введение в иммулогию(25ноя2014).pptx
- Количество слайдов: 153
Alexander Gabibov Лекции 3 -4 Комбинаторные подходы в химии и биологии Каталитические антитела Moscow University, November, 26

Why we need proteins with novel functionality? Limited Natural Repertoires for Biotech & Bio. Pharma purposes

Enzymes – the catalysts of life – have been selected by natural evolution to sustain the development of living cells and organisms in their natural environment. Consequently, natural enzymes do not necessarily have adequate properties, specificity, stability or catalytic activity to make them suitable for biotechnological applications. Jacques Fastrez

Enzyme Evolution How has evolution produced an incredible variety of enzymatic activities from a limited number of protein folds? Enzyme superfamily of different types could be substituted for immunoglobulin superfamily
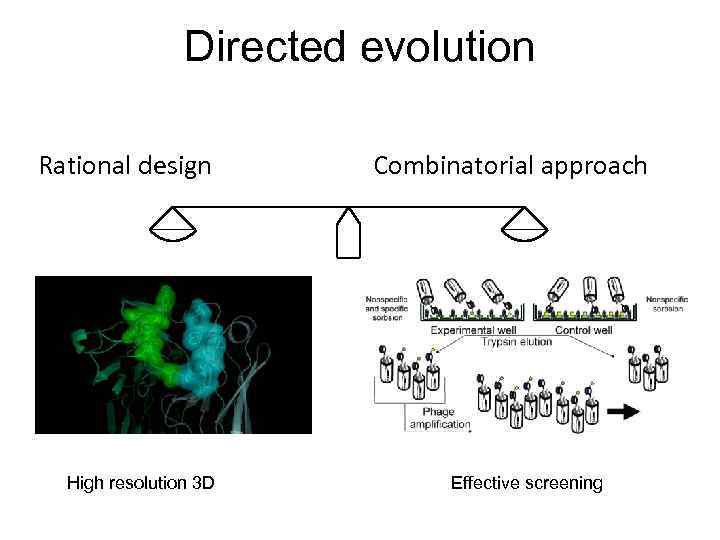
Directed evolution Rational design High resolution 3 D Combinatorial approach Effective screening
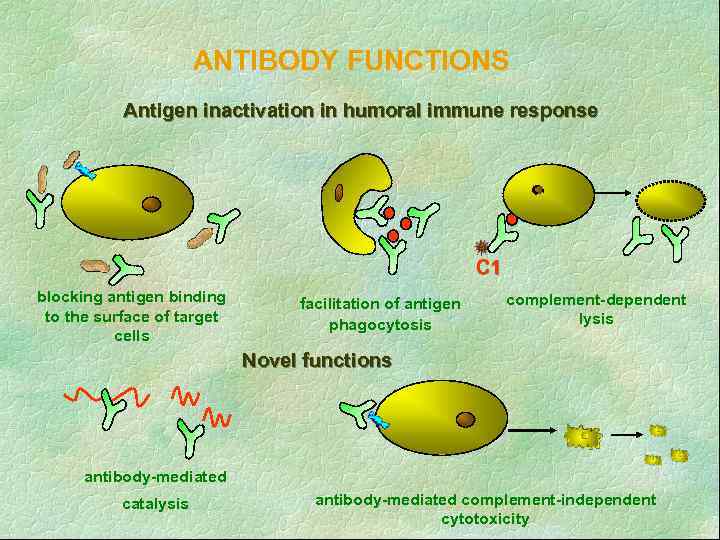
ANTIBODY FUNCTIONS Antigen inactivation in humoral immune response C 1 blocking antigen binding to the surface of target cells facilitation of antigen phagocytosis complement-dependent lysis Novel functions antibody-mediated catalysis antibody-mediated complement-independent cytotoxicity
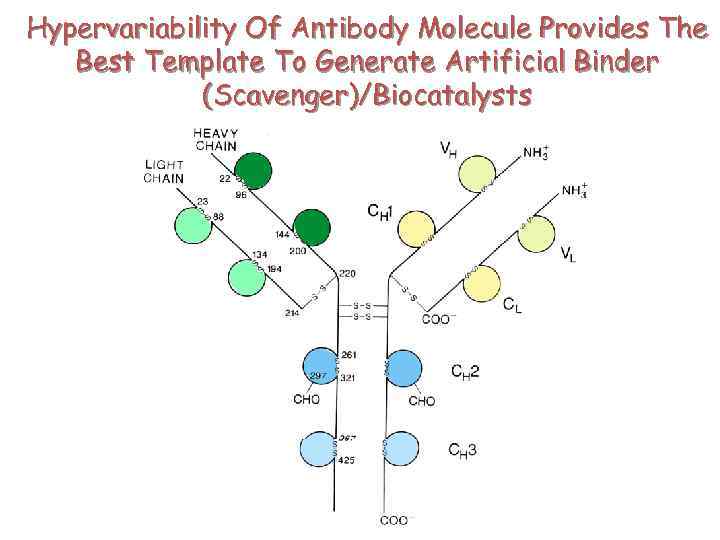
Hypervariability Of Antibody Molecule Provides The Best Template To Generate Artificial Binder (Scavenger)/Biocatalysts

Catalytic Antibodies Anti. Bodies ENzymes ABzymes

MESSAGE To make de novo functional binder/biocatalyst using Ig template we have to: 1)enlarge the repertoire for combinatorial screening, 2)propose the “vector” for selection strategy For these purposes we may use: 1)phage-display libraries (“immunization” and screening in vitro), 2)autoimmune repertoires (in vivo)

Chemical selection of phage display library Enzymes Million years of evolution Artificial Enzymes made by Combinatorial Approaches Several months or weeks of evolution Selection in the test tube from naïve repertoire 108 in mouse to 109 in semisynthetic libraries Enzyme superfamily of different types could be substituted by immunoglobulin superfamily

Three critical aspects in any combinatorial library: üThe chemical units that go to the library üThe technique for generating the library üIdentification of the library members that interact with the biological target of interest
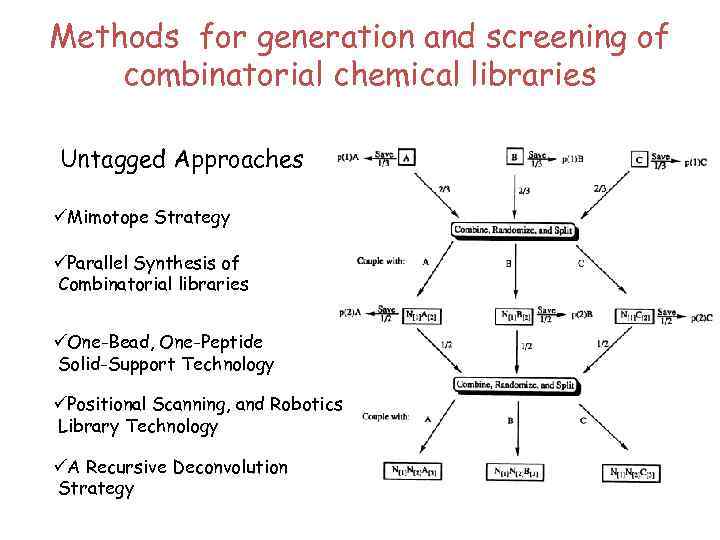
Methods for generation and screening of combinatorial chemical libraries Untagged Approaches üMimotope Strategy üParallel Synthesis of Combinatorial libraries üOne-Bead, One-Peptide Solid-Support Technology üPositional Scanning, and Robotics Library Technology üA Recursive Deconvolution Strategy

