5fb3bd1d4d07a7cfed28c2614258d84e.ppt
- Количество слайдов: 15
 AML in mice after retroviral cell marking Heinrich-Pette-Institute, Hamburg Bernd Schiedlmeier, Martin Forster, Carol Stocking, Anke Wahlers, Oliver Frank, Wolfram Ostertag University Hospital Eppendorf, Hamburg Jochen Duellmann, Axel Zander, Boris Fehse University Freiburg Manfred Schmidt, Christof von Kalle EUFETS AG Klaus Kuehlcke, Hans-Georg Eckert Hannover Medical School Zhixiong Li, Johann Meyer, Christopher Baum CB 02
AML in mice after retroviral cell marking Heinrich-Pette-Institute, Hamburg Bernd Schiedlmeier, Martin Forster, Carol Stocking, Anke Wahlers, Oliver Frank, Wolfram Ostertag University Hospital Eppendorf, Hamburg Jochen Duellmann, Axel Zander, Boris Fehse University Freiburg Manfred Schmidt, Christof von Kalle EUFETS AG Klaus Kuehlcke, Hans-Georg Eckert Hannover Medical School Zhixiong Li, Johann Meyer, Christopher Baum CB 02
 Oncogenic progression related to insertional mutagenesis Risk ~ 10 -7 per insertion in human TF-1 leukemia cells (Stocking et al. , 1993) Insertional mutagenesis promotes tumor formation in numerous animal models, but single insertion never sufficient to explain malignancy No disease induction reported using replication-defective vectors designed for gene therapy in numerous preclinical and clinical trials, probably involving manipulation of >1012 hematopoietic or lymphoid cells Side effects of transgene or active replication required for CB 02 pathogenesis
Oncogenic progression related to insertional mutagenesis Risk ~ 10 -7 per insertion in human TF-1 leukemia cells (Stocking et al. , 1993) Insertional mutagenesis promotes tumor formation in numerous animal models, but single insertion never sufficient to explain malignancy No disease induction reported using replication-defective vectors designed for gene therapy in numerous preclinical and clinical trials, probably involving manipulation of >1012 hematopoietic or lymphoid cells Side effects of transgene or active replication required for CB 02 pathogenesis
