Microsoft_PowerPoint-Pr_228_sentation_neu (2).pptx
- Количество слайдов: 27

Assessing the immunogenicity of various cells, derived by differentiation of the ROSA 26 OVA-expressing murine i. PSCs Arseniy Petrov UMG
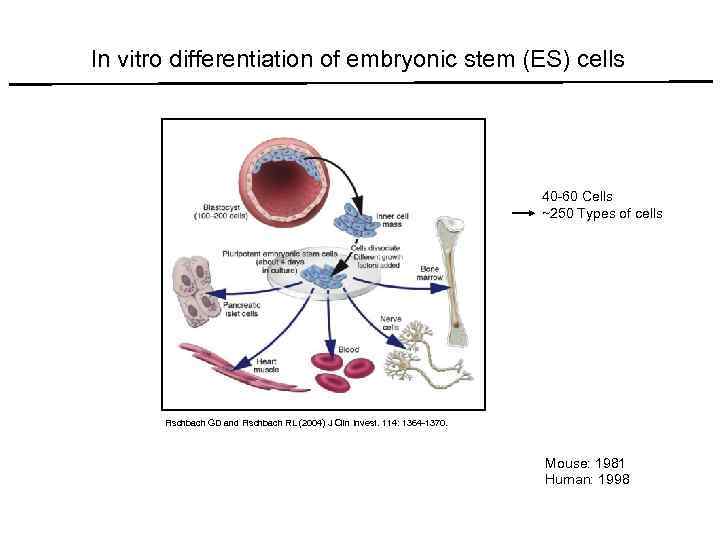
In vitro differentiation of embryonic stem (ES) cells 40 -60 Cells ~250 Types of cells Fischbach GD and Fischbach RL (2004) J Clin Invest. 114: 1364 -1370. Mouse: 1981 Human: 1998

Problems of ESCs in clinical application • The use of ESCs is ethically and legally restricted • ESCs can be used only in an allogeneic setting • ESCs can give rise to teratomas
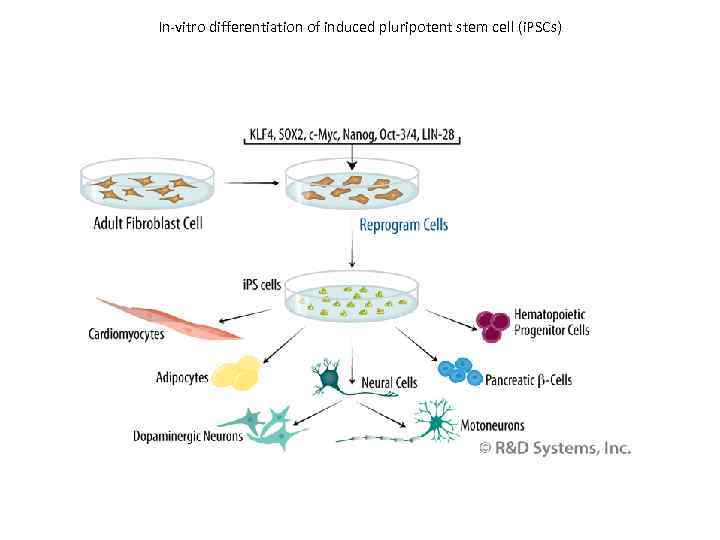
In-vitro differentiation of induced pluripotent stem cell (i. PSCs)

Major obstacles towards usage of the i. PSCs • Tumorigenicity and • Immunogenicity of the i. PSC-derived cells

Background of the study • Many studies suggest that certain cells derived from i. PSCs might be immunogenic in response to autologous or syngeneic immune system. • Therefore, it is critical to establish a physiologically relevant system to evaluate the immunogenicity of the cells derived from i. PSCs • Doktor Monecke has been addressing both problems during his Ph. D-thesis

Results obtained by doctor Monecke In order to analyze the ability of murine PSCs to process and present intracellular antigens, an Ovalbumin (OVA) expression construct was introduced into different PSC lines and as control in RMA cells (murine T-cell lymphoma line) q. RT-PCR and Western blot analysis revealed that the RMA OVA clone expressed higher amounts of the OVA-e. GFP transgene than the majority of tested PSC clones
