a04d522e5bddd29ded79b93838f0ce1d.ppt
- Количество слайдов: 60

Antibody Identification Mohammed Jaber

Antibody Presence of an antibody may be indicated by the following serological tests: 1. A discrepancy in the results of cell and serum ABH grouping. 2. A positive test for unexpected antibodies. 3. A positive direct Coomb’s test. 4. An incompatible major cross match.

The Basics…. . ¡ As we said in the previous lecture, l l Antibody Screens use 2 or 3 Screening Cells to “detect” if antibodies are present in the serum If antibodies are detected, then they should be identified… present Not present

Why do we need to identify? Antibody identification is an important component of compatibility testing ¡ It will identify any unexpected antibodies in the patient’s serum ¡ If a person with an antibody is exposed to donor cells with the corresponding antigen, serious side effects can occur (i. e. transfusion reactions). ¡

Key Concepts ¡ In blood banking, we test “knowns” with “unknowns”: Known: Reagent RBCs + Reagent antisera ¡ Unknown: patient serum + patient RBCs When detecting/screening and/or identifying antibodies, we test patient serum (unknown that contain blood bank antibodies) with reagent RBCs (known)

Reagent RBCs ¡ Screening Red Cells and Panel Red Cells are the same with minor differences: l Screening red cells Antibody detection/screening ¡ Sets of only 2 or 3 vials ¡ l Panel red cells Antibody identification ¡ At least 10 vials/set ¡
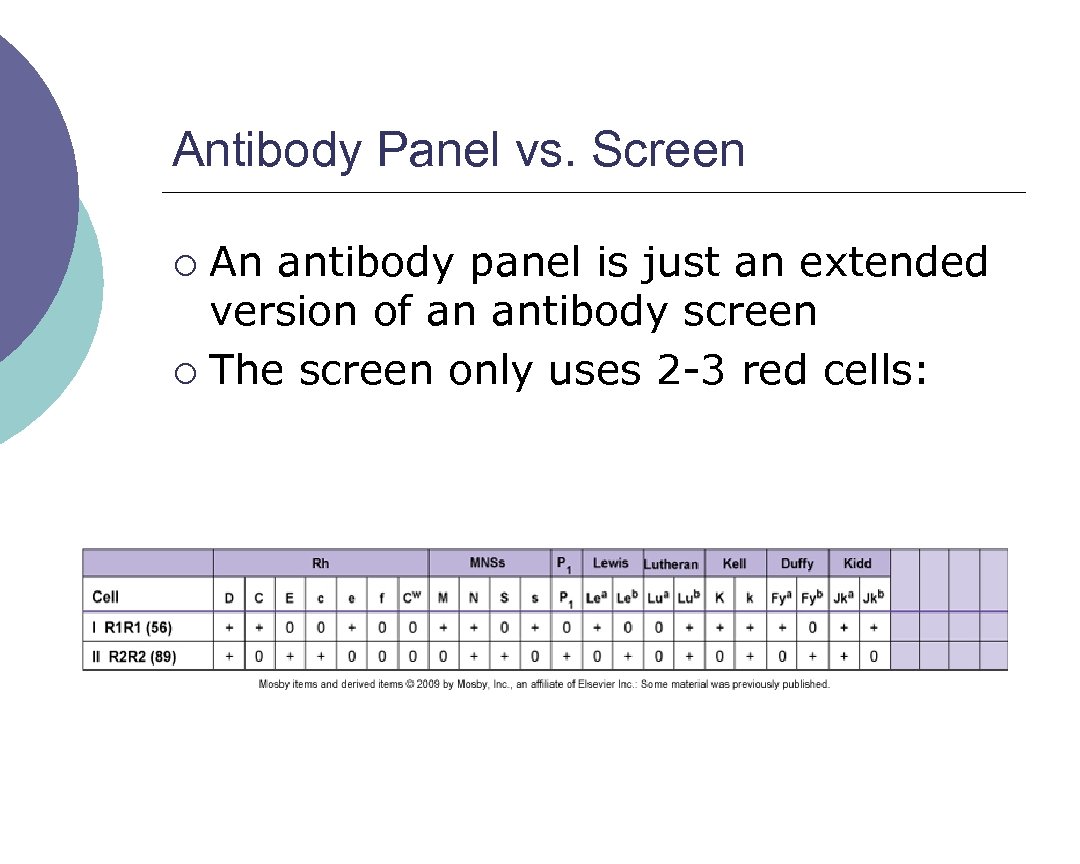
Antibody Panel vs. Screen An antibody panel is just an extended version of an antibody screen ¡ The screen only uses 2 3 red cells: ¡
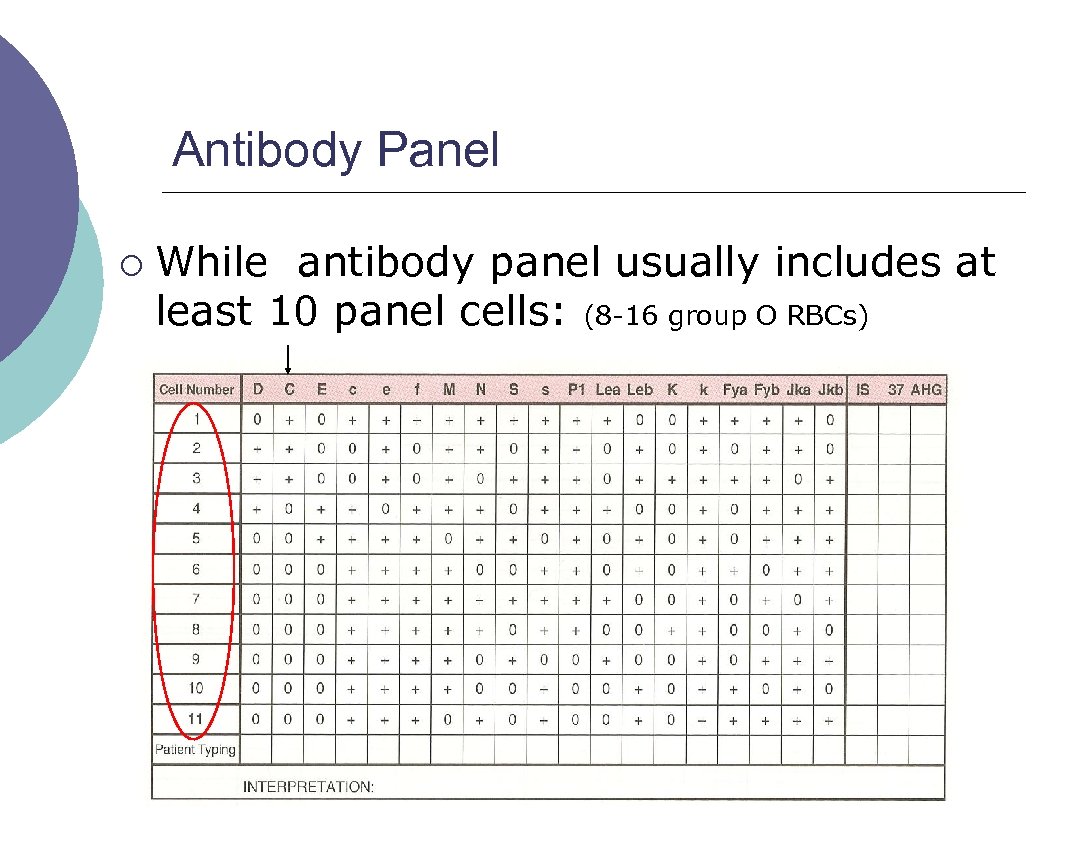
Antibody Panel ¡ While antibody panel usually includes at least 10 panel cells: (8 16 group O RBCs)
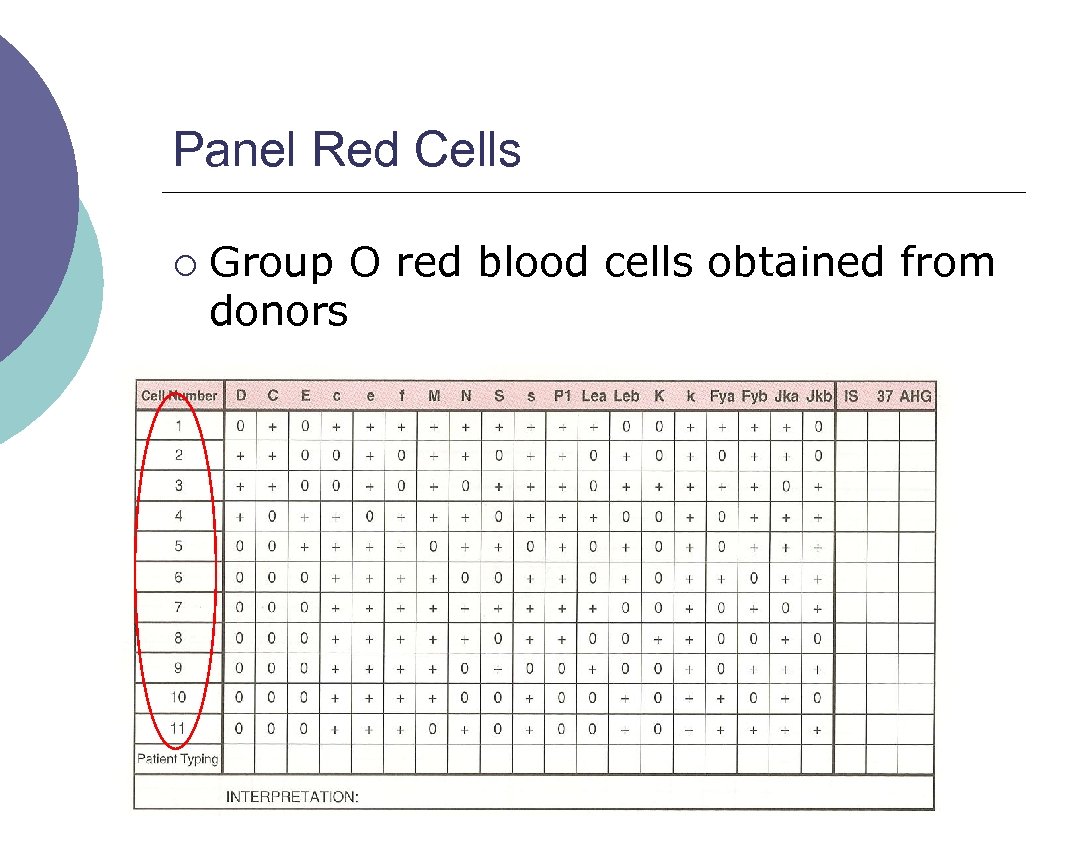
Panel Red Cells ¡ Group O red blood cells obtained from donors
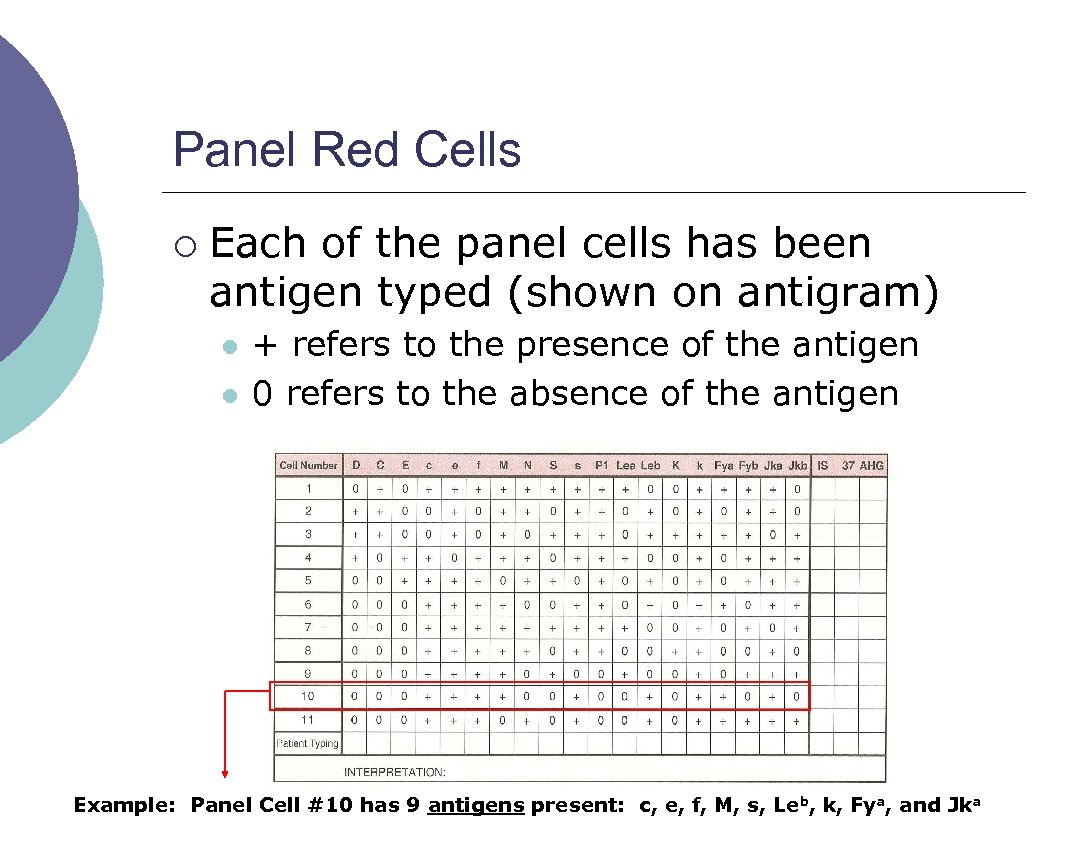
Panel Red Cells ¡ Each of the panel cells has been antigen typed (shown on antigram) l l + refers to the presence of the antigen 0 refers to the absence of the antigen Example: Panel Cell #10 has 9 antigens present: c, e, f, M, s, Leb, k, Fya, and Jka

