41787f3358b008be423cdacc079835b7.ppt
- Количество слайдов: 64


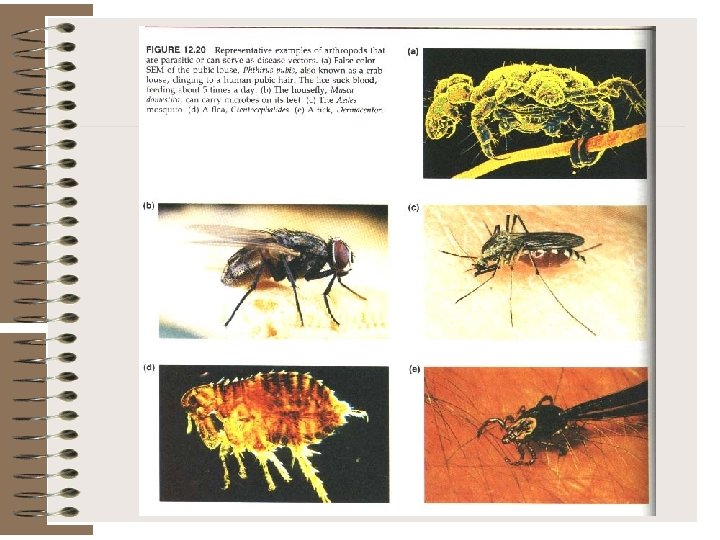
Arthropod-Borne (Arbo) Viral Diseases The arthropod-borne viruses, or arboviruses, are a group of infectious agents that are transmitted by blood-sucking arthropods from one vertebrate host to another. They can multiply in the tissues of the arthropod without evidence of disease or damage. The vector acquires a lifelong infection through the ingestion of blood from a viremic vertebrate. All arboviruses have an RNA genome, and most have a lipid-containing envelope and consequently are inactivated by ether or sodium deoxycholate.
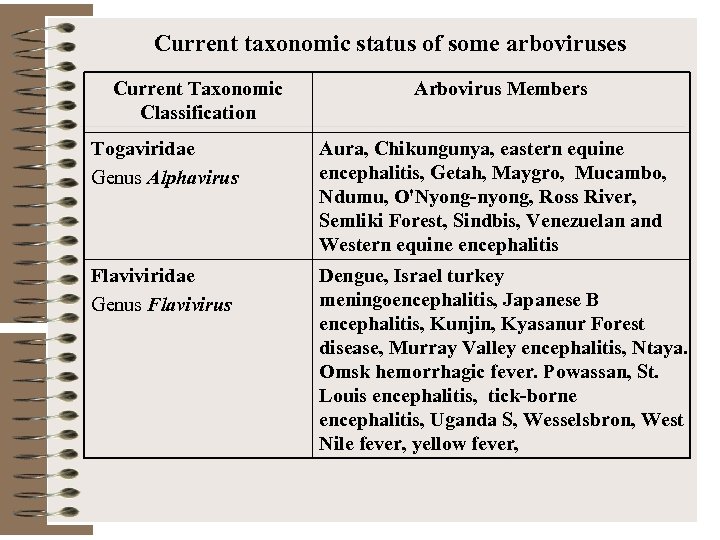
Current taxonomic status of some arboviruses Current Taxonomic Classification Arbovirus Members Togaviridae Genus Alphavirus Aura, Chikungunya, eastern equine encephalitis, Getah, Maygro, Mucambo, Ndumu, O'Nyong-nyong, Ross River, Semliki Forest, Sindbis, Venezuelan and Western equine encephalitis Flaviviridae Genus Flavivirus Dengue, Israel turkey meningoencephalitis, Japanese B encephalitis, Kunjin, Kyasanur Forest disease, Murray Valley encephalitis, Ntaya. Omsk hemorrhagic fever. Powassan, St. Louis encephalitis, tick-borne encephalitis, Uganda S, Wesselsbron, West Nile fever, yellow fever,
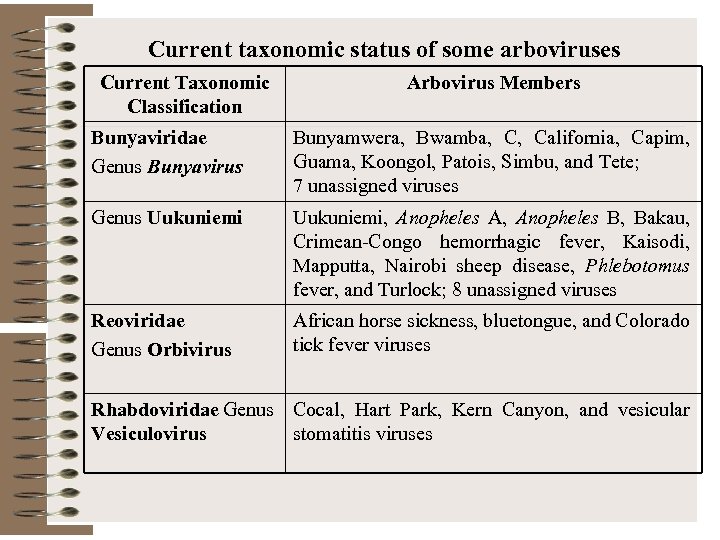
Current taxonomic status of some arboviruses Current Taxonomic Classification Arbovirus Members Bunyaviridae Genus Bunyavirus Bunyamwera, Bwamba, C, California, Capim, Guama, Koongol, Patois, Simbu, and Tete; 7 unassigned viruses Genus Uukuniemi, Anopheles A, Anopheles B, Bakau, Crimean-Congo hemorrhagic fever, Kaisodi, Mapputta, Nairobi sheep disease, Phlebotomus fever, and Turlock; 8 unassigned viruses Reoviridae Genus Orbivirus African horse sickness, bluetongue, and Colorado tick fever viruses Rhabdoviridae Genus Cocal, Hart Park, Kern Canyon, and vesicular Vesiculovirus stomatitis viruses
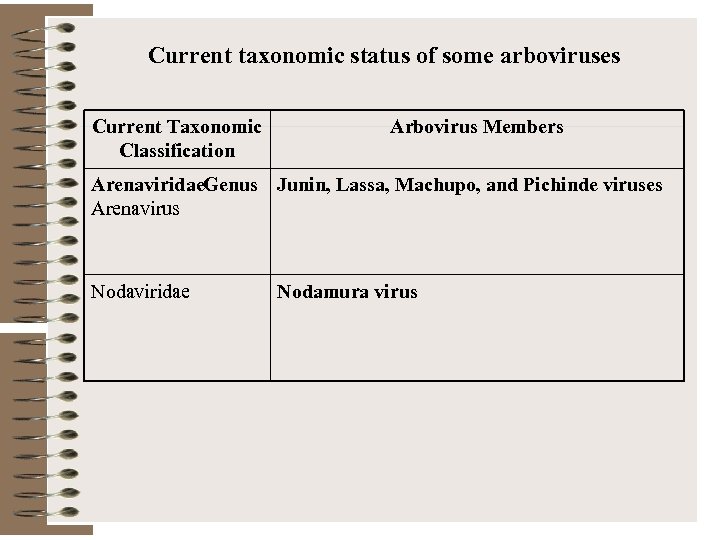