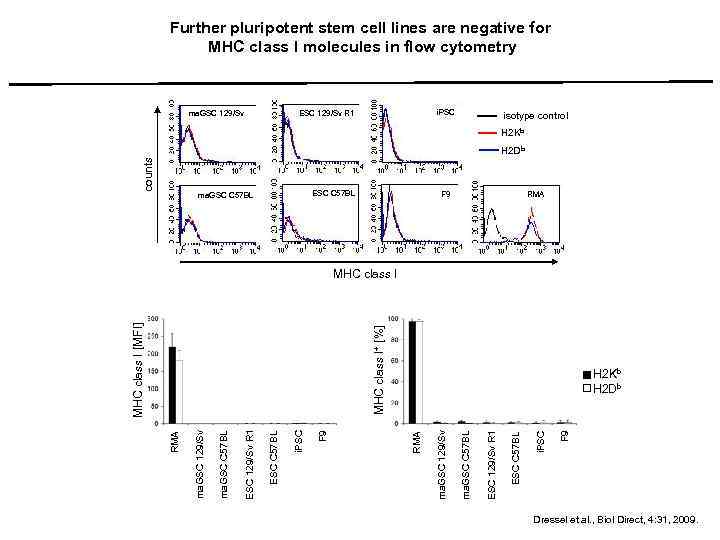
Further pluripotent stem cell lines are negative for MHC class I molecules in flow cytometry i. PSC ESC 129/Sv R 1 ma. GSC 129/Sv isotype control H 2 Kb counts H 2 Db ESC C 57 BL ma. GSC C 57 BL F 9 RMA F 9 i. PSC ESC C 57 BL ESC 129/Sv R 1 ma. GSC C 57 BL ma. GSC 129/Sv H 2 Kb H 2 Db RMA F 9 i. PSC ESC C 57 BL ESC 129/Sv R 1 ma. GSC C 57 BL ma. GSC 129/Sv RMA MHC class I+ [%] MHC class I [MFI] MHC class I Dressel et al. , Biol Direct, 4: 31, 2009.
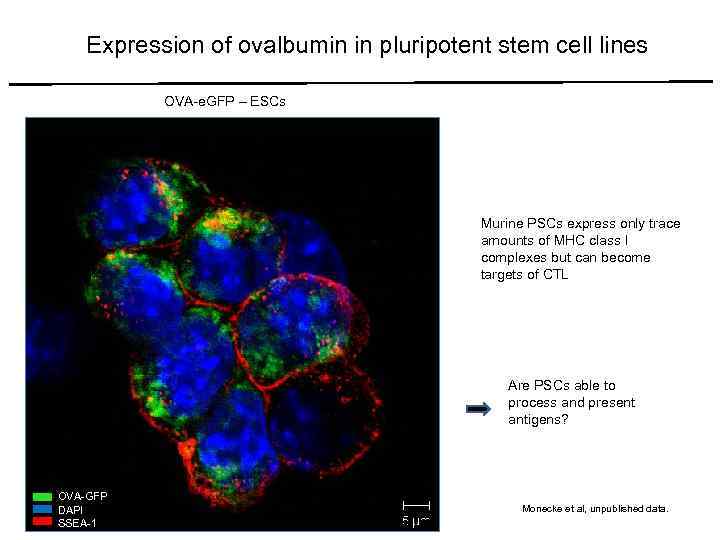
Expression of ovalbumin in pluripotent stem cell lines OVA-e. GFP – ESCs Murine PSCs express only trace amounts of MHC class I complexes but can become targets of CTL Are PSCs able to process and present antigens? OVA-GFP DAPI SSEA-1 Monecke et al, unpublished data.
![relative lysis [%] Susceptibility of OVA-expressing i. PSCs to peptide-specific CTL effector : target relative lysis [%] Susceptibility of OVA-expressing i. PSCs to peptide-specific CTL effector : target](https://present5.com/presentation/62736254_437076575/image-10.jpg)
relative lysis [%] Susceptibility of OVA-expressing i. PSCs to peptide-specific CTL effector : target ratio Monecke et al, unpublished data.