Panel Red Cells ¡ An autocontrol (AC) should also be run with ALL panels Autocontrol Patient RBCs + Patient serum
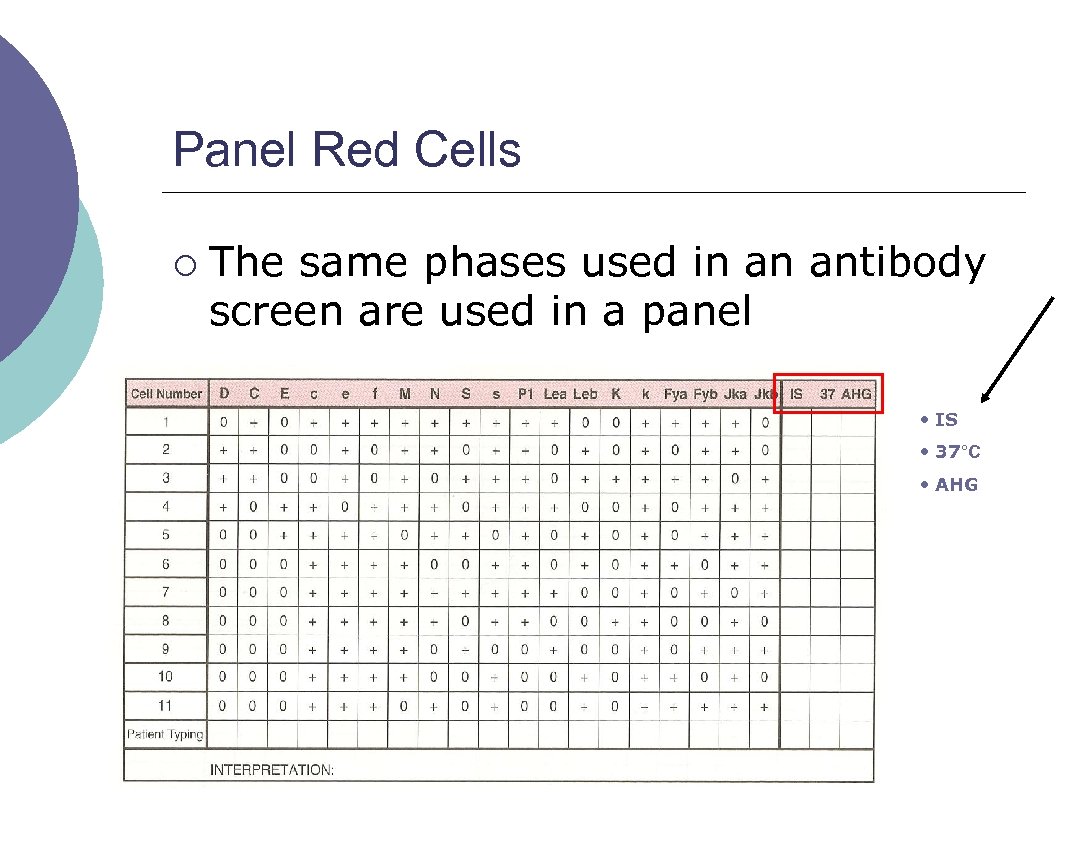
Panel Red Cells ¡ The same phases used in an antibody screen are used in a panel • IS • 37°C • AHG
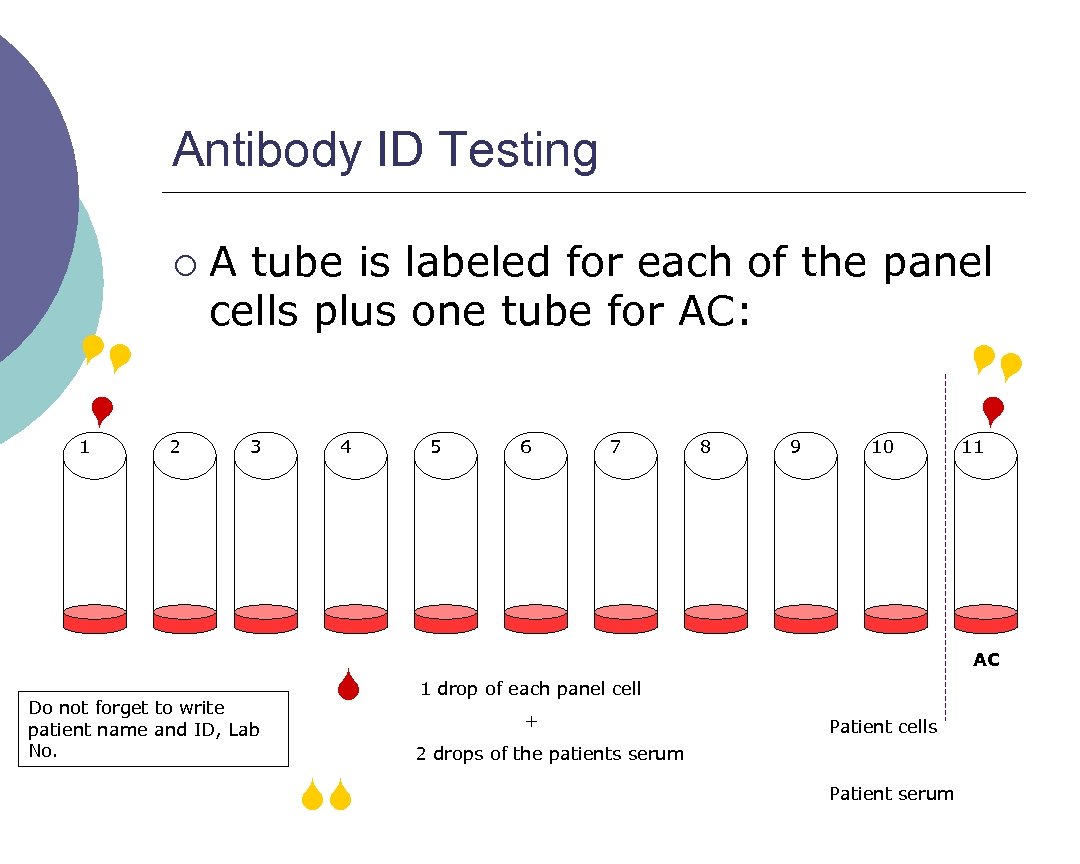
Antibody ID Testing 1 2 A tube is labeled for each of the panel cells plus one tube for AC: 3 Do not forget to write patient name and ID, Lab No. 4 5 6 7 8 9 10 11 AC 1 drop of each panel cell + Patient cells 2 drops of the patients serum ¡ Patient serum