Current taxonomic status of some arboviruses Current Taxonomic Classification Arbovirus Members Arenaviridae. Genus Junin, Lassa, Machupo, and Pichinde viruses Arenavirus Nodaviridae Nodamura virus

Sindbis virus
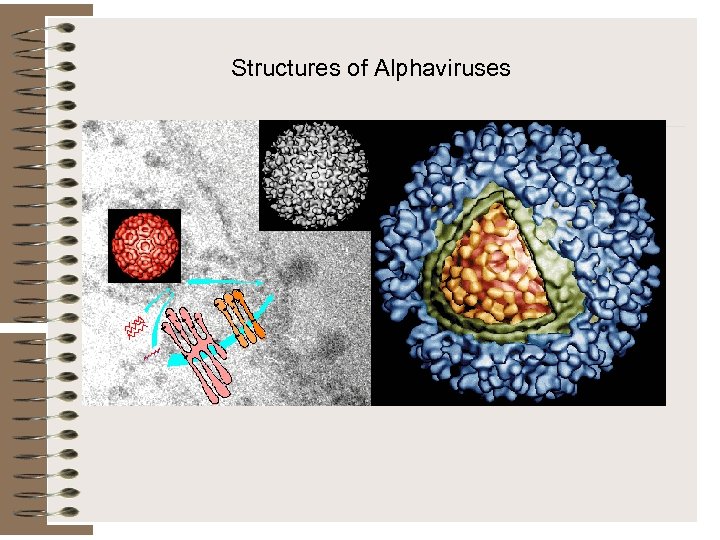
Structures of Alphaviruses
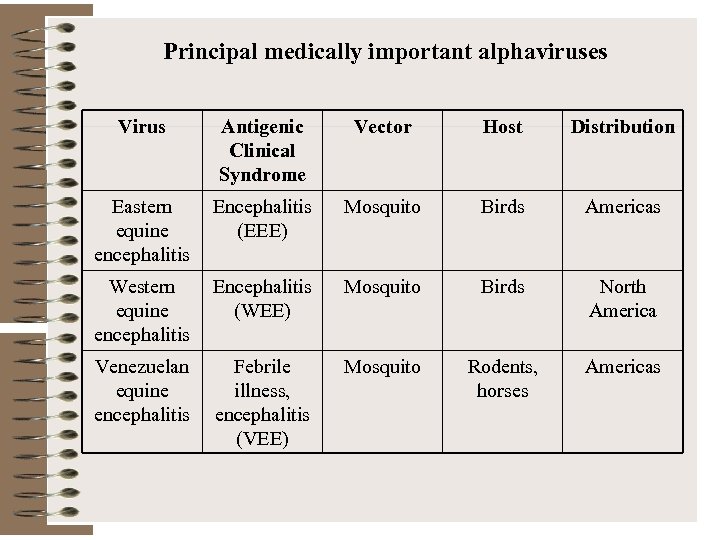
Principal medically important alphaviruses Virus Antigenic Clinical Syndrome Vector Host Distribution Eastern equine encephalitis Encephalitis (EEE) Mosquito Birds Americas Western equine encephalitis Encephalitis (WEE) Mosquito Birds North America Venezuelan equine encephalitis Febrile illness, encephalitis (VEE) Mosquito Rodents, horses Americas
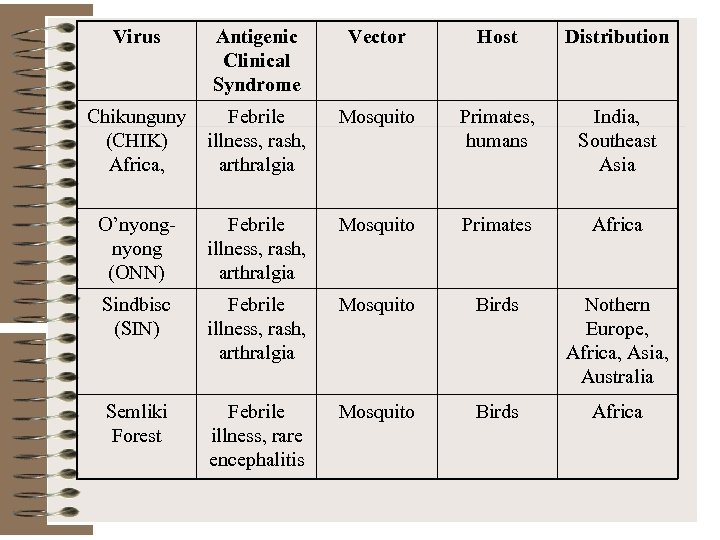
Virus Antigenic Clinical Syndrome Vector Host Distribution Chikunguny (CHIK) Africa, Febrile illness, rash, arthralgia Mosquito Primates, humans India, Southeast Asia O’nyong (ONN) Febrile illness, rash, arthralgia Mosquito Primates Africa Sindbisc (SIN) Febrile illness, rash, arthralgia Mosquito Birds Nothern Europe, Africa, Asia, Australia Semliki Forest Febrile illness, rare encephalitis Mosquito Birds Africa
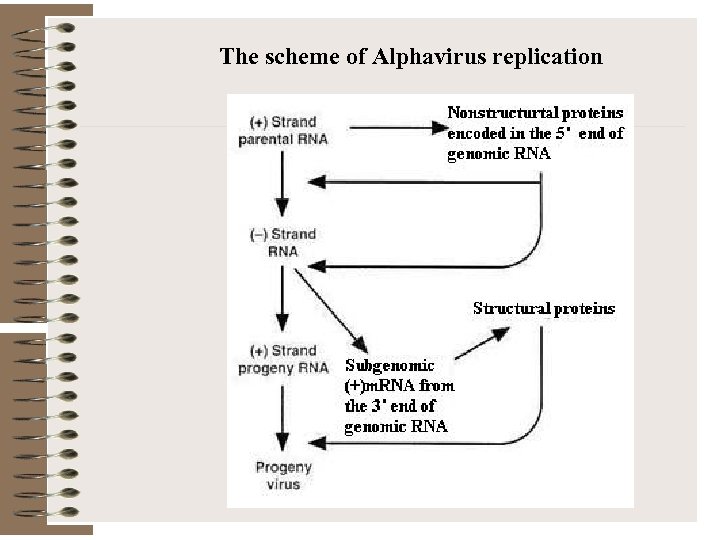
The scheme of Alphavirus replication
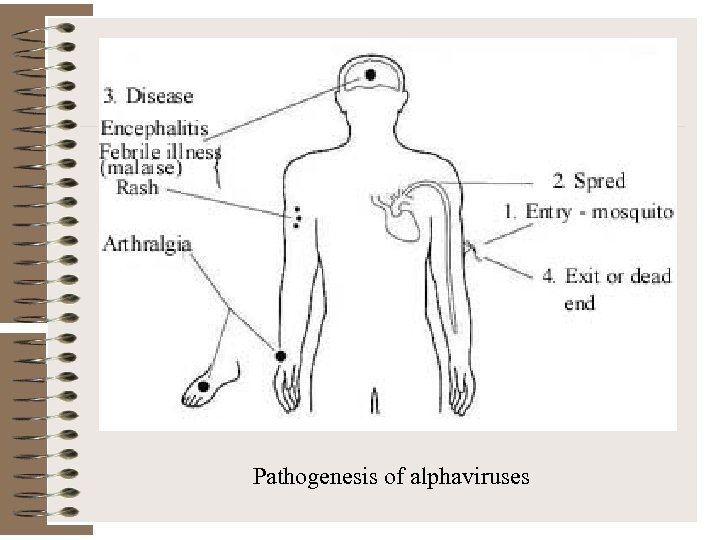
Pathogenesis of alphaviruses
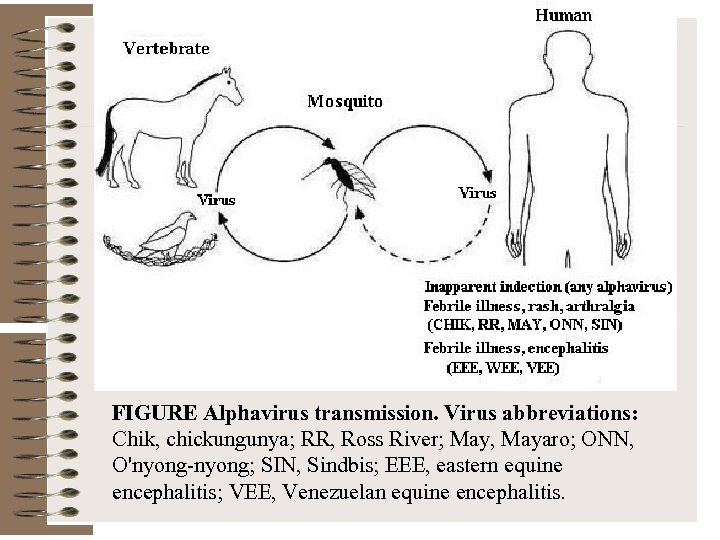
FIGURE Alphavirus transmission. Virus abbreviations: Chik, chickungunya; RR, Ross River; May, Mayaro; ONN, O'nyong-nyong; SIN, Sindbis; EEE, eastern equine encephalitis; VEE, Venezuelan equine encephalitis.

Rubellaviruses Rubella (German measles) is a common mild disease characterized by a rash. It affects children and adolescents worldwide and can also affect young adults. When rubella virus infects susceptible women early in pregnancy, it may be transmitted to the fetus and may cause birth defects. Therefore, accurate diagnosis is critical in pregnancy. The rubella virus is a member of the genus Rubivirus in the family Togaviridae.
