3c98722ce375cad2fad5f919a576c3fc.ppt
- Количество слайдов: 23

ASAQ, a new fixed-dose combination of artesunateamodiaquine: progress and challenges Ann-Marie Sevcsik on behalf of Graciela Diap, Jean-Rene Kiechel & FACT Partners MSF-UK Scientific Day London, June 2007
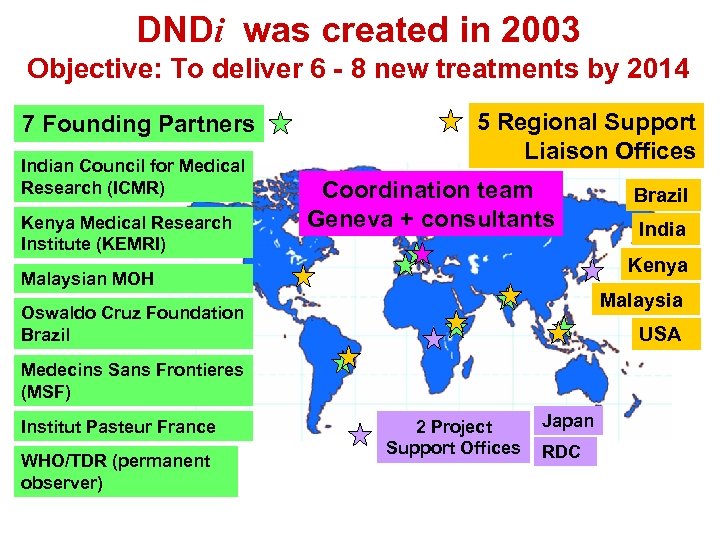
DNDi was created in 2003 Objective: To deliver 6 - 8 new treatments by 2014 7 Founding Partners Indian Council for Medical Research (ICMR) Kenya Medical Research Institute (KEMRI) 5 Regional Support Liaison Offices Coordination team Geneva + consultants Malaysia Oswaldo Cruz Foundation Brazil USA Medecins Sans Frontieres (MSF) WHO/TDR (permanent observer) India Kenya Malaysian MOH Institut Pasteur France Brazil 2 Project Support Offices Japan RDC

2006, WHO strengthens recommendations. ACTS should be: 1. first-line treatment for falciparum malaria everywhere 2. in fixed-dose combinations when possible www. who. int/malaria/docs/Treatment. Guidelines 2006. pdf

