e6252bce93ddd9ecdab6be17e9a58960.ppt
- Количество слайдов: 39
 American College of Surgeons Oncology Group Heidi Nelson, M. D. David M. Ota, M. D. 3048365 -1
American College of Surgeons Oncology Group Heidi Nelson, M. D. David M. Ota, M. D. 3048365 -1
 Who are we? 3048365 -2
Who are we? 3048365 -2
 ACOSOG Operations and Membership Center Members Scientific Leadership Statistics and Data Center 3048365 -3
ACOSOG Operations and Membership Center Members Scientific Leadership Statistics and Data Center 3048365 -3
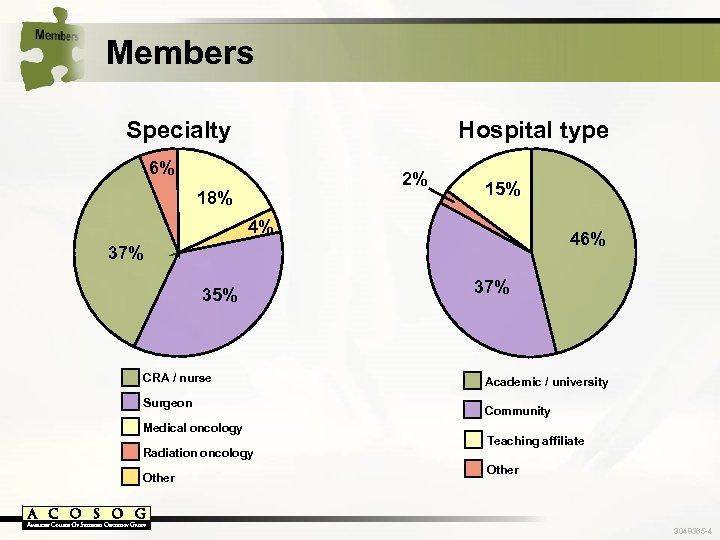 Members Specialty Hospital type 6% 2% 18% 15% 4% 46% 37% 35% CRA / nurse Surgeon Medical oncology Radiation oncology Other 37% Academic / university Community Teaching affiliate Other 3048365 -4
Members Specialty Hospital type 6% 2% 18% 15% 4% 46% 37% 35% CRA / nurse Surgeon Medical oncology Radiation oncology Other 37% Academic / university Community Teaching affiliate Other 3048365 -4
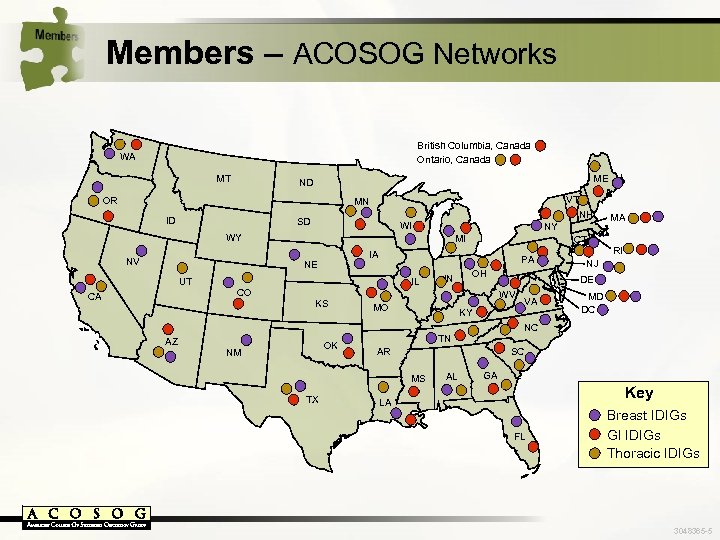 Members – ACOSOG Networks British Columbia, Canada Ontario, Canada WA MT ME ND OR VT NH MN ID SD WI NY WY NV MI UT CA PA IL IN OH CO KS AZ CT IA NE OK NM VA KY MD DC NC TN AR SC MS TX RI NJ DE WV MO MA AL GA Key LA FL Breast IDIGs GI IDIGs Thoracic IDIGs 3048365 -5
Members – ACOSOG Networks British Columbia, Canada Ontario, Canada WA MT ME ND OR VT NH MN ID SD WI NY WY NV MI UT CA PA IL IN OH CO KS AZ CT IA NE OK NM VA KY MD DC NC TN AR SC MS TX RI NJ DE WV MO MA AL GA Key LA FL Breast IDIGs GI IDIGs Thoracic IDIGs 3048365 -5
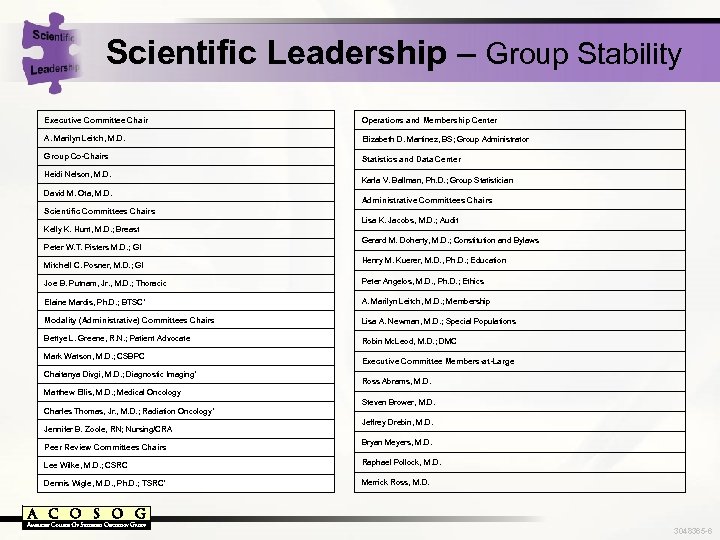 Scientific Leadership – Group Stability Executive Committee Chair Operations and Membership Center A. Marilyn Leitch, M. D. Elizabeth D. Martinez, BS; Group Administrator Group Co-Chairs Statistics and Data Center Heidi Nelson, M. D. David M. Ota, M. D. Karla V. Ballman, Ph. D. ; Group Statistician Administrative Committees Chairs Scientific Committees Chairs Lisa K. Jacobs, M. D. ; Audit Kelly K. Hunt, M. D. ; Breast Peter W. T. Pisters M. D. ; GI Mitchell C. Posner, M. D. ; GI Gerard M. Doherty, M. D. ; Constitution and Bylaws Henry M. Kuerer, M. D. , Ph. D. ; Education Joe B. Putnam, Jr. , M. D. ; Thoracic Peter Angelos, M. D. , Ph. D. ; Ethics Elaine Mardis, Ph. D. ; BTSC* A. Marilyn Leitch, M. D. ; Membership Modality (Administrative) Committees Chairs Lisa A. Newman, M. D. ; Special Populations