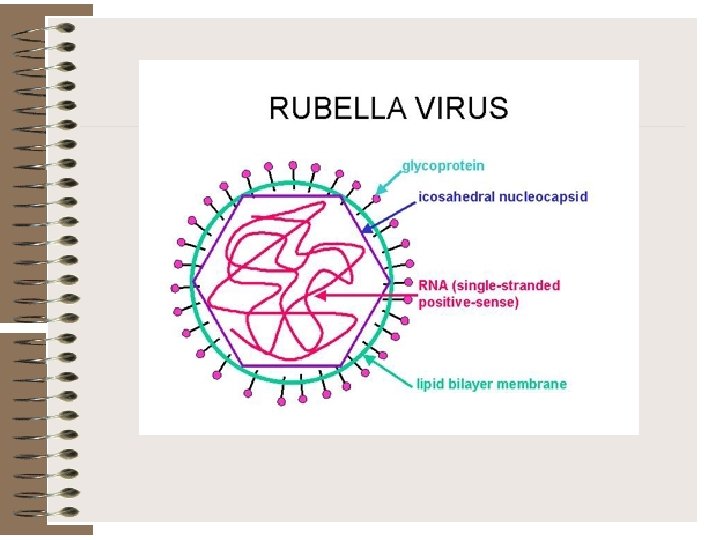
Rubella virus
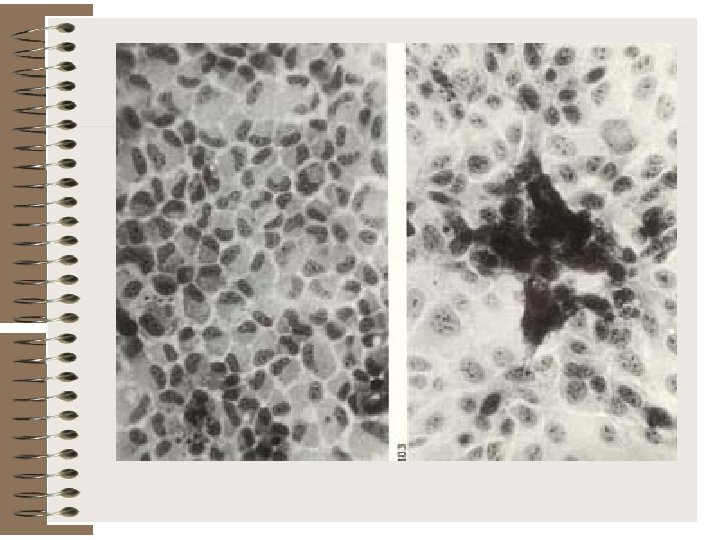
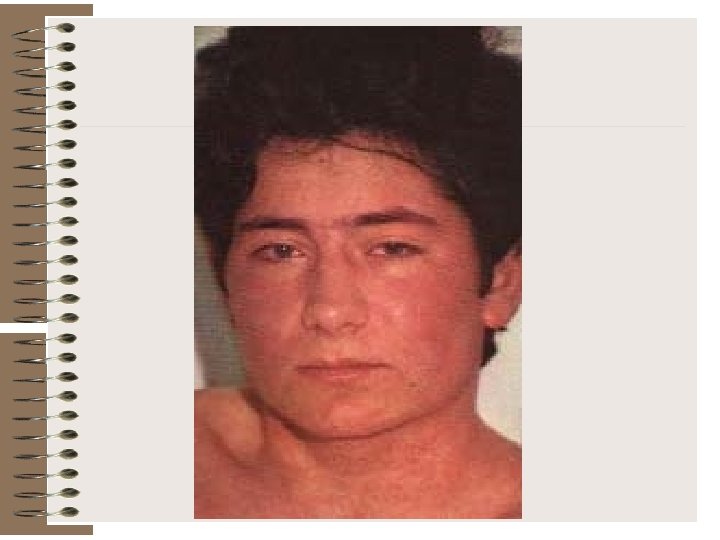
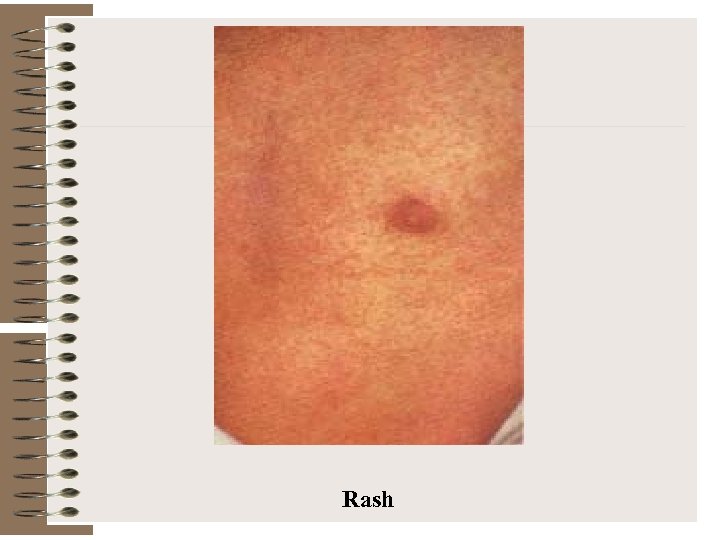
Rash
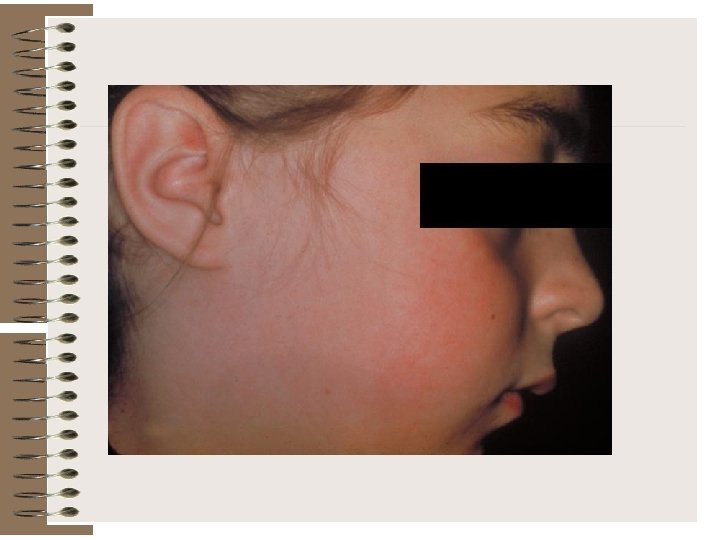
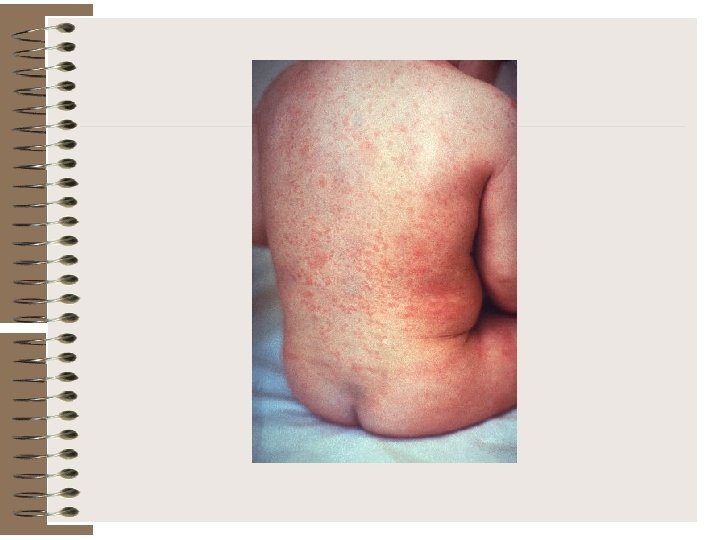
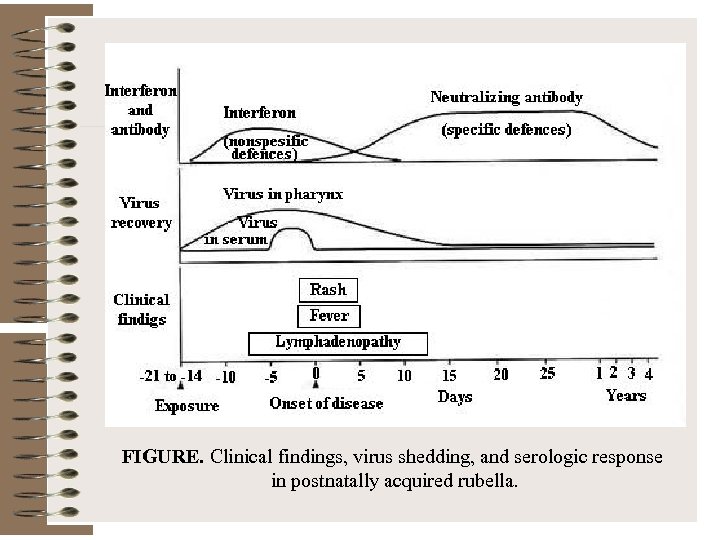
FIGURE. Clinical findings, virus shedding, and serologic response in postnatally acquired rubella.