+ FACT Project partners The product: Artesunate-Amodiaquine Fixed-Dose Combination « ASAQ Winthrop» / « Coarsucam » 1. Clinical results 2. Registration plans / Regulatory process 3. Possibilities for MSF
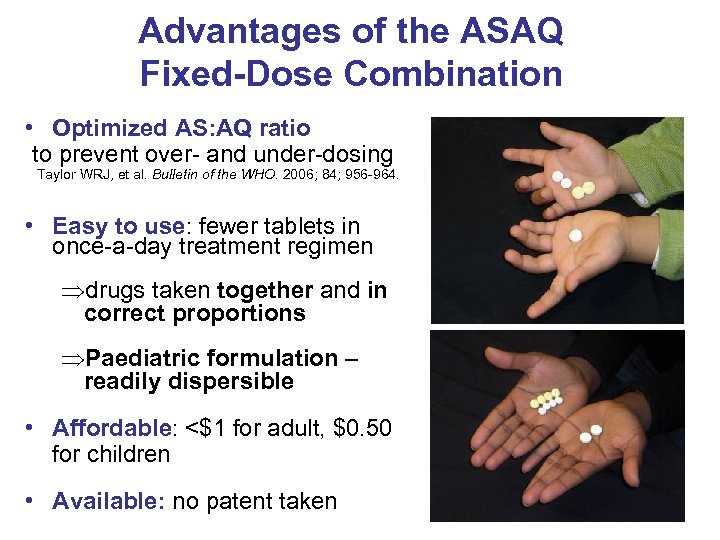
Advantages of the ASAQ Fixed-Dose Combination • Optimized AS: AQ ratio to prevent over- and under-dosing Taylor WRJ, et al. Bulletin of the WHO. 2006; 84; 956 -964. • Easy to use: fewer tablets in once-a-day treatment regimen Þdrugs taken together and in correct proportions ÞPaediatric formulation – readily dispersible • Affordable: <$1 for adult, $0. 50 for children • Available: no patent taken
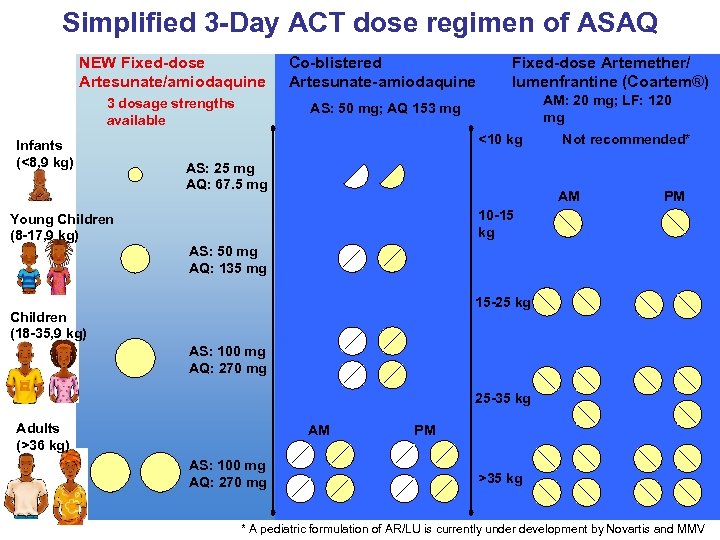
Simplified 3 -Day ACT dose regimen of ASAQ NEW Fixed-dose Artesunate/amiodaquine Co-blistered Artesunate-amiodaquine Fixed-dose Artemether/ lumenfrantine (Coartem®) 3 dosage strengths available AS: 50 mg; AQ 153 mg AM: 20 mg; LF: 120 mg Infants (<8, 9 kg) <10 kg AS: 25 mg AQ: 67. 5 mg Not recommended* AM PM 10 -15 kg Young Children (8 -17, 9 kg) AS: 50 mg AQ: 135 mg 15 -25 kg Children (18 -35, 9 kg) AS: 100 mg AQ: 270 mg 25 -35 kg Adults (>36 kg) AM AS: 100 mg AQ: 270 mg PM >35 kg * A pediatric formulation of AR/LU is currently under development by Novartis and MMV
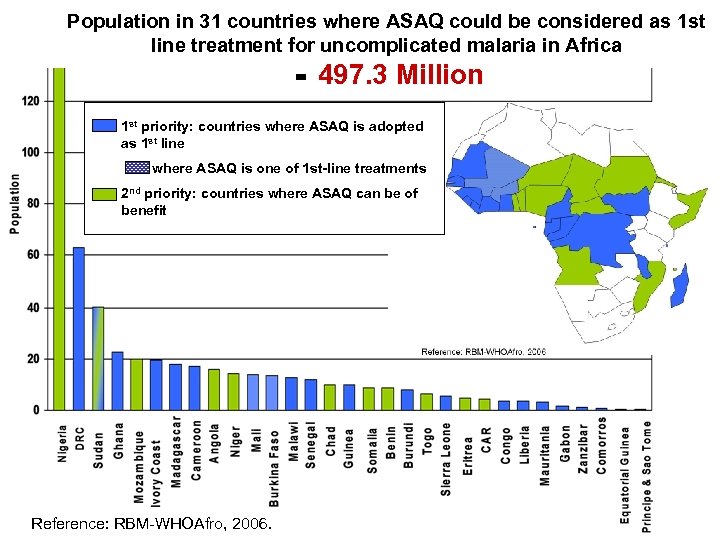
Population in 31 countries where ASAQ could be considered as 1 st line treatment for uncomplicated malaria in Africa = 497. 3 Million 1 st priority: countries where ASAQ is adopted as 1 st line where ASAQ is one of 1 st-line treatments 2 nd priority: countries where ASAQ can be of benefit Reference: RBM-WHOAfro, 2006.