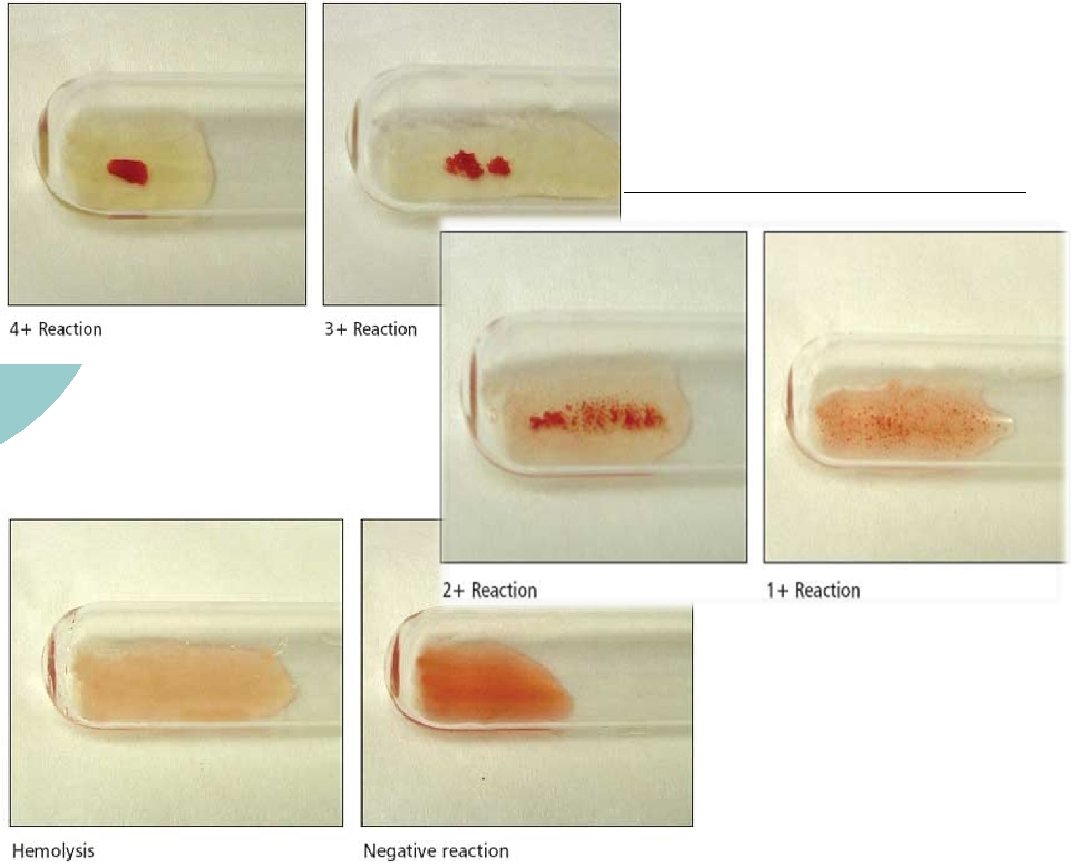
IS Phase ¡ ¡ Perform immediate spin (IS) and grade agglutination; inspect for hemolysis Record the results in the appropriate space as shown: 2+ 0 0 Up to the Last tube

