand Norwegian Standards New Zealand Finland Poland Japanese Standards Vietnam Yugoslavia Brazil Denmark Belgium CENT TC 251 Spain 10
 Why Has There Been So Little Success? 11
Why Has There Been So Little Success? 11
 1. Information Content l Inconsistencies l Different Cultural Aspects l CCR Standards Needs Content Agreed Upon by Medical Specialties EHR Taught by Medical Schools Implemented by Software Vendors 12
1. Information Content l Inconsistencies l Different Cultural Aspects l CCR Standards Needs Content Agreed Upon by Medical Specialties EHR Taught by Medical Schools Implemented by Software Vendors 12
 Everything or Just Relevant Information? l Summary l Referral Data Set l Management-Specific Information 13
Everything or Just Relevant Information? l Summary l Referral Data Set l Management-Specific Information 13
 2. Information Capture l How to get information into the computer? l How to get physicians to use computers in the exam room? l Complex issues EHR 14
2. Information Capture l How to get information into the computer? l How to get physicians to use computers in the exam room? l Complex issues EHR 14
 Is Documentation Like This Acceptable Anywhere Else Than in Health Care? • Legibility • Structure • Meaning Method of 15 Documentation
Is Documentation Like This Acceptable Anywhere Else Than in Health Care? • Legibility • Structure • Meaning Method of 15 Documentation
 ASTM E 31 Standards (cont. ) l E 31. 22 Health Information Transcription and Documentation – Scope: To develop standards for the systems, processes, and management of medical transcription and its integration with other modalities of report generation. l E 31. 22 Standards · E 1902 Standard Guide for Management of the Confidentiality and Security of Dictation, Transcription, and Transcribed Health Records · E 1959 Standard Guide for Requests for Proposals Regarding Medical Transcription Services for Healthcare Institutions · E 2185 Standard Specification for Transferring Digital Voice Data Between Independent Digital Dictation Systems and Workstations · E 2117 Standard Guide for the Identification and Establishment of a Quality Assurance Program for Medical Transcription · E 2364 Standard Guide to Speech Recognition Technology Products in Health Care · E 2344 Standard Guide for Data Capture Through the Dictation Process 16
ASTM E 31 Standards (cont. ) l E 31. 22 Health Information Transcription and Documentation – Scope: To develop standards for the systems, processes, and management of medical transcription and its integration with other modalities of report generation. l E 31. 22 Standards · E 1902 Standard Guide for Management of the Confidentiality and Security of Dictation, Transcription, and Transcribed Health Records · E 1959 Standard Guide for Requests for Proposals Regarding Medical Transcription Services for Healthcare Institutions · E 2185 Standard Specification for Transferring Digital Voice Data Between Independent Digital Dictation Systems and Workstations · E 2117 Standard Guide for the Identification and Establishment of a Quality Assurance Program for Medical Transcription · E 2364 Standard Guide to Speech Recognition Technology Products in Health Care · E 2344 Standard Guide for Data Capture Through the Dictation Process 16
 3. Information Representation l l Inconsistent Meaning of Text Different Code Sets Lack of standards Reimbursement Code Sets – CPT – ICD 9 CM l Clinical Code sets – SNOMED – LOINC – Many Others EHR 17
3. Information Representation l l Inconsistent Meaning of Text Different Code Sets Lack of standards Reimbursement Code Sets – CPT – ICD 9 CM l Clinical Code sets – SNOMED – LOINC – Many Others EHR 17
 Unless a Coherent Framework of Terminology is Used, Interoperability Cannot be Achieved l Framework of Terminology That Allows Mapping to Each Vocabulary or Code Set l Ontology-based Web Language (OWL) 18
Unless a Coherent Framework of Terminology is Used, Interoperability Cannot be Achieved l Framework of Terminology That Allows Mapping to Each Vocabulary or Code Set l Ontology-based Web Language (OWL) 18
 Toward Greater Clinical Specificity HL 7 begins to specify code sets for certain message fields l HL 7 Vocabulary SIG Recommending Code Sets for OBX Segment: l l l LOINC for observation identifier fields SNOMED for use in the value field Should the National Library of Medicine (NLM) include HL 7 codes in the UMLS? Coordinating the development of a common drug code model with several drug code developers 19
Toward Greater Clinical Specificity HL 7 begins to specify code sets for certain message fields l HL 7 Vocabulary SIG Recommending Code Sets for OBX Segment: l l l LOINC for observation identifier fields SNOMED for use in the value field Should the National Library of Medicine (NLM) include HL 7 codes in the UMLS? Coordinating the development of a common drug code model with several drug code developers 19