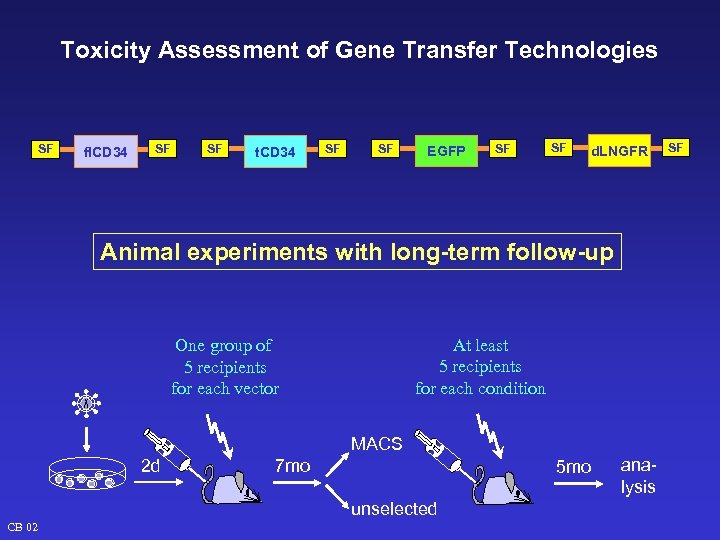 Toxicity Assessment of Gene Transfer Technologies SF fl. CD 34 SF SF t. CD 34 SF SF EGFP SF SF d. LNGFR Animal experiments with long-term follow-up At least 5 recipients for each condition One group of 5 recipients for each vector MACS 2 d 7 mo 5 mo unselected CB 02 analysis SF
Toxicity Assessment of Gene Transfer Technologies SF fl. CD 34 SF SF t. CD 34 SF SF EGFP SF SF d. LNGFR Animal experiments with long-term follow-up At least 5 recipients for each condition One group of 5 recipients for each vector MACS 2 d 7 mo 5 mo unselected CB 02 analysis SF
 d. LNGFR group, 2° recipients (n=10) – AML M 5: n=6 – Overt dysplasia: n=3 – Microscopic lesions: n=1 CB 02
d. LNGFR group, 2° recipients (n=10) – AML M 5: n=6 – Overt dysplasia: n=3 – Microscopic lesions: n=1 CB 02
 AML after Retroviral Gene Marking in Mice Long latency: No overt disease in first cohort (7 mo) 10/10 secondary recipients developed dysplasia or AML M 5 (5 mo) Leukemia is transplantable to 3° cohort (lethal) Monoclonal origin, heterogenous kinetics, however identical entity with reproducible phenotype Aberrant clone has single vector integration Vector is intact and continues to express d. LNGFR Insertional activation of Evi-1 RCR and activation of endogenous MLV excluded CB 02
AML after Retroviral Gene Marking in Mice Long latency: No overt disease in first cohort (7 mo) 10/10 secondary recipients developed dysplasia or AML M 5 (5 mo) Leukemia is transplantable to 3° cohort (lethal) Monoclonal origin, heterogenous kinetics, however identical entity with reproducible phenotype Aberrant clone has single vector integration Vector is intact and continues to express d. LNGFR Insertional activation of Evi-1 RCR and activation of endogenous MLV excluded CB 02