Methods for generation and screening of combinatorial chemical libraries Tagged Methodologies: Based on a physical link between a protein and its encoded gene – the best known system is phage display. üPhage Technology üPeptides on Plasmids üPeptide coded Libraries üElectrophoric Polyhalobenzene Coded Libraries üEncoded Combinatorial Libraries The product of gene 8 (g 8 p) – is a small protein that forms the cylinder of the capsid; its number of copies (2700 for the wild-type phage). The other coat proteins (g 3 p, g 6 p, g 7 p and g 9 p) close the extremities of the cylinder. The product of gene 3 – present in three to five copies – is responsible for phage infectivity. Proteins of interest are usually fused by their C-terminal to g 3 p or to g 8 p. Filamentous phage
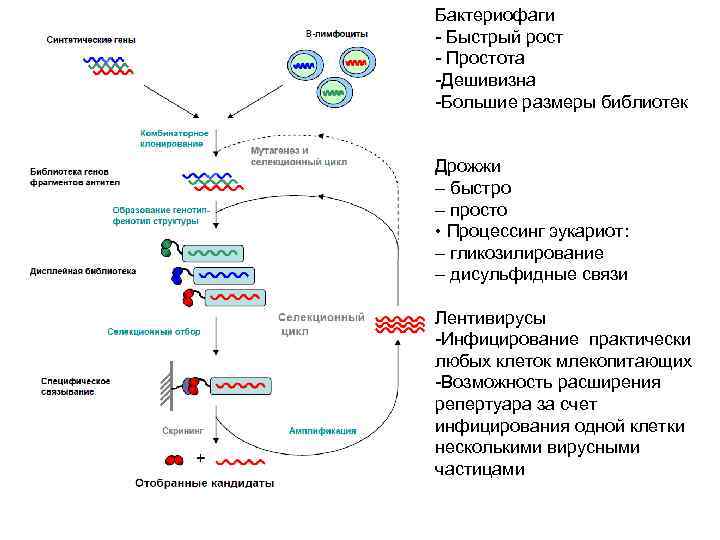
Бактериофаги - Быстрый рост - Простота -Дешивизна -Большие размеры библиотек Дрожжи – быстро – просто • Процессинг эукариот: – гликозилирование – дисульфидные связи Лентивирусы -Инфицирование практически любых клеток млекопитающих -Возможность расширения репертуара за счет инфицирования одной клетки несколькими вирусными частицами
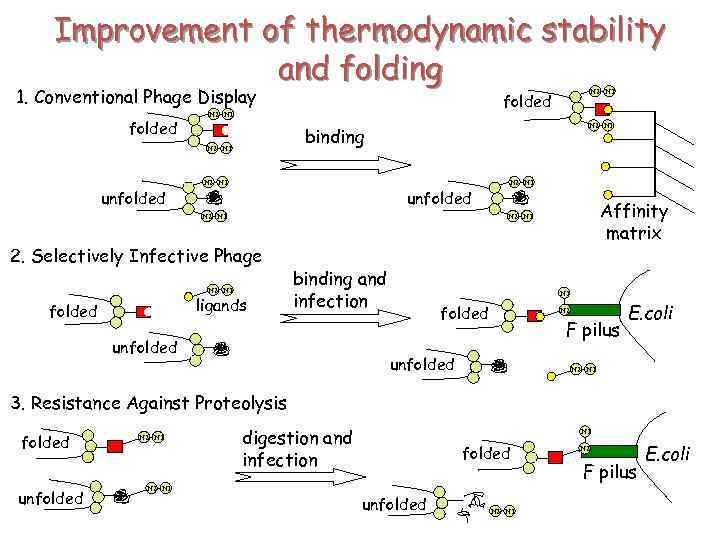
Improvement of thermodynamic stability and folding 1. Conventional Phage Display folded N 2 N 1 binding N 2 N 1 unfolded N 2 N 1 Affinity matrix N 2 N 1 2. Selectively Infective Phage N 2 N 1 ligands folded N 2 N 1 folded binding and infection unfolded N 1 folded N 2 F pilus unfolded E. coli N 2 N 1 3. Resistance Against Proteolysis folded unfolded N 2 N 1 digestion and infection N 1 folded unfolded N 2 N 1 N 2 F pilus E. coli
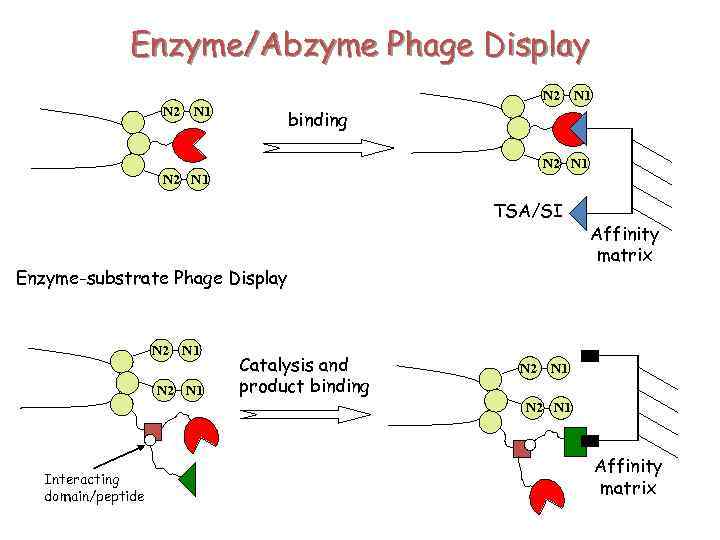
Enzyme/Abzyme Phage Display N 2 N 1 binding N 2 N 1 TSA/SI Enzyme-substrate Phage Display N 2 N 1 Catalysis and product binding Affinity matrix N 2 N 1 Interacting domain/peptide Affinity matrix
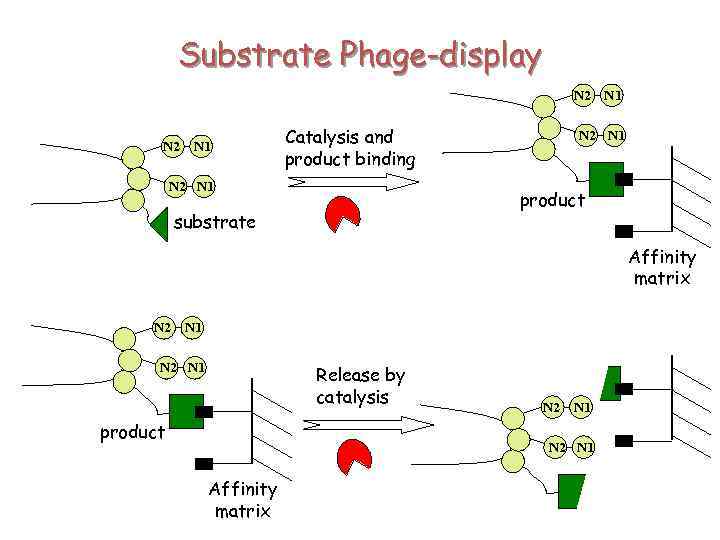
Substrate Phage-display N 2 N 1 Catalysis and product binding N 2 N 1 product substrate Affinity matrix N 2 N 1 Release by catalysis product N 2 N 1 Affinity matrix
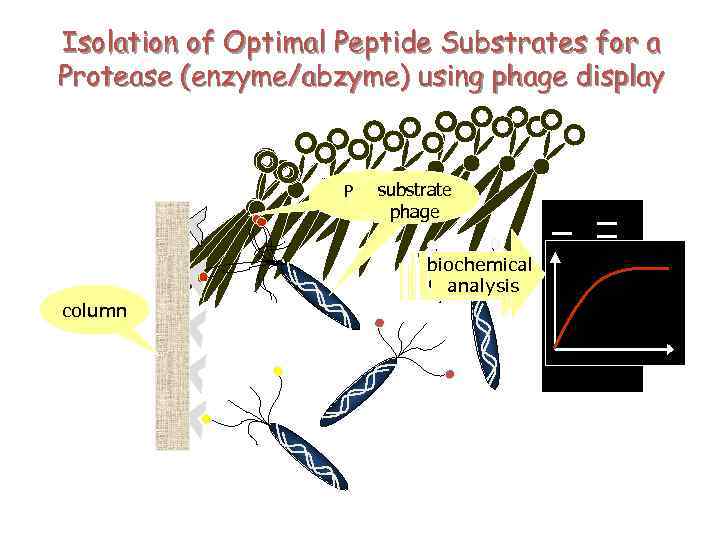
Isolation of Optimal Peptide Substrates for a Protease (enzyme/abzyme) using phage display substrate Protease LF phage biochemical sequencing analysis column
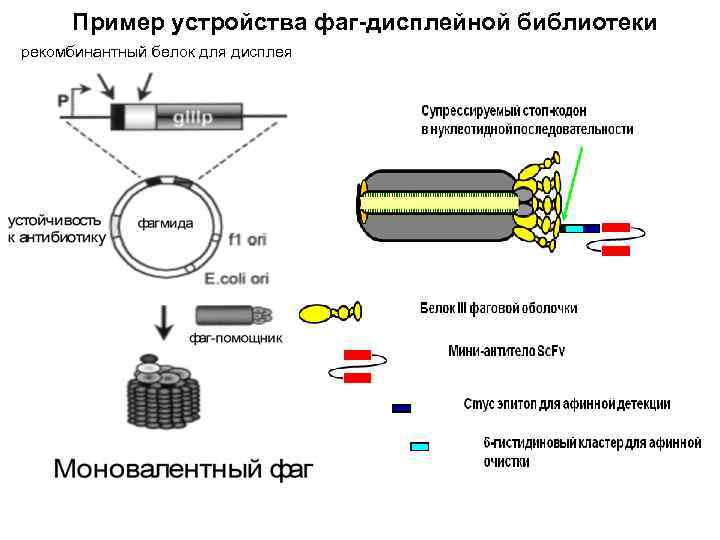
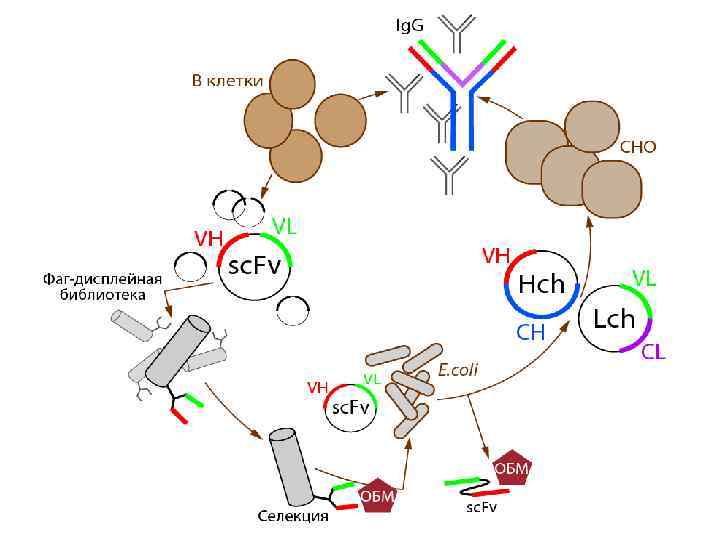
Пример устройства фаг-дисплейной библиотеки рекомбинантный белок для дисплея
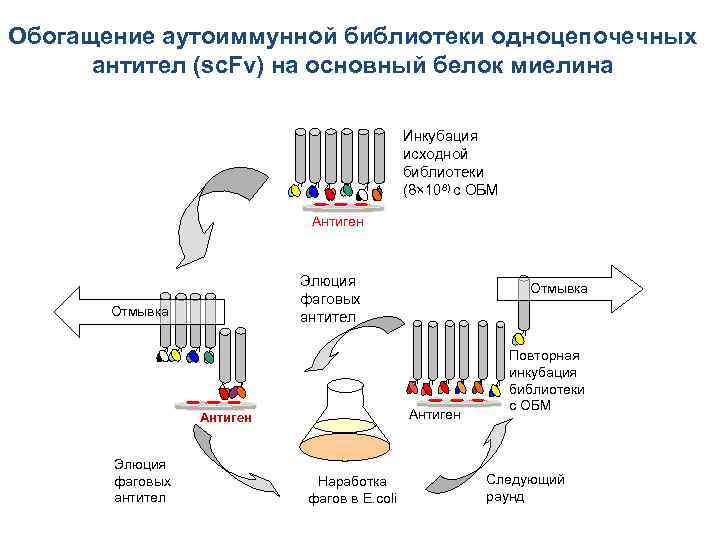
Обогащение аутоиммунной библиотеки одноцепочечных антител (sc. Fv) на основный белок миелина Инкубация исходной библиотеки (8× 108) c ОБМ Антиген Элюция фаговых антител Отмывка Наработка фагов в E. coli Повторная инкубация библиотеки с ОБМ Следующий раунд
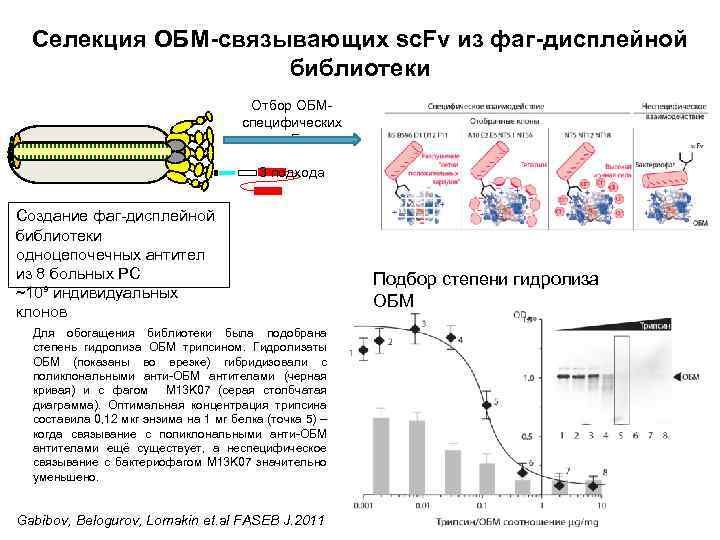
Селекция ОБМ-связывающих sc. Fv из фаг-дисплейной библиотеки Отбор ОБМспецифических sc. Fv 3 подхода Создание фаг-дисплейной библиотеки одноцепочечных антител из 8 больных РС ~10⁹ индивидуальных клонов Для обогащения библиотеки была подобрана степень гидролиза ОБМ трипсином. Гидролизаты ОБМ (показаны во врезке) гибридизовали с поликлональными анти-ОБМ антителами (черная кривая) и с фагом M 13 K 07 (серая столбчатая диаграмма). Оптимальная концентрация трипсина составила 0, 12 мкг энзима на 1 мг белка (точка 5) – когда связывание с поликлональными анти-ОБМ антителами ещё существует, а неспецифическое связывание с бактериофагом M 13 K 07 значительно уменьшено. Gabibov, Belogurov, Lomakin et. al FASEB J. 2011 Подбор степени гидролиза ОБМ
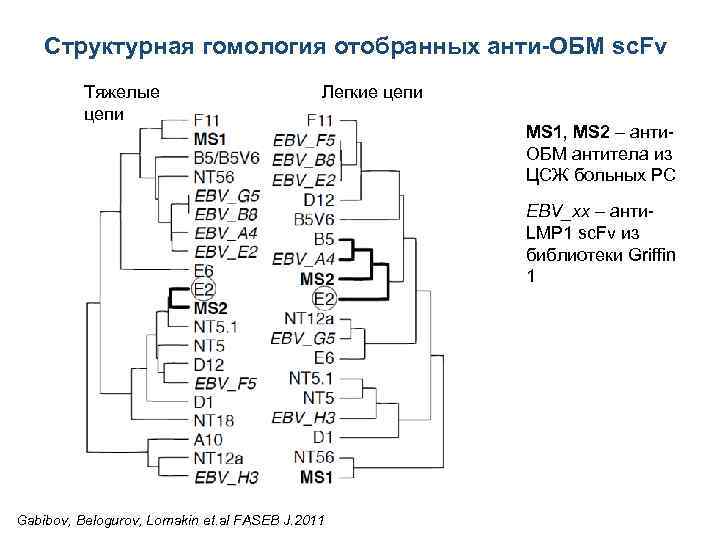
Структурная гомология отобранных анти-ОБМ sc. Fv Тяжелые цепи Легкие цепи MS 1, MS 2 – анти. ОБМ антитела из ЦСЖ больных РС EBV_xx – анти. LMP 1 sc. Fv из библиотеки Griffin 1 Gabibov, Belogurov, Lomakin et. al FASEB J. 2011