 Code Sets l Code Sets becoming more structured and stable – SNOMED’s Reference Terminology Developed l Concept-based hierarchies created l Stable foundation provided – Code Sets converging with SNOMED l ADA Micro-glossary l DICOM Micro-glossary l LOINC Micro-glossary l NANDA Micro-glossary l Others – The NLM’s UMLS becomes a meta-thesaurus 20
Code Sets l Code Sets becoming more structured and stable – SNOMED’s Reference Terminology Developed l Concept-based hierarchies created l Stable foundation provided – Code Sets converging with SNOMED l ADA Micro-glossary l DICOM Micro-glossary l LOINC Micro-glossary l NANDA Micro-glossary l Others – The NLM’s UMLS becomes a meta-thesaurus 20
 Codes and Code Sets (including issues of maintenance) ¶ World Health Organization (WHO) – International Classification of Diseases, Ninth Revision (ICD-9) – International Classification of Diseases, Tenth Revision (ICD-10) · CMS and the National Center for Health Statistics (NCHS) – International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9 -CM) – International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10 -CM) – International Classification of Diseases (ICD) – Disease Classification (code set) ¸ American Medical Association (AMA) – Physicians’ Current Procedural Terminology (CPT) 21
Codes and Code Sets (including issues of maintenance) ¶ World Health Organization (WHO) – International Classification of Diseases, Ninth Revision (ICD-9) – International Classification of Diseases, Tenth Revision (ICD-10) · CMS and the National Center for Health Statistics (NCHS) – International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9 -CM) – International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10 -CM) – International Classification of Diseases (ICD) – Disease Classification (code set) ¸ American Medical Association (AMA) – Physicians’ Current Procedural Terminology (CPT) 21
 Codes and Code Sets (continued. . ) ¹ College of American Pathologists – Systematized Nomenclature of Human and Veterinary Medicine (SNOMED) International º American Dental Association (ADA) – Current Dental Terminology (CDT) » Advisory Committee on Dental Electronic Nomenclature Indexing and Classification (ACODENIC) – Microglossary of SNOMED for Dentistry ¼ Center for Nursing Classification, University of Iowa College of Nursing Interventions Classification (NIC) ½ International Conference on Harmonization – International Medical Terminology (IMT) 22
Codes and Code Sets (continued. . ) ¹ College of American Pathologists – Systematized Nomenclature of Human and Veterinary Medicine (SNOMED) International º American Dental Association (ADA) – Current Dental Terminology (CDT) » Advisory Committee on Dental Electronic Nomenclature Indexing and Classification (ACODENIC) – Microglossary of SNOMED for Dentistry ¼ Center for Nursing Classification, University of Iowa College of Nursing Interventions Classification (NIC) ½ International Conference on Harmonization – International Medical Terminology (IMT) 22
 Codes and Code Sets (continued. . ) ¾ Health Care Claim Adjustment Reason Code/Health Care Claim Status Code Committee – Health Care Claim Adjustment Reason Codes – Health Care Claim Status Codes ¿ Logical Observation Identifier Names and Codes (LOINC) Consortium – Logical Observation Identifier Names and Codes (LOINC) è Georgetown University Home Care Project – Home Health Care Classification (HHCC) System è Perspective on Code Sets Within Transaction Standards 23
Codes and Code Sets (continued. . ) ¾ Health Care Claim Adjustment Reason Code/Health Care Claim Status Code Committee – Health Care Claim Adjustment Reason Codes – Health Care Claim Status Codes ¿ Logical Observation Identifier Names and Codes (LOINC) Consortium – Logical Observation Identifier Names and Codes (LOINC) è Georgetown University Home Care Project – Home Health Care Classification (HHCC) System è Perspective on Code Sets Within Transaction Standards 23
 ASTM Standards l l l E 31. 01 Controlled Vocabularies for Healthcare Informatics Chair: Peter Elkin (elkin. peter@mayo. edu) Scope: 1, Standardize existing High Level principles for the contents and structure of Controlled Health Vocabularies. 2. Develop a description and comparison of existing formalisms for health concept representation. 3. Develop a standard formalism for Controlled Health Vocabularies. a. This implies a natural ordering of the terminology from its formal definitions. b. The standard formalism must abide by the rules established in Task #1. 4. Work toward a standard model for vocabulary evolution, maintenance, and distribution. 5. Work to develop mechanisms to facilitate international use of common underlying formal structures for Controlled Health Vocabularies E 31. 01 Standards · E 1284 Standard Guideline for Construction of a Clinical Nomenclature for the Support of Electronic Health Records 24