What is known about ASAQ? 1. 2. 3. AS+AQ: Tolerability and Drug Interaction in HNV AS vs AQ vs AS/AQ. P. Olliaro, et al. AS+AQ: Tolerability and PK study in HNV of non-fixed and fixed-dose combinations, P. Olliaro et al. Comparative Clinical study of ASAQ vs AS+AQ in Burkina Faso – S. Sirima et al. , Ouagadougou (n=750) 4. AS+AQ for uncomplicated falciparum malaria: A systematic review of safety and efficacy data - P. Olliaro et al. (n=5000) 5. Efficacy & Tolerability of AS + AQ for falciparum malaria in Senegal. 6 -year deployment of a 10 -year survey field survey - P. Brasseur et al. Sénégal (n=3000), 6. Comparative Clinical study of FDC ASAQ vs. Coartem®, March to Dec. 06/Cameroon, Mali, Madagascar & Senegal; (n=941) – Preliminary results presented in Amsterdam, May 07
Burkina Faso: Most relevant study to register ASAQ A comparative clinical assessment of fixed-dose ASAQ vs. non-fixed artesunate-amodiaquine (AS+AQ) Laos -Study coordinated by DNDi with CNFRP from 2004 to 2006 Algérie Sénégal Mali Nigeria Côte Ghana d’Ivoire Bénin -750 children < 5 kg were followed up over 28 days -To demonstrate the noninferiority of fixed-dose artesunate-amodiaquine (ASAQ) versus loose combination (AS+AQ) -Efficacy -Safety
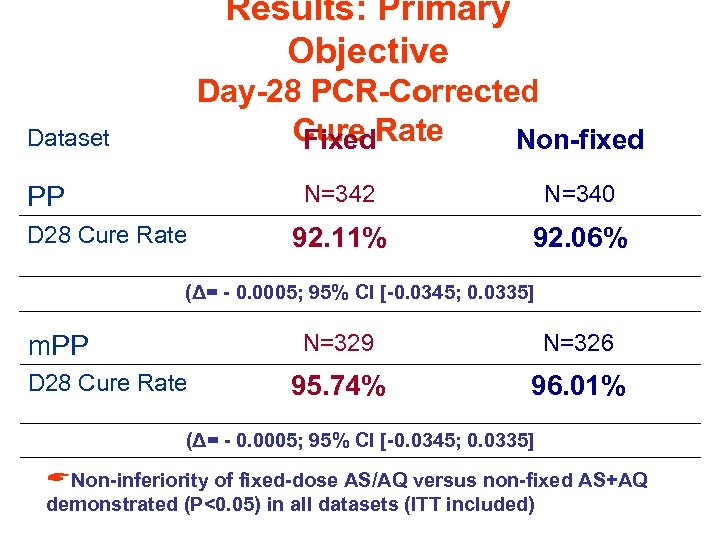
Results: Primary Objective Day-28 PCR-Corrected Cure Rate Fixed Non-fixed Dataset N=342 PP D 28 Cure Rate N=340 92. 11% 92. 06% (Δ= - 0. 0005; 95% CI [-0. 0345; 0. 0335] N=329 m. PP D 28 Cure Rate N=326 95. 74% 96. 01% (Δ= - 0. 0005; 95% CI [-0. 0345; 0. 0335] ANon-inferiority of fixed-dose AS/AQ versus non-fixed AS+AQ demonstrated (P<0. 05) in all datasets (ITT included)