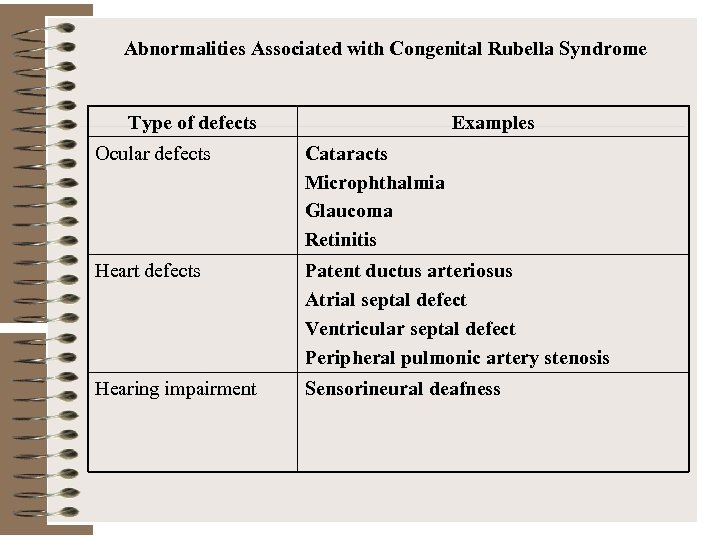
Abnormalities Associated with Congenital Rubella Syndrome Type of defects Examples Ocular defects Cataracts Microphthalmia Glaucoma Retinitis Heart defects Patent ductus arteriosus Atrial septal defect Ventricular septal defect Peripheral pulmonic artery stenosis Hearing impairment Sensorineural deafness
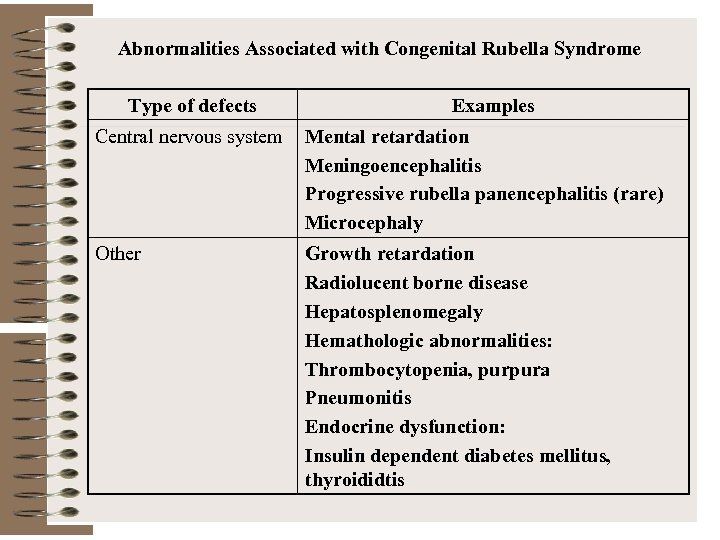
Abnormalities Associated with Congenital Rubella Syndrome Type of defects Examples Central nervous system Mental retardation Meningoencephalitis Progressive rubella panencephalitis (rare) Microcephaly Other Growth retardation Radiolucent borne disease Hepatosplenomegaly Hemathologic abnormalities: Thrombocytopenia, purpura Pneumonitis Endocrine dysfunction: Insulin dependent diabetes mellitus, thyroididtis
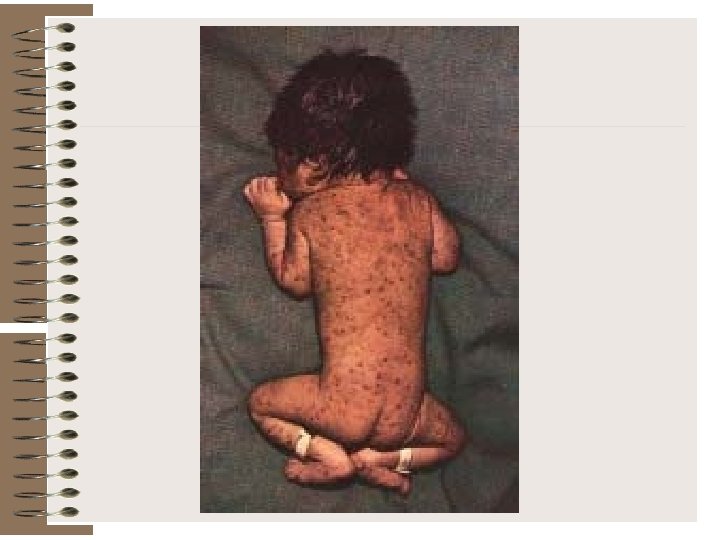
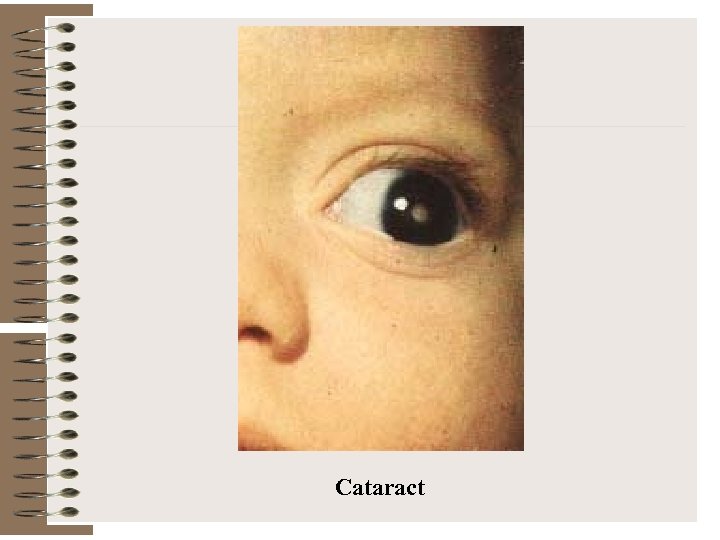
Cataract
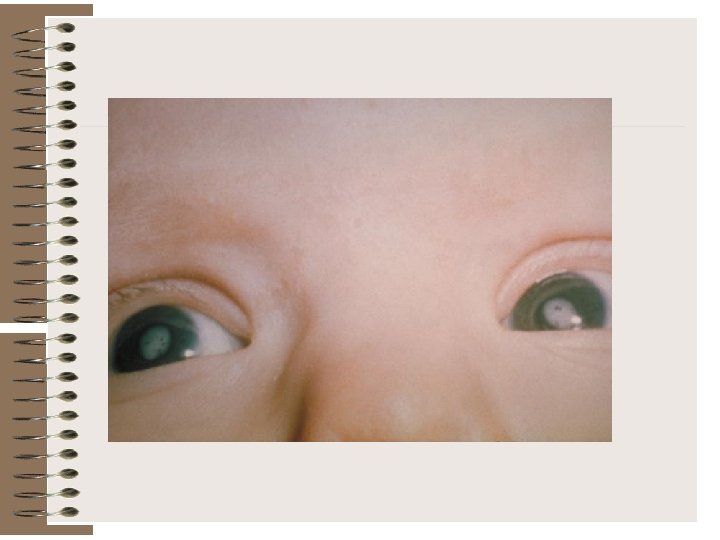
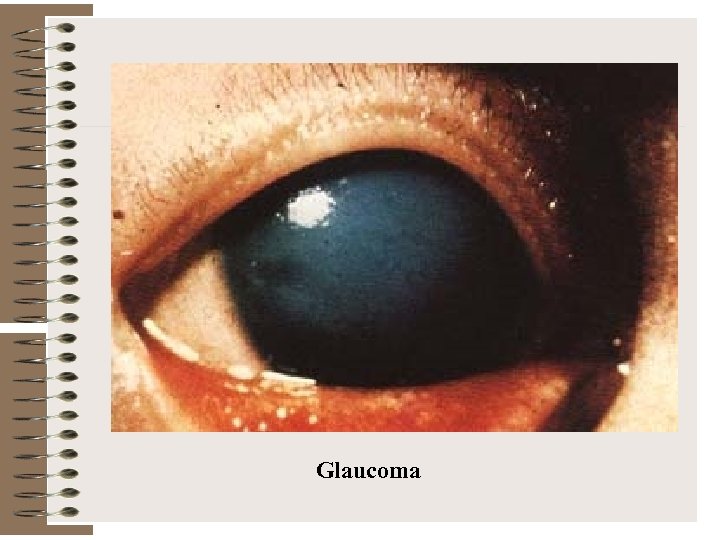
Glaucoma

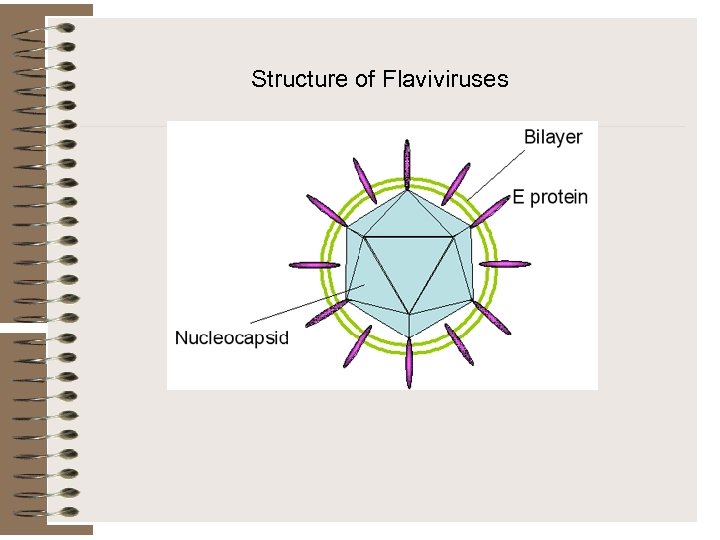
Structure of Flaviviruses
Flavivirus
Tick-born encephalitis virus
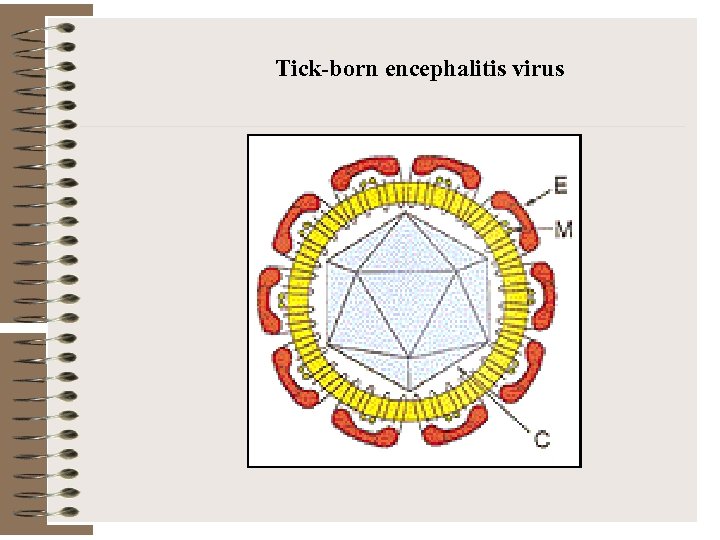