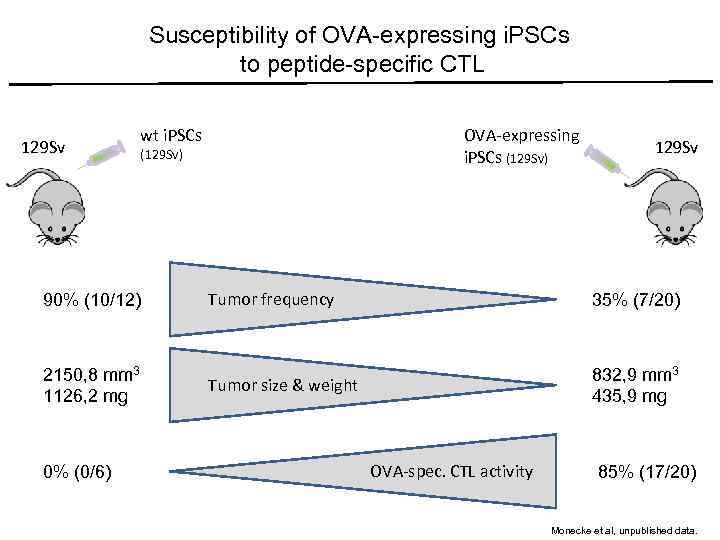
Susceptibility of OVA-expressing i. PSCs to peptide-specific CTL 129 Sv wt i. PSCs (129 Sv) 90% (10/12) 2150, 8 mm 3 1126, 2 mg 0% (0/6) OVA-expressing i. PSCs (129 Sv) 129 Sv Tumor frequency 35% (7/20) Tumor size & weight 832, 9 mm 3 435, 9 mg OVA-spec. CTL activity 85% (17/20) Monecke et al, unpublished data.
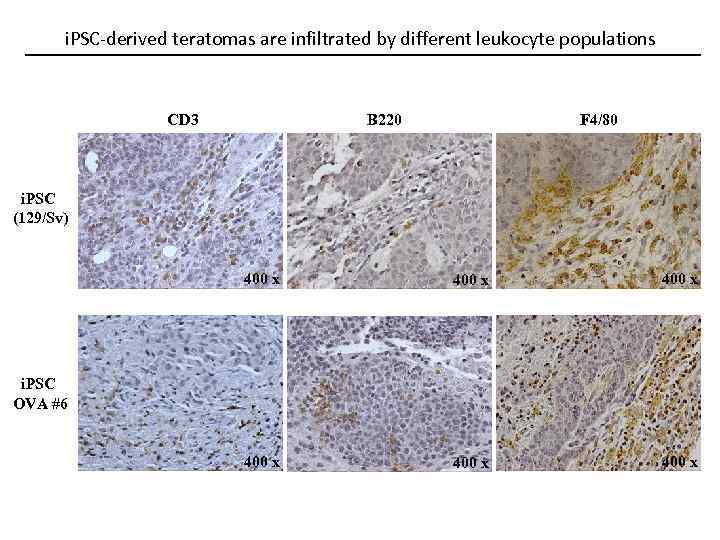
i. PSC-derived teratomas are infiltrated by different leukocyte populations CD 3 B 220 F 4/80 i. PSC (129/Sv) 400 x 400 x i. PSC OVA #6
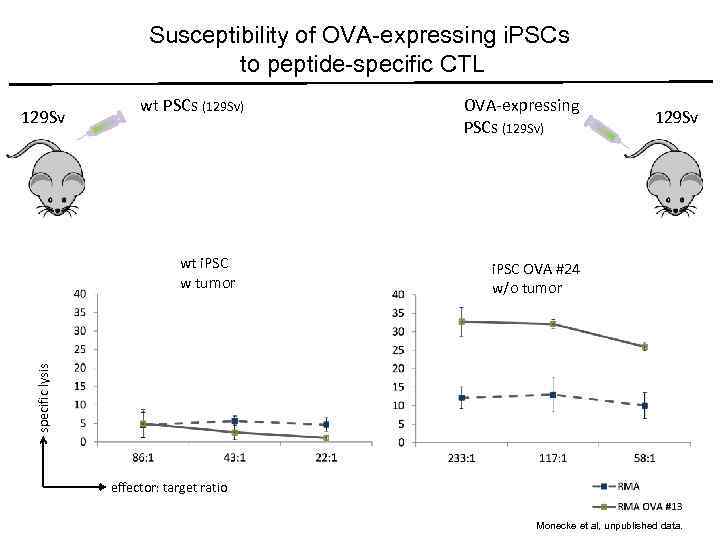
Susceptibility of OVA-expressing i. PSCs to peptide-specific CTL 129 Sv wt PSCs (129 Sv) 129 Sv i. PSC OVA #24 w/o tumor specific lysis wt i. PSC w tumor OVA-expressing PSCs (129 Sv) effector: target ratio Monecke et al, unpublished data.