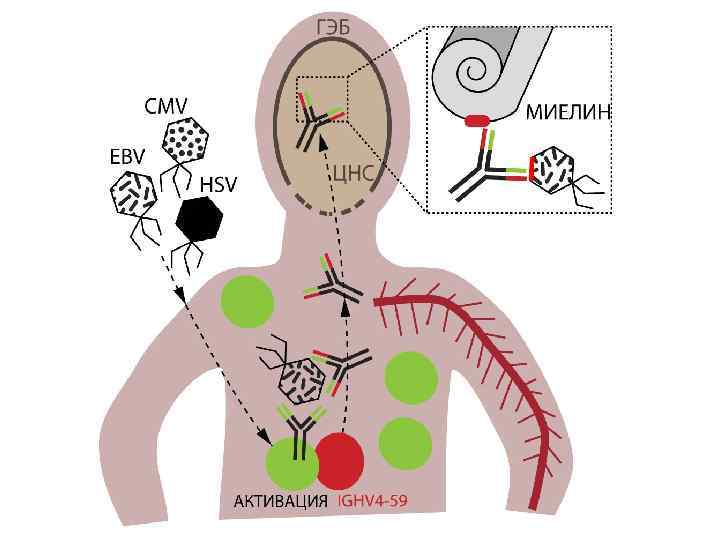
EBV как возможный триггер РС The New England Journal of Medicine, 2003. Hartmut Wekerle
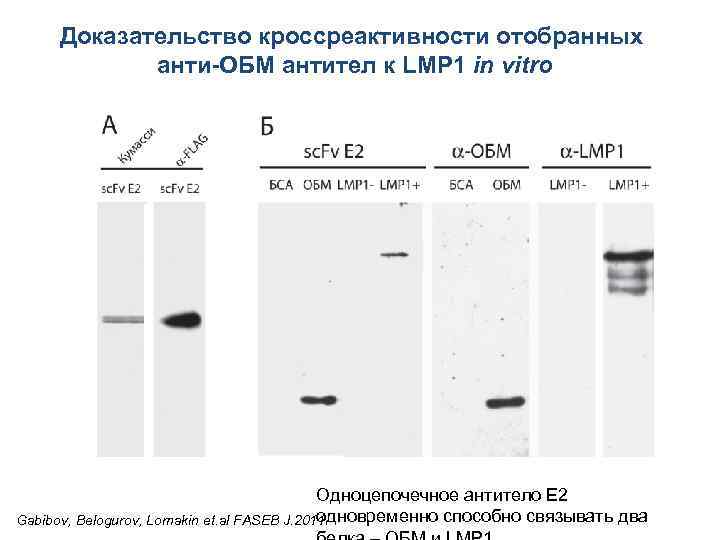
Доказательство кроссреактивности отобранных анти-ОБМ антител к LMP 1 in vitro Одноцепочечное антитело E 2 одновременно способно связывать два Gabibov, Belogurov, Lomakin et. al FASEB J. 2011
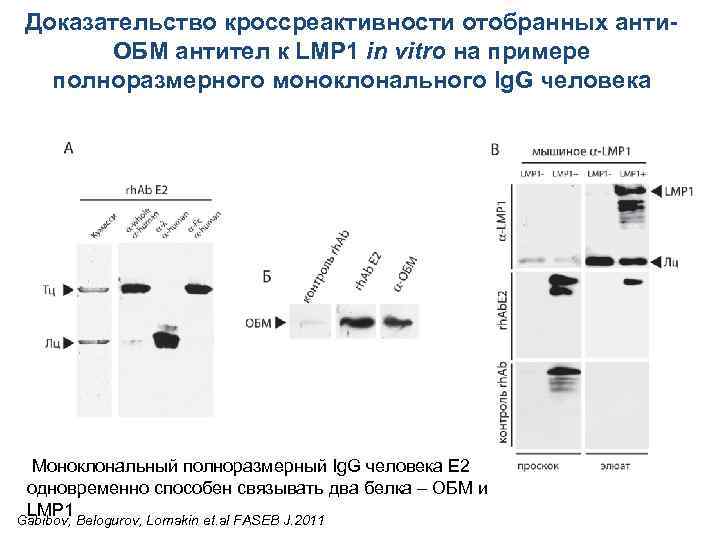
Доказательство кроссреактивности отобранных анти. ОБМ антител к LMP 1 in vitro на примере полноразмерного моноклонального Ig. G человека Моноклональный полноразмерный Ig. G человека E 2 одновременно способен связывать два белка – ОБМ и LMP 1 Gabibov, Belogurov, Lomakin et. al FASEB J. 2011
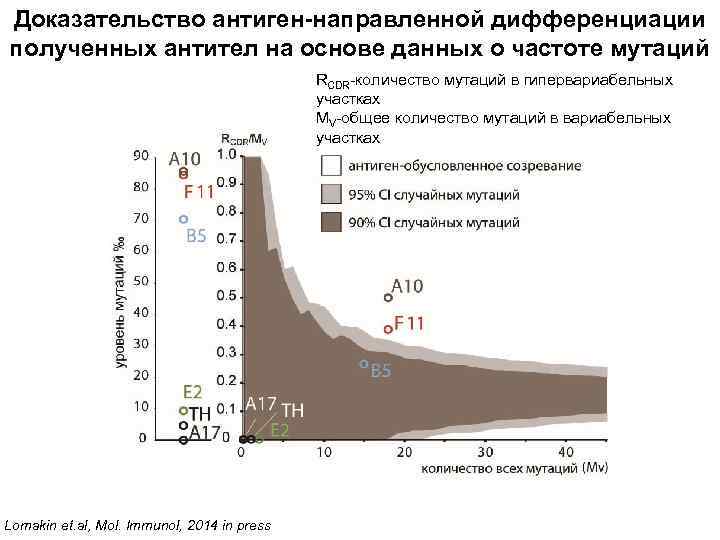
Доказательство антиген-направленной дифференциации полученных антител на основе данных о частоте мутаций RCDR-количество мутаций в гипервариабельных участках MV-общее количество мутаций в вариабельных участках Lomakin et. al, Mol. Immunol, 2014 in press
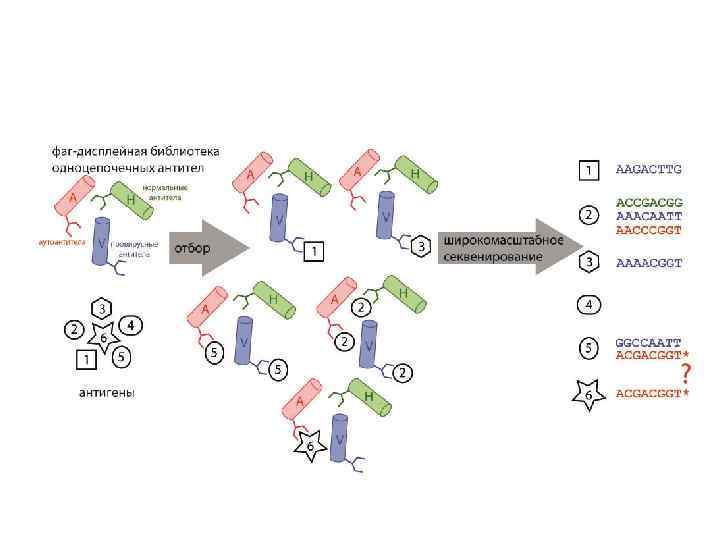
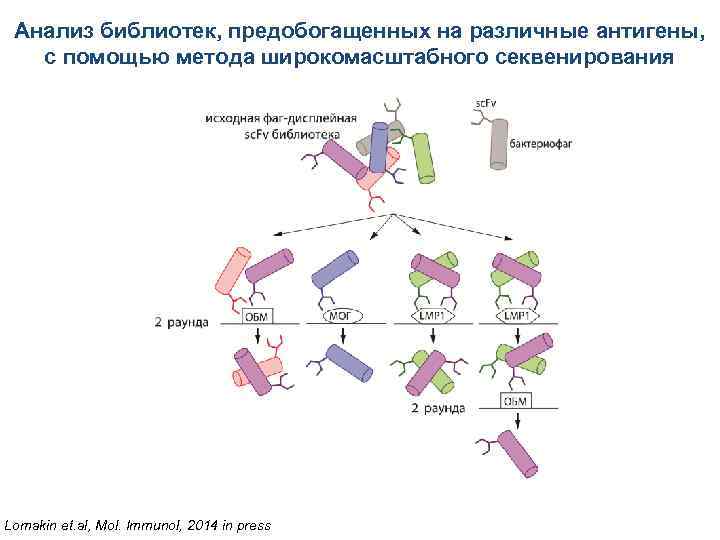
Анализ библиотек, предобогащенных на различные антигены, с помощью метода широкомасштабного секвенирования Lomakin et. al, Mol. Immunol, 2014 in press
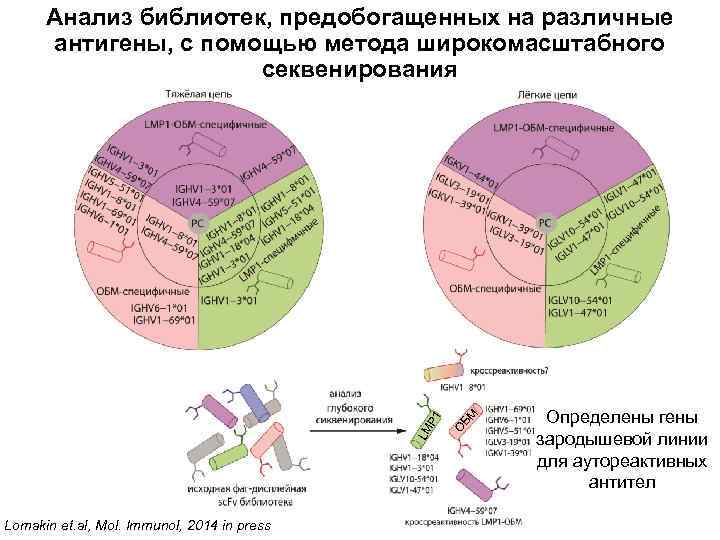
Lomakin et. al, Mol. Immunol, 2014 in press БМ О LM P 1 Анализ библиотек, предобогащенных на различные антигены, с помощью метода широкомасштабного секвенирования Определены гены зародышевой линии для аутореактивных антител
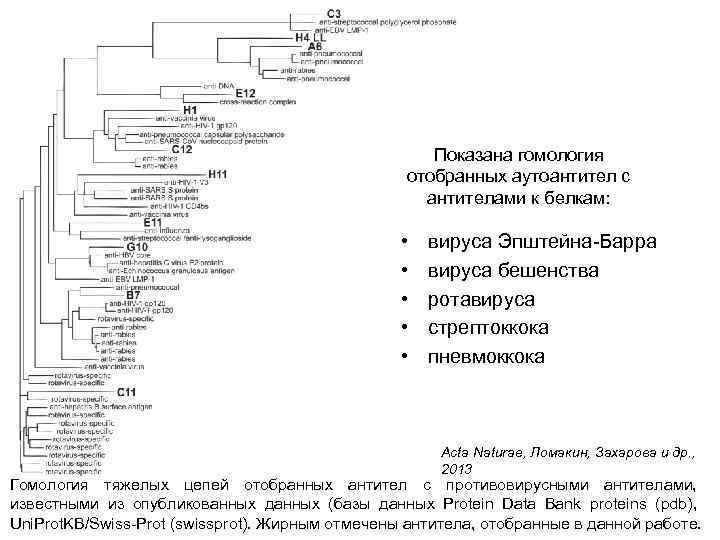
Показана гомология отобранных аутоантител с антителами к белкам: • • • вируса Эпштейна-Барра вируса бешенства ротавируса стрептоккока пневмоккока Acta Naturae, Ломакин, Захарова и др. , 2013 Гомология тяжелых цепей отобранных антител с противовирусными антителами, известными из опубликованных данных (базы данных Protein Data Bank proteins (pdb), Uni. Prot. KB/Swiss-Prot (swissprot). Жирным отмечены антитела, отобранные в данной работе.
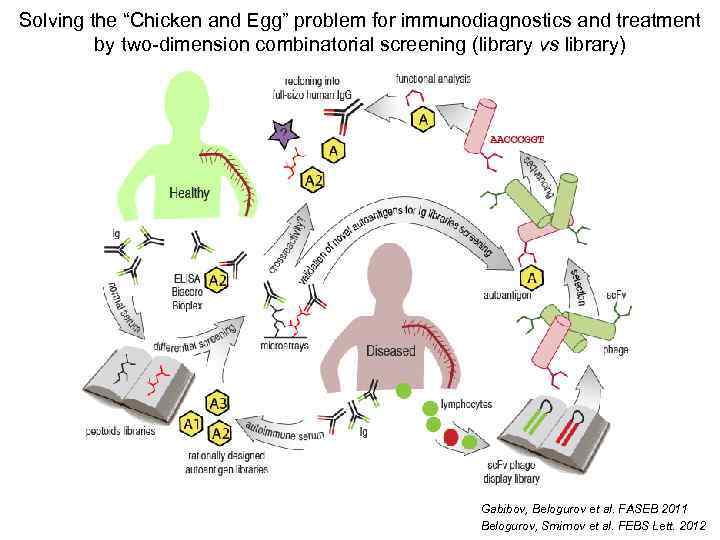
Solving the “Chicken and Egg” problem for immunodiagnostics and treatment by two-dimension combinatorial screening (library vs library) Gabibov, Belogurov et al. FASEB 2011 Belogurov, Smirnov et al. FEBS Lett. 2012
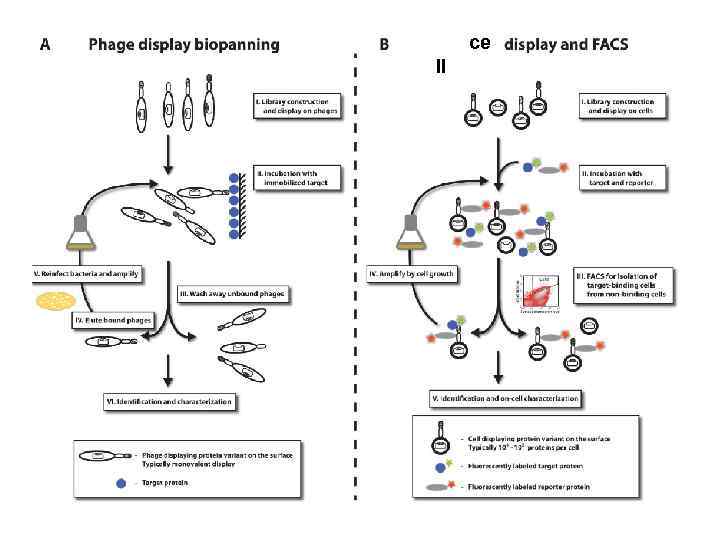
ce ll
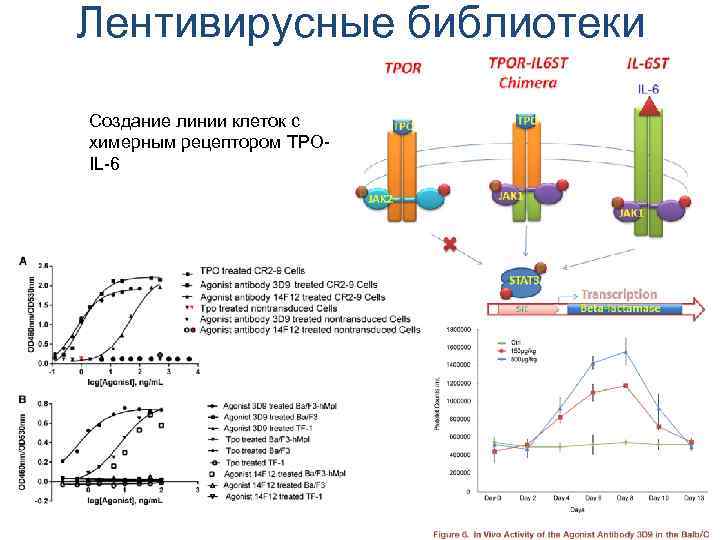
Лентивирусные библиотеки Создание линии клеток с химерным рецептором TPOIL-6
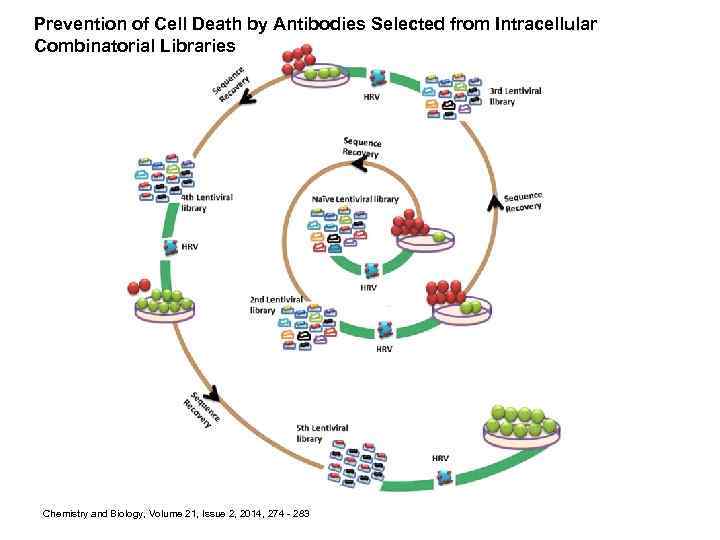
Prevention of Cell Death by Antibodies Selected from Intracellular Combinatorial Libraries Chemistry and Biology, Volume 21, Issue 2, 2014, 274 - 283
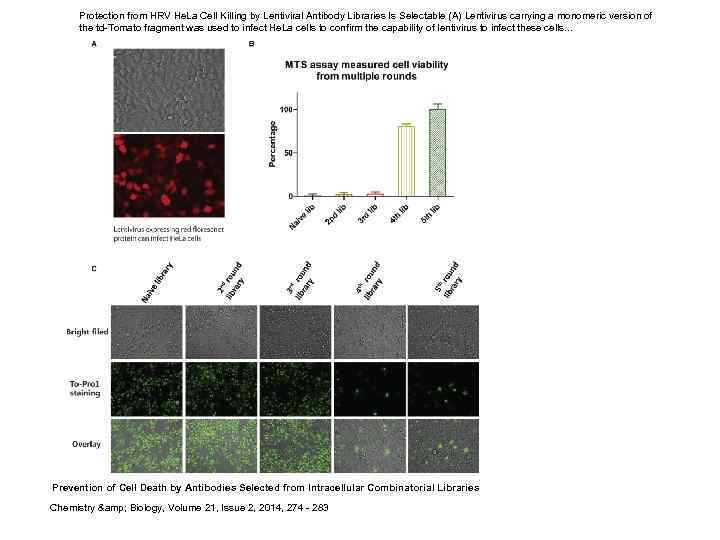
Protection from HRV He. La Cell Killing by Lentiviral Antibody Libraries Is Selectable (A) Lentivirus carrying a monomeric version of the td-Tomato fragment was used to infect He. La cells to confirm the capability of lentivirus to infect these cells. . . Prevention of Cell Death by Antibodies Selected from Intracellular Combinatorial Libraries Chemistry & Biology, Volume 21, Issue 2, 2014, 274 - 283
Phenotype-information-phenotype cycle.
Antigen display on bacterial surfaces.
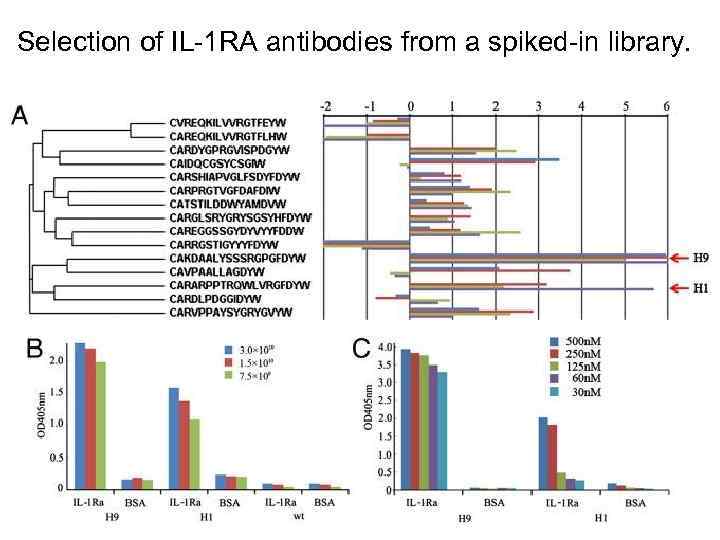
Selection of IL-1 RA antibodies from a spiked-in library.
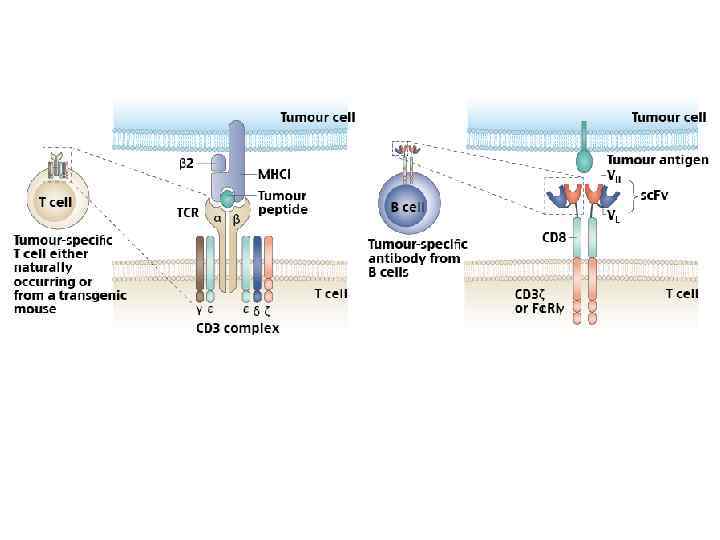
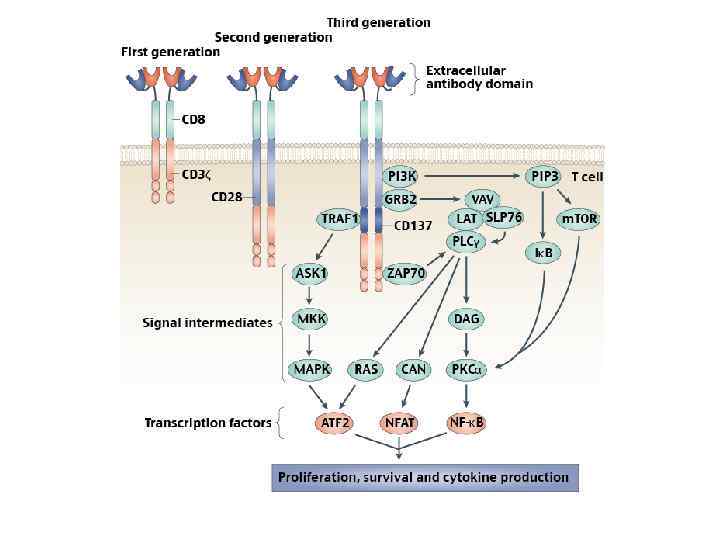
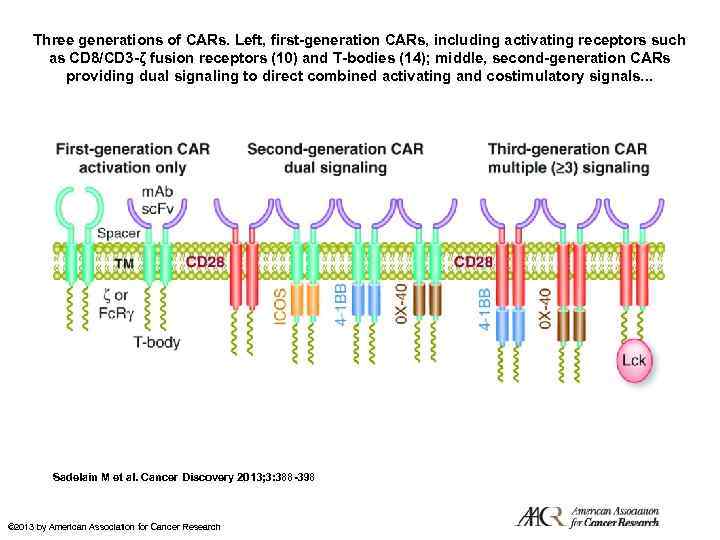
Three generations of CARs. Left, first-generation CARs, including activating receptors such as CD 8/CD 3 -ζ fusion receptors (10) and T-bodies (14); middle, second-generation CARs providing dual signaling to direct combined activating and costimulatory signals. . . Sadelain M et al. Cancer Discovery 2013; 3: 388 -398 © 2013 by American Association for Cancer Research
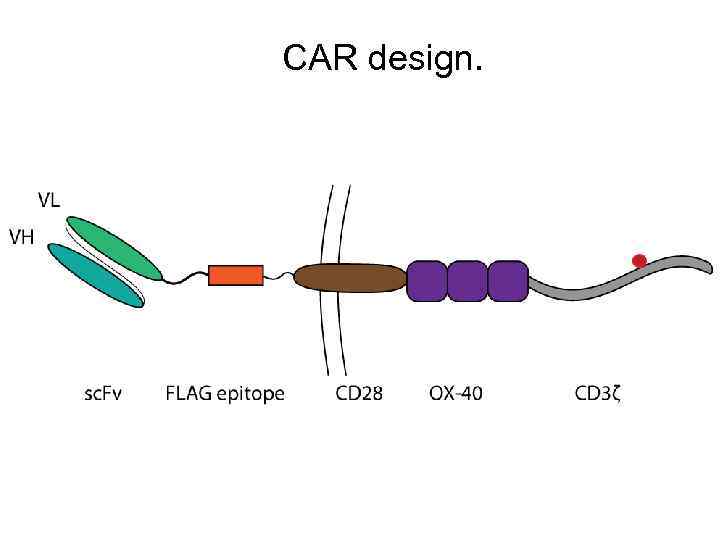
CAR design.
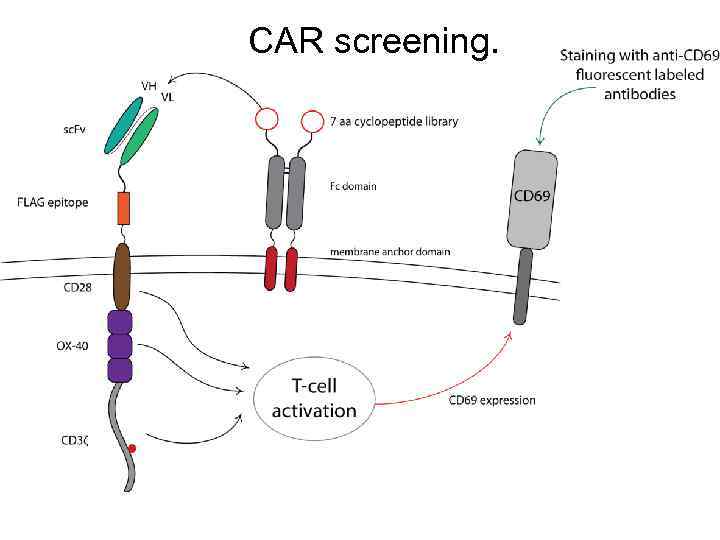
CAR screening.

