dd8c706d073695b68ca95e1aae8e777e.ppt
- Количество слайдов: 57

ANSI/AAMI ST 91: 2015 Flexible and semi-rigid endoscope processing in health care facilities Cathy Rocco, RN, MSN, CNOR 039877 -150908

Disclaimer This presentation reflects the techniques, approaches and opinions of individual presenter. This Advanced Sterilization Products (“ASP”) sponsored presentation is not intended to be used as a training guide. Before using any medical device, review all relevant package inserts with particular attention to the indications, contraindications, warning and precautions, and steps for the use of the device(s). I, Cathy Rocco, am compensated by and presenting on behalf of ASP, and must present information in accordance with applicable FDA requirements. Third party trademarks used herein are trademarks of their respective owners. 039877 -150908 page 2

Objectives: By the end of this presentation, the learner should be able to: • Understand ST 91 as it relates to Flexible Endoscope Reprocessing • Determine best practices for your facility utilizing national standards and recommended practices • List the differences between AAMI ST 91 and other national standards and guidelines 039877 -150908 page 3

AAMI • The Association for the Advancement of Medical Instrumentation (AAMI), founded in 1967, is a nonprofit, international membership organization dedicated to improving the safety and efficacy of medical instrumentation and healthcare technologies • AAMI is the recognized developer of medical device standards worldwide – managing over 150 technical committees and publishing more than 100 technical documents 039877 -150908 page 4

AAMI’s Membership Industry representatives Statewide biomedical societies page 5 Government agency representatives (FDA) Individuals/Users Students Professional organizations (AORN, IAHCSMM, APIC, SGNA)

American National Standards Institute (ANSI) • Formed in 1918 from 5 engineering societies and 3 government agencies • Coordinates and promotes consensus of U. S. standards of American products used worldwide • Does not develop standards – accredits the procedures for openness, balance, consensus and due process 039877 -150908 page 6

Understanding Recommended Practices Standards and Recommended Practices (RP) can contribute significantly to the enhancement of cleaning and reprocessing medical instruments. By definition: “A recommended practice provides guidelines for the use, care and/or processing of a medical device or system” (AAMI, 2015). RPs enhance procedures and practices that will help ensure a device is used safely and effectively and that its performance can be maintained. 039877 -150908 page 7

Common Terminology in AAMI Standards Shall: Indicates requirements to be followed strictly to conform to the standard Should: Indicated that among several possibilities one is recommended as particularly suitable but does not exclude others 039877 -150908 page 8

Common Terminology (continued) May: Used to indicate that a course of action is permissible within the limits of the standard Can: Used as a statement of possibility and capability Must: Used only to describe “unavoidable” situations including those mandated by government regulation 039877 -150908 page 9

Why Develop AAMI National Standard for Flexible Endoscope Reprocessing? At least 11 million gastrointestinal endoscopies are performed each year and the number of procedures is increasing (Cullen et al, 2009; SGNA, 2012). • Risks of failures… • • • Breach of host barriers Person-to-person transmission of pathogens Environmental pathogens Device damage Inefficient use of the device Toxic reactions in patients 039877 -150908 page 10

Why develop ? (continued) This document specifically addresses: • Functional and physical design criteria for endoscope reprocessing area • Education, training, competency verification, and other personnel considerations • Processing recommendations • Installation, care, and maintenance of AERs • Quality control and quality process improvement 039877 -150908 page 11

Design of Endoscope Reprocessing Area Space “should” facilitate both infection prevention and patient and employee safety. • Work Flow (Unidirectional) & Traffic Flow • • Negative pressure/positive pressure/air flow/ventilation Appropriate lighting to facilitate proper inspection Eye wash station locations Required utilities – medical grade air, water quality • Patient Volume/types of endoscopes • Quantity and type(s) of processing equipment • Location of AER • Scopes/equipment storage requirements • Temperature/humidity monitoring • Supply/chemical storage requirements 039877 -150908 page 12

Personnel Guidelines Provides guidelines for policies and procedures including: • Education • Certification “recommended” • Training and competency verification • Hand hygiene criteria • Immunizations • Hepatitis B • Attire • Proper PPE • No exam gloves for decontamination 039877 -150908 page 13

Training, Competency & Education “Should” be completed… • Upon initial hire • Annually • At designated intervals • When new endoscope models, updated IFUs, processing equipment or chemicals are introduced 039877 -150908 page 14

