2d028cc3645ead45690b730d010e580b.ppt
- Количество слайдов: 25

ANALYTICAL SEPARATIONS GROUP Megan Bennett, Ashlee Crable, Sherry Faye, Narek Gharibyan, Julie Gostic, and Chris Klug Subgroup Leader: Ralf Sudowe

COMMON RESEARCH GOALS Develop better separation schemas for various radioisotopes (Sr, An, Transactinides) in aqueous systems Basic Science Applications Environmental Emergency Response Nuclear Forensics Sorption/Desorption Studies Characterizing various forms of chromatographic separation procedures

HEAVY ELEMENT CHEMISTRY (MEGAN BENNETT &JULIE GOSTIC) Chemical characterization of transactinides: elements 104 and 105 Studying the nuclear and chemical properties of the heavy elements or transactinides provides validation of predicted periodic trends and illustrates the importance of relativistic effects as a causality for deviations in periodicity. Reaction Products Target Nucleus Hot Fusion or Projectile Compound Nucleus Fission Products Figure adapted from presentations by Dawn Shaughnessy and Ken Moody

ELEMENT 104 & 105 CHEMISTRY Objective Develop separation methods that will allow us to separate a few atoms from a sea of other constituents Analytical Challenges Rapid Large number of exchange steps Highly Selective Continuous process Samples easily prepared for α spec Investigation Using Group IV/V chemical homologs, we can determine which extraction chromatography resins are the best candidates
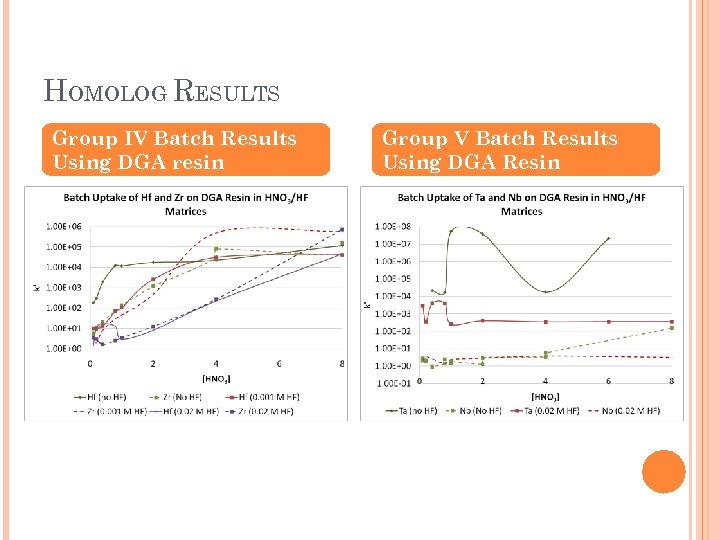
HOMOLOG RESULTS Group IV Batch Results Using DGA resin Group V Batch Results Using DGA Resin
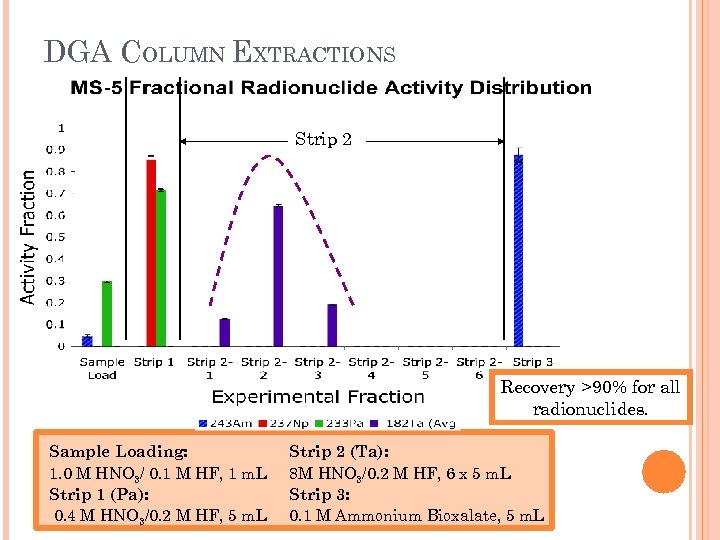
DGA COLUMN EXTRACTIONS Strip 2 Recovery >90% for all radionuclides. Sample Loading: 1. 0 M HNO 3/ 0. 1 M HF, 1 m. L Strip 1 (Pa): 0. 4 M HNO 3/0. 2 M HF, 5 m. L Strip 2 (Ta): 8 M HNO 3/0. 2 M HF, 6 x 5 m. L Strip 3: 0. 1 M Ammonium Bioxalate, 5 m. L