Bettye L. Greene, R. N. ; Patient Advocate Robin Mc. Leod, M. D. ; DMC Mark Watson, M. D. ; CSBPC Chaitanya Divgi, M. D. ; Diagnostic Imaging* Executive Committee Members-at-Large Ross Abrams, M. D. Matthew Ellis, M. D. ; Medical Oncology Steven Brower, M. D. Charles Thomas, Jr. , M. D. ; Radiation Oncology* Jennifer B. Zoole, RN; Nursing/CRA Peer Review Committees Chairs Jeffrey Drebin, M. D. Bryan Meyers, M. D. Lee Wilke, M. D. ; CSRC Raphael Pollock, M. D. Dennis Wigle, M. D. , Ph. D. ; TSRC* Merrick Ross, M. D. 3048365 -6
Scientific Leadership – Group Stability Executive Committee Chair Operations and Membership Center A. Marilyn Leitch, M. D. Elizabeth D. Martinez, BS; Group Administrator Group Co-Chairs Statistics and Data Center Heidi Nelson, M. D. David M. Ota, M. D. Karla V. Ballman, Ph. D. ; Group Statistician Administrative Committees Chairs Scientific Committees Chairs Lisa K. Jacobs, M. D. ; Audit Kelly K. Hunt, M. D. ; Breast Peter W. T. Pisters M. D. ; GI Mitchell C. Posner, M. D. ; GI Gerard M. Doherty, M. D. ; Constitution and Bylaws Henry M. Kuerer, M. D. , Ph. D. ; Education Joe B. Putnam, Jr. , M. D. ; Thoracic Peter Angelos, M. D. , Ph. D. ; Ethics Elaine Mardis, Ph. D. ; BTSC* A. Marilyn Leitch, M. D. ; Membership Modality (Administrative) Committees Chairs Lisa A. Newman, M. D. ; Special Populations Bettye L. Greene, R. N. ; Patient Advocate Robin Mc. Leod, M. D. ; DMC Mark Watson, M. D. ; CSBPC Chaitanya Divgi, M. D. ; Diagnostic Imaging* Executive Committee Members-at-Large Ross Abrams, M. D. Matthew Ellis, M. D. ; Medical Oncology Steven Brower, M. D. Charles Thomas, Jr. , M. D. ; Radiation Oncology* Jennifer B. Zoole, RN; Nursing/CRA Peer Review Committees Chairs Jeffrey Drebin, M. D. Bryan Meyers, M. D. Lee Wilke, M. D. ; CSRC Raphael Pollock, M. D. Dennis Wigle, M. D. , Ph. D. ; TSRC* Merrick Ross, M. D. 3048365 -6
 American College of Surgeons The American College of Surgeons is a scientific and educational association of surgeons that was founded in 1913 to improve the quality of care for the surgical patient by setting high standards for surgical education and practice. 3048365 -7
American College of Surgeons The American College of Surgeons is a scientific and educational association of surgeons that was founded in 1913 to improve the quality of care for the surgical patient by setting high standards for surgical education and practice. 3048365 -7