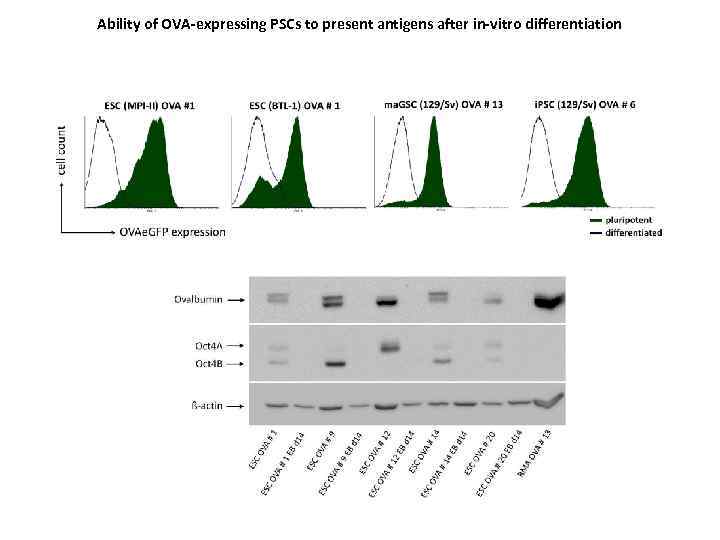
Ability of OVA-expressing PSCs to present antigens after in-vitro differentiation
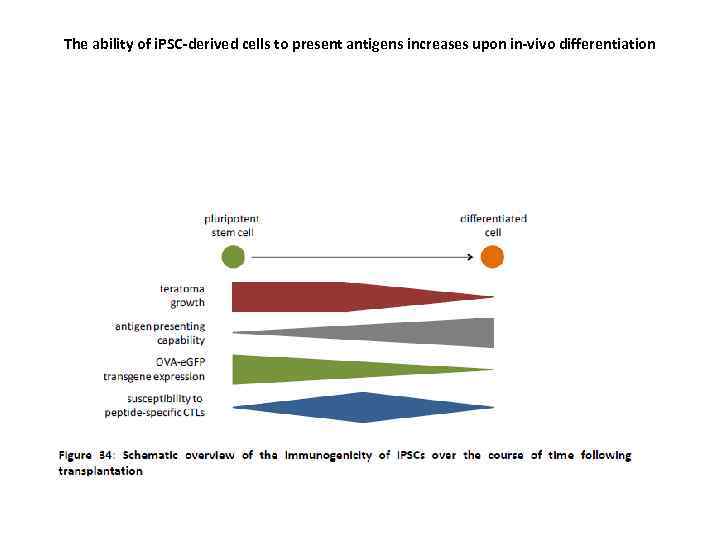
The ability of i. PSC-derived cells to present antigens increases upon in-vivo differentiation

Conclusions of doctor Monecke’s study • Pluripotent stem cells (PSCs) can be killed by activated CTL despite low MHC class I expression • PSCs are defective in antigen processing • PSCs inhibit the activation of CTL in vitro • In vivo, CTL become activated against an antigen expressed in PSCs • A single mismatched antigen can impair the engraftment of PSCs • Impairment of engraftment correlates with the antigen-specific CTL activity not with the antigen-specfic antibody response

Objectives of current Study and Hypotheses to be tested • • Basing on previous work done, OVA has been proven to represent a suitable model– antigen for studying immunogenicity of the i. PSCs But we still do not have a suitable model, which would allow ubiquitous expression of the model antigen in the differentiated tissues. The present task is to create such a model with stable OVA-transgene expression, retained upon in-vitro differentiation, allowing to perform various functional immunogenicity tests. ROSA 26 i. PSC´s with OVA-e. GFP