ASTM Standards l l l E 31. 01 Controlled Vocabularies for Healthcare Informatics Chair: Peter Elkin (elkin. peter@mayo. edu) Scope: 1, Standardize existing High Level principles for the contents and structure of Controlled Health Vocabularies. 2. Develop a description and comparison of existing formalisms for health concept representation. 3. Develop a standard formalism for Controlled Health Vocabularies. a. This implies a natural ordering of the terminology from its formal definitions. b. The standard formalism must abide by the rules established in Task #1. 4. Work toward a standard model for vocabulary evolution, maintenance, and distribution. 5. Work to develop mechanisms to facilitate international use of common underlying formal structures for Controlled Health Vocabularies E 31. 01 Standards · E 1284 Standard Guideline for Construction of a Clinical Nomenclature for the Support of Electronic Health Records 24
 4. Data Models and Operational Conformity In order to achieve interoperability, a standardized model must be applied to as well as a standardized data model l Current competing models are RIM, FAM, GEHR (Open. EHR), etc. l CDA l EHR 25
4. Data Models and Operational Conformity In order to achieve interoperability, a standardized model must be applied to as well as a standardized data model l Current competing models are RIM, FAM, GEHR (Open. EHR), etc. l CDA l EHR 25
 ASTM’s View 1990 -1998 Messaging 1998 -2005 From Messages to Documents 2005 From Documents to Authenticated Data 26
ASTM’s View 1990 -1998 Messaging 1998 -2005 From Messages to Documents 2005 From Documents to Authenticated Data 26
 5. Clinical Practice l Integrating Guidelines and Protocols l Disease Management l Pathways l Software and Patient Management EHR 27
5. Clinical Practice l Integrating Guidelines and Protocols l Disease Management l Pathways l Software and Patient Management EHR 27
 6. Decision Support l Standardized Decision Support – Admission Systems – Eligibility – Diagnostic Support – Order Entry and Test EHR Results – Etc. 28
6. Decision Support l Standardized Decision Support – Admission Systems – Eligibility – Diagnostic Support – Order Entry and Test EHR Results – Etc. 28
 7. Confidentiality/Security l General Security l Authentication l Data Integrity l Accessibility l Auditability EHR 29
7. Confidentiality/Security l General Security l Authentication l Data Integrity l Accessibility l Auditability EHR 29
 Security Standards l US HIPAA l ASTM E 31 Standards l ISO TC 215 PKI Standard l CEN TC 251 Security Standards 30
Security Standards l US HIPAA l ASTM E 31 Standards l ISO TC 215 PKI Standard l CEN TC 251 Security Standards 30
 Signature Standards l Different in each Country: – US – UK – Germany – Australia – Sweden 31
Signature Standards l Different in each Country: – US – UK – Germany – Australia – Sweden 31
 Security, Safeguards and Electronic Signatures ASTM E 31 Standards Committee on Healthcare Informatics l ACR NEMA / DICOM l Accredited Standards Committee (ASC) X 12 l CEN TC 251 Working Group 6 on Security, Privacy, Quality and Safety l Health Level Seven (HL-7) l IEEE l National Council for Prescription Drug Programs (NCPDP) l 32
Security, Safeguards and Electronic Signatures ASTM E 31 Standards Committee on Healthcare Informatics l ACR NEMA / DICOM l Accredited Standards Committee (ASC) X 12 l CEN TC 251 Working Group 6 on Security, Privacy, Quality and Safety l Health Level Seven (HL-7) l IEEE l National Council for Prescription Drug Programs (NCPDP) l 32
 ASTM E 31 Standards (cont. ) l l E 31. 17 Privacy, Confidentiality and Access Scope: To develop standards that address access, privacy, confidentiality and data security of health information in its many forms and locations. l E 31. 17 Standards · E 1869 Guide for Confidentiality, Privacy, Access and Data Security Principles for Health Information Including Computer Based Patient Records · E 1986 Standard Guide for Information Access Privileges to Health Information · E 1987 Standard Guide for Individual Rights Regarding Health Information · E 1988 Standard Guide for the Training Persons Who Have Access to Health Information · PS 115 Provisional Standard Specification for Security Audit and Disclosure Logs for Use in Health Information Systems · PS 105 Provisional Standard Guide for Amendments to Health Information l Standards Under Development · Draft Standard for Utilization and Retention of Encrypted Signature Certificates 33
ASTM E 31 Standards (cont. ) l l E 31. 17 Privacy, Confidentiality and Access Scope: To develop standards that address access, privacy, confidentiality and data security of health information in its many forms and locations. l E 31. 17 Standards · E 1869 Guide for Confidentiality, Privacy, Access and Data Security Principles for Health Information Including Computer Based Patient Records · E 1986 Standard Guide for Information Access Privileges to Health Information · E 1987 Standard Guide for Individual Rights Regarding Health Information · E 1988 Standard Guide for the Training Persons Who Have Access to Health Information · PS 115 Provisional Standard Specification for Security Audit and Disclosure Logs for Use in Health Information Systems · PS 105 Provisional Standard Guide for Amendments to Health Information l Standards Under Development · Draft Standard for Utilization and Retention of Encrypted Signature Certificates 33