 American College of Surgeons • 73, 000 U. S. members • Several cancer programs or initiatives • Commission on Cancer • National Cancer Database • AJCC Staging • ACOSOG 3048365 -8
American College of Surgeons • 73, 000 U. S. members • Several cancer programs or initiatives • Commission on Cancer • National Cancer Database • AJCC Staging • ACOSOG 3048365 -8
 American College of Surgeons Commission on Cancer • Established by the American College of Surgeons in 1922 • Consortium of 50 professional organizations • 1, 500 hospitals with Co. C-accredited cancer programs • Network of more than 1, 600 volunteer Cancer Liaison Physicians • ACOSOG – Co. C Goals: • To establish, disseminate and monitor new clinical practice standards based on emerging clinical trial evidence • To develop and implement skills verification programs • To serve as research arm of ACS including for emerging technologies 3048365 -9
American College of Surgeons Commission on Cancer • Established by the American College of Surgeons in 1922 • Consortium of 50 professional organizations • 1, 500 hospitals with Co. C-accredited cancer programs • Network of more than 1, 600 volunteer Cancer Liaison Physicians • ACOSOG – Co. C Goals: • To establish, disseminate and monitor new clinical practice standards based on emerging clinical trial evidence • To develop and implement skills verification programs • To serve as research arm of ACS including for emerging technologies 3048365 -9
 What do we do? 3048365 -10
What do we do? 3048365 -10
 Mission ACOSOG is dedicated to improving the care of the surgical oncology patient • Increase response and cure rates • Reduce morbidities and disabilities • Better understand the biologic basis of early-stage disease and its treatment 3048365 -11
Mission ACOSOG is dedicated to improving the care of the surgical oncology patient • Increase response and cure rates • Reduce morbidities and disabilities • Better understand the biologic basis of early-stage disease and its treatment 3048365 -11
 Theme 1 Investigate novel surgical and targeted therapies to maintain oncologic outcomes while reducing toxicities and disabilities • Key Scientific Highlights: Z 9001, Z 0030, Z 0011 Test molecular and imaging profiling to enhance the accuracy of risk stratification Apply neoadjuvant therapies to improve overall response rates and monitor individual responses 3048365 -12