Catalytic Antibodies Anti. Bodies ENzymes ABzymes

Catalytic Antibodies. Historical Background Emil Fisher, Berichte Deutsh. Chem. Ges. , 1894, vol. 27, p. 2985 “The Lock and Key” approach William Jencks, Catalysis in Chemistry and Enzymology, Mc. Grow Hill, 1969 Catalytic properties of anti-transition state antibodies Richard Lerner, Stephen Benkovic, Peter Shultz Science, 1991, 252(5006): 659 -67. At the crossroads of chemistry and immunology: catalytic antibodies. Linus Pauling, Am. Sci. , 1948, vol. 36, p. 51 ”Transition state” concept Niels Jerne, Ann. Inst. Pasteur. Immunol. , 125 C, 1974, p. 373 Network theory of Immune System
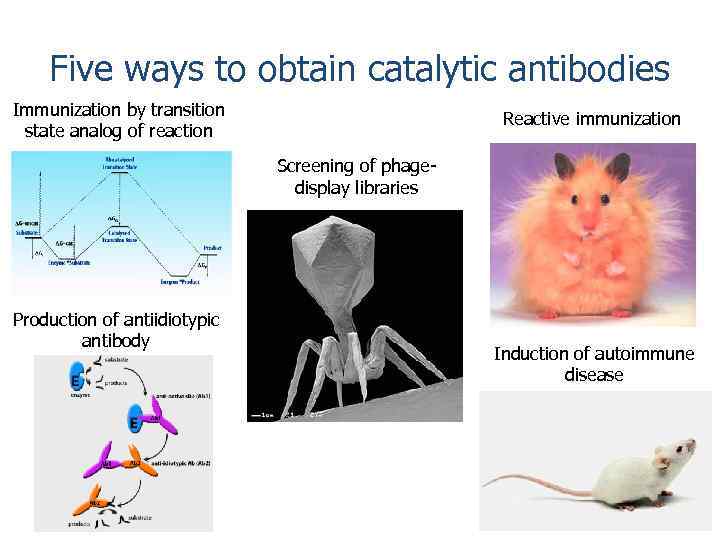
Five ways to obtain catalytic antibodies Immunization by transition state analog of reaction Reactive immunization Screening of phagedisplay libraries Production of antiidiotypic antibody Induction of autoimmune disease

• PLAN OF THE LECTURE • Rational design • -TSA approach - Antiidiotypic catalytic antibodies with amidase and esterase activities -Combinatorial approach - Artificial abzymes toward OPC • induction of anti-gp 120 catalytic antibodies by reactive immunization (catalytic vaccines) • Catalytic antibodies against autoantigens - Natural DNA-abzymes - MBP-hydrolyzing antibodies in Multiple Sclerosis • Catalytic antibodies as “Double-Edge Sword”
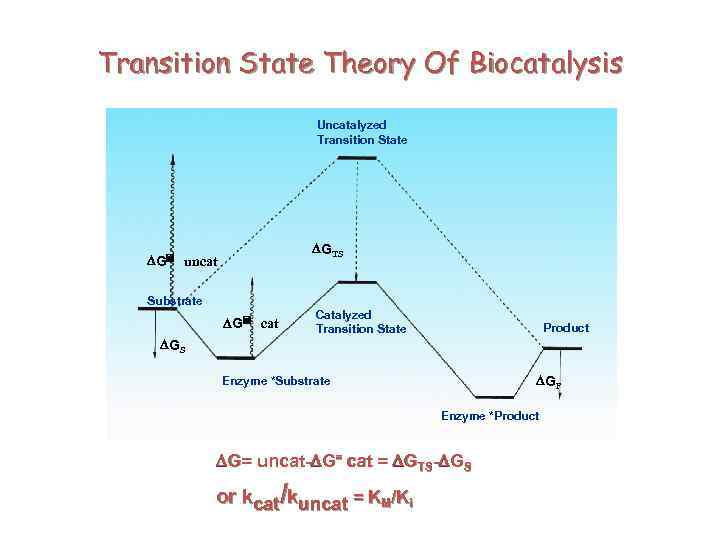
Transition State Theory Of Biocatalysis Uncatalyzed Transition State DGTS DG= uncat Substrate DG= cat Catalyzed Transition State Product DGS DGP Enzyme *Substrate Enzyme *Product DG= uncat-DG= cat = DGTS-DGS or kcat/kuncat = KM/Ki
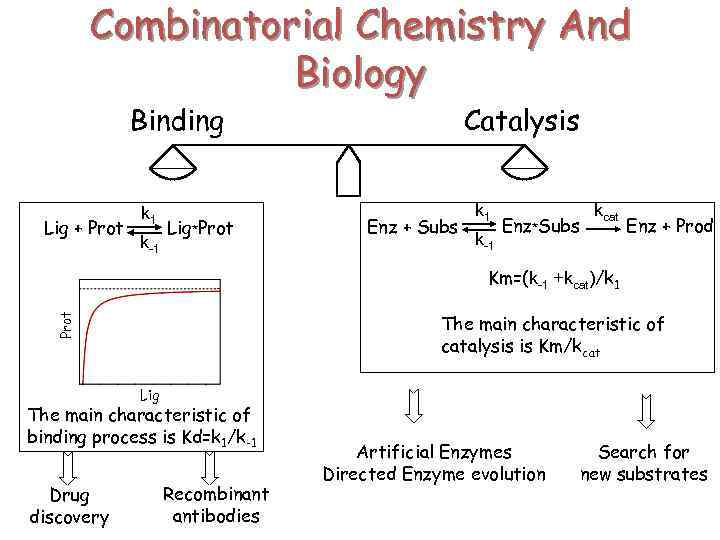
Combinatorial Chemistry And Biology Binding Lig + Prot k 1 k-1 Lig*Prot Catalysis Enz + Subs k 1 k-1 Enz*Subs kcat Enz + Prod Km=(k-1 +kcat)/k 1 Prot The main characteristic of catalysis is Km/kcat Lig The main characteristic of binding process is Kd=k 1/k-1 Drug discovery Recombinant antibodies Artificial Enzymes Directed Enzyme evolution Search for new substrates
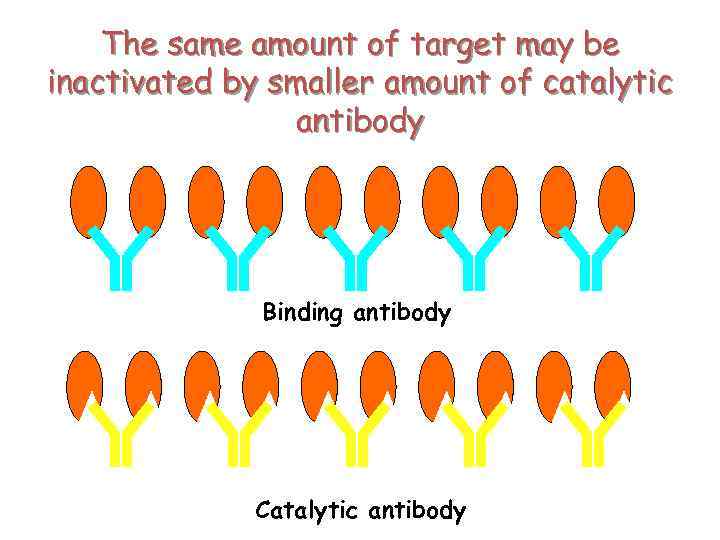
The same amount of target may be inactivated by smaller amount of catalytic antibody Binding antibody Catalytic antibody