ANALYSIS OF BONE ASH AND OTHER MATRICES (ASHLEE CRABLE) Developing more efficient separation methods for Sr and Actinides in various environmental matrices Problem Statement The current analytical methods that exist for determining total strontium contamination in various matrices are greatly influenced by the presence of other matrix constituents such as calcium and phosphates. This presents a particular problem for determining total deposition in bone (hydroxyapatite). Preliminary Objective To determine the separation efficiency of 90 Sr using vacuum-assisted extraction chromatography (Sr·Spec resin cartridges) in the presence of Ca 2+

SEM image of bone ash
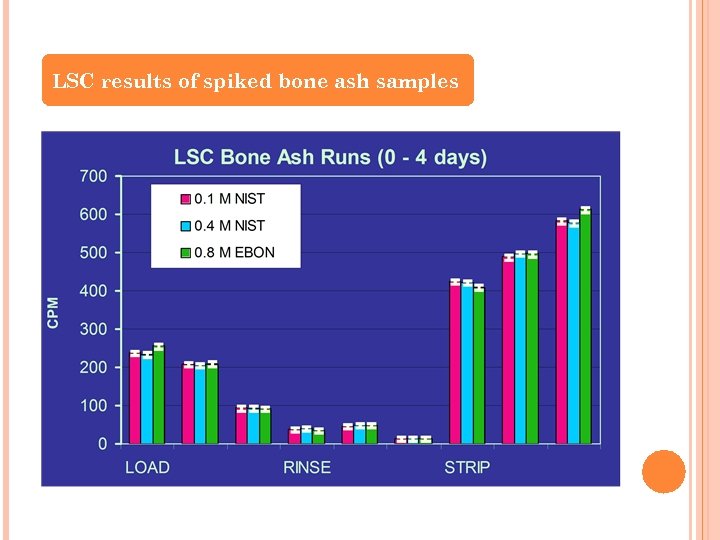
LSC results of spiked bone ash samples

EFFECT OF BACTERIA ON SORPTION OF RN TO SOIL (SHERRY FAYE) Sorption of 241 Am and 233 U to Volcanic Tuff in the Presence of Shewanella oneidensis (MR-1) Objectives: To obtain data on sorption kinetics, equilibrium and fundamental surface interactions of radionuclides to volcanic tuff, commonly found in the Southern Nevada areas of Yucca Mountain and the Nevada Test site. To obtain a better understanding of surface interactions of the Shewanella oneidensis (MR-1) culture with tuff and radionuclides.
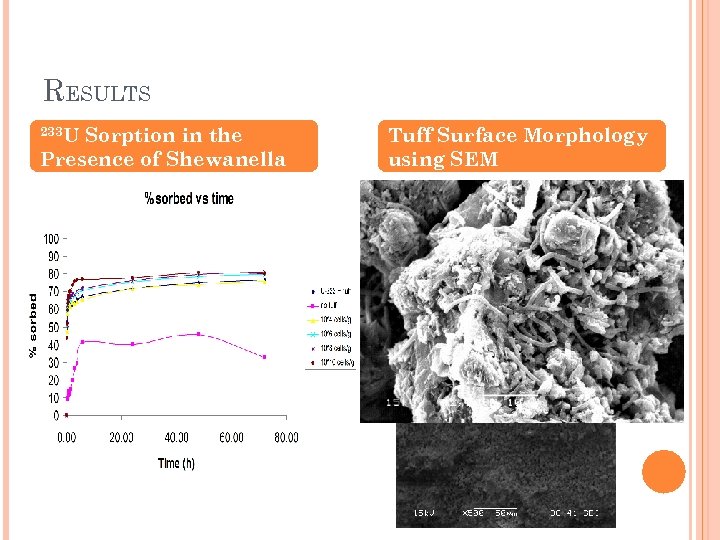
RESULTS 233 U Sorption in the Presence of Shewanella Tuff Surface Morphology using SEM