Theme 1 Investigate novel surgical and targeted therapies to maintain oncologic outcomes while reducing toxicities and disabilities • Key Scientific Highlights: Z 9001, Z 0030, Z 0011 Test molecular and imaging profiling to enhance the accuracy of risk stratification Apply neoadjuvant therapies to improve overall response rates and monitor individual responses 3048365 -12
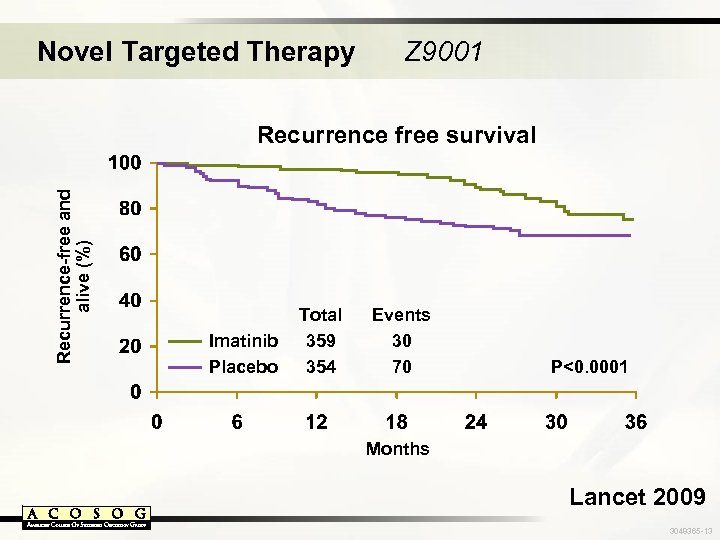 Novel Targeted Therapy Z 9001 Recurrence-free and alive (%) Recurrence free survival Imatinib Placebo Total 359 354 Events 30 70 P<0. 0001 Months Lancet 2009 3048365 -13
Novel Targeted Therapy Z 9001 Recurrence-free and alive (%) Recurrence free survival Imatinib Placebo Total 359 354 Events 30 70 P<0. 0001 Months Lancet 2009 3048365 -13
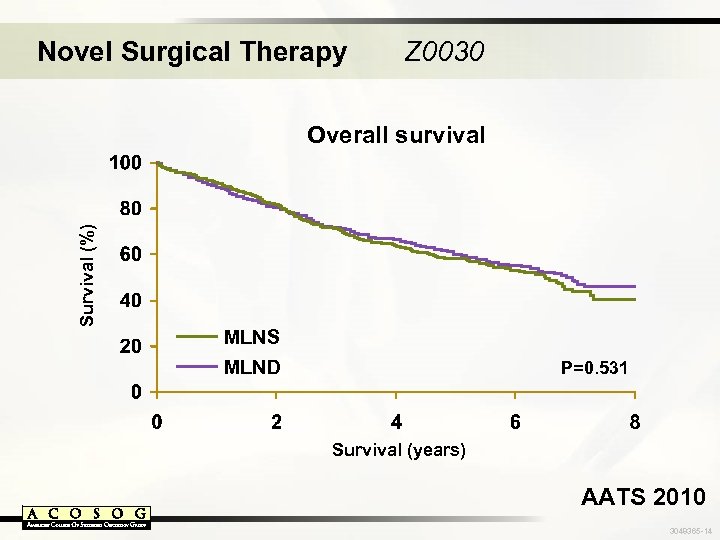 Novel Surgical Therapy Z 0030 Survival (%) Overall survival MLNS MLND P=0. 531 Survival (years) AATS 2010 3048365 -14
Novel Surgical Therapy Z 0030 Survival (%) Overall survival MLNS MLND P=0. 531 Survival (years) AATS 2010 3048365 -14
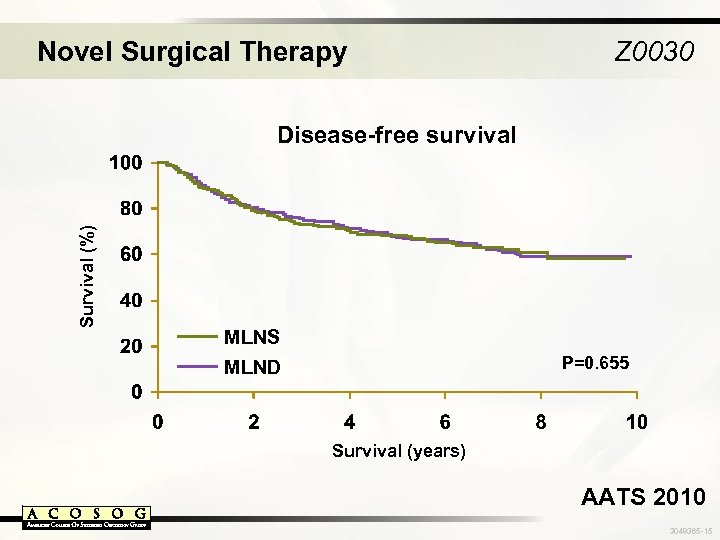 Novel Surgical Therapy Z 0030 Survival (%) Disease-free survival MLNS MLND P=0. 655 Survival (years) AATS 2010 3048365 -15
Novel Surgical Therapy Z 0030 Survival (%) Disease-free survival MLNS MLND P=0. 655 Survival (years) AATS 2010 3048365 -15
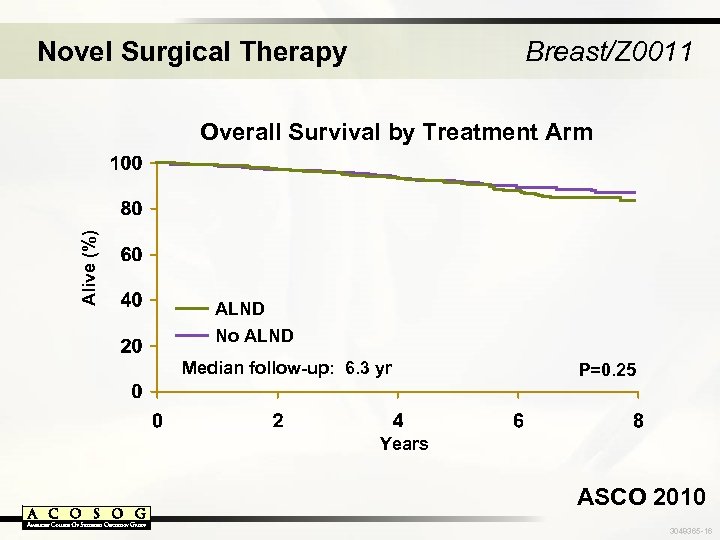 Novel Surgical Therapy Breast/Z 0011 Alive (%) Overall Survival by Treatment Arm ALND No ALND Median follow-up: 6. 3 yr P=0. 25 Years ASCO 2010 3048365 -16
Novel Surgical Therapy Breast/Z 0011 Alive (%) Overall Survival by Treatment Arm ALND No ALND Median follow-up: 6. 3 yr P=0. 25 Years ASCO 2010 3048365 -16
 Theme 2 Investigate novel surgical and targeted therapies to maintain oncologic outcomes while reducing toxicities and disabilities Test molecular and imaging profiling to enhance the accuracy of risk stratification • Key Scientific Highlights: Z 9001, Z 0010, Z 0040 Apply neoadjuvant therapies to improve overall response rates and monitor individual responses 3048365 -17
Theme 2 Investigate novel surgical and targeted therapies to maintain oncologic outcomes while reducing toxicities and disabilities Test molecular and imaging profiling to enhance the accuracy of risk stratification • Key Scientific Highlights: Z 9001, Z 0010, Z 0040 Apply neoadjuvant therapies to improve overall response rates and monitor individual responses 3048365 -17