(LISS) 37°C Phase 2 drops of LISS are added, mixed and the mixture is incubated for 10 15 minutes ¡ Centrifuge and check for agglutination ¡ Record results as previous but now fill the 37°C lane. ¡
Agglutination Viewer
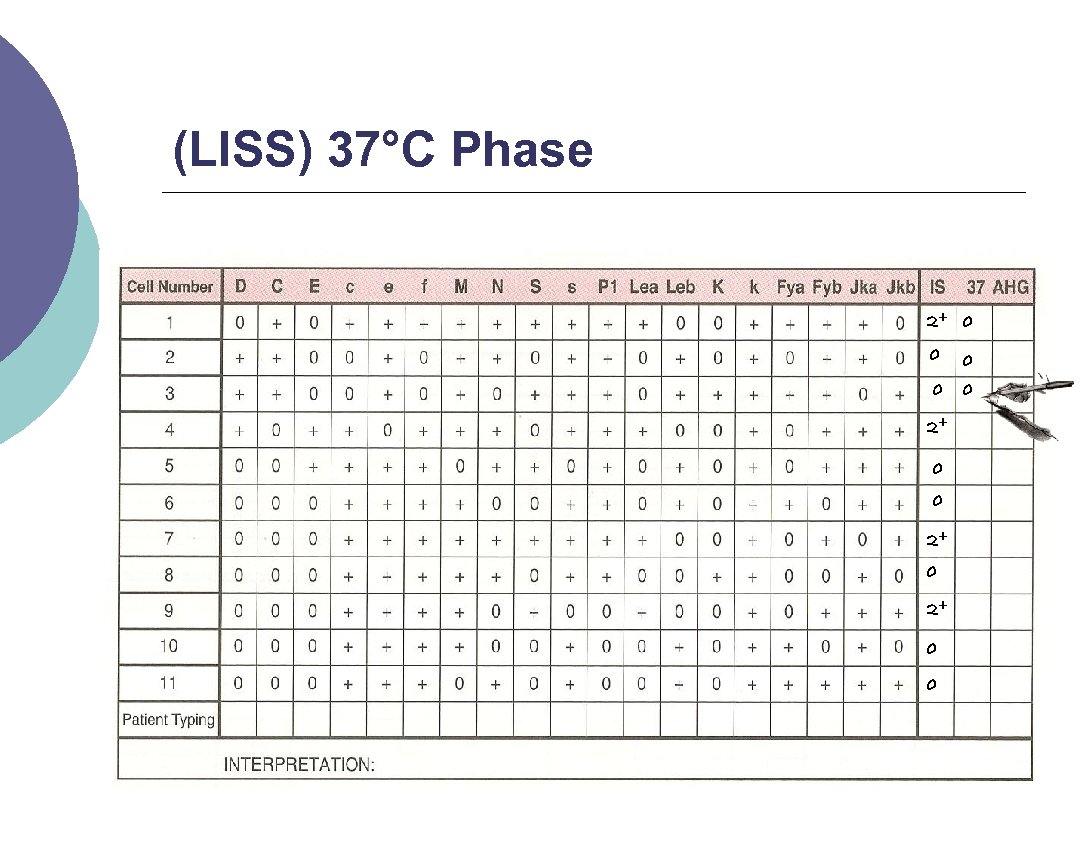
(LISS) 37°C Phase 2+ 0 0 0 2+ 0 0

IAT Phase (or AHG) Indirect Antiglobulin Test (IAT) – we’re testing whether or not possible antibodies in patient’s serum will react with RBCs in vitro ¡ To do this we use the Anti Human Globulin reagent (AHG) ¡ l l l Polyspecific AHG Monospecific Anti Ig. G Monospecific Anti Complement

IAT (AHG) Phase Wash red cells 3 X with saline (manual or automated (cell washer)) ¡ Add 2 drops of AHG and gently mix ¡ l l l Centrifuge Read for agglutination Record reactions
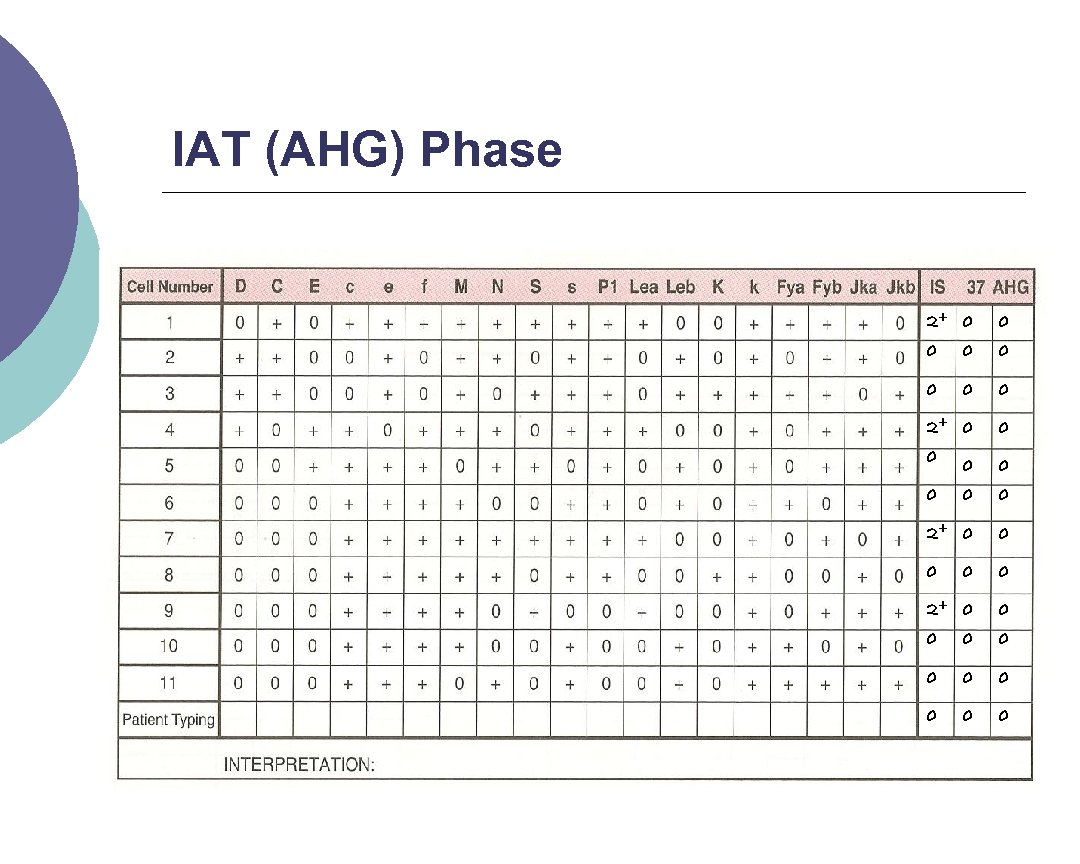
IAT (AHG) Phase 2+ 0 0 0 0 2+ 0 0 0 0