MEASUREMENT OF NEUTRON CAPTURE ONAM-241 (NAREK GHARIBYAN) Objective Separation of curium from americium for neutron capture cross section and isomeric ratio measurements (242 m+g. Am from 241 Am) Nuclear reactions (n, γ) 241 Am 141 y IT 242 Am Investigation 16. 02 h β 242 Cm 162. 8 d Am/Cm separation methods with extraction chromatography resins from Eichrom that would not require changing Am (III) oxidation state.
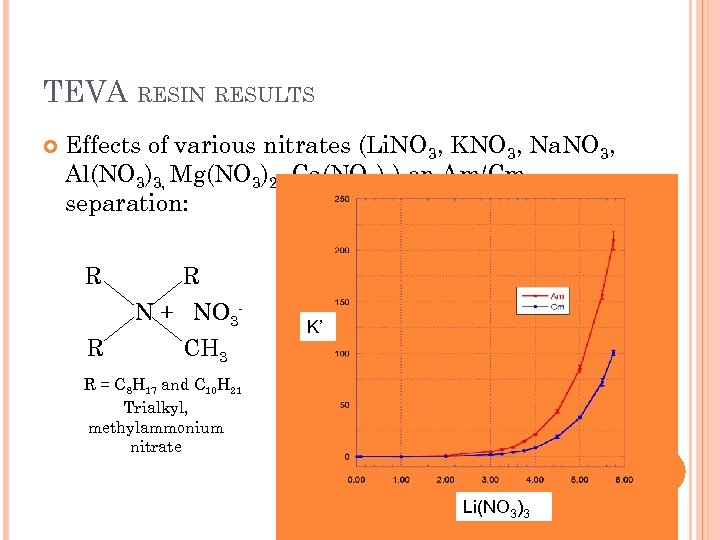
TEVA RESIN RESULTS Effects of various nitrates (Li. NO 3, KNO 3, Na. NO 3, Al(NO 3)3, Mg(NO 3)2, Ca(NO 3)2) on Am/Cm separation: R R N + NO 3 - R CH 3 K’ R = C 8 H 17 and C 10 H 21 Trialkyl, methylammonium nitrate Li(NO 3)3
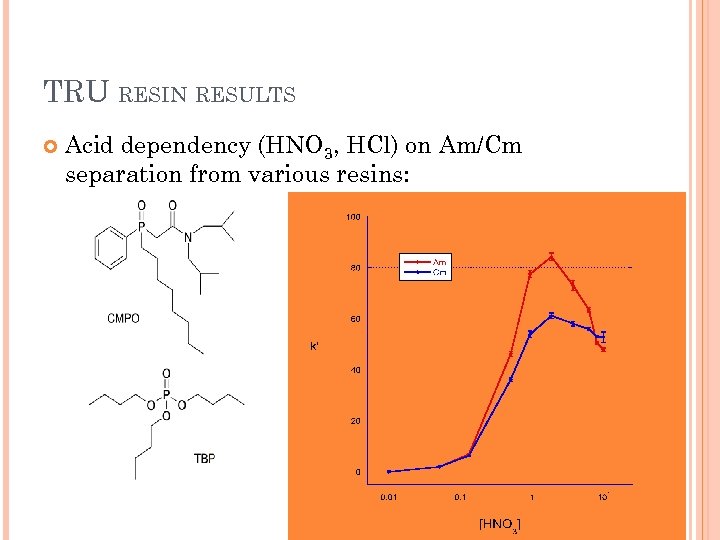
TRU RESIN RESULTS Acid dependency (HNO 3, HCl) on Am/Cm separation from various resins:
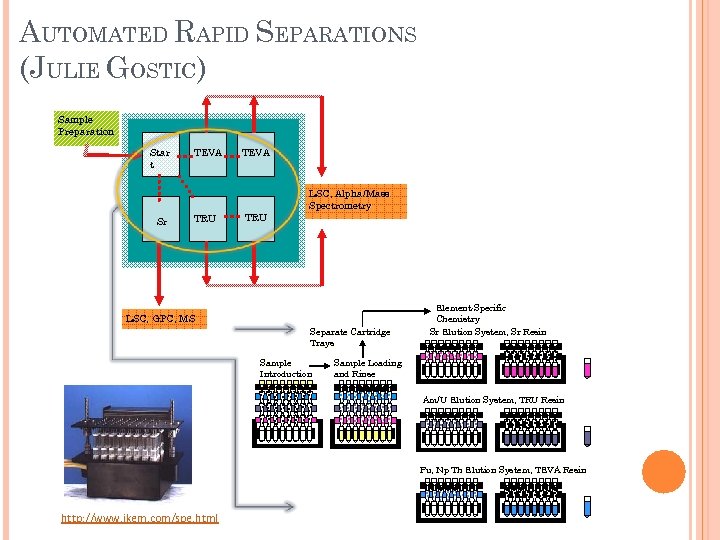
AUTOMATED RAPID SEPARATIONS (JULIE GOSTIC) Sample Preparation Star t Sr TEVA TRU LSC, Alpha/Mass Spectrometry LSC, GPC, MS Separate Cartridge Trays Sample Introduction Element-Specific Chemistry Sr Elution System, Sr Resin Sample Loading and Rinse Am/U Elution System, TRU Resin Pu, Np Th Elution System, TEVA Resin Pu, http: //www. jkem. com/spe. html