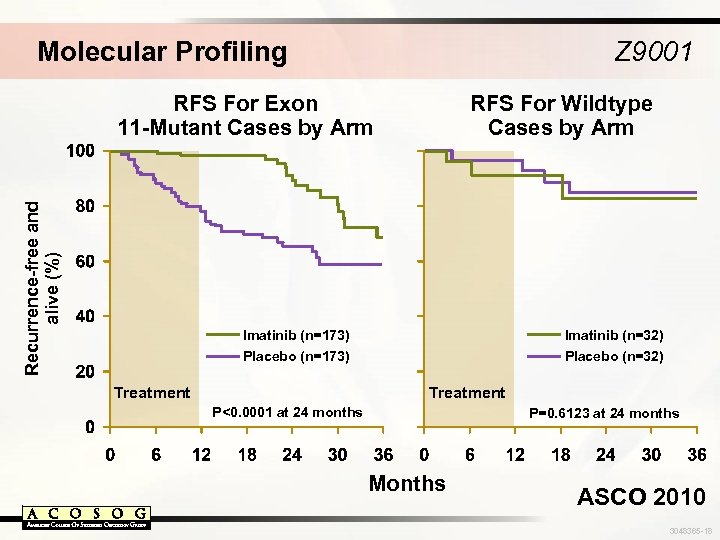 Molecular Profiling Z 9001 Recurrence-free and alive (%) RFS For Exon 11 -Mutant Cases by Arm RFS For Wildtype Cases by Arm Imatinib (n=173) Imatinib (n=32) Placebo (n=173) Placebo (n=32) Treatment P<0. 0001 at 24 months P=0. 6123 at 24 months Months ASCO 2010 3048365 -18
Molecular Profiling Z 9001 Recurrence-free and alive (%) RFS For Exon 11 -Mutant Cases by Arm RFS For Wildtype Cases by Arm Imatinib (n=173) Imatinib (n=32) Placebo (n=173) Placebo (n=32) Treatment P<0. 0001 at 24 months P=0. 6123 at 24 months Months ASCO 2010 3048365 -18
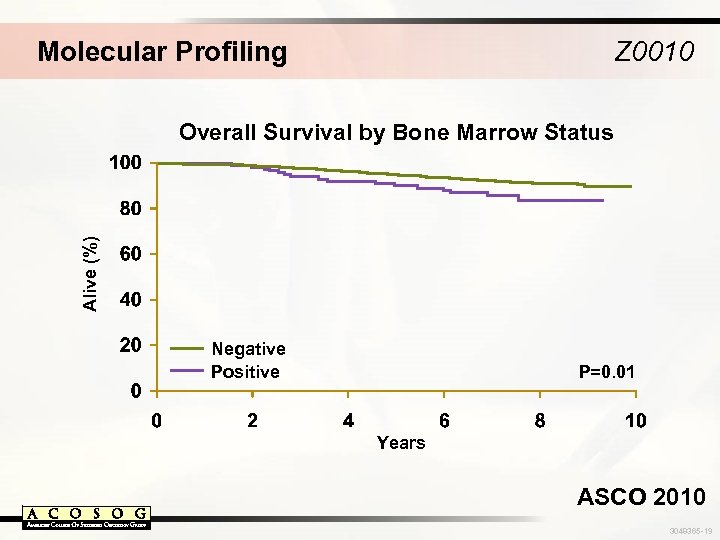 Molecular Profiling Z 0010 Alive (%) Overall Survival by Bone Marrow Status Negative Positive P=0. 01 Years ASCO 2010 3048365 -19
Molecular Profiling Z 0010 Alive (%) Overall Survival by Bone Marrow Status Negative Positive P=0. 01 Years ASCO 2010 3048365 -19
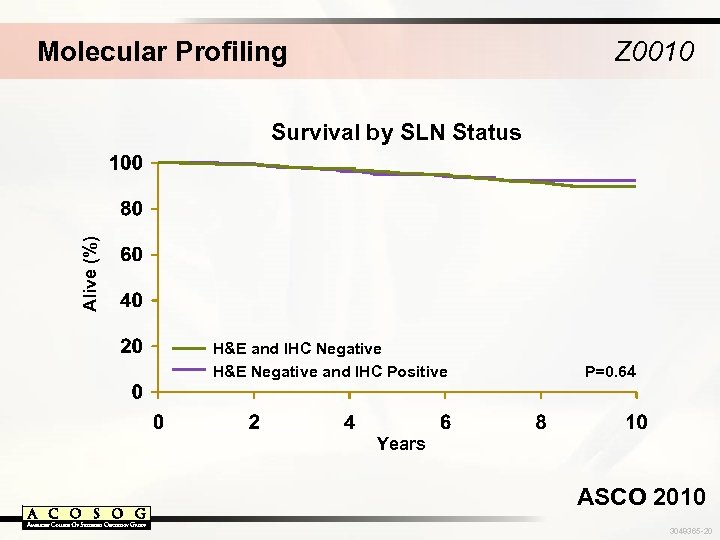 Molecular Profiling Z 0010 Alive (%) Survival by SLN Status H&E and IHC Negative H&E Negative and IHC Positive P=0. 64 Years ASCO 2010 3048365 -20
Molecular Profiling Z 0010 Alive (%) Survival by SLN Status H&E and IHC Negative H&E Negative and IHC Positive P=0. 64 Years ASCO 2010 3048365 -20
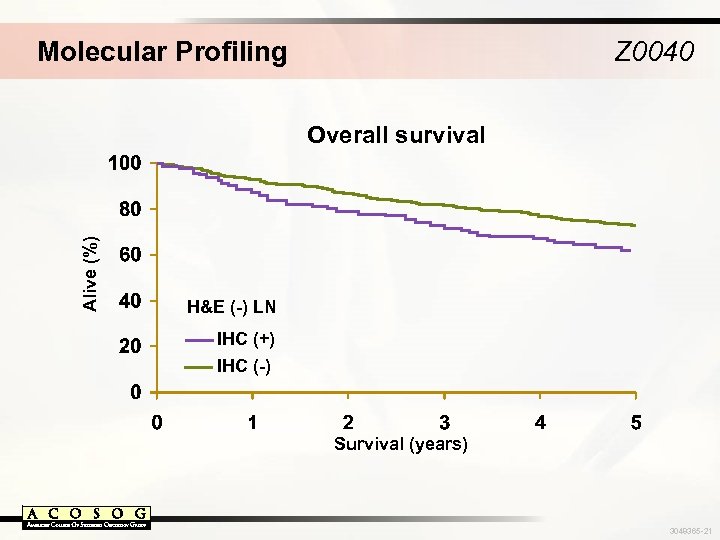 Molecular Profiling Z 0040 Alive (%) Overall survival H&E (-) LN IHC (+) IHC (-) Survival (years) 3048365 -21
Molecular Profiling Z 0040 Alive (%) Overall survival H&E (-) LN IHC (+) IHC (-) Survival (years) 3048365 -21
 Theme 3 Investigate novel surgical and targeted therapies to maintain oncologic outcomes while reducing toxicities and disabilities Test molecular and imaging profiling to enhance the accuracy of risk stratification Apply neoadjuvant therapies to improve overall response rates and monitor individual responses • Key Scientific Highlights: Z 6041, Z 1031 3048365 -22
Theme 3 Investigate novel surgical and targeted therapies to maintain oncologic outcomes while reducing toxicities and disabilities Test molecular and imaging profiling to enhance the accuracy of risk stratification Apply neoadjuvant therapies to improve overall response rates and monitor individual responses • Key Scientific Highlights: Z 6041, Z 1031 3048365 -22