And don’t forget…. …. add “check” cells to any negative AHG !
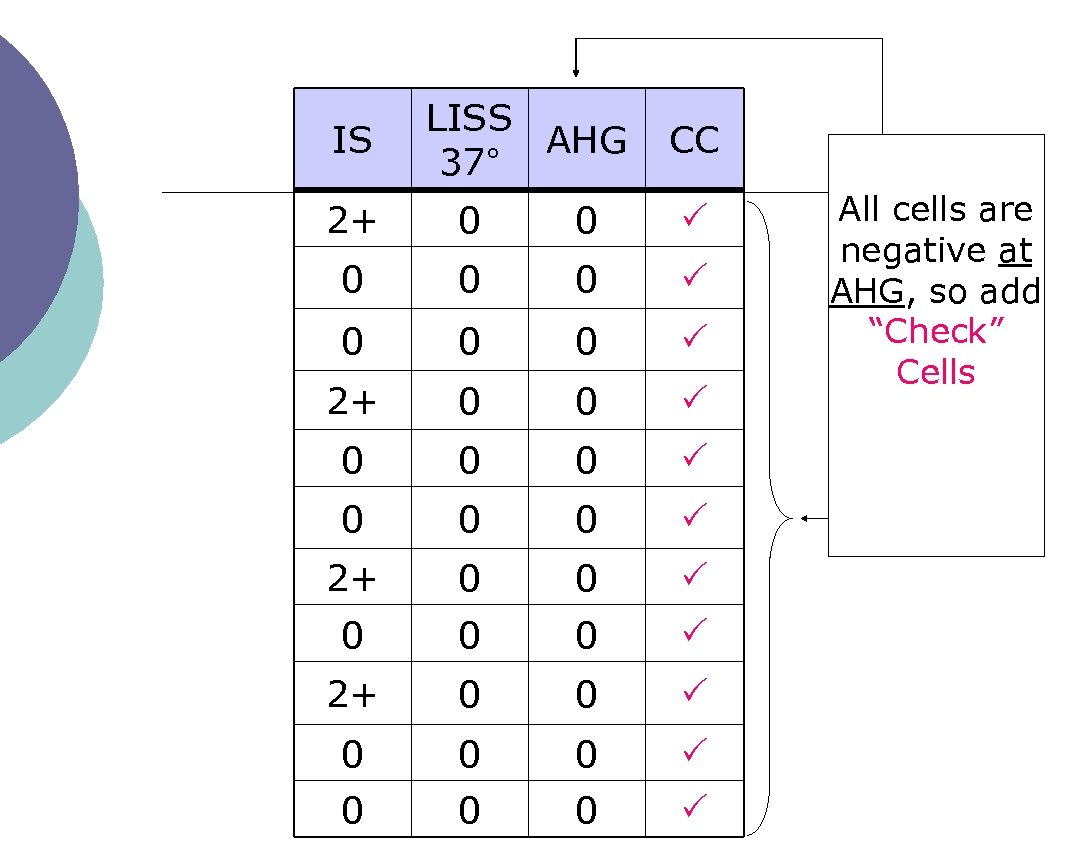
IS LISS AHG 37° CC 2+ 0 0 0 0 0 0 0 0 All cells are negative at AHG, so add “Check” Cells

You have agglutination…now what? CC 2+ 0 0 0 0 0 0 0 0 ? ?

Interpreting Antibody Panels ¡ There a few basic steps to follow when interpreting panels 1. 2. 3. 4. “Ruling out” means crossing out antigens that did not react Circle the antigens that are not crossed out Consider antibody’s (from the circled) usual reactivity Look for a matching pattern

Always remember: An antibody will only react with cells that have the corresponding antigen; antibodies will not react with cells that do not have the antigen