Most frequently reported adverse events (AEs) • Intensity was mild to moderate FIXED N=375 LOOSE N=374 – Pyrexia (fever) 18. 1 % 18. 7 % – Anaemia 5. 3 % 6. 4 % – Diarrhea 1. 9 % 2. 7 % • Nausea and vomiting, typically seen, were <2% over course of study (Days 1 -28)
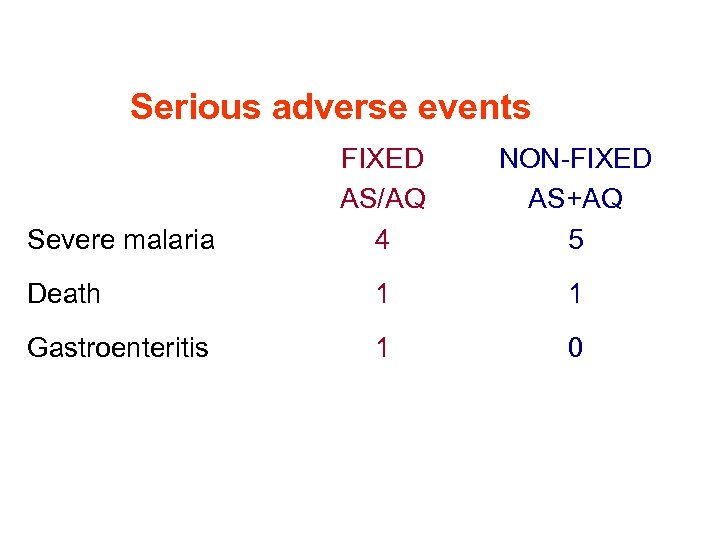
Serious adverse events FIXED AS/AQ 4 NON-FIXED AS+AQ 5 Death 1 1 Gastroenteritis 1 0 Severe malaria

Conclusions § Comparable efficacy of fixed-dose ASAQ vs non-fixed AS-AQ § Non-inferiority demonstrated § Easy to use § Well tolerated

AS + AQ Meta-Analyses • Meta-analysis on safety and efficacy of AS + AQ studies around the world (P. Olliaro et al. ) – N ≥ 5000 patients – 31 Studies conducted between 1999 -2006 • 27 comparative, 3 non-comparative, 1 PK only – 18 African countries – Most following WHO guidelines – Patients largely children (<5 yr in 18 studies); • only 5 enrolled also adults – Preliminary results presented at ASTMH 2006, Atlanta

Conclusions Efficacy & Safety • Efficacy generally >90% after genotyping correction • AS+AQ: – more effective than monotherapy and non-ACTs – similar to AS+SP – compares well with AM+LF, DH+PQ after PCR correction but more re-infections • Safety: crudely comparable safety profiles – Broader safety data needed

AS+AQ Individual Patient Meta-Analysis Julien Zwang (Nosten + Olliaro) AS+AQ Efficacy in the Treatment of Uncomplicated falciparum malaria • > 4000 patients treated with AS+AQ 66% under 5 y. o. • 24 African sites 12 African Countries (Uganda 1283, Senegal 966) • 28 to 63 days follow up • AS+AQ efficacy PCR corrected 91% • Median time of parasite recrudescence 21 days • Tolerability analysis pending full compilation • Revision by PI’s June 2007 • Publication December 2007