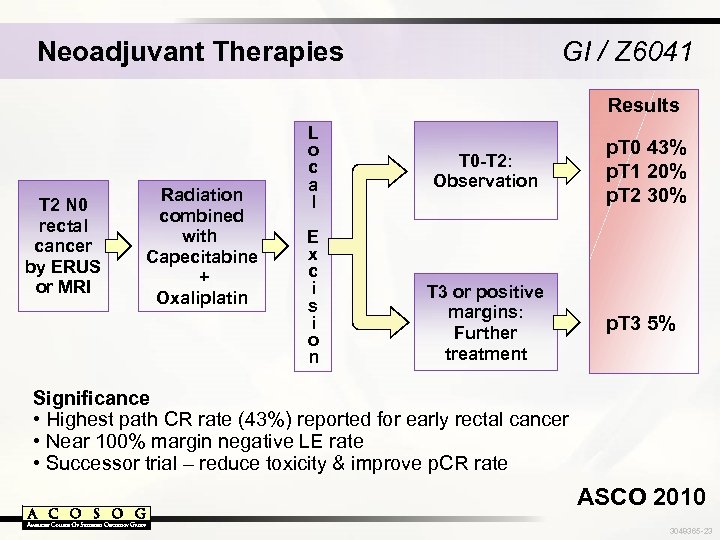 Neoadjuvant Therapies GI / Z 6041 Results T 2 N 0 rectal cancer by ERUS or MRI Radiation combined with Capecitabine + Oxaliplatin L o c a l E x c i s i o n T 0 -T 2: Observation T 3 or positive margins: Further treatment p. T 0 43% p. T 1 20% p. T 2 30% p. T 3 5% Significance • Highest path CR rate (43%) reported for early rectal cancer • Near 100% margin negative LE rate • Successor trial – reduce toxicity & improve p. CR rate ASCO 2010 3048365 -23
Neoadjuvant Therapies GI / Z 6041 Results T 2 N 0 rectal cancer by ERUS or MRI Radiation combined with Capecitabine + Oxaliplatin L o c a l E x c i s i o n T 0 -T 2: Observation T 3 or positive margins: Further treatment p. T 0 43% p. T 1 20% p. T 2 30% p. T 3 5% Significance • Highest path CR rate (43%) reported for early rectal cancer • Near 100% margin negative LE rate • Successor trial – reduce toxicity & improve p. CR rate ASCO 2010 3048365 -23
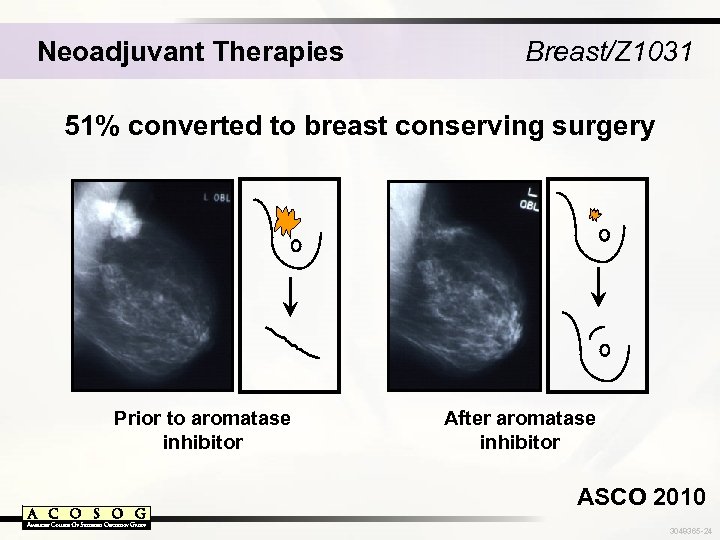 Neoadjuvant Therapies Breast/Z 1031 51% converted to breast conserving surgery Prior to aromatase inhibitor After aromatase inhibitor ASCO 2010 3048365 -24
Neoadjuvant Therapies Breast/Z 1031 51% converted to breast conserving surgery Prior to aromatase inhibitor After aromatase inhibitor ASCO 2010 3048365 -24
 Z 1031 Specimen Acquisition, Processing and Analysis 3048365 -25
Z 1031 Specimen Acquisition, Processing and Analysis 3048365 -25
 Z 1031 Specimen Acquisition, Processing and Analysis Genome Consent ocurement Banking Tracking Specimen Procurement Banking and Tracking Pathology Review and Analysis of Cellularity 3048365 -26
Z 1031 Specimen Acquisition, Processing and Analysis Genome Consent ocurement Banking Tracking Specimen Procurement Banking and Tracking Pathology Review and Analysis of Cellularity 3048365 -26
 Z 1031 Specimen Acquisition, Processing and Analysis Genome Consent Specimen Procurement Banking and Tracking Pathology Review and Analysis of Cellularity 3048365 -27
Z 1031 Specimen Acquisition, Processing and Analysis Genome Consent Specimen Procurement Banking and Tracking Pathology Review and Analysis of Cellularity 3048365 -27
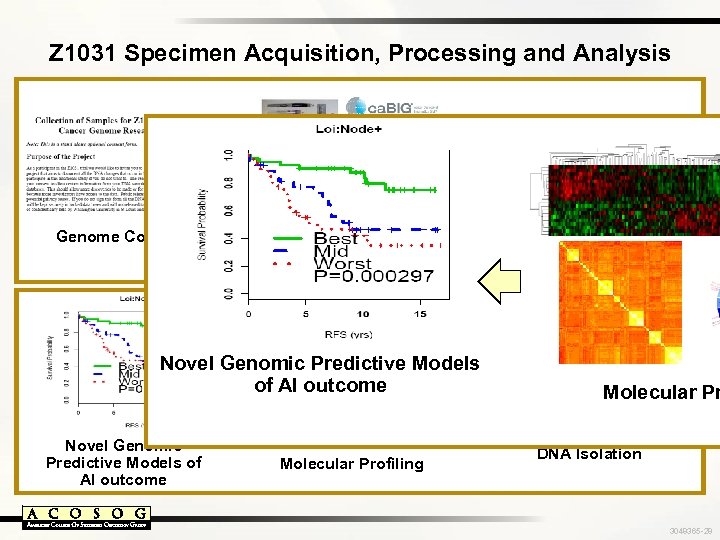 Z 1031 Specimen Acquisition, Processing and Analysis Genome Consent Specimen Procurement Banking and Tracking Novel Genomic Predictive Models of AI outcome Molecular Profiling Pathology Review and Analysis of Cellularity Molecular Pr High Quality RNA and DNA Isolation 3048365 -28