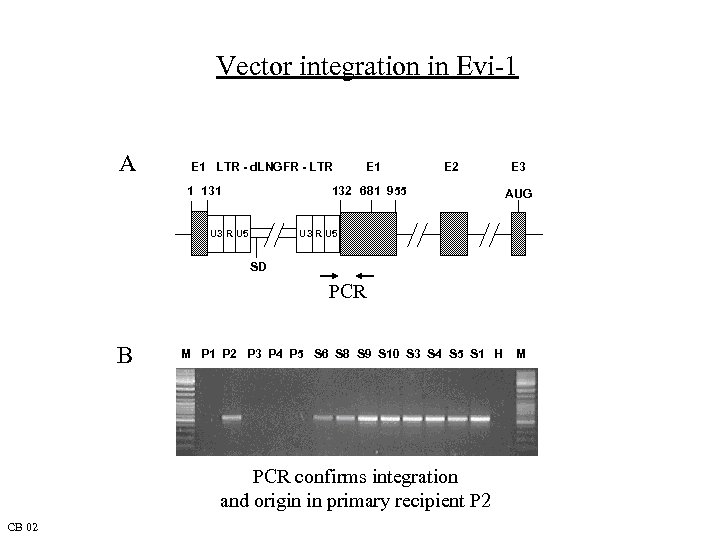 Vector integration in Evi-1 A E 1 LTR - d. LNGFR - LTR 1 131 E 2 132 681 955 E 3 AUG U 3 R U 5 SD PCR B M P 1 P 2 P 3 P 4 P 5 S 6 S 8 S 9 S 10 S 3 S 4 S 5 S 1 H PCR confirms integration and origin in primary recipient P 2 CB 02 M
Vector integration in Evi-1 A E 1 LTR - d. LNGFR - LTR 1 131 E 2 132 681 955 E 3 AUG U 3 R U 5 SD PCR B M P 1 P 2 P 3 P 4 P 5 S 6 S 8 S 9 S 10 S 3 S 4 S 5 S 1 H PCR confirms integration and origin in primary recipient P 2 CB 02 M
 Evi-1 Transcription factor, known oncogene Endogenous expression in primitive stem cells Ectopic expression blocks granulocytic and erythroid differentiation promotes megakaryocytic hematopoiesis Activation implicated in MDS and AML (usually immature phenotype) Tg mice at increased risk for leukemia (dysplastic hematopoiesis) Not sufficient to explain AML M 5 CB 02
Evi-1 Transcription factor, known oncogene Endogenous expression in primitive stem cells Ectopic expression blocks granulocytic and erythroid differentiation promotes megakaryocytic hematopoiesis Activation implicated in MDS and AML (usually immature phenotype) Tg mice at increased risk for leukemia (dysplastic hematopoiesis) Not sufficient to explain AML M 5 CB 02
 d. LNGFR: variant of p 75 NTR d. LNGFR Ligand binding domain Juxtamembrane domain Death domain Differentiation Apoptosis CB 02
d. LNGFR: variant of p 75 NTR d. LNGFR Ligand binding domain Juxtamembrane domain Death domain Differentiation Apoptosis CB 02
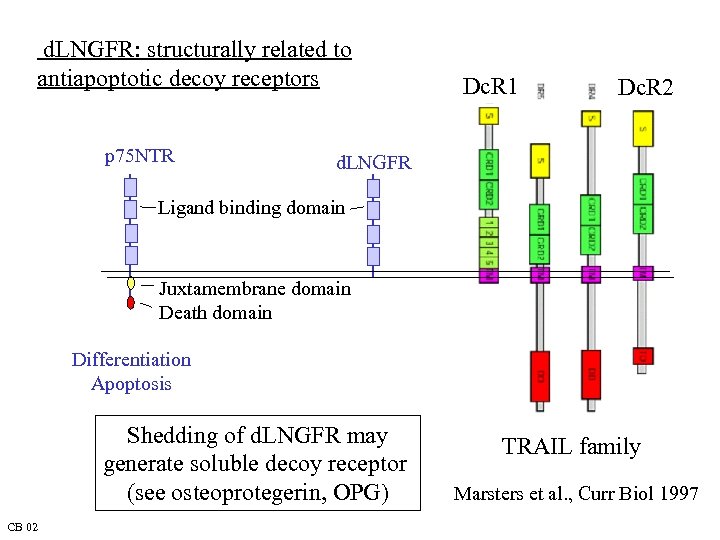 d. LNGFR: structurally related to antiapoptotic decoy receptors p 75 NTR Dc. R 1 Dc. R 2 d. LNGFR Ligand binding domain Juxtamembrane domain Death domain Differentiation Apoptosis Shedding of d. LNGFR may generate soluble decoy receptor (see osteoprotegerin, OPG) CB 02 TRAIL family Marsters et al. , Curr Biol 1997