transgene isnserted by homologous recombination, might represent a potential model for transgene expression upon differentiation, because targeting of genes to the ROSA 26 locus has been shown to achieve ubiquitous expression during development or in the adult. (1). And this model has been used for Studying Cellular (De)Differentiation(2) • 1. Generalized lac. Z expression with the ROSA 26 Cre reporter strain. Soriano P. • 2. The ROSA 26 -i. PSC Mouse: A Conditional, Inducible, and Exchangeable Resourcefor Studying Cellular (De)Differentiation. Lieven Haenebalcke, 1, 2 Steven Goossens, 1, 2 Pieterjan Dierickx, 1, 2 Sonia Bartunkova, 1, 2 Jinke D’Hont, 1, 2 Katharina Haigh, 1, 2 Tino Hochepied, 2 Dagmar Wirth, 3 Andras Nagy, 4 and Jody J. Haigh 1, 2, *)
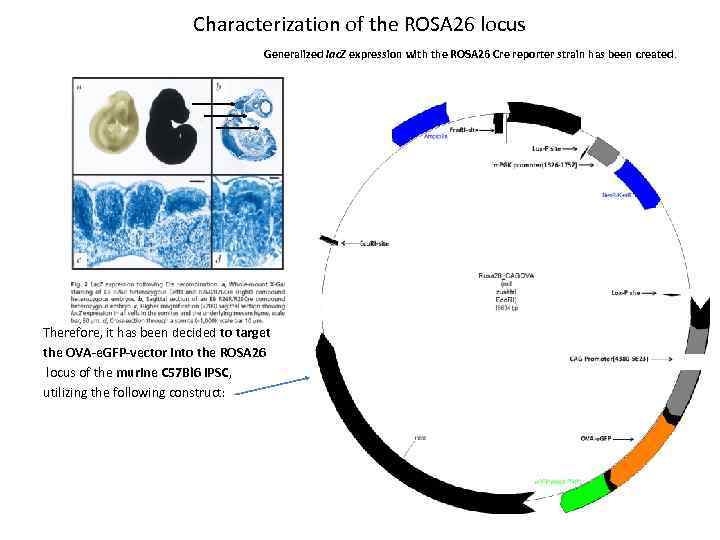
Characterization of the ROSA 26 locus Generalized lac. Z expression with the ROSA 26 Cre reporter strain has been created. Therefore, it has been decided to target the OVA-e. GFP-vector into the ROSA 26 locus of the murine C 57 Bl 6 i. PSC, utilizing the following construct:
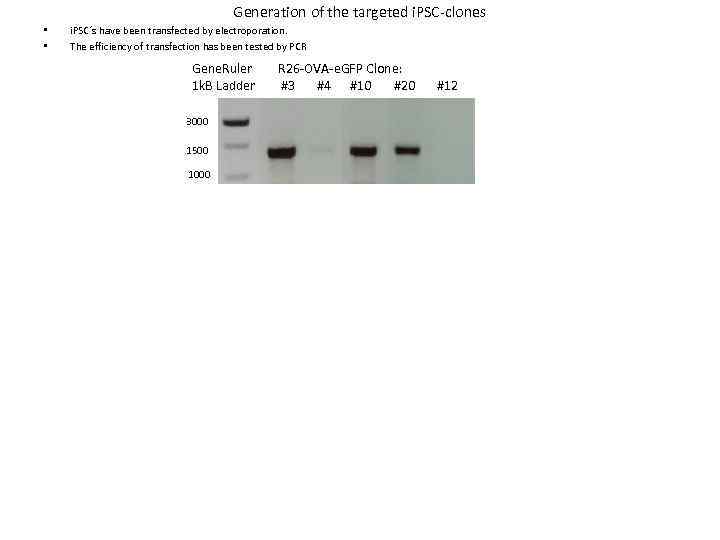
Generation of the targeted i. PSC-clones • • i. PSC´s have been transfected by electroporation. The efficiency of transfection has been tested by PCR Gene. Ruler 1 k. B Ladder 3000 1500 1000 R 26 -OVA-e. GFP Clone: #3 #4 #10 #20 #12