Tick-born encephalitis virus
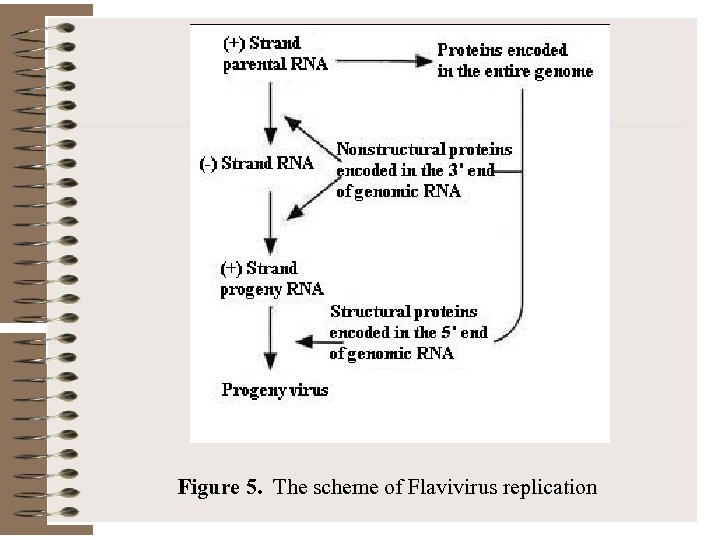
Figure 5. The scheme of Flavivirus replication
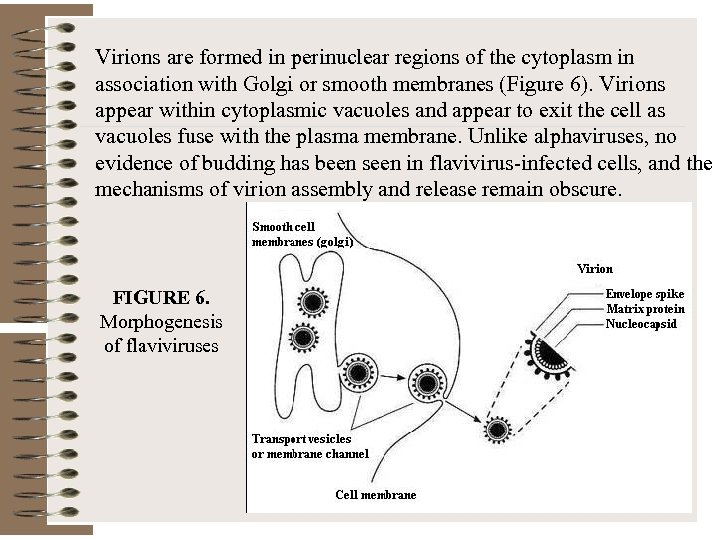
Virions are formed in perinuclear regions of the cytoplasm in association with Golgi or smooth membranes (Figure 6). Virions appear within cytoplasmic vacuoles and appear to exit the cell as vacuoles fuse with the plasma membrane. Unlike alphaviruses, no evidence of budding has been seen in flavivirus-infected cells, and the mechanisms of virion assembly and release remain obscure. FIGURE 6. Morphogenesis of flaviviruses
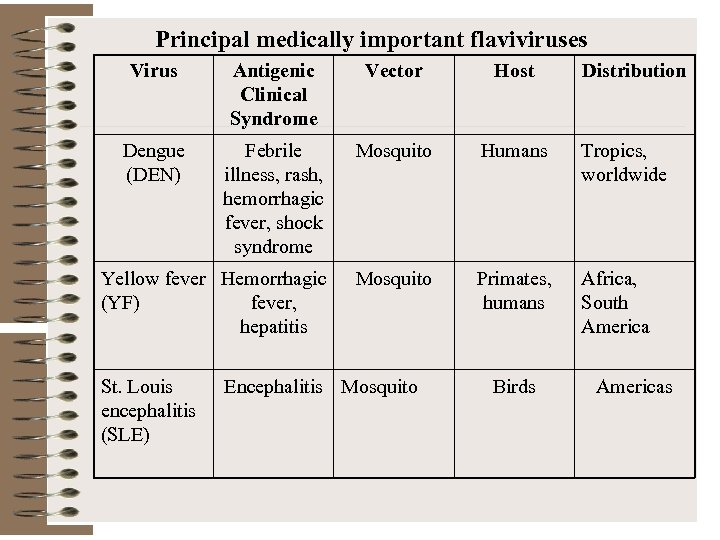
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Dengue (DEN) Febrile illness, rash, hemorrhagic fever, shock syndrome Mosquito Humans Tropics, worldwide Yellow fever Hemorrhagic (YF) fever, hepatitis Mosquito Primates, humans Africa, South America St. Louis encephalitis (SLE) Encephalitis Mosquito Birds Distribution Americas
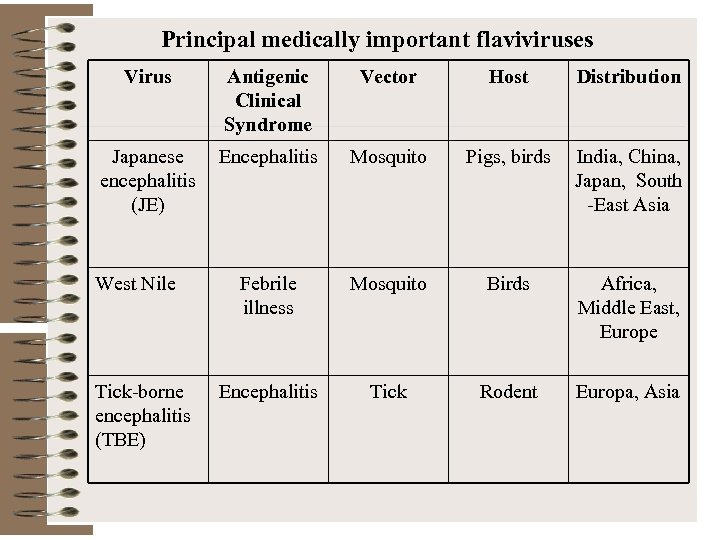