Who can train employees? Facility personnel providing orientation, competencies or training “should”: • Complete facility-specific education and competency verification related to the role of “educator” • Maintain competence including new technologies • Use regulatory and evidence-based professional guidelines • Participate in ongoing activities related to education of personnel • Periodically re-educate and re-assess competency of processing personnel with documentation of training 039877 -150908 page 15

Education and Training “should” include… • Procedures for cleaning, disinfecting or sterilizing, packaging and storing endoscopes including connectors for cleaning/AER connections • Knowledge of and access to current National Standards • Decontamination processes • Operation of automated processes and equipment • Workplace safety • Documentation of quality monitoring results 039877 -150908 page 16

039877 -150908 page 17

Hand Hygiene is not only hand washing… • P & P “should” be developed and communicated • Co-written/Co-approved through IP • Fingernails “should” be short and clean…no artificial nails or nail tips • Each facility “should” develop their own policy regarding nail polish as this subject remains unresolved and requires further studies 039877 -150908 page 18

Personnel Attire All personnel “should” wear clean uniforms provided by and donned at the facility and laundered by a health care-accredited facility • Changed daily or as needed and not worn outside facility • All head and facial hair, except eyelashes and eyebrows, should be covered with a surgical type hair covering • No jewelry or wrist watches • Shoes should have non-skid soles and sturdy enough to prevent injury if something is dropped on the foot • Cover apparel “should” be worn when outside the processing area 039877 -150908 page 19

Cleaning and High-Level Disinfection (HLD) “Meticulous” attention to all steps is critical • Pre-cleaning at the point of use • Transporting • Leak testing (wet/dry per manufacturers IFU) • Cleaning • Rinsing • Inspecting or testing for cleaning verification • HLD and monitoring the process • Rinsing • Drying and alcohol flush • Storage 039877 -150908 page 20

Transportation of Dirty Endoscopes Each endoscope should be isolated and transported to the processing area • Maintain components of the scope with the scope • Transport in a closed system • System should be marked as biohazard meeting OSHA guidelines • System should be large enough to loosely coil endoscope • Forceps/wires/sharp objects should be transported in a separate container 039877 -150908 page 21

Automated Endoscope Reprocessors (AER) Designed for cleaning and HLD for endoscopes and components. • More efficient than manual processing • Standardizes required rinses for chemicals • If cycle is interrupted, repeat entire cycle • Quality of rinse water varies and “may” re-contaminate endoscopes • Ensure quality testing of AER is performed according to manufacturer’s IFU 039877 -150908 page 22

Qualities to consider when purchasing an AER • What features does the AER provide? • Is it FDA cleared? • Can it reprocess devices in your current inventory? • What accessories can the AER process? • Is the HLD/LCS solution FDA cleared? • Do I have enough space for the AER? • Specific utilities/specifications/circulation? • Recommended preventive maintenance? • Who is certified to perform PM? 039877 -150908 page 23

Qualities to Consider (continued) • Does the AER have cleaning claims? • Is it clear how those cleaning claims are applied to current practice? Are there limitations to the cleaning claim? • Proper rinses per HLD/LCS IFU? • Self-disinfect cycle? • Drying/alcohol flush capabilities? • Built in safety alarms? • MEC Verification? • Data verification of each cycle performed? • Printout with required documentation of each completed cycle 039877 -150908 page 24

Processing Endoscope Accessories Ex: air/water/suction valves, biopsy port covers, water bottles, tubing, etc. • The same standard of inspection, cleaning and HLD is “required” • Reusable devices that break the mucosal barrier “require” mechanical cleaning and sterilization • Components disassembled and cleaned • Water bottles per the manufacturer’s IFU or at least daily 039877 -150908 page 25

Storage of Reprocessed Endoscopes • Hung vertically with distal tip hanging freely • Well-ventilated, clean area • Sufficient space between/around other endoscopes • Angulation in open position • Removable parts detached from endoscope AORN (2016) “recommends” endoscopes stored in a closed cabinet with venting without touching the bottom of the cabinet and sufficient space for storage of multiple endoscopes without touching. 039877 -150908 page 26

Storage (continued) • Temperature and humidity “should” be monitored where endoscopes are stored • Do not use manufacturer-provided case for storage • Internal channels “should” be dry with medical grade forced air (appropriate PSI according to IFU) • Detachable parts “should” be stored as a unique set with specific endoscopes for traceability • Valves “should” be dried and lubricated according to the manufacturer’s IFU • If sterilized, store in container/packaging they were sterilized in with an agreed upon maximum shelf life 039877 -150908 page 27

