452e80dc6f4ab6f8a05d0a688e860c70.ppt
- Количество слайдов: 18
 Animal Health Products Identification Standard A first step towards traceability 1
Animal Health Products Identification Standard A first step towards traceability 1
 What is IFAH? • IFAH is the global representative body of manufacturers of veterinary medicines, vaccines and other animal health products in both developed and developing countries across five continents • IFAH is an international non-profit organisation registered under Belgian law and is based in Brussels, Belgium • IFAH represents both Animal Health Companies and Associations. These associations comprise both local medium-size enterprises (SMEs) and international companies. They represent most of the global market for animal health products 2
What is IFAH? • IFAH is the global representative body of manufacturers of veterinary medicines, vaccines and other animal health products in both developed and developing countries across five continents • IFAH is an international non-profit organisation registered under Belgian law and is based in Brussels, Belgium • IFAH represents both Animal Health Companies and Associations. These associations comprise both local medium-size enterprises (SMEs) and international companies. They represent most of the global market for animal health products 2
 IFAH Mission Statement • The mission of IFAH is to promote a predictable, harmonised, science-based and innovative market place for the provision of quality animal medicines, vaccines and other animal health products. These contribute to a healthy and safe food supply and to a high standard of health and welfare for animals and people 3
IFAH Mission Statement • The mission of IFAH is to promote a predictable, harmonised, science-based and innovative market place for the provision of quality animal medicines, vaccines and other animal health products. These contribute to a healthy and safe food supply and to a high standard of health and welfare for animals and people 3
 IFAH Goal • To ensure that the contribution of the Animal Health Industry to the health and quality of animal and human life through the advancement of sound science is understood by society at large 4
IFAH Goal • To ensure that the contribution of the Animal Health Industry to the health and quality of animal and human life through the advancement of sound science is understood by society at large 4
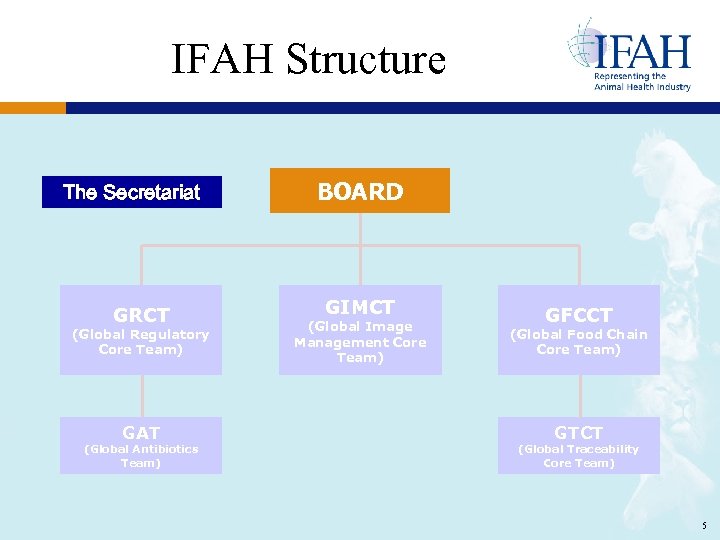 IFAH Structure The Secretariat GRCT (Global Regulatory Core Team) GAT (Global Antibiotics Team) BOARD GIMCT (Global Image Management Core Team) GFCCT (Global Food Chain Core Team) GTCT (Global Traceability Core Team) 5
IFAH Structure The Secretariat GRCT (Global Regulatory Core Team) GAT (Global Antibiotics Team) BOARD GIMCT (Global Image Management Core Team) GFCCT (Global Food Chain Core Team) GTCT (Global Traceability Core Team) 5
 IFAH Members Member Associations EUROPE Belgium PHARMA. BE Croatia Vetcro Denmark VIF France SIMV Germany Bf. T Greece HAVEPHARM Ireland APHA Israel MAI Italy AISA The Netherlands FIDIN Portugal APIFARMA Slovenia WAH (SPMA) Spain VETERINDUSTRIA Switzerland SGCI/SSIC United Kingdom NOAH 6
IFAH Members Member Associations EUROPE Belgium PHARMA. BE Croatia Vetcro Denmark VIF France SIMV Germany Bf. T Greece HAVEPHARM Ireland APHA Israel MAI Italy AISA The Netherlands FIDIN Portugal APIFARMA Slovenia WAH (SPMA) Spain VETERINDUSTRIA Switzerland SGCI/SSIC United Kingdom NOAH 6
 IFAH Members Member Associations NORTH AMERICA Canada United States CAHI 7
IFAH Members Member Associations NORTH AMERICA Canada United States CAHI 7
 IFAH Members Member Associations CENTRAL & SOUTH AMERICA Argentina CAPROVE Brazil SINDAN Mexico INFARVET-CANIFARMA 8
IFAH Members Member Associations CENTRAL & SOUTH AMERICA Argentina CAPROVE Brazil SINDAN Mexico INFARVET-CANIFARMA 8