Enzyme Catalysis in Organic Synthesis. Chapter 14. Catalytic antibodies. Smirnov I. , Belogurov A. , Gabibov A. Wiley-VCH, 2012
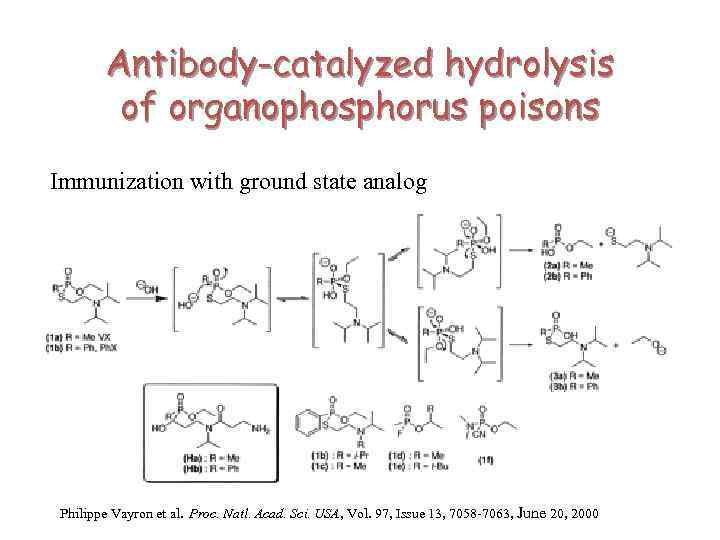
Antibody-catalyzed hydrolysis of organophosphorus poisons Immunization with ground state analog Philippe Vayron et al. Proc. Natl. Acad. Sci. USA, Vol. 97, Issue 13, 7058 -7063, June 20, 2000
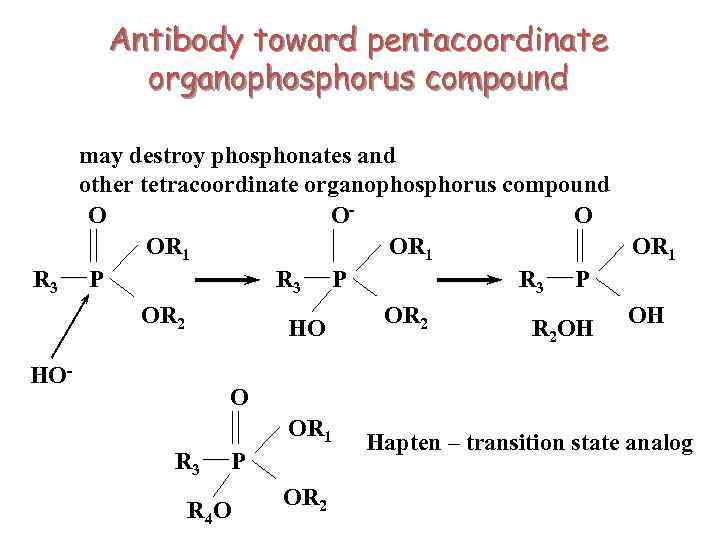
Antibody toward pentacoordinate organophosphorus compound R 3 may destroy phosphonates and other tetracoordinate organophosphorus compound O OO OR 1 R 3 P P OR 2 HO HO- OR 2 R 2 OH OH O OR 1 R 3 P R 4 O OR 2 Hapten – transition state analog
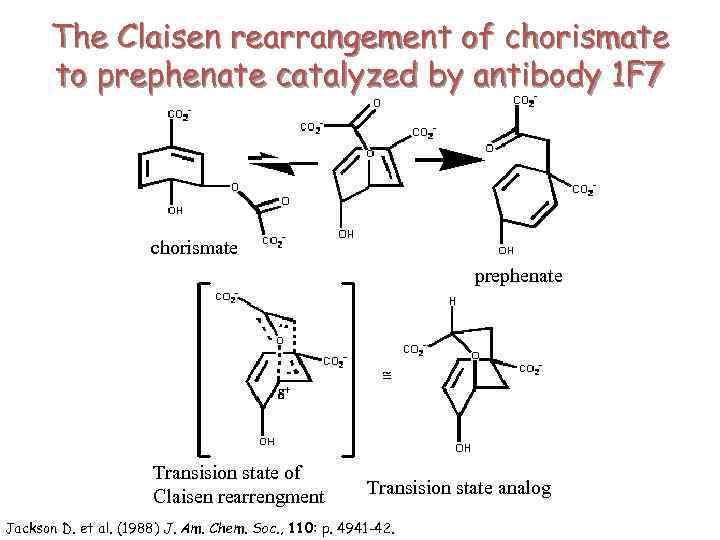
The Claisen rearrangement of chorismate to prephenate catalyzed by antibody 1 F 7 chorismate prephenate Transision state of Claisen rearrengment Transision state analog Jackson D. et al. (1988) J. Am. Chem. Soc. , 110: p. 4941 -42.
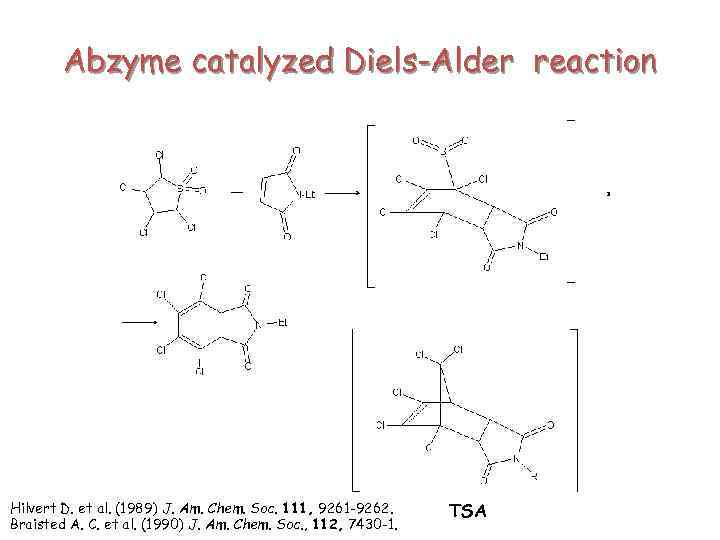
Abzyme catalyzed Diels-Alder reaction Hilvert D. et al. (1989) J. Am. Chem. Soc. 111, 9261 -9262. Braisted A. C. et al. (1990) J. Am. Chem. Soc. , 112, 7430 -1. TSA
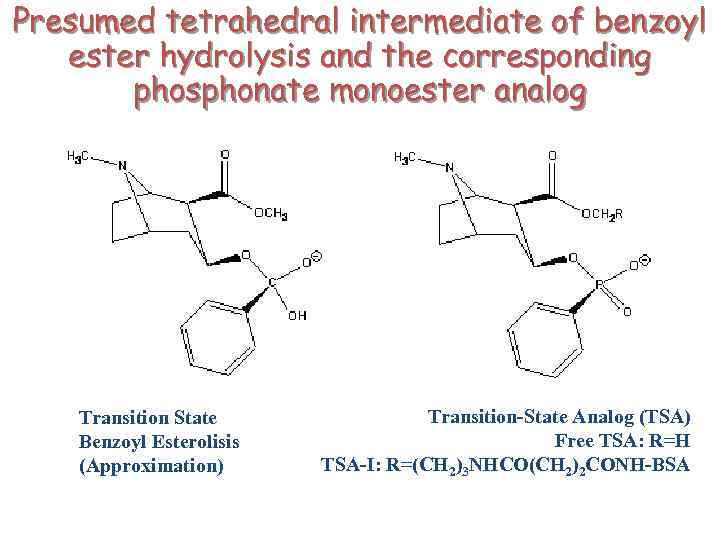
Presumed tetrahedral intermediate of benzoyl ester hydrolysis and the corresponding phosphonate monoester analog Transition State Benzoyl Esterolisis (Approximation) Transition-State Analog (TSA) Free TSA: R=H TSA-I: R=(CH 2)3 NHCO(CH 2)2 CONH-BSA
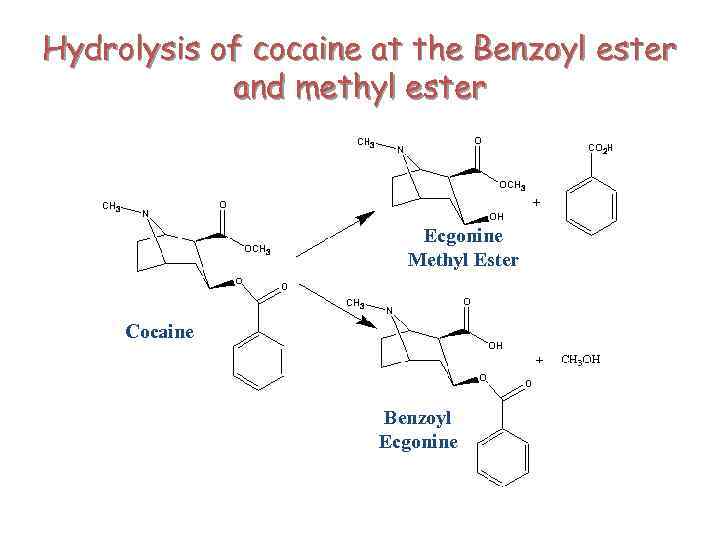
Hydrolysis of cocaine at the Benzoyl ester and methyl ester Ecgonine Methyl Ester Cocaine Benzoyl Ecgonine
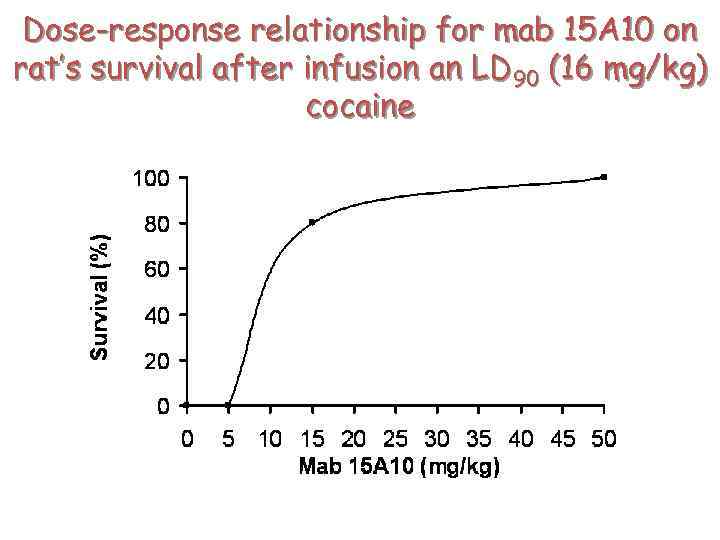
Dose-response relationship for mab 15 A 10 on rat’s survival after infusion an LD 90 (16 mg/kg) cocaine
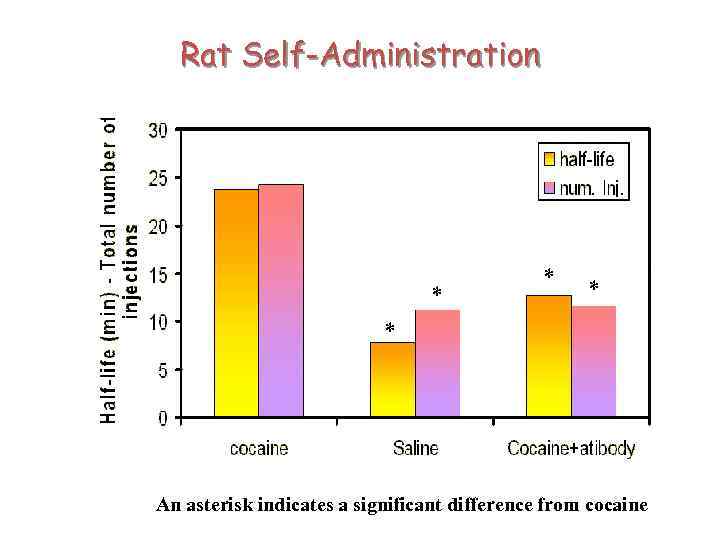
Rat Self-Administration * * An asterisk indicates a significant difference from cocaine
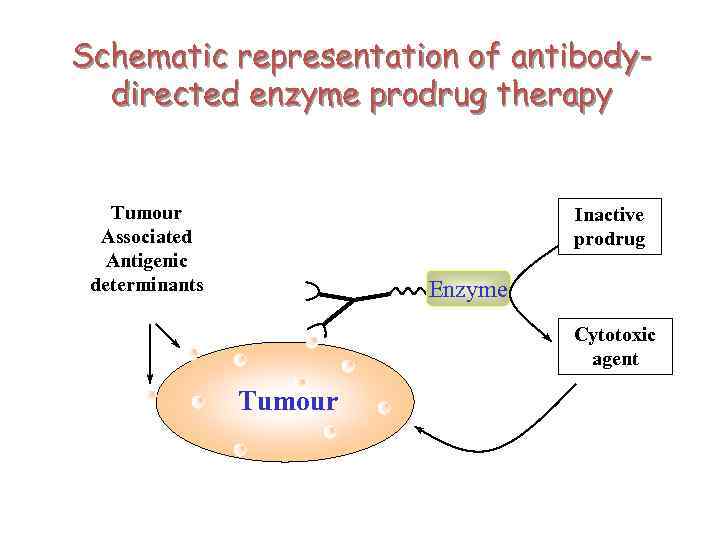
Schematic representation of antibodydirected enzyme prodrug therapy Tumour Associated Antigenic determinants Inactive prodrug Enzyme Cytotoxic agent Tumour
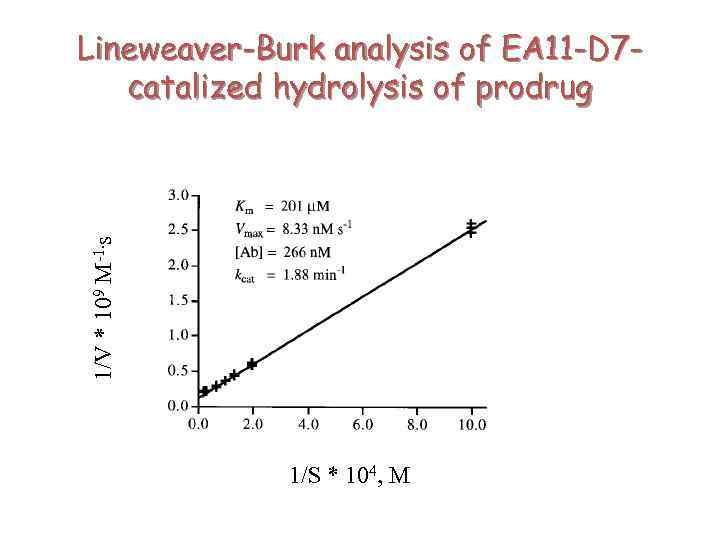
1/V * 109 M-1. s Lineweaver-Burk analysis of EA 11 -D 7 catalized hydrolysis of prodrug 1/S * 104, M
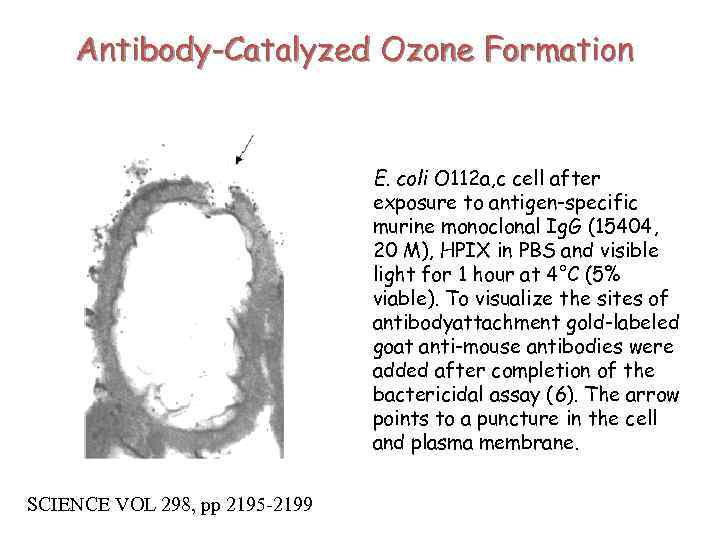
Antibody-Catalyzed Ozone Formation E. coli O 112 a, c cell after exposure to antigen-specific murine monoclonal Ig. G (15404, 20 M), HPIX in PBS and visible light for 1 hour at 4°C (5% viable). To visualize the sites of antibodyattachment gold-labeled goat anti-mouse antibodies were added after completion of the bactericidal assay (6). The arrow points to a puncture in the cell and plasma membrane. SCIENCE VOL 298, pp 2195 -2199
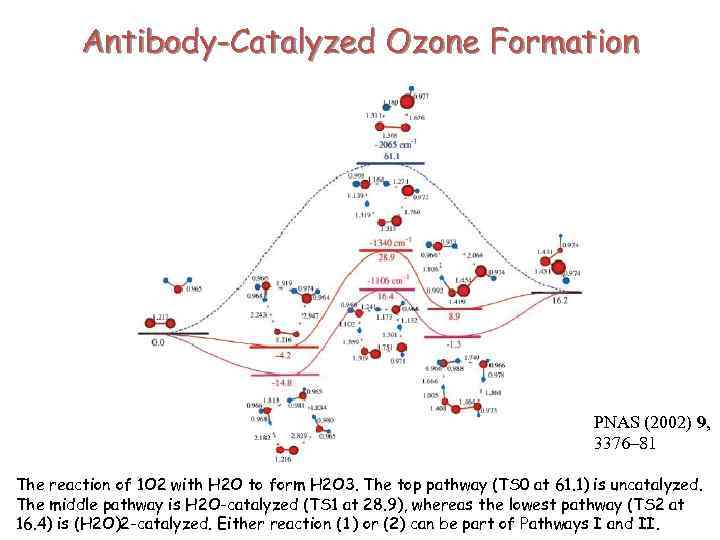
Antibody-Catalyzed Ozone Formation PNAS (2002) 9, 3376– 81 The reaction of 1 O 2 with H 2 O to form H 2 O 3. The top pathway (TS 0 at 61. 1) is uncatalyzed. The middle pathway is H 2 O-catalyzed (TS 1 at 28. 9), whereas the lowest pathway (TS 2 at 16. 4) is (H 2 O)2 -catalyzed. Either reaction (1) or (2) can be part of Pathways I and II.
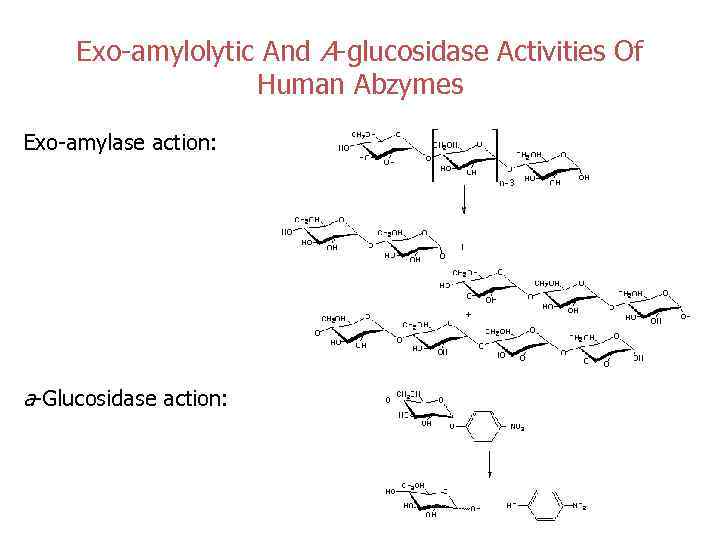
Exo-amylolytic And A-glucosidase Activities Of Human Abzymes Exo-amylase action: a-Glucosidase action:

Catalysis by Anti-Idiotypic Antibodies Ö properties of antigens seems very promising to us and opens up great possibilities for development of different directions in molecular biology: from the investigations of mechanisms of regulation of genetic processes to that new field of enzymology known as “catalytic antibodies” or “abzymes” Bronstein et all. Molecular Biology (Russian) 1989, v. 223, (6) pp. 1553 -1557
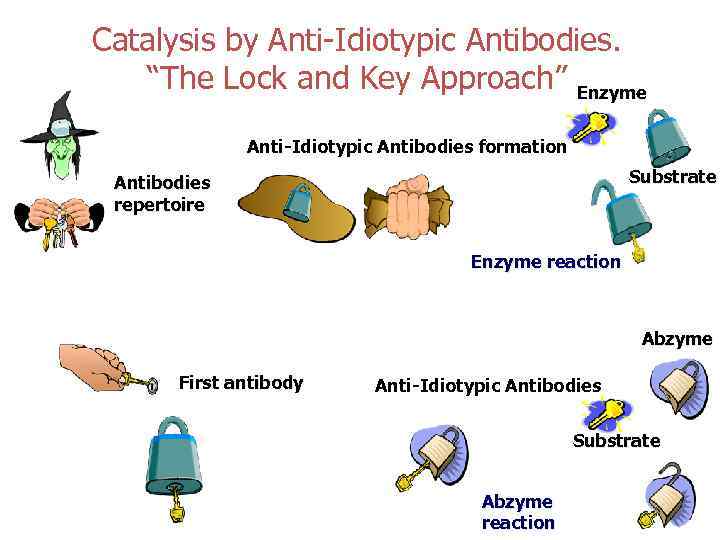
Catalysis by Anti-Idiotypic Antibodies. “The Lock and Key Approach” Enzyme Anti-Idiotypic Antibodies formation Substrate Antibodies repertoire Enzyme reaction Abzyme First antibody Anti-Idiotypic Antibodies Substrate Abzyme reaction
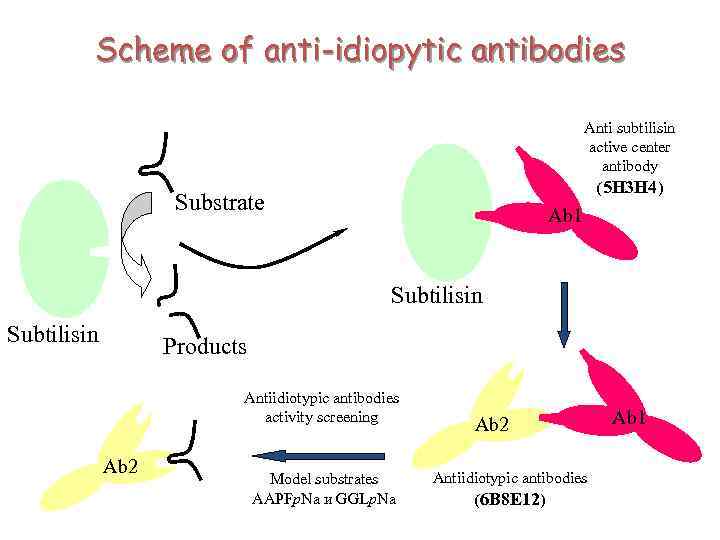
Scheme of anti-idiopytic antibodies Anti subtilisin active center antibody (5 H 3 H 4) Substrate Ab 1 Subtilisin Products Antiidiotypic antibodies activity screening Ab 2 Model substrates AAPFp. Na и GGLp. Na Ab 2 Antiidiotypic antibodies (6 B 8 E 12) Ab 1
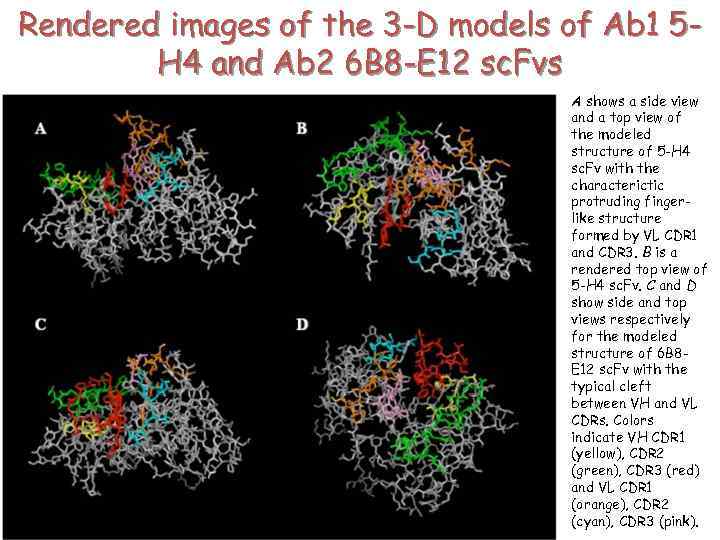
Rendered images of the 3 -D models of Ab 1 5 H 4 and Ab 2 6 B 8 -E 12 sc. Fvs A shows a side view and a top view of the modeled structure of 5 -H 4 sc. Fv with the characterictic protruding fingerlike structure formed by VL CDR 1 and CDR 3. B is a rendered top view of 5 -H 4 sc. Fv. C and D show side and top views respectively for the modeled structure of 6 B 8 E 12 sc. Fv with the typical cleft between VH and VL CDRs. Colors indicate VH CDR 1 (yellow), CDR 2 (green), CDR 3 (red) and VL CDR 1 (orange), CDR 2 (cyan), CDR 3 (pink).
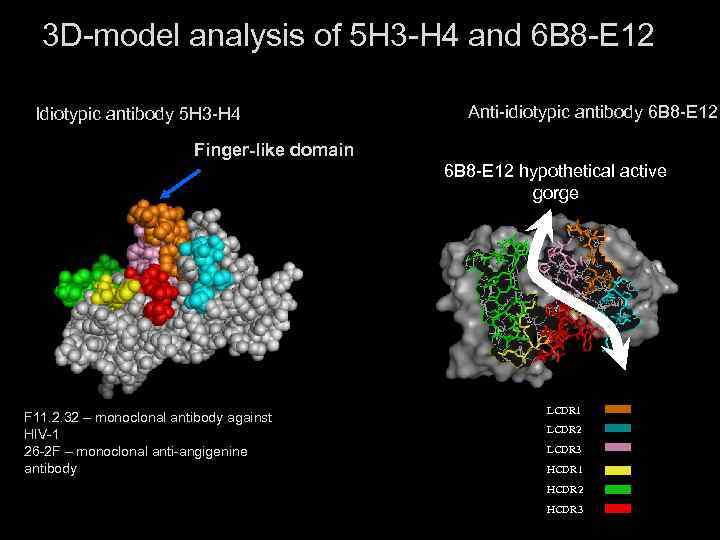
3 D-model analysis of 5 H 3 -H 4 and 6 B 8 -E 12 Idiotypic antibody 5 H 3 -H 4 Anti-idiotypic antibody 6 B 8 -E 12 Finger-like domain 6 B 8 -E 12 hypothetical active gorge F 11. 2. 32 – monoclonal antibody against HIV-1 26 -2 F – monoclonal anti-angigenine antibody LCDR 1 LCDR 2 LCDR 3 HCDR 1 HCDR 2 HCDR 3
Hyper Variability Of Antibody Molecule Provides The Best Template To Generate Artificial Binder (Scavenger)/Biocatalysts
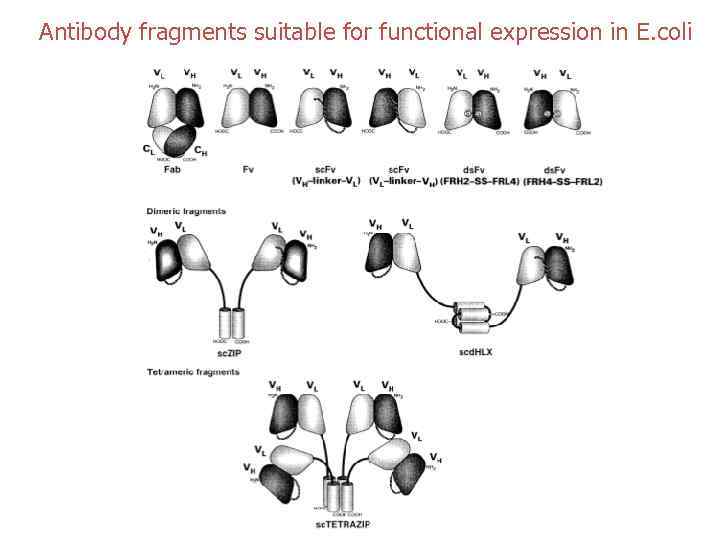
Antibody fragments suitable for functional expression in E. coli
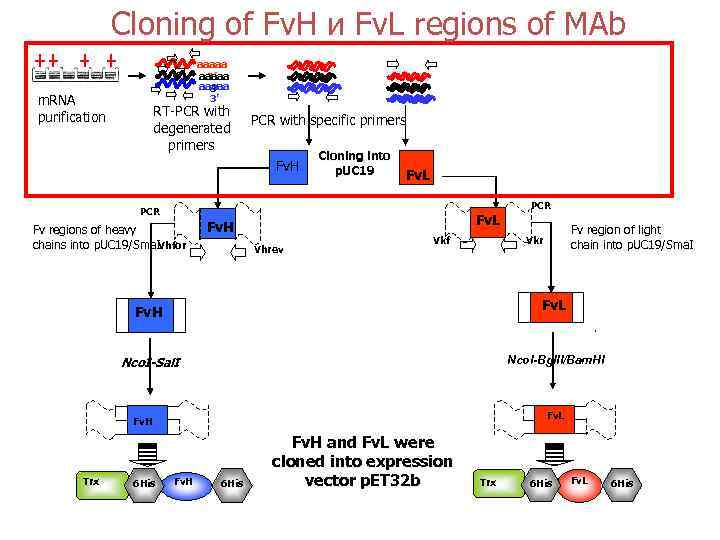
Cloning of Fv. Н и Fv. L regions of MAb aaaaa 3’ 3’ m. RNA purification RT-PCR with degenerated primers PCR with specific primers Fv. H Cloning into p. UC 19 Fv. L PCR Fv regions of heavy chains into p. UC 19/Sma. IVhfor Fv. L Fv. H Vhrev Vkf Fv. L Nco. I-Bgl. II/Bam. HI Nco. I-Sal. I Fv. L Fv. H 6 His Fv region of light chain into p. UC 19/Sma. I Vkr Fv. H Trx PCR Fv. H 6 His Fv. H and Fv. L were cloned into expression vector p. ET 32 b Trx 6 His Fv. L 6 His
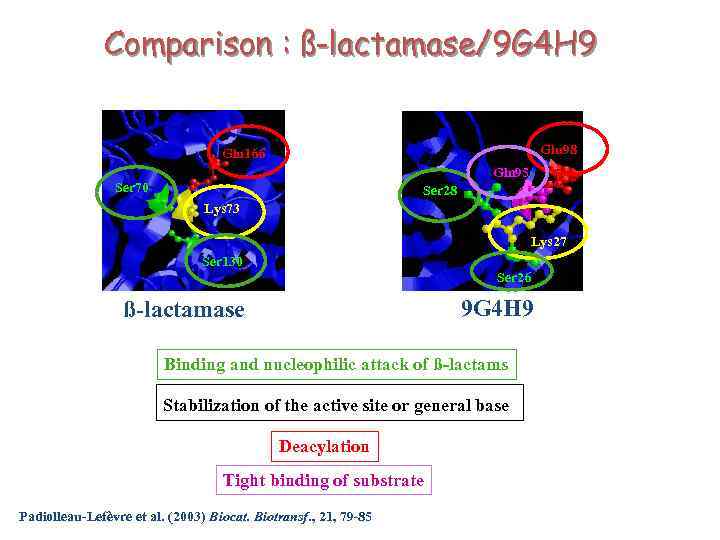
Comparison : ß-lactamase/9 G 4 H 9 Glu 98 Glu 166 Gln 95 Ser 70 Ser 28 Lys 73 Lys 27 Ser 130 Ser 26 9 G 4 H 9 ß-lactamase Binding and nucleophilic attack of ß-lactams Stabilization of the active site or general base Deacylation Tight binding of substrate Padiolleau-Lefèvre et al. (2003) Biocat. Biotransf. , 21, 79 -85
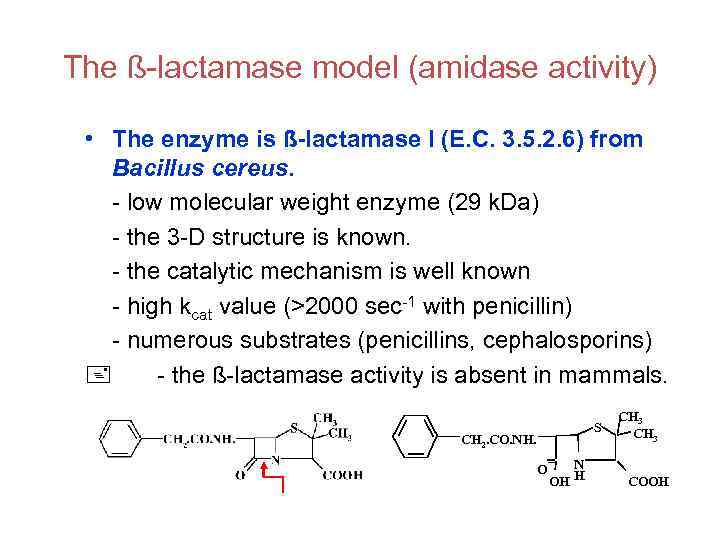
The ß-lactamase model (amidase activity) • The enzyme is ß-lactamase I (E. C. 3. 5. 2. 6) from Bacillus cereus. - low molecular weight enzyme (29 k. Da) - the 3 -D structure is known. - the catalytic mechanism is well known - high kcat value (>2000 sec-1 with penicillin) - numerous substrates (penicillins, cephalosporins) - the ß-lactamase activity is absent in mammals. S CH 2. CO. NH. O N H OH CH 3 COOH
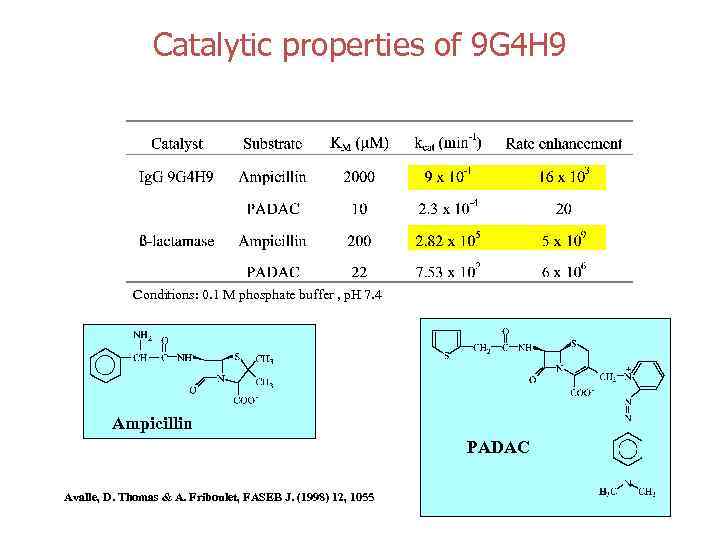
Catalytic properties of 9 G 4 H 9 Conditions: 0. 1 M phosphate buffer , p. H 7. 4 Ampicillin PADAC Avalle, D. Thomas & A. Friboulet, FASEB J. (1998) 12, 1055
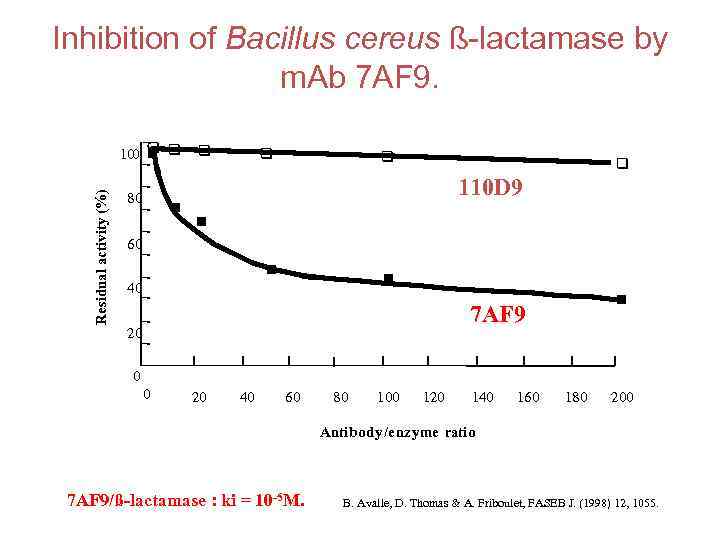
Inhibition of Bacillus cereus ß-lactamase by m. Ab 7 AF 9. Residual activity (%) n 100 q q n 80 n q q 110 D 9 n 60 n n 40 n 7 AF 9 20 0 0 20 40 60 80 100 120 140 160 180 200 Antibody/enzyme ratio 7 AF 9/ß-lactamase : ki = 10 -5 M. B. Avalle, D. Thomas & A. Friboulet, FASEB J. (1998) 12, 1055.