A multinational, randomized Phase III study to assess the efficacy and tolerability of FDC AS+AQ once or twice daily vs. AL for uncomplicated falciparum malaria - To demonstrate the non-inferiority of ASAQ versus AL in Cameroon, Mali, Senegal & Madagascar, from Mar to Dec 2006 - 941 patients including 437 children - Preliminary results*: - >95% PCR-corrected cure rate at D 28 for all groups - Good clinical & biological safety seen among all treatment arms *Presented at 5 th ECTMIH, Amsterdam, 24 -28 May 2007
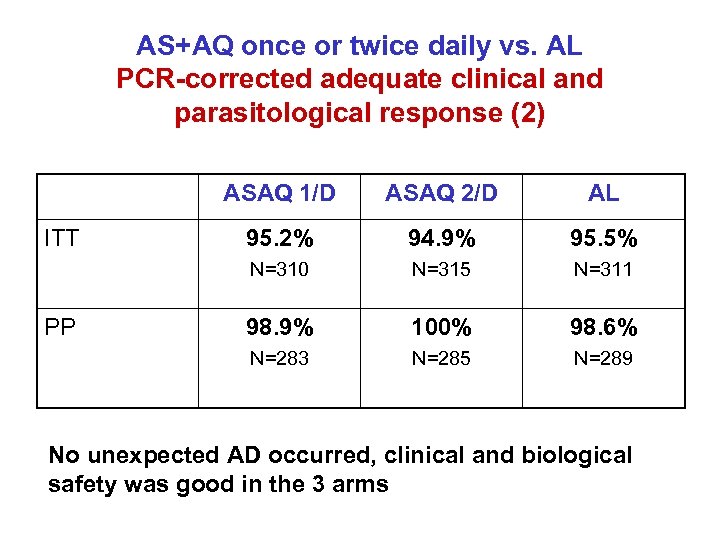
AS+AQ once or twice daily vs. AL PCR-corrected adequate clinical and parasitological response (2) ASAQ 1/D 95. 2% 94. 9% 95. 5% N=315 N=311 98. 9% 100% 98. 6% N=283 PP AL N=310 ITT ASAQ 2/D N=285 N=289 No unexpected AD occurred, clinical and biological safety was good in the 3 arms

Registration File – Fixed-Dose Formulation International CTD format • Approved in Morocco, February 2007 • Submitted to the WHO prequalification process, February 2007 • Registration process undertaken in 23 sub-Saharan African countries • Approved in 14 sub-Saharan African countries – Benin, Burkina Faso, Congo, Côte d’Ivoire, Gabon, Guinea, Kenya, Mali, Mauritania, Democratic Republic of the Congo, Togo, Zanzibar, Ghana & Madagascar

How can ASAQ be ordered? • In many countries, Coarsucam® needs to be registered before Arte. Sunate Amodia. Quine Winthrop® can be submitted to national regulatory agencies. • Until Artesunate Amodiaquine Winthrop® is available, NGOs and other non-for profit institutions interested in obtaining ASAQ can purchase Coarsucam® Impact Malaria at a “no profit-no loss” price under the following conditions : – – Coarsucam® Impact Malaria is registered in the country of interest Minimum order of 5000€ Qualified person (“pharmacist responsible") required Storage facility available at country level

How can MSF contribute? - Anticipate orders: estimate ASAQ needs for use in the field considering the 4 different presentations - Get special authorisation to use ASAQ in countries out of the s-a 1 st priority (Liberia, Sierra Leone, Malawi, South Sudan) - Document and analyse the use of ASAQ in the field - Tolerability - Adherence - Efficacy - Drug interaction - Special group of patients - Share experience and knowledge as part of the MSF advocacy

Thank you! Looking forward to more partners who could help to deliver innovation to the patients!

Acknowledgements • EC INCO-Dev Program • FACT Consortium and Team Members – – – – • • Fondation Médecins Sans Frontières Tropival, Université de Bordeaux 2 Centre for Tropical Medicine, University of Oxford Centre for Drug Research, Universiti Sains Malaysia Faculty of Tropical Medicine, Mahidol University Instituto de Technologia em Farmacos, Farmanguinhos WHO Special Programme on Research & Training in Tropical Diseases Centre National de Recherche et de Formation sur le Paludisme (Burkina Faso) P. Olliaro, W. R. J. Taylor (WHO/TDR) Colleagues from DNDi and MSF Ellipse, Cardinal Systems, Quintiles, Cardinal Health… Abbott (Knoll); Pfizer (Parke-Davis); Roche; sanofi-aventis
3c98722ce375cad2fad5f919a576c3fc.ppt