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Distribution Japanese encephalitis (JE) Encephalitis Mosquito Pigs, birds India, China, Japan, South -East Asia Febrile illness Mosquito Birds Africa, Middle East, Europe Encephalitis Tick Rodent Europa, Asia West Nile Tick-borne encephalitis (TBE)
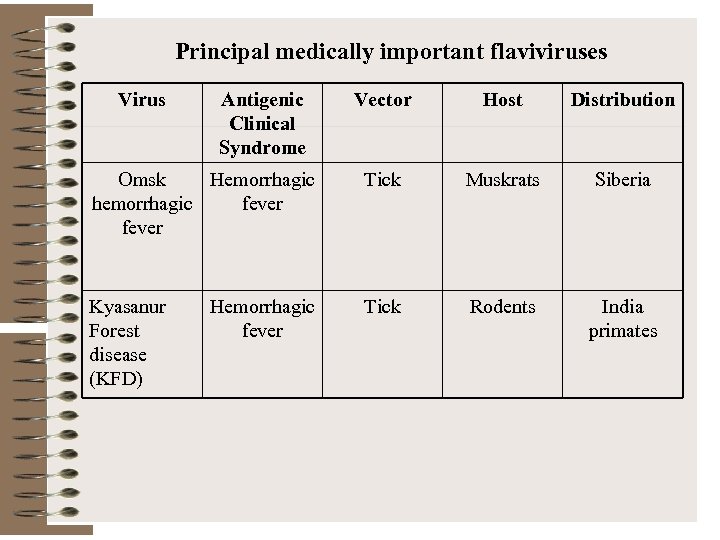
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Distribution Omsk Hemorrhagic hemorrhagic fever Tick Muskrats Siberia Kyasanur Forest disease (KFD) Tick Rodents India primates Hemorrhagic fever
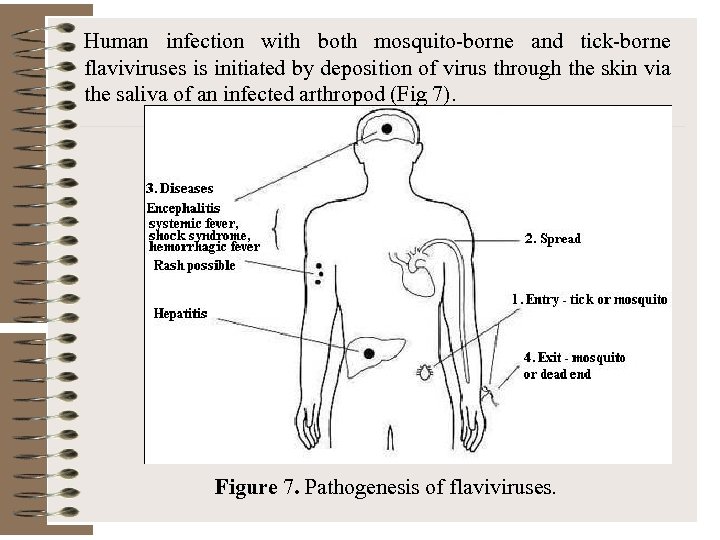
Human infection with both mosquito-borne and tick-borne flaviviruses is initiated by deposition of virus through the skin via the saliva of an infected arthropod (Fig 7). Figure 7. Pathogenesis of flaviviruses.

Tick
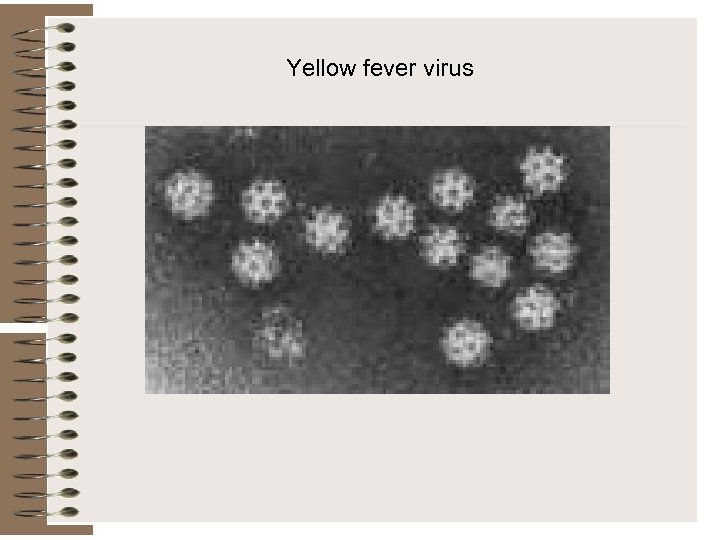
Yellow fever virus
Yellow fever
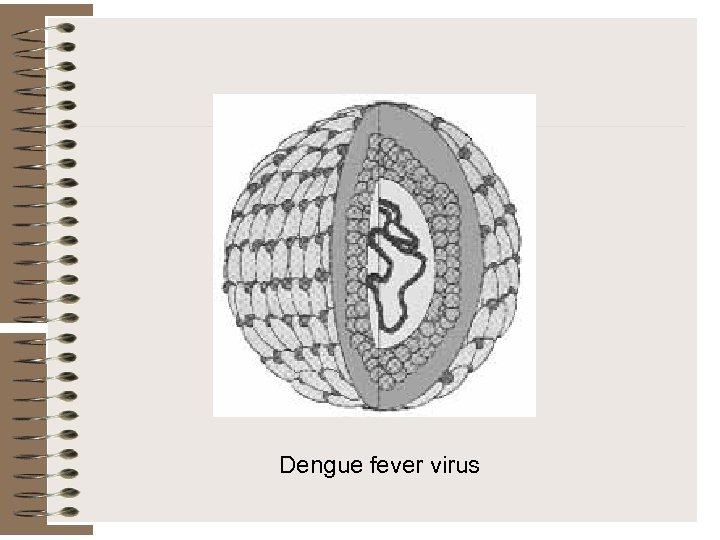
Dengue fever virus
Dengue fever


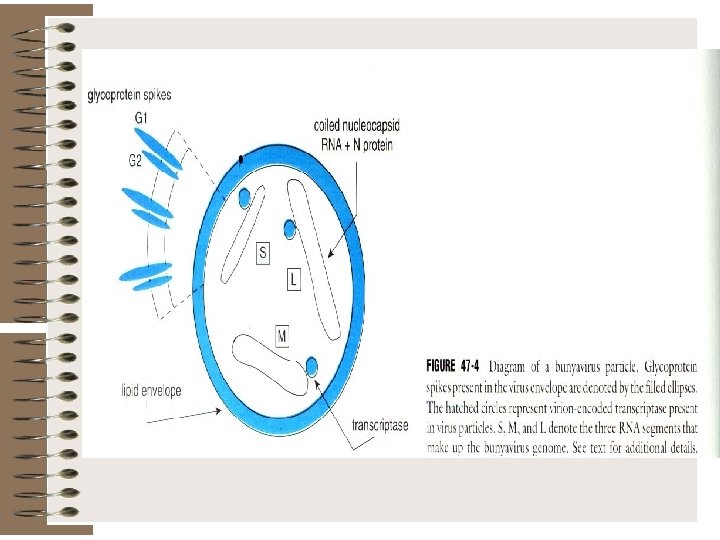
Bunyaviridae is a family of arthropod-borne or rodent-borne, spherical, enveloped RNA viruses. Bunyaviruses are responsible for a number of febrile diseases in humans and other vertebrates. They have either a rodent host or an arthropod vector and a vertebrate host.