Here’s an example:
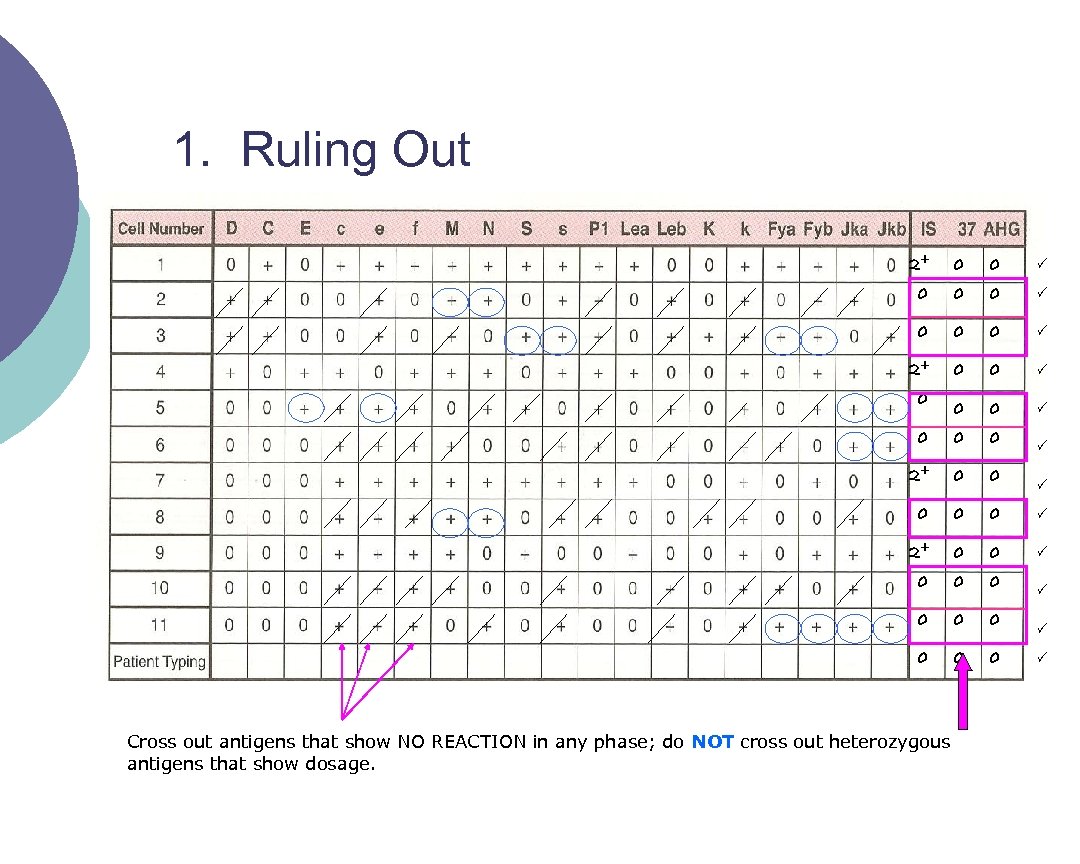
1. Ruling Out 2+ 0 0 0 0 2+ 0 0 0 0 Cross out antigens that show NO REACTION in any phase; do NOT cross out heterozygous antigens that show dosage.
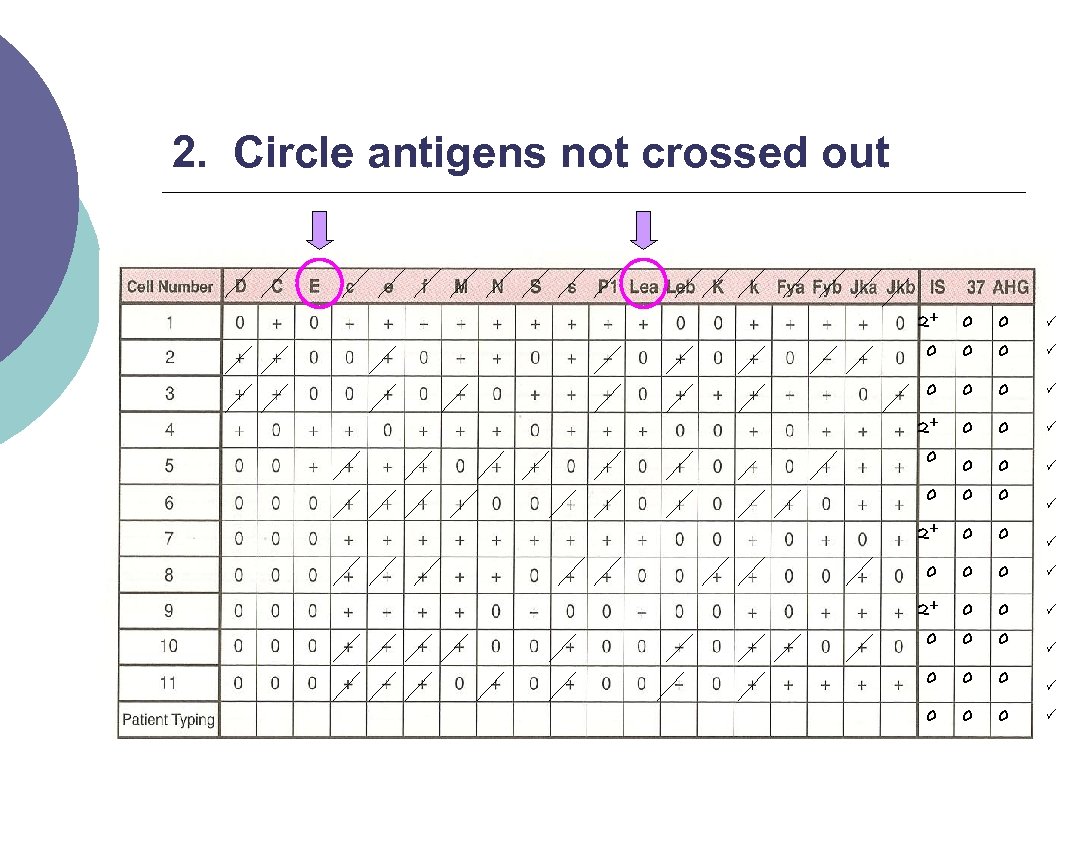
2. Circle antigens not crossed out 2+ 0 0 0 0 2+ 0 0 0 0
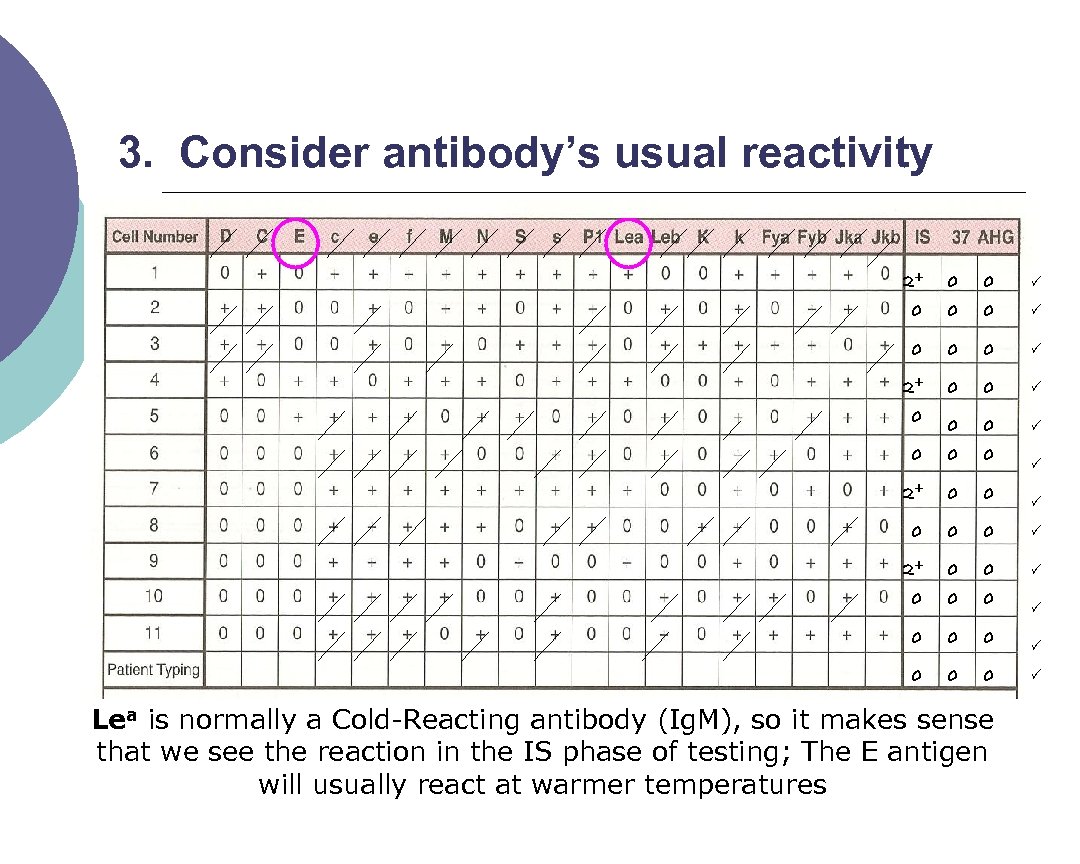
3. Consider antibody’s usual reactivity 2+ 0 0 0 0 2+ 0 0 0 0 Lea is normally a Cold Reacting antibody (Ig. M), so it makes sense that we see the reaction in the IS phase of testing; The E antigen will usually react at warmer temperatures
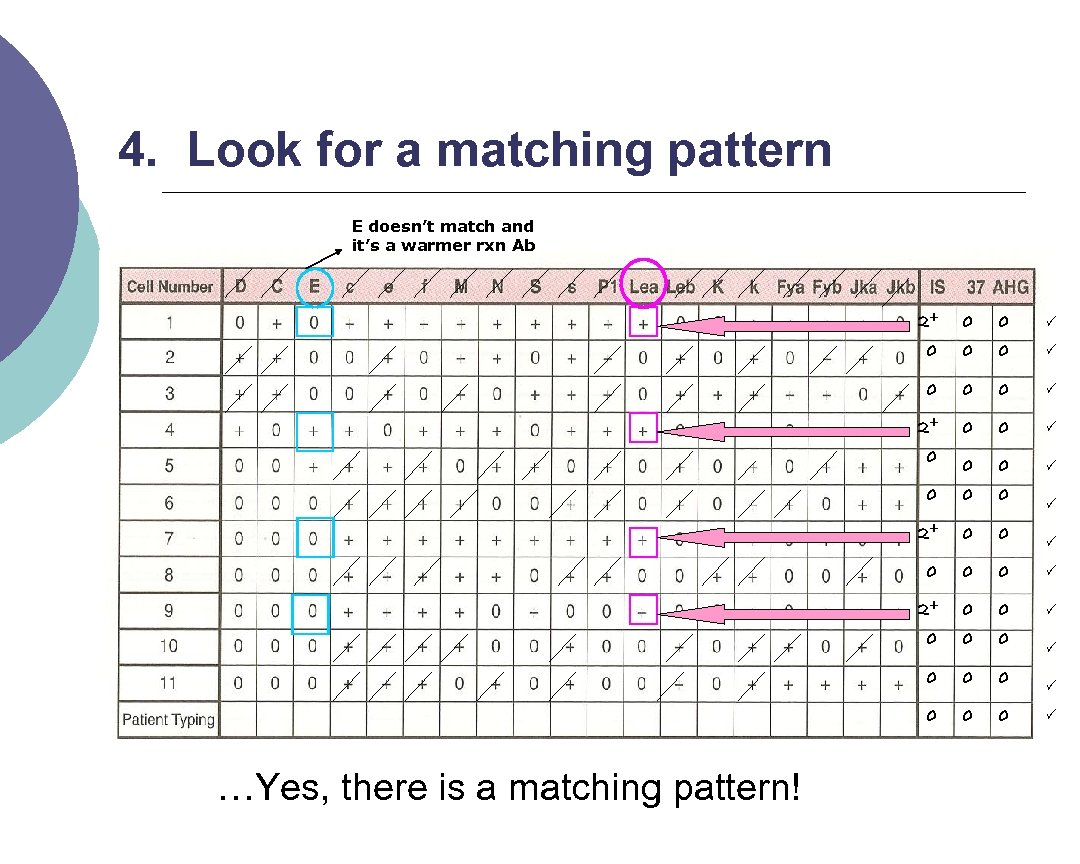
4. Look for a matching pattern E doesn’t match and it’s a warmer rxn Ab 2+ 0 0 0 0 2+ 0 0 0 0 …Yes, there is a matching pattern!
Interpretation: antia Le

Guidelines ¡ Again, it’s important to look at: l Autocontrol ¡ ¡ l Phases ¡ ¡ ¡ l Negative alloantibody Positive – autoantibody or DTR (i. e. alloantibodies) IS – cold (Ig. M) 37° cold (some have higher thermal range) or warm reacting AHG – warm (Ig. G)…significant!! Reaction strength ¡ ¡ 1 consistent strength – one antibody Different strengths – multiple antibodies or dosage