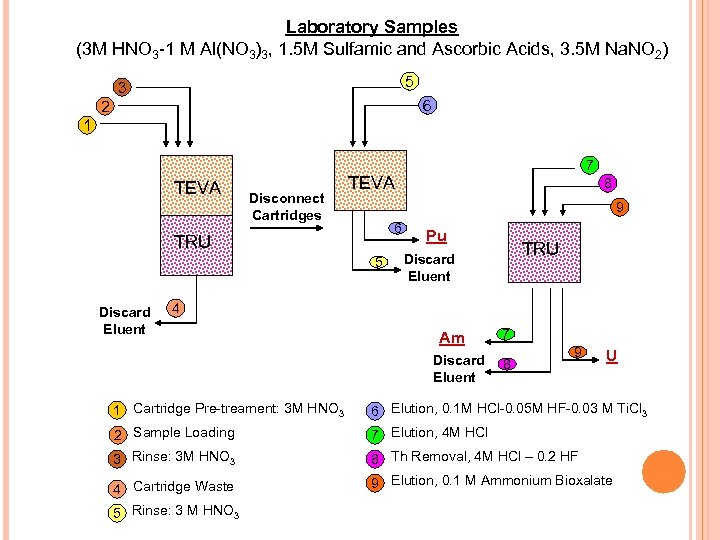
Laboratory Samples (3 M HNO 3 -1 M Al(NO 3)3, 1. 5 M Sulfamic and Ascorbic Acids, 3. 5 M Na. NO 2) 5 3 6 2 1 TEVA Disconnect Cartridges TEVA 8 9 6 TRU 5 Discard Eluent 7 Pu TRU Discard Eluent 4 Am Discard Eluent 7 8 9 U 1 Cartridge Pre-treament: 3 M HNO 3 6 Elution, 0. 1 M HCl-0. 05 M HF-0. 03 M Ti. Cl 3 2 Sample Loading 7 Elution, 4 M HCl 3 Rinse: 3 M HNO 3 8 Th Removal, 4 M HCl – 0. 2 HF 9 Elution, 0. 1 M Ammonium Bioxalate 4 Cartridge Waste 5 Rinse: 3 M HNO 3
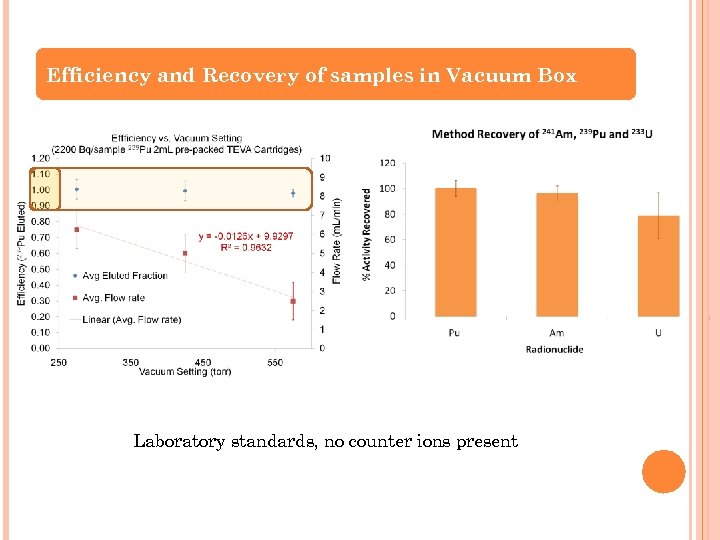
Efficiency and Recovery of samples in Vacuum Box Laboratory standards, no counter ions present
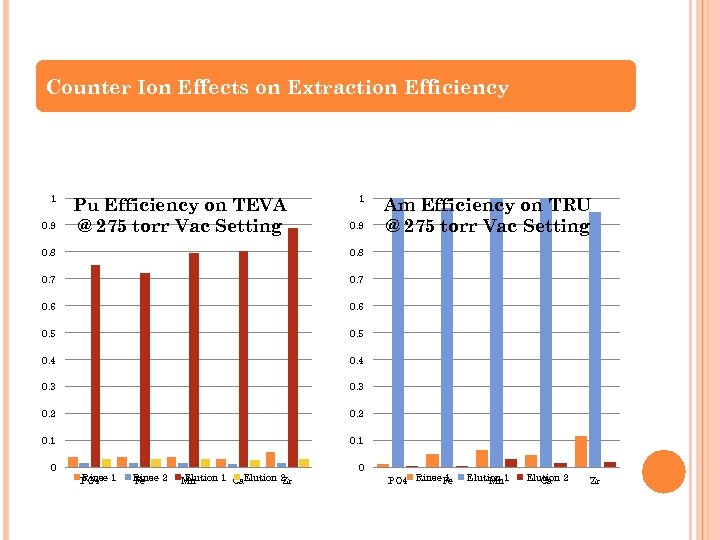
Counter Ion Effects on Extraction Efficiency 1 0. 9 Pu Efficiency on TEVA @ 275 torr Vac Setting 1 0. 9 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 Am Efficiency on TRU @ 275 torr Vac Setting 0. 1 0 Rinse PO 4 1 Rinse 2 Fe Elution 1 Ca. Elution 2 Mn Zr 0 1 PO 4 Rinse. Fe Elution 1 Mn Elution 2 Ca Zr