d. LNGFR: structurally related to antiapoptotic decoy receptors p 75 NTR Dc. R 1 Dc. R 2 d. LNGFR Ligand binding domain Juxtamembrane domain Death domain Differentiation Apoptosis Shedding of d. LNGFR may generate soluble decoy receptor (see osteoprotegerin, OPG) CB 02 TRAIL family Marsters et al. , Curr Biol 1997
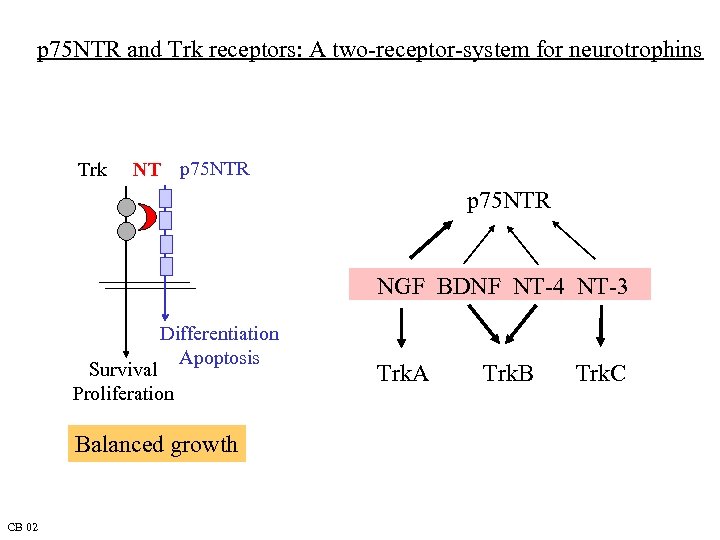 p 75 NTR and Trk receptors: A two-receptor-system for neurotrophins Trk NT p 75 NTR NGF BDNF NT-4 NT-3 Differentiation Apoptosis Survival Proliferation Balanced growth CB 02 Trk. A Trk. B Trk. C
p 75 NTR and Trk receptors: A two-receptor-system for neurotrophins Trk NT p 75 NTR NGF BDNF NT-4 NT-3 Differentiation Apoptosis Survival Proliferation Balanced growth CB 02 Trk. A Trk. B Trk. C
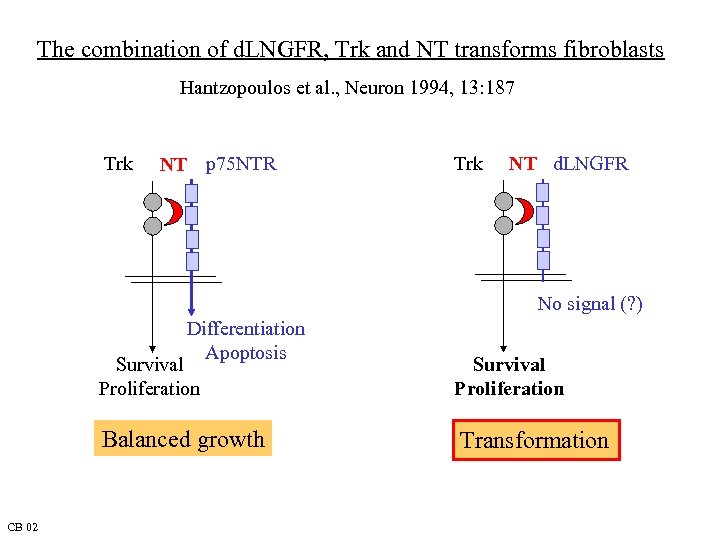 The combination of d. LNGFR, Trk and NT transforms fibroblasts Hantzopoulos et al. , Neuron 1994, 13: 187 Trk NT p 75 NTR Trk NT d. LNGFR No signal (? ) Differentiation Apoptosis Survival Proliferation Balanced growth CB 02 Survival Proliferation Transformation
The combination of d. LNGFR, Trk and NT transforms fibroblasts Hantzopoulos et al. , Neuron 1994, 13: 187 Trk NT p 75 NTR Trk NT d. LNGFR No signal (? ) Differentiation Apoptosis Survival Proliferation Balanced growth CB 02 Survival Proliferation Transformation
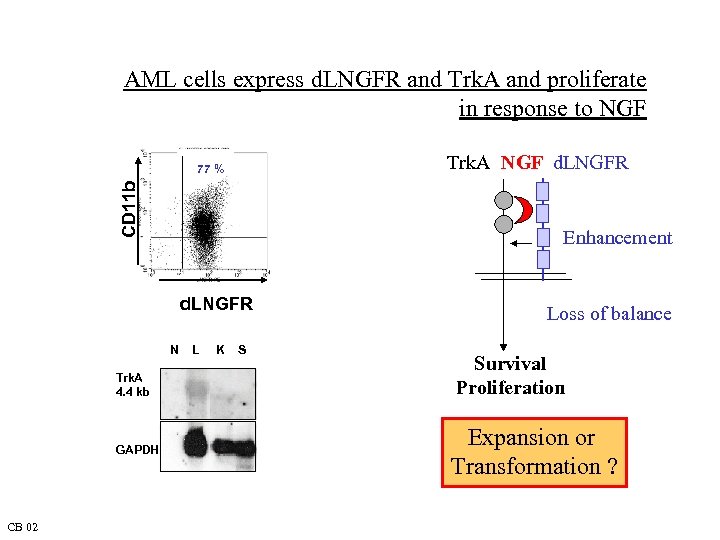 AML cells express d. LNGFR and Trk. A and proliferate in response to NGF Trk. A NGF d. LNGFR CD 11 b 77 % Enhancement d. LNGFR N Trk. A 4. 4 kb GAPDH CB 02 L K S Loss of balance Survival Proliferation Expansion or Transformation ?
AML cells express d. LNGFR and Trk. A and proliferate in response to NGF Trk. A NGF d. LNGFR CD 11 b 77 % Enhancement d. LNGFR N Trk. A 4. 4 kb GAPDH CB 02 L K S Loss of balance Survival Proliferation Expansion or Transformation ?