Z 1031 Specimen Acquisition, Processing and Analysis Genome Consent Specimen Procurement Banking and Tracking Novel Genomic Predictive Models of AI outcome Molecular Profiling Pathology Review and Analysis of Cellularity Molecular Pr High Quality RNA and DNA Isolation 3048365 -28
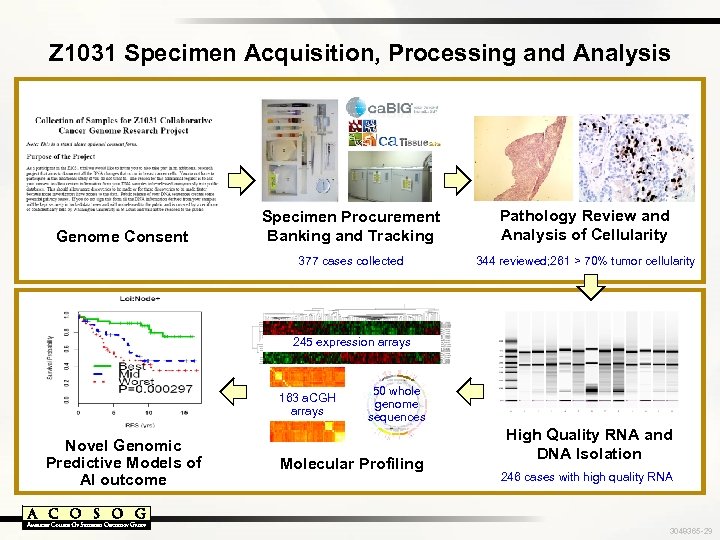 Z 1031 Specimen Acquisition, Processing and Analysis Pathology Review and Analysis of Cellularity 377 cases collected Genome Consent Specimen Procurement Banking and Tracking 344 reviewed; 261 > 70% tumor cellularity 245 expression arrays 163 a. CGH arrays Novel Genomic Predictive Models of AI outcome 50 whole genome sequences Molecular Profiling High Quality RNA and DNA Isolation 246 cases with high quality RNA 3048365 -29
Z 1031 Specimen Acquisition, Processing and Analysis Pathology Review and Analysis of Cellularity 377 cases collected Genome Consent Specimen Procurement Banking and Tracking 344 reviewed; 261 > 70% tumor cellularity 245 expression arrays 163 a. CGH arrays Novel Genomic Predictive Models of AI outcome 50 whole genome sequences Molecular Profiling High Quality RNA and DNA Isolation 246 cases with high quality RNA 3048365 -29
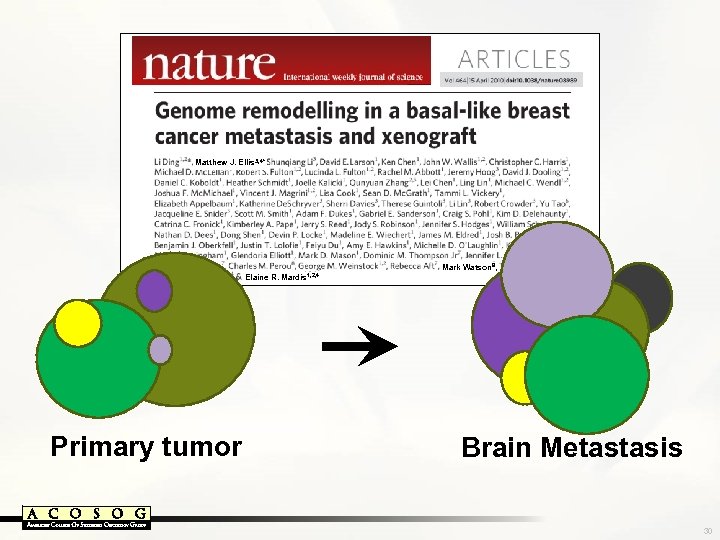 Matthew J. Ellis 3, 4* Mark Watson 9, Elaine R. Mardis 1, 2, 4 Primary tumor Brain Metastasis 30
Matthew J. Ellis 3, 4* Mark Watson 9, Elaine R. Mardis 1, 2, 4 Primary tumor Brain Metastasis 30
 How do we do it? 3048365 -31
How do we do it? 3048365 -31
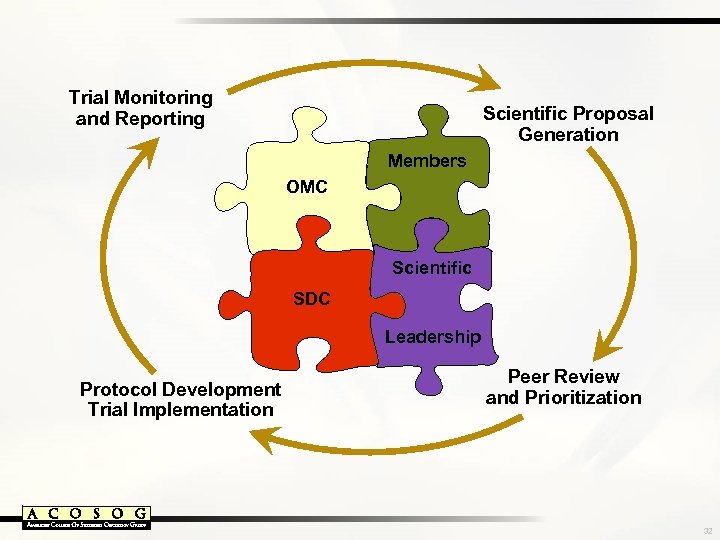 Trial Monitoring and Reporting Scientific Proposal Generation Members OMC Scientific SDC Leadership Protocol Development Trial Implementation Peer Review and Prioritization 32
Trial Monitoring and Reporting Scientific Proposal Generation Members OMC Scientific SDC Leadership Protocol Development Trial Implementation Peer Review and Prioritization 32
 Scientific Proposal Generation Idea generation – Scientific Committee Study team development of concept • Multidisciplinary and statistical input • External collaborations (QARC, other groups…) Feasibility estimates • National Cancer Database – case numbers • Network surveys – MD interest • Patient Advocacy input – patient interest 3048365 -33