DEVELOPING A NOVEL EXTRACTION RESIN (CHRIS KLUG) Project Goal Characterize a new extraction resin for trivalent actinide separations Current Objective Some commercially available resins use extractants from 1970 s, 1960 s, and earlier. Use molecules designed more recently for trivalent actinide separations in solvent extraction to maximize extraction properties. Secondary Objective Compare performance of our resins to commercially available resins and to solvent extraction systems The novel resin will follow the “CHNO rule” – P or S can make incineration troublesome
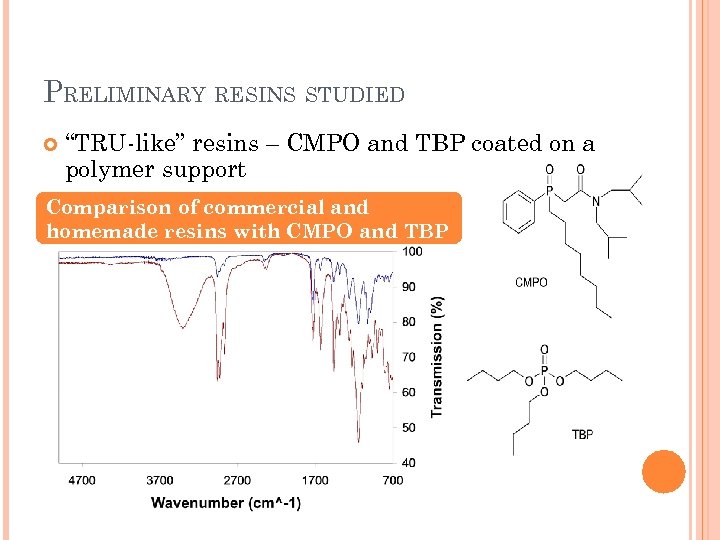
PRELIMINARY RESINS STUDIED “TRU-like” resins – CMPO and TBP coated on a polymer support Comparison of commercial and homemade resins with CMPO and TBP
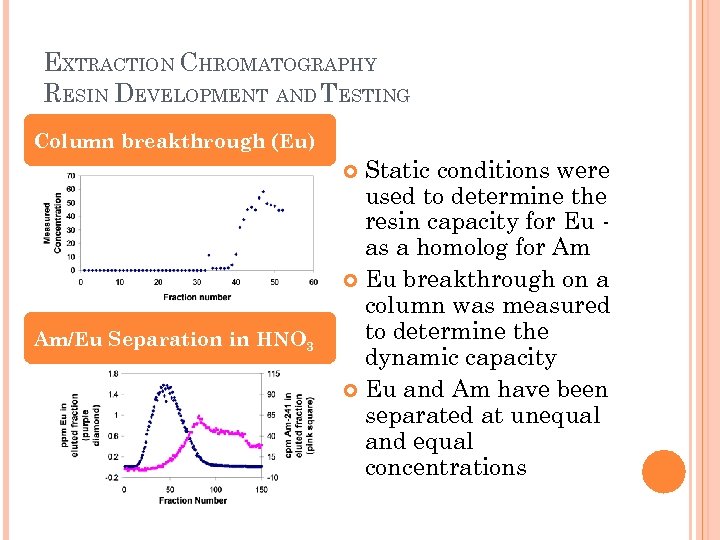
EXTRACTION CHROMATOGRAPHY RESIN DEVELOPMENT AND TESTING Column breakthrough (Eu) Static conditions were used to determine the resin capacity for Eu as a homolog for Am Eu breakthrough on a column was measured to determine the dynamic capacity Eu and Am have been separated at unequal and equal concentrations Am/Eu Separation in HNO 3