 ASTM E 31 Standards (cont. ) l l E 31. 20 Data and System Security for Health Information Scope: To develop standards addressing security of health information data and systems and the process for authentication in computer-based patient records systems. l E 31. 20 Standards · E 1714 Standard Guide for the Properties of a Universal Healthcare Identifier (UHID) · E 1762 Standard Guide for Electronic Authentication of Health Care Information · E 1985 Standard Guide for User Authentication and Authorization · PS 100 Provisional Standard Specification for Authentication of Healthcare Information Using Digital Signatures · PS 101 Provisional Standard Guideline on a Security Framework for Healthcare Information · PS 102 Provisional Standard Guide for Internet and Intranet Security l Standards Under Development · Draft Standard Specification for Transmission of Healthcare Information Using Secure Messaging Protocols · Draft Standard for Data, System, Network and Device Integrity, Security, Availability, Reliability and Permanence 34
ASTM E 31 Standards (cont. ) l l E 31. 20 Data and System Security for Health Information Scope: To develop standards addressing security of health information data and systems and the process for authentication in computer-based patient records systems. l E 31. 20 Standards · E 1714 Standard Guide for the Properties of a Universal Healthcare Identifier (UHID) · E 1762 Standard Guide for Electronic Authentication of Health Care Information · E 1985 Standard Guide for User Authentication and Authorization · PS 100 Provisional Standard Specification for Authentication of Healthcare Information Using Digital Signatures · PS 101 Provisional Standard Guideline on a Security Framework for Healthcare Information · PS 102 Provisional Standard Guide for Internet and Intranet Security l Standards Under Development · Draft Standard Specification for Transmission of Healthcare Information Using Secure Messaging Protocols · Draft Standard for Data, System, Network and Device Integrity, Security, Availability, Reliability and Permanence 34
 8. Performance l The most-overlooked criterion – Affects Selection of Systems No Standards exist. EHR 35
8. Performance l The most-overlooked criterion – Affects Selection of Systems No Standards exist. EHR 35
 Reliability No Standards 36
Reliability No Standards 36
 9. Technical Interoperability l Which interoperability system will succeed in health care? – OSI – Microsoft EHR – CORBAmed – GEHR/Open. EHR – HL 7 – Generic Internet: XML with Ontology 37
9. Technical Interoperability l Which interoperability system will succeed in health care? – OSI – Microsoft EHR – CORBAmed – GEHR/Open. EHR – HL 7 – Generic Internet: XML with Ontology 37
 Continuity of Care Record Standard l. A core data set of the most relevant current and past information about a patient’s health status and healthcare treatment l Organized and transportable l Prepared by a practitioner at the conclusion of a healthcare encounter l Enables the next practitioner to readily access such information 38
Continuity of Care Record Standard l. A core data set of the most relevant current and past information about a patient’s health status and healthcare treatment l Organized and transportable l Prepared by a practitioner at the conclusion of a healthcare encounter l Enables the next practitioner to readily access such information 38
 Unique Standards Development Effort Consortium of sponsoring organizations l l l ASTM International E 31 Health Informatics Committee Massachusetts Medical Society HIMSS American Academy of Family Physicians American Academy of Pediatrics American Medical Association Patient Safety Institute American Health Care Association National Association for the Support of LTC Mobile Healthcare Alliance (Mo. HCA) Medical Group Management Association American Academy of Osteopathic Family Physicians 39
Unique Standards Development Effort Consortium of sponsoring organizations l l l ASTM International E 31 Health Informatics Committee Massachusetts Medical Society HIMSS American Academy of Family Physicians American Academy of Pediatrics American Medical Association Patient Safety Institute American Health Care Association National Association for the Support of LTC Mobile Healthcare Alliance (Mo. HCA) Medical Group Management Association American Academy of Osteopathic Family Physicians 39
 Sponsors represent… l l l l ANSI-recognized standards development organization Over 500, 000 practitioners Over 13, 000 IT professionals Over 19, 000 managers of over 11, 000 organizations in which 240, 000 physicians practice Over 12, 000 institutions in the long-term care community providing care to over 1. 5 million elderly and disabled Major stakeholders in m-Health Patients, patient advocates, data sources, 40 corporations, provider institutions….