 Expression of Neurotrophins and their Receptors in Human Hematopoiesis (Labouyrie et al. , AJP 1999, 154: 411) Progenitors p 75 NTR absent B cells (mouse mast cells) Trk. A erythroblasts mono, baso, mast, B cells Trk. Bi erythroblasts eo meg Trk. Ci myeloblasts eo, meg, granulo NGF BDNF NT-3 NT-4/5 CB 02 Mature Cells bone marrow stroma cells, monocytic cells osteoblasts, osteoclasts, mast cells, B cells (T cells ? )
Expression of Neurotrophins and their Receptors in Human Hematopoiesis (Labouyrie et al. , AJP 1999, 154: 411) Progenitors p 75 NTR absent B cells (mouse mast cells) Trk. A erythroblasts mono, baso, mast, B cells Trk. Bi erythroblasts eo meg Trk. Ci myeloblasts eo, meg, granulo NGF BDNF NT-3 NT-4/5 CB 02 Mature Cells bone marrow stroma cells, monocytic cells osteoblasts, osteoclasts, mast cells, B cells (T cells ? )
 Trk receptors and human leukemia Trk. A was detected in some leukemic cell lines, such as UT-7(acute megakaryoblastic leukemia), K 562 and TF 1 (erythroleukemia), and myeloid cell lines HEL, HL 60 and KG 1, but not in myeloid cell lines U 937 and THP-1 (Chevalier et al. , 1994, Auffray et al. , 1996, Kaebisch et al. , 1996). So far, there are only 3 reports on expression of p 75 NTR and Trk receptors in primary leukemia: • 44% Trk. A gene expression in patients with AML (Kaebisch et al. , 1996). • A translocation t(12; 15) (p 13; q 25) was found in an AML patient, which resulted in a fusion RNA ETV 6 -Trk. C (Eguchi et al. , 1999). • A deleted form of Trk. A, DTrk. A, was identified in AML patients. 75 -aa deletion in the extracellular domain resulted in constitutive tyrosine phosphorylation of the protein, which also transforms fibroblasts (Reuther et al. , 2000). These data suggest a possible role of Trk receptors and their mutant forms in leukemia development (however, so far no evidence for transformation of lymphatic cells). CB 02
Trk receptors and human leukemia Trk. A was detected in some leukemic cell lines, such as UT-7(acute megakaryoblastic leukemia), K 562 and TF 1 (erythroleukemia), and myeloid cell lines HEL, HL 60 and KG 1, but not in myeloid cell lines U 937 and THP-1 (Chevalier et al. , 1994, Auffray et al. , 1996, Kaebisch et al. , 1996). So far, there are only 3 reports on expression of p 75 NTR and Trk receptors in primary leukemia: • 44% Trk. A gene expression in patients with AML (Kaebisch et al. , 1996). • A translocation t(12; 15) (p 13; q 25) was found in an AML patient, which resulted in a fusion RNA ETV 6 -Trk. C (Eguchi et al. , 1999). • A deleted form of Trk. A, DTrk. A, was identified in AML patients. 75 -aa deletion in the extracellular domain resulted in constitutive tyrosine phosphorylation of the protein, which also transforms fibroblasts (Reuther et al. , 2000). These data suggest a possible role of Trk receptors and their mutant forms in leukemia development (however, so far no evidence for transformation of lymphatic cells). CB 02
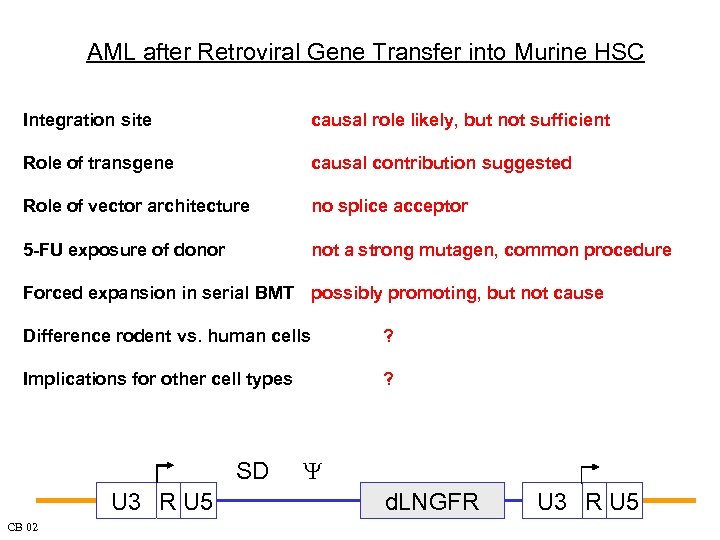 AML after Retroviral Gene Transfer into Murine HSC Integration site causal role likely, but not sufficient Role of transgene causal contribution suggested Role of vector architecture no splice acceptor 5 -FU exposure of donor not a strong mutagen, common procedure Forced expansion in serial BMT possibly promoting, but not cause Difference rodent vs. human cells ? Implications for other cell types ? SD U 3 R U 5 CB 02 Y d. LNGFR U 3 R U 5
AML after Retroviral Gene Transfer into Murine HSC Integration site causal role likely, but not sufficient Role of transgene causal contribution suggested Role of vector architecture no splice acceptor 5 -FU exposure of donor not a strong mutagen, common procedure Forced expansion in serial BMT possibly promoting, but not cause Difference rodent vs. human cells ? Implications for other cell types ? SD U 3 R U 5 CB 02 Y d. LNGFR U 3 R U 5