UNLV DEEP BURN REPOSITORY PERFORMANCE TASKS (YOU? ? ? ) Project Summary SNF Source Term Models Based on LWR Fuel Cladding Failure UO 2 Dissolution Kinetic Release Model Particle Size Surface Area Release to Near Field TRISO Fuel Small oxide particles Intrinsic Transport Barrier Goal: Develop Source Term Model for TRISO fuels

PREDICTING REPOSITORY PERFORMANCE WORK PLANNED AT UNLV TRISO Repository Behavior Actinide Sorption to Graphite Determination of Equilibrium Sorption Evaluation of Sorption Kinetics Degradation of Irradiated Graphite Evaluation of Degradation Rate for Irradiated Graphite Determination of Degradation Mechanisms TRISO Fuel Performance Modeling Develop Source Term Model Sorption-controlled release vs. degradation of graphite matrix? Equilibrium Sorption vs. Desorption-kinetics controlled release? Implement Model for Performance Assessment

CONCLUSIONS Focusing on extraction chromatography protocols Simple, high selectivity, fast kinetics, lower waste stream volume, and automatable Environmental sorption studies Microbial activity should be considered for actinide transport Sequential extraction studies will be conducted to investigate actinide sorption in soils Develop more efficient methods for the isolation/separation of actinides in various matrices Lessons from bone ash can be applied to cement and other construction materials

CONCLUSIONS Basic Science Applications Develop new resins for actinide separations Develop methods suited for heavy element chemistry Emergency Response Developing an automatable radioanalytical protocol Testing chromatography method on samples containing WG-Pu particulates Forensics Capabilities Different interpretation of the same data Same samples, different analysis methods Isotopic information
2d028cc3645ead45690b730d010e580b.ppt