Sponsors represent… l l l l ANSI-recognized standards development organization Over 500, 000 practitioners Over 13, 000 IT professionals Over 19, 000 managers of over 11, 000 organizations in which 240, 000 physicians practice Over 12, 000 institutions in the long-term care community providing care to over 1. 5 million elderly and disabled Major stakeholders in m-Health Patients, patient advocates, data sources, 40 corporations, provider institutions….
 CCR Body l Patient administrative and clinical data sections – Payers – Advance Directives – Medical Equipment – Support – Immunizations – Functional Status – Vital Signs – Problems – Results – Family History – Procedures – Social History – Encounters – Alerts – Plan of Care – Medications – Healthcare Providers 41
CCR Body l Patient administrative and clinical data sections – Payers – Advance Directives – Medical Equipment – Support – Immunizations – Functional Status – Vital Signs – Problems – Results – Family History – Procedures – Social History – Encounters – Alerts – Plan of Care – Medications – Healthcare Providers 41
 ASTM E 31 Evolution l Physician-driven l Sponsor opportunities l Practical interoperability – vendor involvement l Involved in Re-organization l Involved in Harmonization l International opportunities 42
ASTM E 31 Evolution l Physician-driven l Sponsor opportunities l Practical interoperability – vendor involvement l Involved in Re-organization l Involved in Harmonization l International opportunities 42
 On The Side. . . l Practical Use of CCR for Consumer Empowerment by Mo. HCA – Consumer Health Manager 43
On The Side. . . l Practical Use of CCR for Consumer Empowerment by Mo. HCA – Consumer Health Manager 43
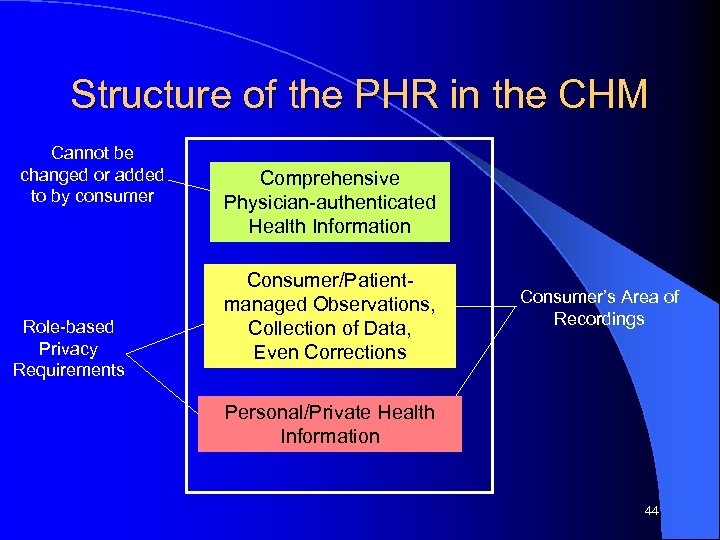 Structure of the PHR in the CHM Cannot be changed or added to by consumer Role-based Privacy Requirements Comprehensive Physician-authenticated Health Information Consumer/Patientmanaged Observations, Collection of Data, Even Corrections Consumer’s Area of Recordings Personal/Private Health Information 44
Structure of the PHR in the CHM Cannot be changed or added to by consumer Role-based Privacy Requirements Comprehensive Physician-authenticated Health Information Consumer/Patientmanaged Observations, Collection of Data, Even Corrections Consumer’s Area of Recordings Personal/Private Health Information 44
 Thank You Copies of these slides may be obtained from peter@medrecinst. com 45
Thank You Copies of these slides may be obtained from peter@medrecinst. com 45