Have you ever felt like this? ? ? How am I ever going to remember all these steps? ? ? 039877 -150908 page 28

How do you know if an endoscope is clean and ready for patient use? CDC “recommends” a policy and procedure ensuring the end user can determine whether a scope has been cleaned and HLD or sterilized. ● For quality assurance, a tag or label “should” be attached to each endoscope and “should” include: • Date of processing • Name(s) of personnel who performed the processing • Date of HLD/sterilization Now for the question that is on everyone’s mind……. 039877 -150908 page 29

“Hang Time” for HLD Endoscopes “The accepted time interval for the storage of processed endoscopes before they can no longer be considered safe for patient use is not well defined” (AAMI, 2015). • Limited number of studies • “Available data suggests risk of contamination is “negligible” when storage is done according to the endoscope manufacturer’s IFU and/or standards related to endoscope processing” (AAMI, 2015). • Currently there are no studies that show prolonged “hang time” as a risk factor to patient safety 039877 -150908 page 30

Existing “Hang Time” Guidelines • SGNA standards “recommend” reprocessing after 7 days • Veterans Affairs follows a directive to reprocess after 12 days of hang time • Gastro Assoc of Australia when 72 hours has elapsed, except for duodenoscopes, bronchoscopes and linear EUS scopes when 12 hours has elapsed. Enteroscopes when 12 hours has elapsed if no continuous air flow provided during storage • New Zealand, Scotland Ireland – up to 72 hours if stored correctly • Canadian Standards Assoc – “should” be reprocessed if storage exceeds 7 days 039877 -150908 page 31

Risk Assessment for Maximum “Hang Time” Guidelines and RPs provide recommendations for maximum duration of storage times before next patient use. • • Complexity and type of endoscope Condition of endoscope after processing Transportation method from processing to storage Condition of storage environment Manufacturer’s IFU for storage Research studies Patient population Quality of final rinse water 039877 -150908 page 32

Risk Assessment (continued) • Based on the risk assessment, each facility “should” develop policies and procedures to address maximum hang times. • Each facility “should” address specifics of what to do when maximum hang times have been exceeded. • Limited data for definitive answers to best practices so…. . Do what works best for your facility…. follow your policy and procedure once established! 039877 -150908 page 33
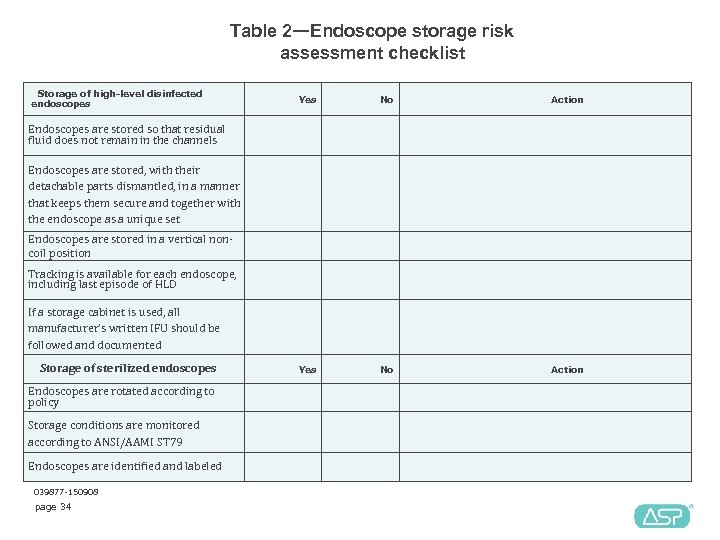
Table 2—Endoscope storage risk assessment checklist Storage of high-level disinfected endoscopes Endoscopes are stored so that residual fluid does not remain in the channels Yes No Action Endoscopes are stored, with their detachable parts dismantled, in a manner that keeps them secure and together with the endoscope as a unique set Endoscopes are stored in a vertical non coil position Tracking is available for each endoscope, including last episode of HLD If a storage cabinet is used, all manufacturer's written IFU should be followed and documented Storage of sterilized endoscopes Yes No Action Endoscopes are rotated according to policy Storage conditions are monitored according to ANSI/AAMI ST 79 Endoscopes are identified and labeled 039877 -150908 page 34

Routine Testing of Stored Endoscopes Numerous incidents of contaminated endoscopes have been traced to a failure in processing… • User errors related to HLD procedures and/or drying procedures • Physically compromised endoscopes or AERs • Contaminated water supply during processing 039877 -150908 page 35