Scientific Proposal Generation Idea generation – Scientific Committee Study team development of concept • Multidisciplinary and statistical input • External collaborations (QARC, other groups…) Feasibility estimates • National Cancer Database – case numbers • Network surveys – MD interest • Patient Advocacy input – patient interest 3048365 -33
 Peer Review and Prioritization Clinical Scientific Review Committee Translational Science Review Committee Central Specimen Bank and Pathology Committee Basic and Translational Science Committee • Opportunities: biospecimen acquisition, basic and correlative studies Scientific Steering Committee • Prioritization: portfolio and resource balance 3048365 -34
Peer Review and Prioritization Clinical Scientific Review Committee Translational Science Review Committee Central Specimen Bank and Pathology Committee Basic and Translational Science Committee • Opportunities: biospecimen acquisition, basic and correlative studies Scientific Steering Committee • Prioritization: portfolio and resource balance 3048365 -34
 Protocol Development/Trial Implementation NCI Steering Committee review Protocol development • Protocol Editor • Study Team • Statistics Engagement of ACOSOG networks 3048365 -35
Protocol Development/Trial Implementation NCI Steering Committee review Protocol development • Protocol Editor • Study Team • Statistics Engagement of ACOSOG networks 3048365 -35
 Trial Monitoring - Suite of Tools 3048365 -36
Trial Monitoring - Suite of Tools 3048365 -36
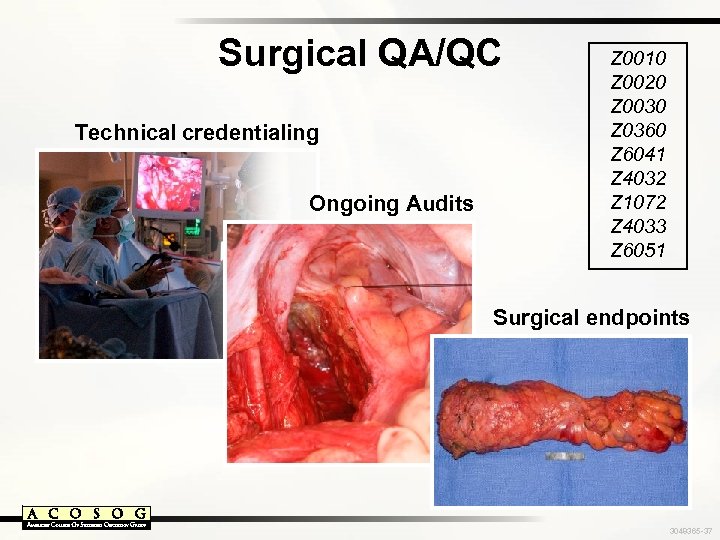 Surgical QA/QC Technical credentialing Ongoing Audits Z 0010 Z 0020 Z 0030 Z 0360 Z 6041 Z 4032 Z 1072 Z 4033 Z 6051 Surgical endpoints 3048365 -37
Surgical QA/QC Technical credentialing Ongoing Audits Z 0010 Z 0020 Z 0030 Z 0360 Z 6041 Z 4032 Z 1072 Z 4033 Z 6051 Surgical endpoints 3048365 -37
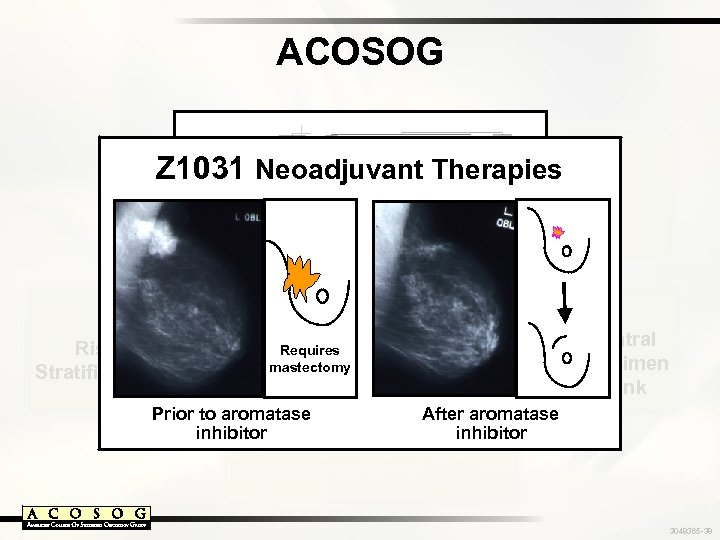 ACOSOG Z 1031 Individualized Response Monitoring Neoadjuvant Therapies Z 6051 Risk Stratification Requires mastectomy Novel Surgical Therapies Molecular Profiling Prior to aromatase inhibitor Central Specimen Bank After aromatase inhibitor 3048365 -38
ACOSOG Z 1031 Individualized Response Monitoring Neoadjuvant Therapies Z 6051 Risk Stratification Requires mastectomy Novel Surgical Therapies Molecular Profiling Prior to aromatase inhibitor Central Specimen Bank After aromatase inhibitor 3048365 -38
 Thank You 3048365 -39
Thank You 3048365 -39