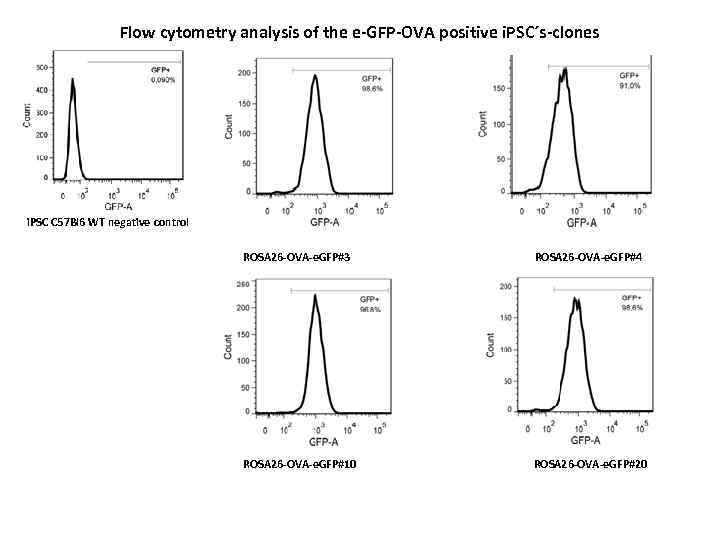
Flow cytometry analysis of the e-GFP-OVA positive i. PSC´s-clones i. PSC C 57 Bl 6 WT negative control ROSA 26 -OVA-e. GFP#3 ROSA 26 -OVA-e. GFP#4 ROSA 26 -OVA-e. GFP#10 ROSA 26 -OVA-e. GFP#20
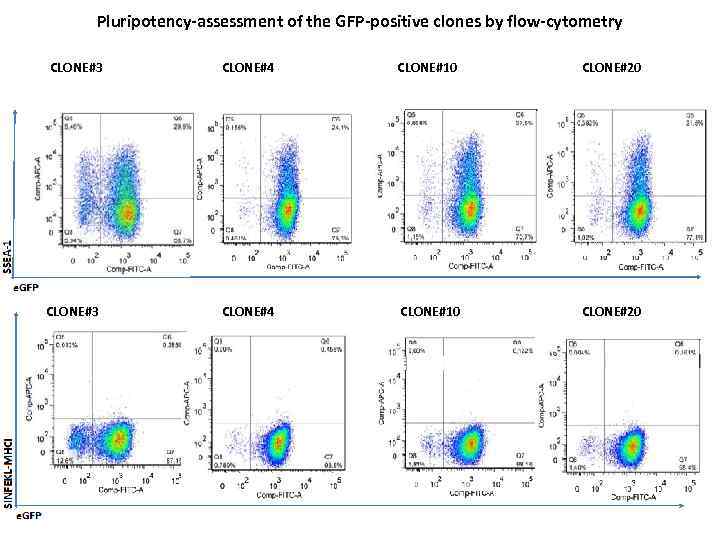
Pluripotency-assessment of the GFP-positive clones by flow-cytometry CLONE#3 CLONE#4 CLONE#10 CLONE#20

Conclusions regarding pluripotency • Inconsistent data of flow-cytometry experiments: despite large populations of cells are SSEA-1 negative, • Cells are able to form teratomas upon transplantation into SCID-mice (data not included) • and differentiate into various cells in-vitro, as shown later

In-vitro undirected differentiation Western blot analysis of the EB-clones and their parental i. PSC-clones
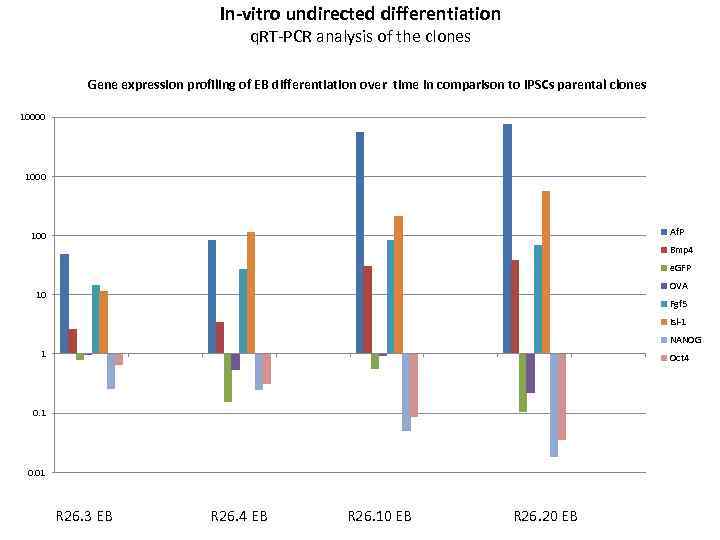
In-vitro undirected differentiation q. RT-PCR analysis of the clones Gene expression profiling of EB differentiation over time in comparison to i. PSCs parental clones 10000 1000 Af. P 100 Bmp 4 e. GFP OVA 10 Fgf 5 Isl-1 NANOG 1 Oct 4 0. 1 0. 01 R 26. 3 EB R 26. 4 EB R 26. 10 EB R 26. 20 EB
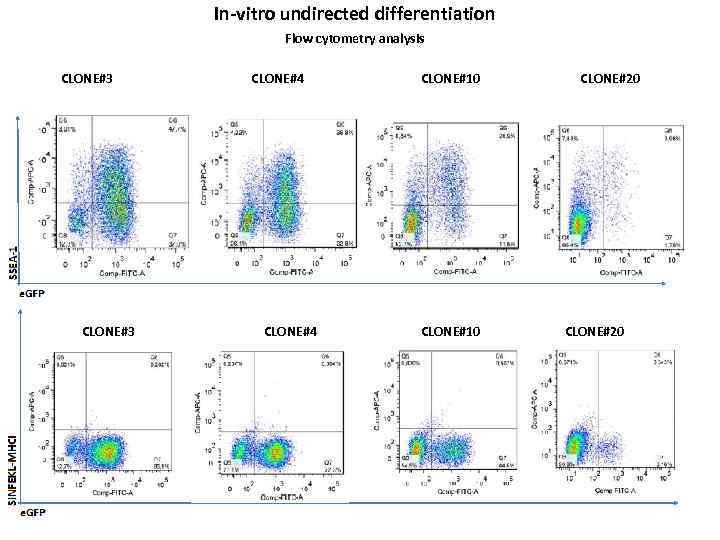
In-vitro undirected differentiation Flow cytometry analysis CLONE#3 CLONE#4 CLONE#10 CLONE#20

Conclusions and plans • • We hope to obtain consistent results in the upcoming differentiation experiments, with unambiguous data of Western blot analyses, flow-cytometry and PCR-expression profiles. In order to elucidate how transgene expression is being altered in course of differentiation, further differentiation experiments will be performed, with samples collected at different time-points during differentiation, allowing cells to acchieve more mature state. We seek to prove, that R 26 -i. PSCs will retain transgene expression to some substantial extent, rendering them useful for immunogenicity tests. But one should consider, that there are reports, describing that the incorporation of exogenous promoters to the ROSA 26 locus are prone to silencing effects. [1, 2, 3, 4, 5] It could explain the massive reduction of transgene-expression we observed in differentiated clones. 1. Nyabi, O. , Naessens, M. , Haigh, K. , et al. (2009). 2. Haenebalcke, L. , Goossens, S. , Dierickx, P. , et al. (2013). 3. Strathdee, D. , Ibbotson, H. , & Grant, S. G. (2006). 4. Chen, C. M. , Krohn, J. , Bhattacharya, S. , & Davies, B. (2011). 5. Tchorz, J. S. , Suply, T. , Ksiazek, I. , et al. (2012).

Microsoft_PowerPoint-Pr_228_sentation_neu (2).pptx