 IFAH Members Member Associations ASIA/PACIFIC Australia Indonesia AVCARE ASOHI Japan JVPA Korea KAHPA New Zealand AGCARM AFRICA South Africa SAAHA 9
IFAH Members Member Associations ASIA/PACIFIC Australia Indonesia AVCARE ASOHI Japan JVPA Korea KAHPA New Zealand AGCARM AFRICA South Africa SAAHA 9
 OCTOBER, 26 th, 2004 THE COMPLETION OF A CYCLE • CEESA Manulog and the Traceability Working Group of IFAH have merged into the IFAH’s Global Traceability Core Team • The GTCT is composed of experts designated by their respective CEOs – – – AHI R. Phillips BAYER H. Malik BOEHRINGER INGELHEIM CEESA H. Marion CEVA P. Bonassieux/ M. Chaton-Schaffner – ELANCO M. Mitchelson – FORT DODGE B. Conover/ (S. Verouden) – INTERVET R. Janssen – JANSSEN D. Mann B. Van Merris/ (W. Peeters) – MERIAL JC. Muller (chair) – MONSANTO T. Helscher – NOVARTIS R. Billo – PFIZER R. Hollander (M. Walchak) – VETOQUINOL Y. Lambert – VIRBAC L. Cesar – SCHERING PLOUGH I. Forrester – SIMV F. Lasvergères • The GTCT met several times and agreed by consensus the recommendation 10
OCTOBER, 26 th, 2004 THE COMPLETION OF A CYCLE • CEESA Manulog and the Traceability Working Group of IFAH have merged into the IFAH’s Global Traceability Core Team • The GTCT is composed of experts designated by their respective CEOs – – – AHI R. Phillips BAYER H. Malik BOEHRINGER INGELHEIM CEESA H. Marion CEVA P. Bonassieux/ M. Chaton-Schaffner – ELANCO M. Mitchelson – FORT DODGE B. Conover/ (S. Verouden) – INTERVET R. Janssen – JANSSEN D. Mann B. Van Merris/ (W. Peeters) – MERIAL JC. Muller (chair) – MONSANTO T. Helscher – NOVARTIS R. Billo – PFIZER R. Hollander (M. Walchak) – VETOQUINOL Y. Lambert – VIRBAC L. Cesar – SCHERING PLOUGH I. Forrester – SIMV F. Lasvergères • The GTCT met several times and agreed by consensus the recommendation 10
 OCTOBER, 26 th, 2004 THE COMPLETION OF A CYCLE The Board of Directors of IFAH endorsed the GTCT recommendation for products identification and for the traceability printed support as the worldwide standard for Animal Health Industry. 11
OCTOBER, 26 th, 2004 THE COMPLETION OF A CYCLE The Board of Directors of IFAH endorsed the GTCT recommendation for products identification and for the traceability printed support as the worldwide standard for Animal Health Industry. 11
 AN URGENT NEED IN EU, BUT A GLOBAL INVESTMENT • Trend in EU is to require, from a regulatory point of view, optical readable information containing the batch number, imposing in-line printing of the said information – Decree 306 dated 16/05/2001 in Italy, originally in force in Sept. 05 postponed up to end of 2007, – Decree 263/2003 in France requiring record at every step as of March 2006 admitted 80% end of 2007, completed end of 2009, other countries moving to the same direction – Article 18 of EU regulation 178/2002 – Introduction in Portugal of batch traceability at human pharma first – In preparation in Belgium 12
AN URGENT NEED IN EU, BUT A GLOBAL INVESTMENT • Trend in EU is to require, from a regulatory point of view, optical readable information containing the batch number, imposing in-line printing of the said information – Decree 306 dated 16/05/2001 in Italy, originally in force in Sept. 05 postponed up to end of 2007, – Decree 263/2003 in France requiring record at every step as of March 2006 admitted 80% end of 2007, completed end of 2009, other countries moving to the same direction – Article 18 of EU regulation 178/2002 – Introduction in Portugal of batch traceability at human pharma first – In preparation in Belgium 12
 AN URGENT NEED IN EU, BUT A GLOBAL INVESTMENT • Increasing number of multilingual packs • Products are moving through the borders: produced here, sold there • Uniformity of the system and uniqueness of numbering is essential • A single system to avoid 25 different ones. 13