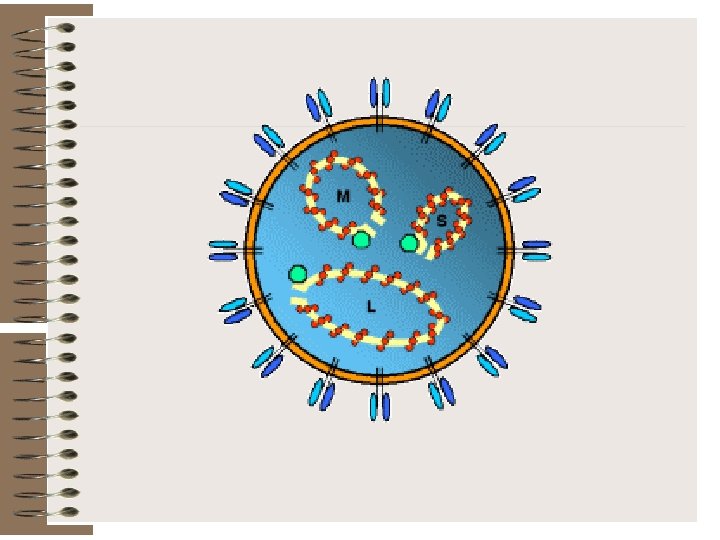
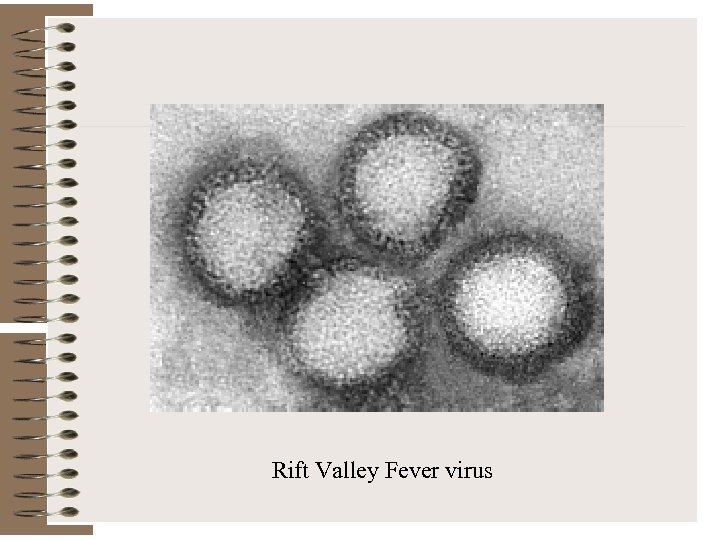
Rift Valley Fever virus
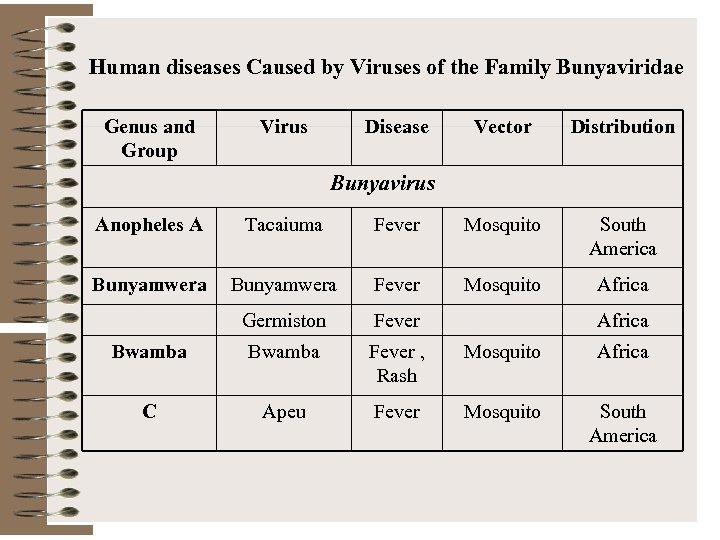
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Bunyavirus Anopheles A Tacaiuma Fever Mosquito South America Bunyamwera Fever Mosquito Africa Germiston Fever Bwamba Fever , Rash Mosquito Africa C Apeu Fever Mosquito South America Africa
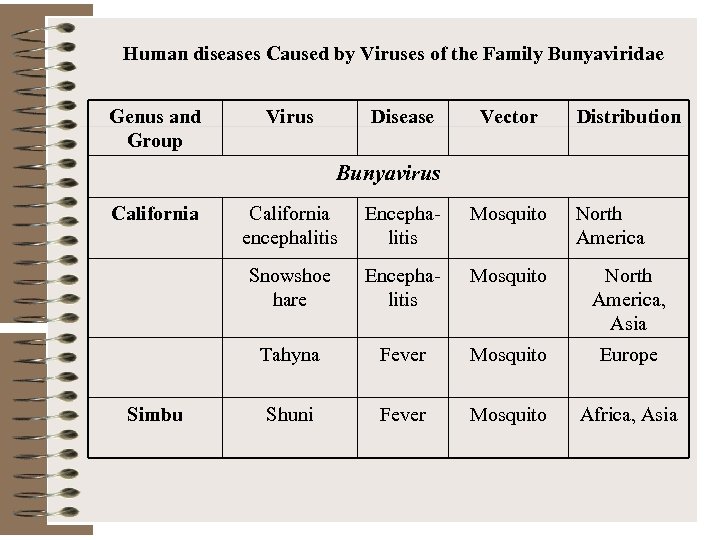
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Bunyavirus California Encephalitis Mosquito Snowshoe hare Encephalitis Mosquito North America, Asia Tahyna Simbu California encephalitis North America Fever Mosquito Europe Shuni Fever Mosquito Africa, Asia
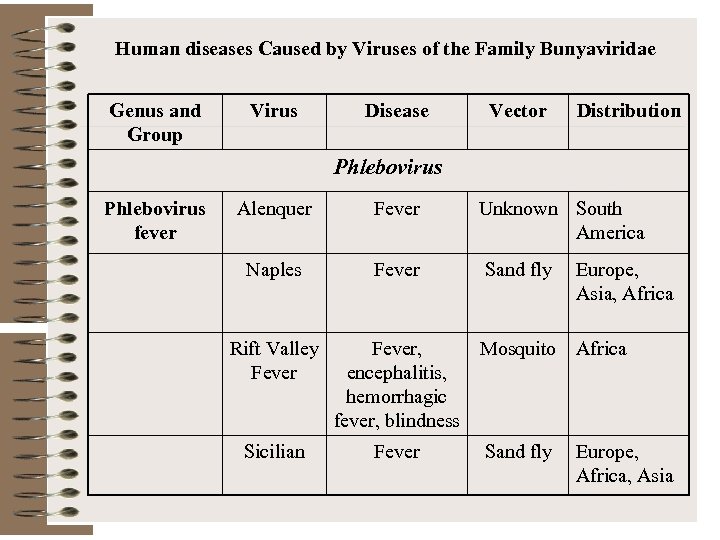
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Phlebovirus fever Alenquer Fever Unknown South America Naples Fever Sand fly Rift Valley Fever Sicilian Europe, Asia, Africa Fever, Mosquito Africa encephalitis, hemorrhagic fever, blindness Fever Sand fly Europe, Africa, Asia
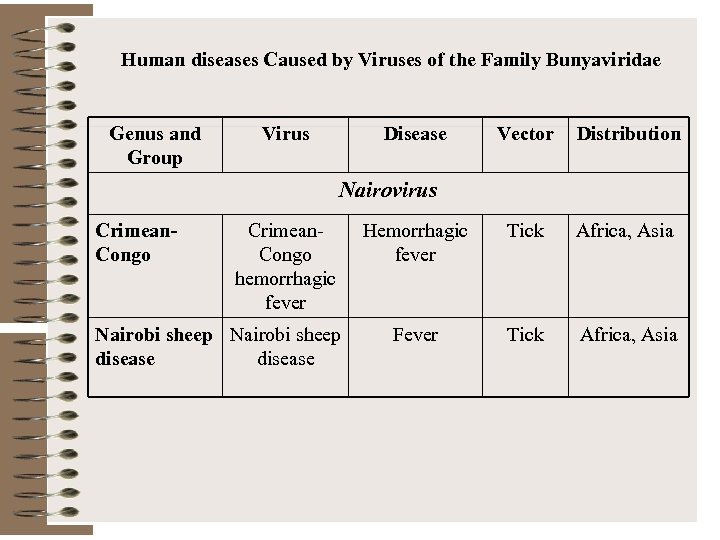
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Hemorrhagic fever Tick Africa, Asia Fever Tick Africa, Asia Nairovirus Crimean. Congo hemorrhagic fever Nairobi sheep disease