Organophosphorus Poisons
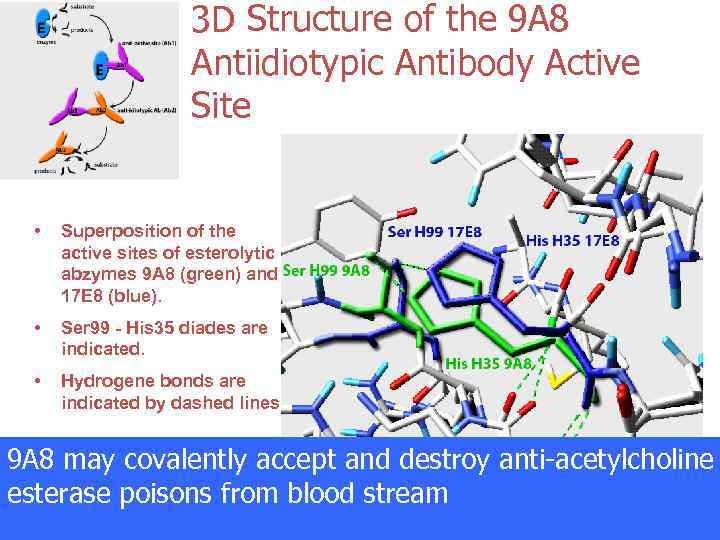
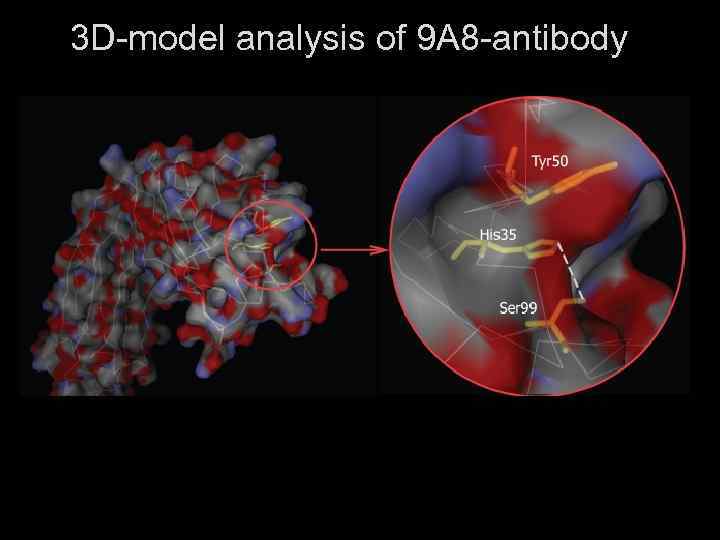
3 D Structure of the 9 A 8 Antiidiotypic Antibody Active Site • Superposition of the active sites of esterolytic abzymes 9 A 8 (green) and 17 E 8 (blue). • Ser 99 - His 35 diades are indicated. • Hydrogene bonds are indicated by dashed lines. 9 A 8 may covalently accept and destroy anti-acetylcholine esterase poisons from blood stream Kolesnikov et al, PNAS 2000
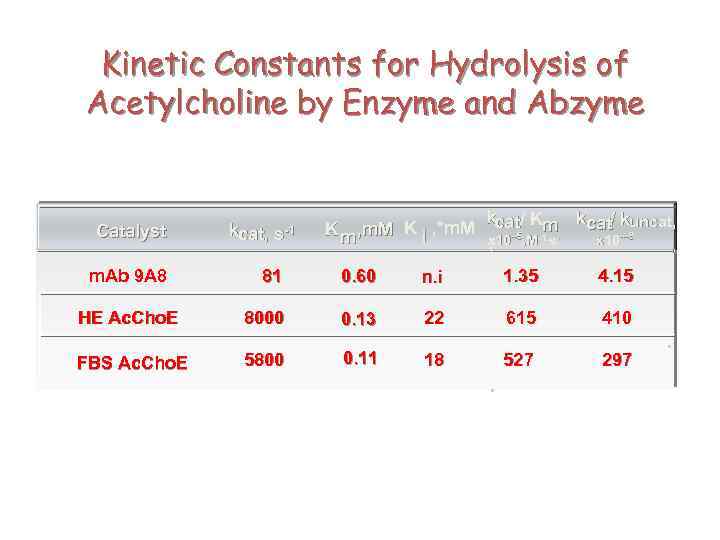
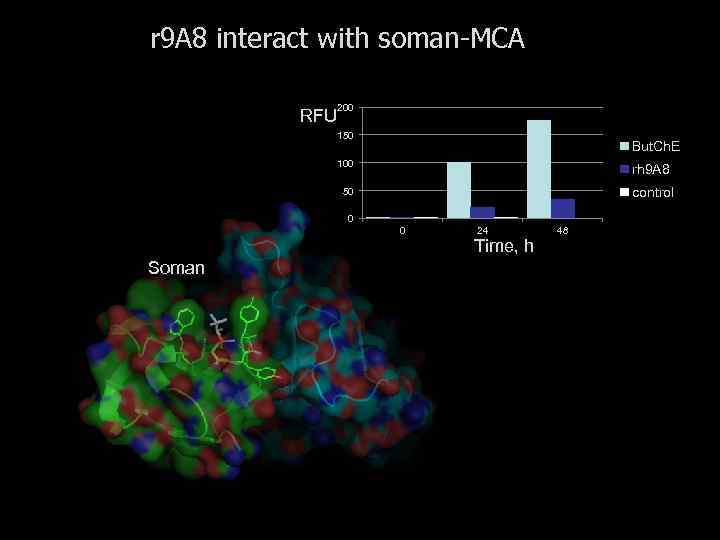
Kinetic Constants for Hydrolysis of Acetylcholine by Enzyme and Abzyme Catalyst kcat, s-1 K m , m. M K i , *m. M kcat/ Km kcat/ kuncat, x 10 --5, M-1. s- x 10 ---8 1 81 0. 60 n. i 1. 35 4. 15 HE Ac. Cho. E 8000 0. 13 22 615 410 FBS Ac. Cho. E 5800 0. 11 18 527 297 m. Ab 9 A 8