AN URGENT NEED IN EU, BUT A GLOBAL INVESTMENT • Increasing number of multilingual packs • Products are moving through the borders: produced here, sold there • Uniformity of the system and uniqueness of numbering is essential • A single system to avoid 25 different ones. 13
 THE CONSENSUS RECOMMENDATION • • Use the EAN/UCC worldwide standards Use GTIN for product identification: uniqueness Code with EAN Application Identifiers Standard Support with Datamatrix ECC 200 symbology registered by EAN in July, 2004 • DMECC 200 allows for: – An extended flexibility/possibility to carry additional data – Very low printing errors – Error check and correcting system allowing reading even with a high level of damage (Reed Solomon) – Limited artwork redesign (can be printed on end flaps) – Small size – In-line printing with high speed inkjet, laser and heat transfer 14
THE CONSENSUS RECOMMENDATION • • Use the EAN/UCC worldwide standards Use GTIN for product identification: uniqueness Code with EAN Application Identifiers Standard Support with Datamatrix ECC 200 symbology registered by EAN in July, 2004 • DMECC 200 allows for: – An extended flexibility/possibility to carry additional data – Very low printing errors – Error check and correcting system allowing reading even with a high level of damage (Reed Solomon) – Limited artwork redesign (can be printed on end flaps) – Small size – In-line printing with high speed inkjet, laser and heat transfer 14
 ENCODATION OF DATA • The coding standard used is EAN/UCC 128 standard using the special character FNC 1 as starter and group separator • Example of concateneted coding string: FNC 1(02)01234567890128(17)YYMMDD(10)ABC 123456 FNC 1(37)10 This code means a group of 10 units of the product bearing code: 01234567890128, batch number ABC 123456, expiring YYMMDD 15
ENCODATION OF DATA • The coding standard used is EAN/UCC 128 standard using the special character FNC 1 as starter and group separator • Example of concateneted coding string: FNC 1(02)01234567890128(17)YYMMDD(10)ABC 123456 FNC 1(37)10 This code means a group of 10 units of the product bearing code: 01234567890128, batch number ABC 123456, expiring YYMMDD 15
 SYMBOLIC REPRESENTATION • Datamatrix ECC 200 symbol as support • Printed on line Datamatrix with id, exp date, batch n°, (plus quantity and serial number for test) on visible side of flap (50 characters) Real size 7 x 7 mm Per comparison, an EAN 128 with only id and batch nb. (20 characters) Product id Expiry date Batch number Quantity (01)3400969846935(17)090504(10)L 123456(37)1 16
SYMBOLIC REPRESENTATION • Datamatrix ECC 200 symbol as support • Printed on line Datamatrix with id, exp date, batch n°, (plus quantity and serial number for test) on visible side of flap (50 characters) Real size 7 x 7 mm Per comparison, an EAN 128 with only id and batch nb. (20 characters) Product id Expiry date Batch number Quantity (01)3400969846935(17)090504(10)L 123456(37)1 16
 SIZE COMPARISON Plenty of codes exist. The best fitting on our pharmaceutical products is Datamatrix. It is the smallest size at the moment for the same content. For example: Micro. PDF 417 0. 66” wide 0. 16” high 6. 7 mil X-dimension, 3: 1 Y: X ratio (Minimum height configuration) 4 columns x 8 rows Data Matrix ECC 200 0. 18” wide 0. 18” high 10 mil cell size, 18 X 18 matrix Micro. PDF 417 is 4 times larger than Datamatrix ECC 200 17
SIZE COMPARISON Plenty of codes exist. The best fitting on our pharmaceutical products is Datamatrix. It is the smallest size at the moment for the same content. For example: Micro. PDF 417 0. 66” wide 0. 16” high 6. 7 mil X-dimension, 3: 1 Y: X ratio (Minimum height configuration) 4 columns x 8 rows Data Matrix ECC 200 0. 18” wide 0. 18” high 10 mil cell size, 18 X 18 matrix Micro. PDF 417 is 4 times larger than Datamatrix ECC 200 17
 Thanking you for your attention Any question? 18
Thanking you for your attention Any question? 18