About reaction strengths…… ¡ Strength of reaction may be due to “dosage” l l ¡ If panel cells are homozygous, a strong reaction may be seen If panel cells are heterozygous, reaction may be weak or even non reactive Panel cells that are heterozygous for an antigen should not be crossed out because antibody may be too weak to react (see previous example)

Guidelines (continued) ¡ Matching the pattern l l Single antibodies usually shows a pattern that matches one of the antigens (see previous panel example) Multiple antibodies are more difficult to match because they often show mixed reaction strengths

Rule of three The rule of three must be met to confirm the presence of the antibody ¡ A p value ≤ 0. 05 must be observed ¡ This gives a 95% confidence interval ¡ How is it demonstrated? ¡ l Patient serum MUST be: Positive with 3 panel cells with the antigen, +ve reaction. ¡ Negative with 3 cells without the antigen and should not be reacting. ¡
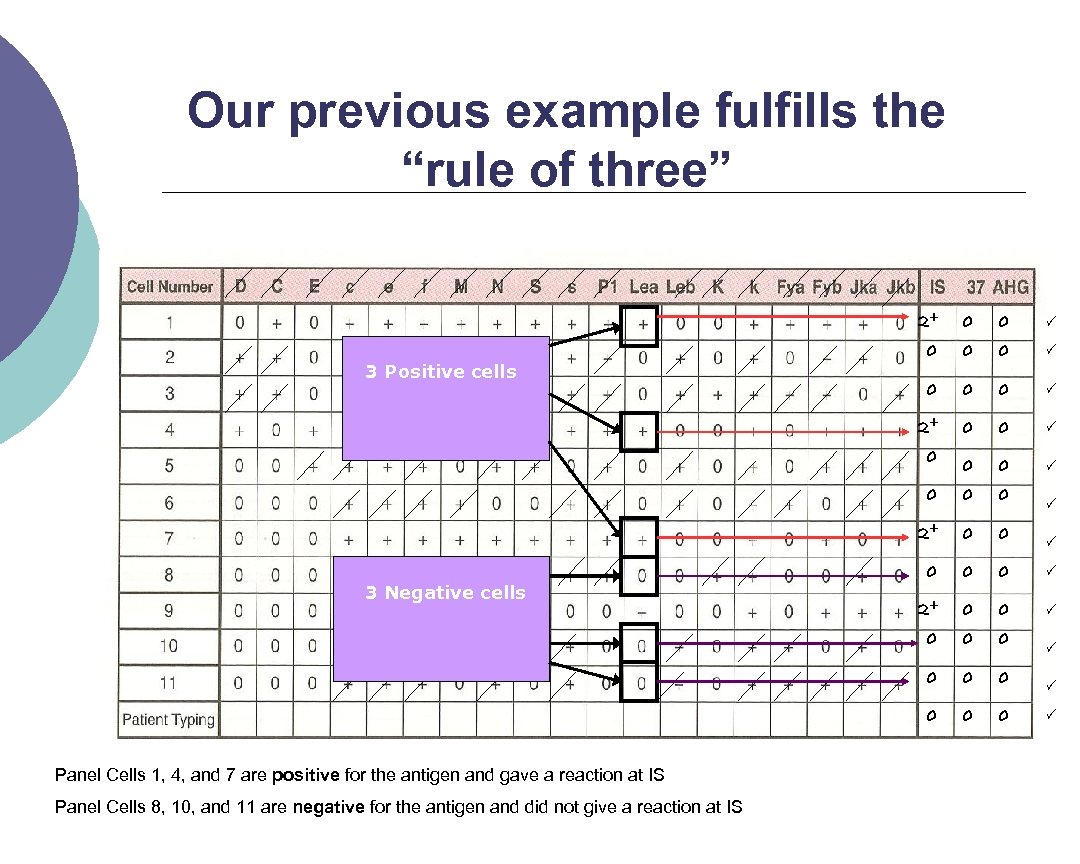
Our previous example fulfills the “rule of three” 2+ 0 0 0 0 0 0 0 3 Positive cells 0 0 3 Negative cells Panel Cells 1, 4, and 7 are positive for the antigen and gave a reaction at IS Panel Cells 8, 10, and 11 are negative for the antigen and did not give a reaction at IS

What if the “rule of three” is not fulfilled? If there are not enough cells in the panel to fulfill the rule, then additional cells from another panel could be used ¡ Most good (I do not know if we are good or not!!) labs carry different lot numbers of panel cells ¡

Patient Antigen Typing (Phenotyping) In addition to the rule of three, antigen typing the patient red cells can also confirm an antibody ¡ How is this done? ¡ l l Only perform this if the patient has NOT been recently transfused (donor cells could react (chimera)). If reagent antisera (of the suspected antibody) is added to the patient RBCs, a negative reaction should result…Why?

Remember Landsteiner’s Rule Individuals DO NOT make allo antibodies against antigens they have

Multiple antibodies may be more of a challenge than a single antibody ¡ Why? ¡ l l Reaction strengths can vary Matching the pattern is difficult

So what we have to do? ¡ Several procedures can be performed to identify multiple antibodies l l l Selected Cells Neutralization Chemical treatment Proteolytic enzymes ¡ Sulfhydryl reagents ¡ ZZAP ¡

1 - Selected Red Cells Selected cells are chosen from other panel or screening cells to confirm or eliminate the antibody. ¡ The cells are “selected” from other panels because of their characteristics. ¡ The number of selected cells needed depends on how may antibodies are identified. ¡

Selected Red Cells …. . Cont’d Every cell should be positive only for each of the antibodies and negative for the remaining suspicious antibodies ¡ For example: ¡ l l Let’s say you ran a panel and identified 3 different antibodies (you cannot rule out): anti. S, anti-Jka, and anti-P 1 Selected cells could help…
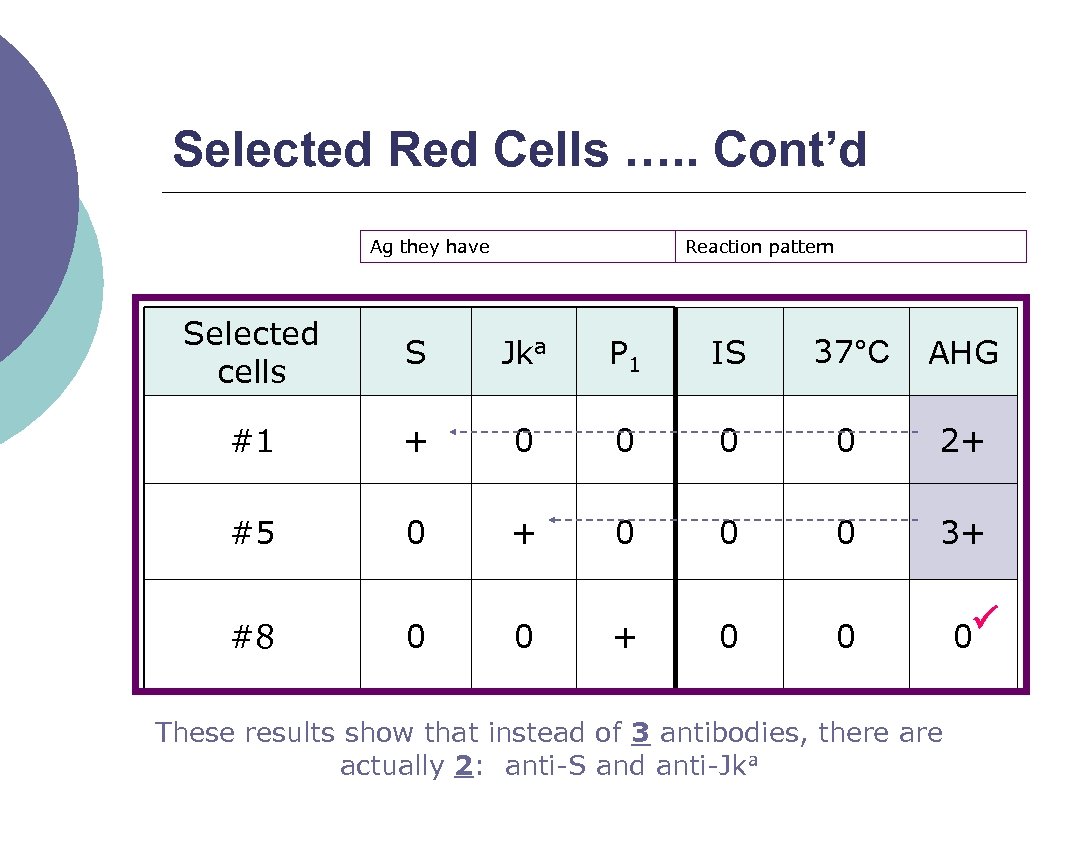
Selected Red Cells …. . Cont’d Ag they have Reaction pattern Selected cells S Jka P 1 IS 37°C AHG #1 + 0 0 2+ #5 0 + 0 0 0 3+ #8 0 0 + 0 0 These results show that instead of 3 antibodies, there actually 2: anti S and anti Jka 0