Routine Testing (continued) • Currently no universal consensus of the value of routine testing • Numerous studies have identified non-environmental pathogens likely to be found in improperly reprocessed endoscopes • Involve Risk Management, Laboratory and Infection Prevention as part of a Quality Assurance program • At the time of printing for this document, AAMI is aware of the CDC considering microbial sampling of endoscopes 039877 -150908 page 36

Quality Control • Product identification and traceability • Documentation and record-keeping • Verification and monitoring of the cleaning process adhering to national standards and guidelines • Monitoring of HLD/sterilization • Personnel performance and work practices • Product recalls • Quality process improvement 039877 -150908 page 37

Quality Assurance & Safety Programs At a minimum, facilities “should” establish comprehensive quality assurance programs to include but not limited to: • Identity of personnel involved • Identify all areas within facility that process endoscopes • Keep accurate inventory records of equipment • Identify storage location • Compatibility of endoscopes with reprocessing protocols (AERs) 039877 -150908 page 38

Quality Assurance (continued) • Visual inspections and testing of equipment • Process monitors for equipment • Documentation for patients and personnel • Education, training and competency verification programs 039877 -150908 page 39

Documentation • AER ID, soaking container and cycle number • Load contents • Patient’s name and unique patient identifier • Procedure, physician, serial number of item • Shelf-life date, lot # and date opened of HLD/LCS 039877 -150908 page 40

Documentation (continued) • Exposure time and temperature • Date and time of cycle, HLD/LCS type and concentration • Name or initials of operator • Results of MEC/BI/CI/spore test strip/quality control of test strips 039877 -150908 page 41

Verification and Monitoring of the Cleaning Process Several technologies have the capability of measuring residual organic soil and microbial contamination. Consideration “should” be given to the use of an appropriate monitoring verification process depending on the IFU of the product used Published studies have evaluated specific markers to determine cleaning efficacy… ü ü ü Proteins Carbohydrates Hemoglobin Adenosine Triphosphate (ATP) Enzyme that detects specific bacteria 039877 -150908 page 42

Verification (continued) • Basic components should include establishing a reasonable benchmark and rapid, easy-to-perform reliable methods. • Benchmarks “may” become more definitive as more data becomes available. • Each facility “should” formulate policies and procedures for verification of cleaning efficacy. • Frequency of testing “should” occur on a regular basis, weekly or preferably daily (Drosnock 2014, Alfa 2014). 039877 -150908 page 43

Inadequate Processing and Recalls • Supervisor, Risk Manager and/or Infection Prevention notification • Time & date of cycle in question • Identification of HLD modality • CI and any other useful information for validity 039877 -150908 page 44

Inadequate Processing (continued) • Root cause analysis • Remove equipment from service if necessary • Determine if recall is necessary • Have written protocol in place for recall 039877 -150908 page 45

Recall Procedure, Order and Summary A recall procedure “should”: • Be written • Outline the circumstances for issuing a recall order • Designate the person/people authorized to issue a recall order; and • Designate responsible personnel for reporting on the execution of a recall order 039877 -150908 page 46

Recall Order “should”: • Include all items processed back to the last negative BI or failed MEC indicator (if applicable) • Be immediately communicated to affected areas and followed by a written order • Identify products to be recalled by lot number, product or patient name or other information • Identify the people or areas to whom the order is addressed 039877 -150908 page 47

Recall Order (continued) • Require the recording, kind and quantity, or products in the recall, and • Specify the action to be taken by the people receiving the order. Outline the circumstances for issuing a recall order • Designate the person/people authorized to issue a recall order; and • Designate responsible personnel for reporting on the execution of a recall order 039877 -150908 page 48

Recall Summary Report “should”: • Identify the circumstances that prompted the recall order • Specify the corrective actions taken to prevent a recurrence • State the percentage of products actually in the recall in terms of number of products intended to be recalled, and • Provide verification that the recalled items were reprocessed or destroyed 039877 -150908 page 49

Risk Analysis = Risk Assessment + Risk Management + Risk Communication Risk Assessment – Identification of the source of a failure, assessing the consequences and how prepared a facility will be to manage the failure. Risk Management – Determining which failures require management and selecting/implementing action plans to control the failures. Risk Communication – Interactive dialogue between SPD personnel, Endoscopy and Infection Prevention actively informing other concerned parties, including patients (recall procedure). 039877 -150908 page 50