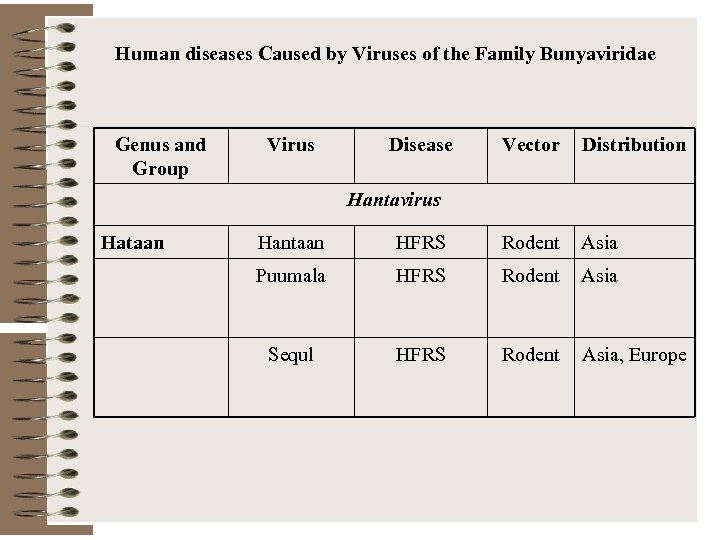
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Hantavirus Hataan Hantaan HFRS Rodent Asia Puumala HFRS Rodent Asia Sequl HFRS Rodent Asia, Europe
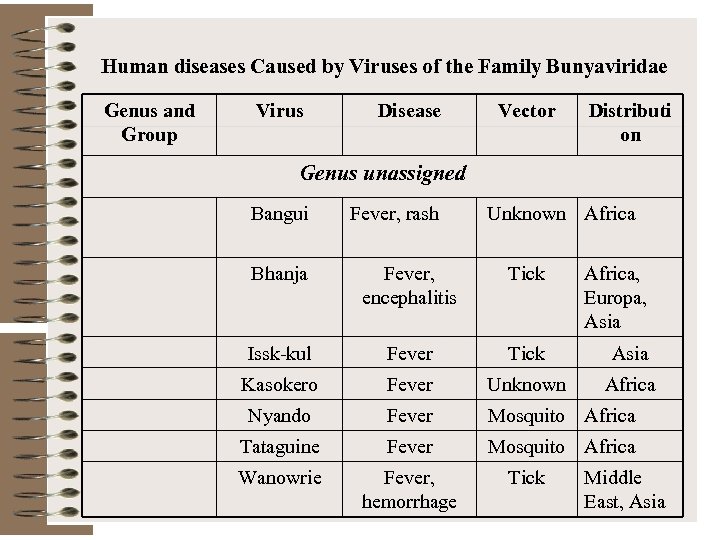
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distributi on Genus unassigned Bangui Fever, rash Unknown Africa Bhanja Fever, encephalitis Tick Africa, Europa, Asia Issk-kul Fever Tick Asia Kasokero Fever Unknown Africa Nyando Fever Mosquito Africa Tataguine Fever Mosquito Africa Wanowrie Fever, hemorrhage Tick Middle East, Asia
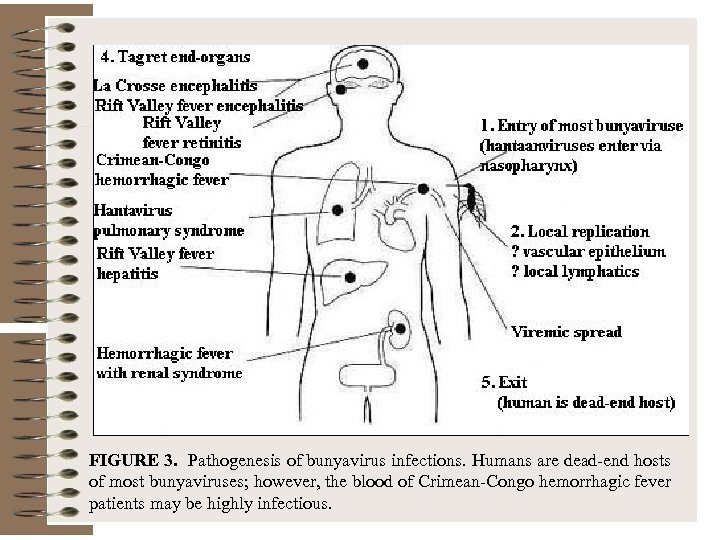
FIGURE 3. Pathogenesis of bunyavirus infections. Humans are dead-end hosts of most bunyaviruses; however, the blood of Crimean-Congo hemorrhagic fever patients may be highly infectious.
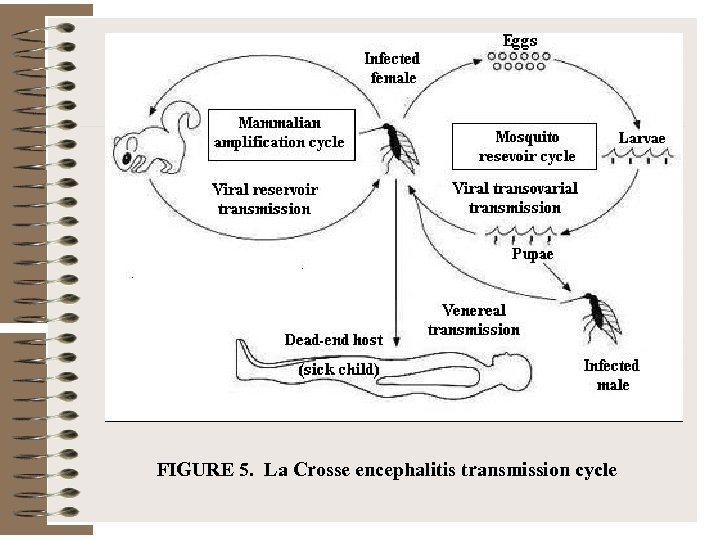
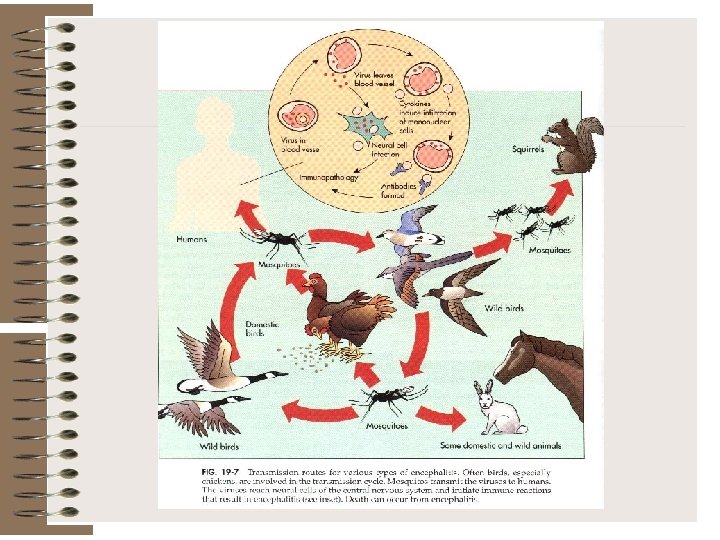
FIGURE 5. La Crosse encephalitis transmission cycle
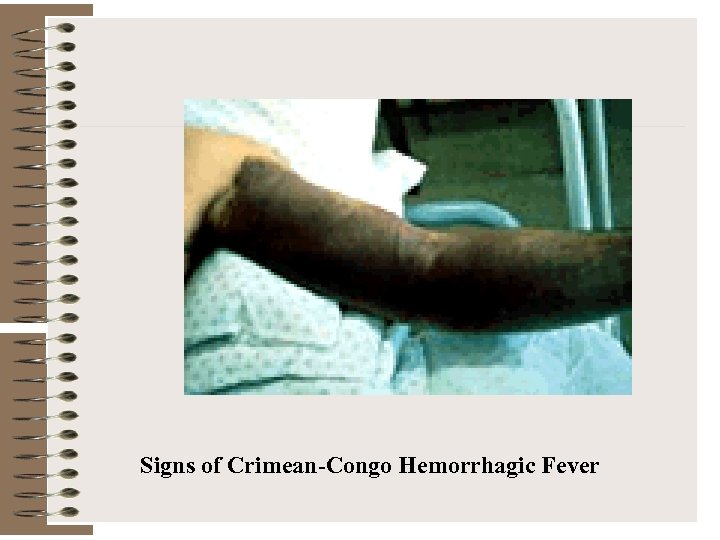
Signs of Crimean-Congo Hemorrhagic Fever
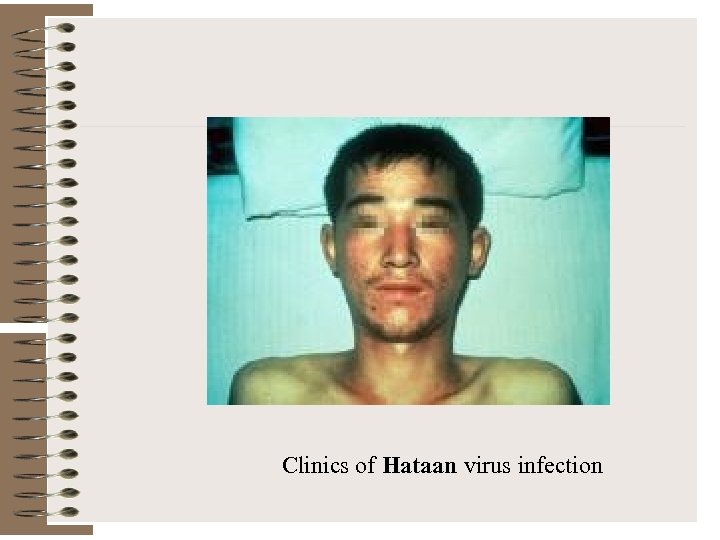
Clinics of Hataan virus infection
41787f3358b008be423cdacc079835b7.ppt