2 - Neutralization Some antibodies may be neutralized as a way of confirmation ¡ Commercial “substances” bind to the antibodies in the patient serum, causing them to show no reaction when tested with the corresponding antigen (in panel) ¡

Neutralization …. . Cont’d ¡ Manufacturer’s directions should be followed and a positive dilutional control should always be used l l The positive dilutional control contains saline and serum (no substance is added) and should remain positive with panel cells. A control shows that a loss of reactivity is due to the neutralization and not to the dilution of the antibody strength when the substance is added

Neutralization …. . Cont’d ¡ Common substances l l l P 1 substance (derived from hydatid cyst fluid) Lea and Leb substance (soluble antigen found in plasma and saliva) I substance can be found in breast milk **you should be aware that many of these substances neutralize COLD antibodies; Cold antibodies can sometimes mask more clinically significant antibodies (Ig. G), an important reason to use neutralization techniques

3 - Again: Proteolytic Enzymes Can be used to enhance or destroy certain blood group antigens ¡ Several enzymes exist: ¡ l l l ¡ Ficin (figs) Bromelin (pineapple) Papain (papaya) In addition, enzyme procedures may be l l One step Two step

Enzymes ¡ Enzymes remove the sialic acid from the RBC membrane, thus “destroying” it and allowing other antigens to be “enhanced” Antigens destroyed: M, N, S, s, Duffy ¡ Antigens enhanced: Rh, Kidd, Lewis, I, and P ¡

Enzyme techniques ¡ One stage l ¡ Enzyme is added directly to the serum/panel cell mixture Two stage l l Panel cells are pre treated with an enzyme, and washed Patient serum is added to treated panel cells and tested

Enzyme techniques ¡ If there is no agglutination after treatment, then it is assumed the enzymes destroyed the antigen
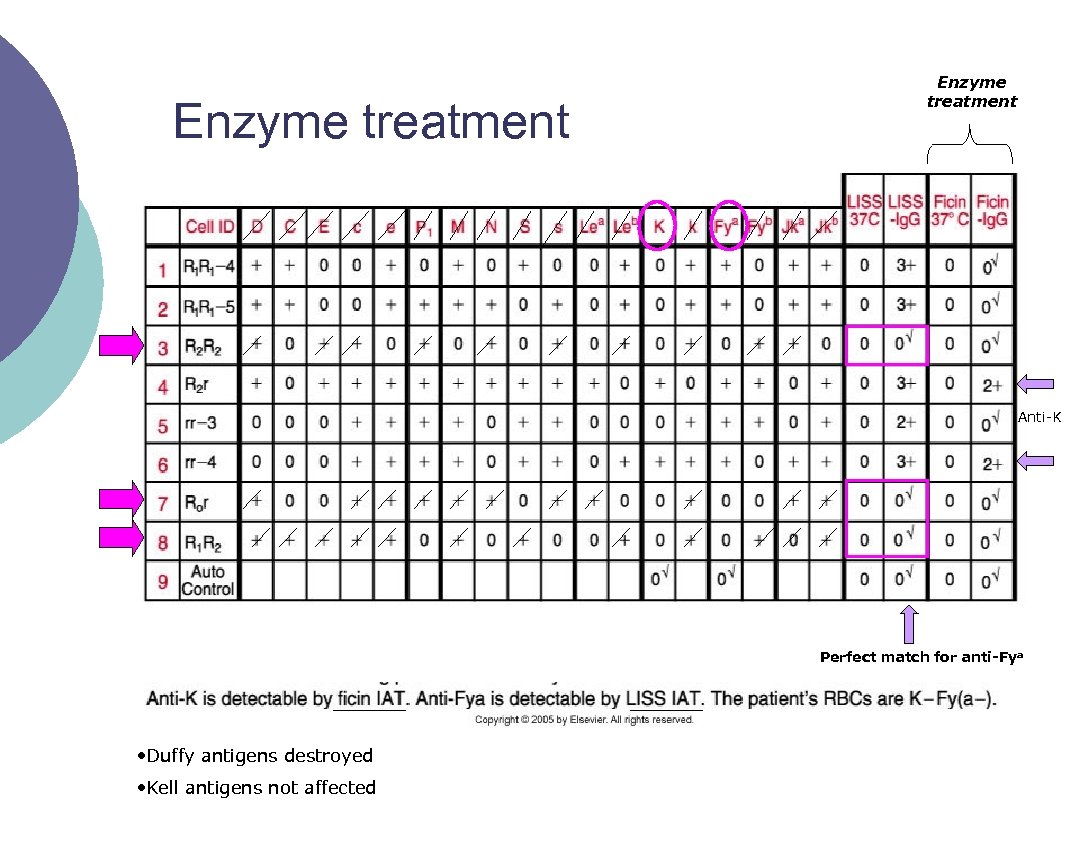
Enzyme treatment Anti K Perfect match for anti-Fya • Duffy antigens destroyed • Kell antigens not affected

Summary If an unexpected antibody is detected in a patient’s serum or plasma it must be identified. ¡ Once identified the clinical significance must be determined. ¡

Summary If the antibody is clinically significant antigen negative donors must be found and crossmatched for the patient, a Coomb’s crossmatch must be done. ¡ If the antibody is not clinically significant it is not necessary to provide antigen negative blood, but the donors must be compatible by the Coomb’s crossmatch. ¡

Providing Compatible Donor Units ¡ ¡ Once an antibody has been identified, the next task is to provide appropriate units of RBCs for transfusion. When clinically insignificant antibodies are detected, use of crossmatch compatible RBCs is appropriate.

Providing Compatible Donor Units ¡ ¡ No further testing is needed to confirm compatibility when the antibody is anti M, anti N, anti Pi. Lea, or Leb. However, when a clinically significant antibody is identified, the blood must be cross match compatible and confirmed as antigen negative with reagent antisera.

Example ¡ ¡ Knowledge of the incidence of antigens is useful for determining how many units of blood to screen or cross match for patients with antibodies. If a patient with an anti Jk(a ) needed 4 units of blood, how many units would need to be tested to find them? Jk (a+)= 0. 77 Jk (a ) = 0. 23 4 units Jk(a-) blood needed = 17. 4 units 0. 23 incidence of Jk(a-) In this case, testing 17 or 18 random units should yield 4 Jk(a ) units.

Example 2 ¡ ¡ The same calculations can be used when multiple antibodies are present if the antigen frequencies are first multiplied together. E. g. a patient with an anti K and anti Jka, 10 random units would need to be tested to find 2 that are compatible. Jk(a+) = 0. 77 K positive = 0. 09 Jk(a ) = 0. 23 K negative = 0. 91 ¡ Jk(a-) (0. 23) X K negative (0. 91) = 0. 20 Jk(a-) and K negative ¡ 2 units needed = 10 units 0. 20 Jk(a-) and Kell negative

THE END!!
a04d522e5bddd29ded79b93838f0ce1d.ppt