Risk Analysis (continued) The Processing Risk Analysis “should” be a part of each facility’s Infection Prevention and Risk Control Analysis. • “Should” be performed at least annually • “Should” be re-evaluated whenever significant changes occur 039877 -150908 page 51

Evaluating and Monitoring Effectiveness of the HLD Process • “Should” be an ongoing effort to maintain control and determine methods for improvement. • “Should” be re-evaluated whenever significant changes occur • “Should” be audited for compliance • Development of a CQI Program to include: • • • Training, continuing education and competency verification Product ID and traceability Monitoring cleaning/HLD/sterilization effectiveness Product testing and product recalls Workplace safety training 039877 -150908 page 52
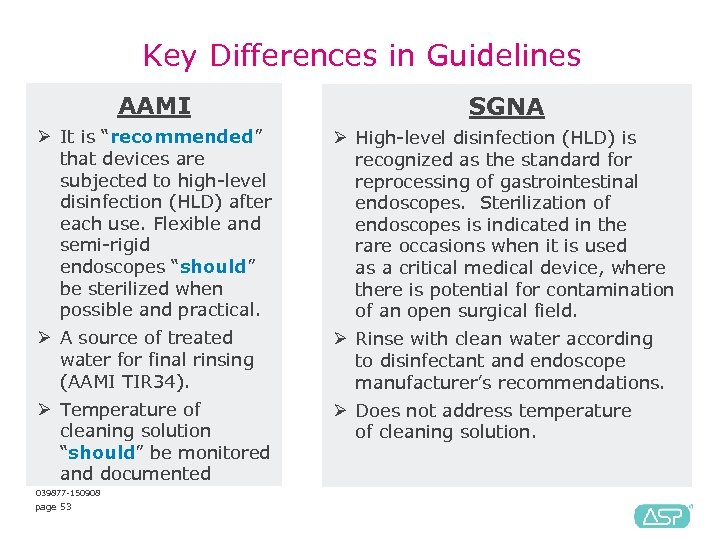
Key Differences in Guidelines AAMI SGNA Ø It is “recommended” Ø High-level disinfection (HLD) is that devices are recognized as the standard for subjected to high-level reprocessing of gastrointestinal disinfection (HLD) after endoscopes. Sterilization of each use. Flexible and endoscopes is indicated in the semi-rigid rare occasions when it is used endoscopes “should” as a critical medical device, where be sterilized when there is potential for contamination possible and practical. of an open surgical field. Ø A source of treated Ø Rinse with clean water according water for final rinsing to disinfectant and endoscope (AAMI TIR 34). manufacturer’s recommendations. Ø Temperature of Ø Does not address temperature cleaning solution of cleaning solution. “should” be monitored and documented 039877 -150908 page 53
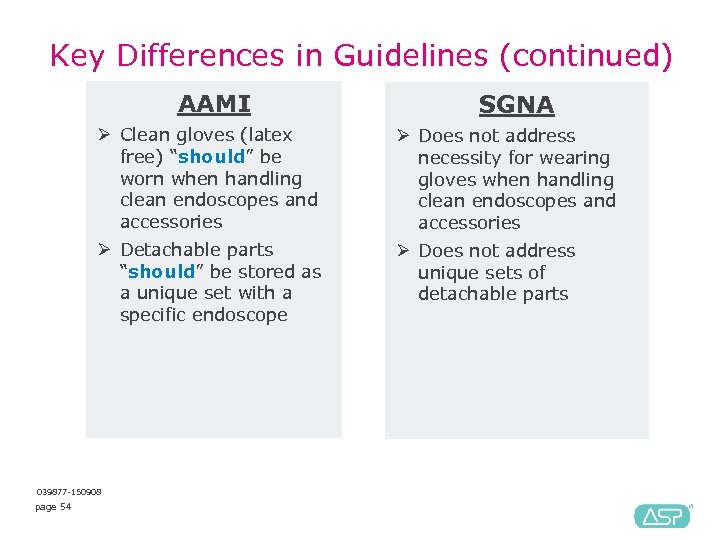
Key Differences in Guidelines (continued) AAMI SGNA Ø Clean gloves (latex free) “should” be worn when handling clean endoscopes and accessories Ø Does not address necessity for wearing gloves when handling clean endoscopes and accessories Ø Detachable parts “should” be stored as a unique set with a specific endoscope Ø Does not address unique sets of detachable parts 039877 -150908 page 54

Questions? 039877 -150908 page 55

039877 -150908 page 56

039877 -150908 page 57 Ethicon US, LLC. 2015
dd8c706d073695b68ca95e1aae8e777e.ppt