Acetylcholine Esterase-like Antiidiotypic 9 A 8 Antibody 9 A 8 may covalently accept and delete acetylcholine esterase poisons from blood stream • Fv fragment of 9 А 8 antibody (blue) • Predicted catalytic diade in green
3 D-model analysis of 9 A 8 -antibody
r 9 A 8 interact with soman-MCA RFU 200 150 But. Ch. E 100 rh 9 A 8 50 control 0 0 Soman 24 Time, h 48
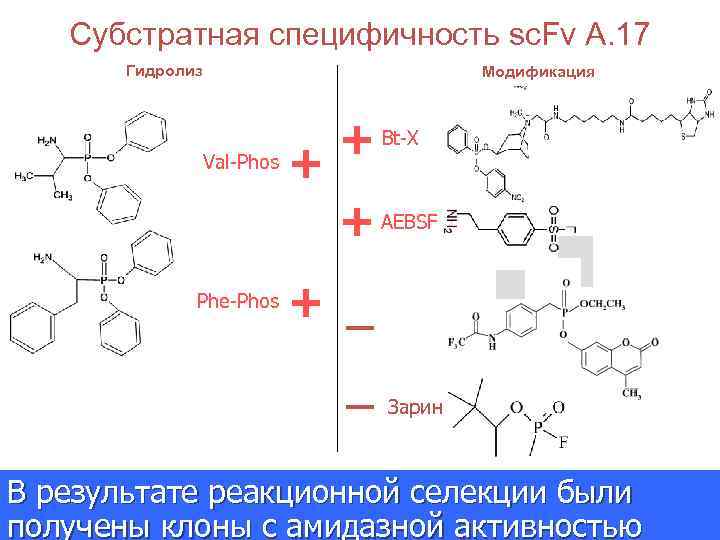
Субстратная специфичность sc. Fv A. 17 Гидролиз Модификация Val-Phos + + Bt-X + AEBSF Phe-Phos + _ _ Зарин В результате реакционной селекции были Journal of the American Chemical Society получены клоны с амидазной активностью 2007
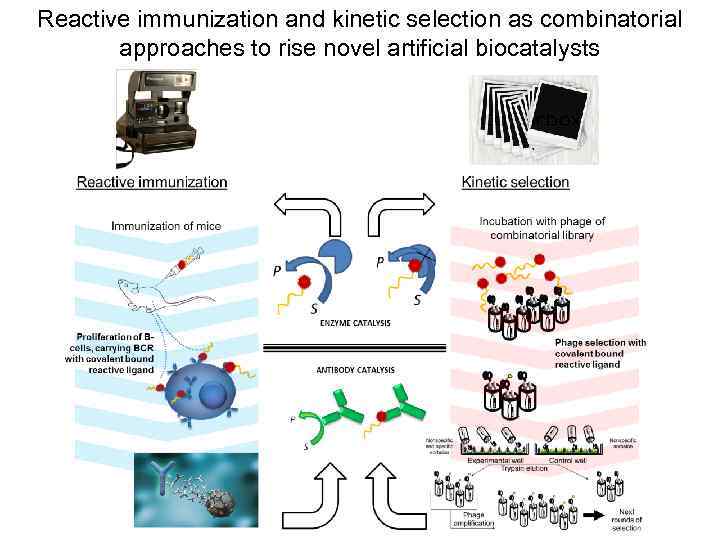
Reactive immunization and kinetic selection as combinatorial approaches to rise novel artificial biocatalysts
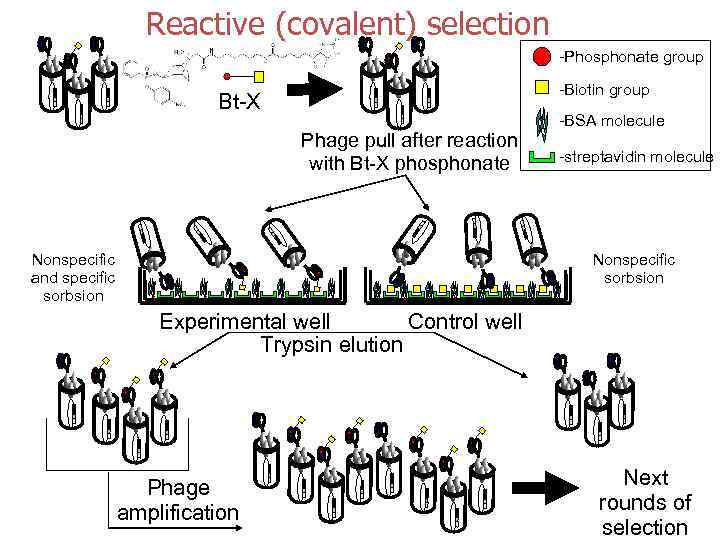
Reactive (covalent) selection -Phosphonate group -Biotin group Bt-X Phage pull after reaction with Bt-X phosphonate Nonspecific and specific sorbsion -BSA molecule -streptavidin molecule Nonspecific sorbsion Experimental well Control well Trypsin elution Phage amplification Next rounds of selection
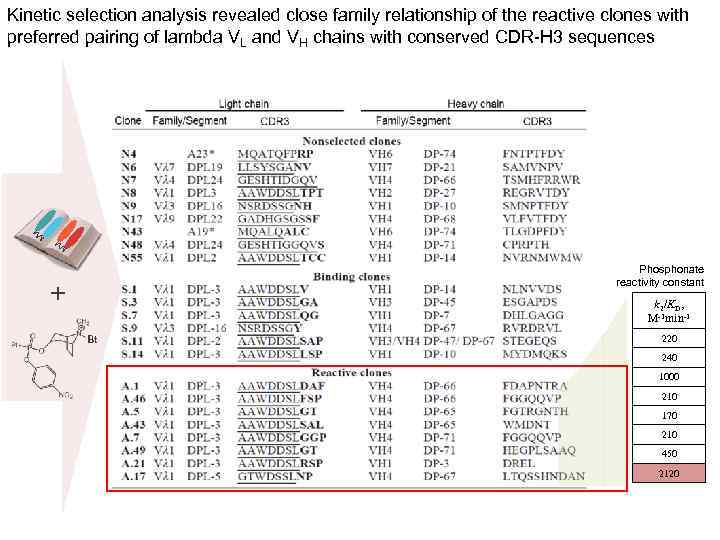
Kinetic selection analysis revealed close family relationship of the reactive clones with preferred pairing of lambda VL and VH chains with conserved CDR-H 3 sequences Phosphonate reactivity constant + k 2/KD, М-1 min-1 Bt 220 240 1000 210 170 210 450 2120

MATURATION IN SILICO
Reactive immunization and kinetic selection as combinatorial approaches to rise novel artificial biocatalysts
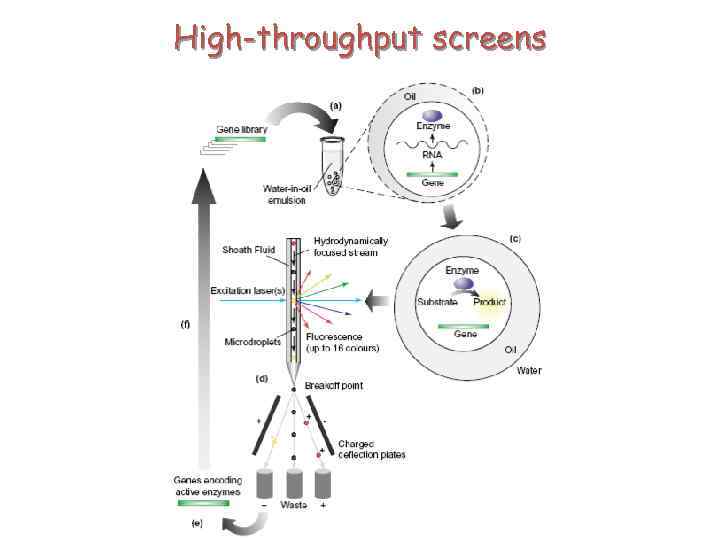
High-throughput screens

MESSAGE To make de novo functional binder/biocatalyst using Ig template we have to: 1)enlarge the repertoire for combinatorial screening, 2)propose the “vector” for selection strategy For these purposes we may use: 1)phage-display libraries (“immunization” and screening in vitro), 2)autoimmune repertoires (in vivo)
MESSAGE: Combination of instrumental and computational methods may be regarded as an efficient strategy to obtain artificial biocatalysts de novo Modification Hybrid QM/MM and MD approach Covalent intermediate Turnover
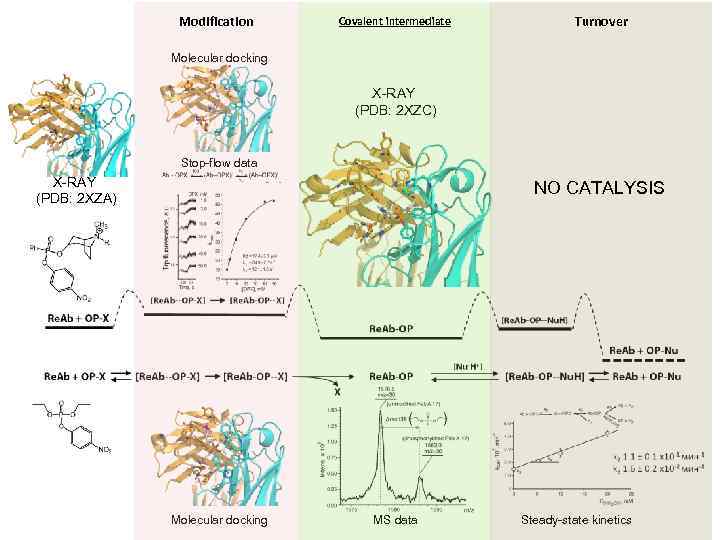
Modification Covalent intermediate Turnover Molecular docking X-RAY (PDB: 2 XZC) Stop-flow data X-RAY (PDB: 2 XZA) NO CATALYSIS Molecular docking MS data Steady-state kinetics
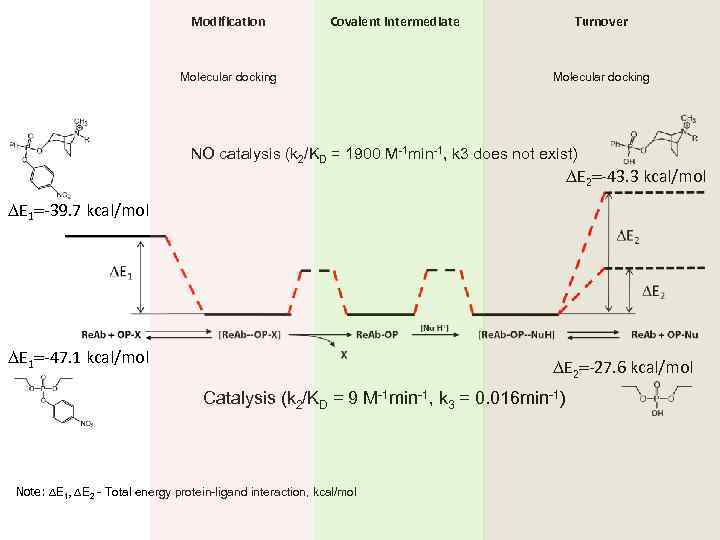
Modification Covalent intermediate Molecular docking Turnover Molecular docking NO catalysis (k 2/KD = 1900 M-1 min-1, k 3 does not exist) DE 2=-43. 3 kcal/mol DE 1=-39. 7 kcal/mol DE 1=-47. 1 kcal/mol DE 2=-27. 6 kcal/mol Catalysis (k 2/KD = 9 M-1 min-1, k 3 = 0. 016 min-1) Note: DE 1, DE 2 - Total energy protein-ligand interaction, kcal/mol
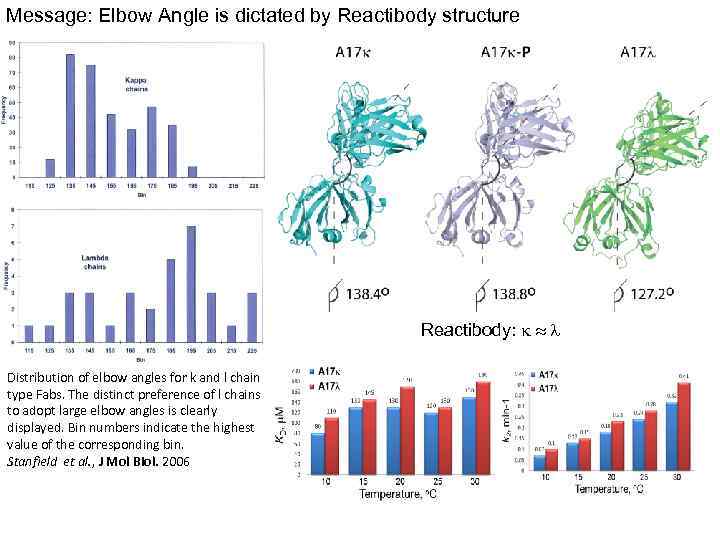
Message: Elbow Angle is dictated by Reactibody structure Reactibody: k l Distribution of elbow angles for k and l chain type Fabs. The distinct preference of l chains to adopt large elbow angles is clearly displayed. Bin numbers indicate the highest value of the corresponding bin. Stanfield et al. , J Mol Biol. 2006
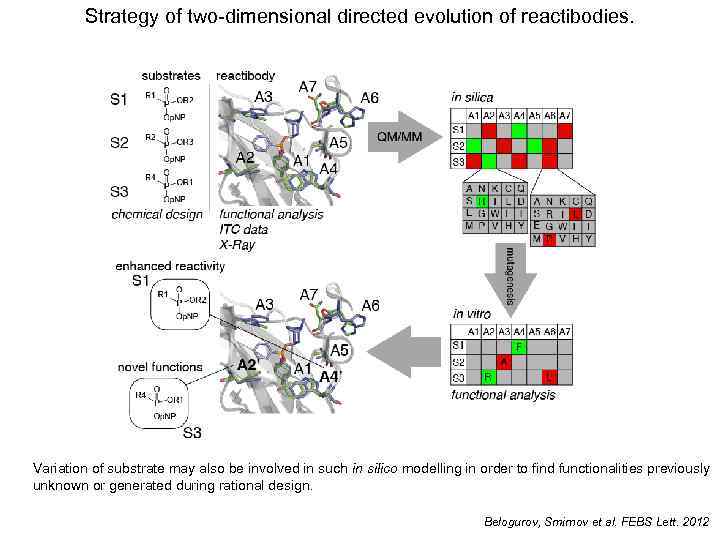
Strategy of two-dimensional directed evolution of reactibodies. Variation of substrate may also be involved in such in silico modelling in order to find functionalities previously unknown or generated during rational design. Belogurov, Smirnov et al. FEBS Lett. 2012
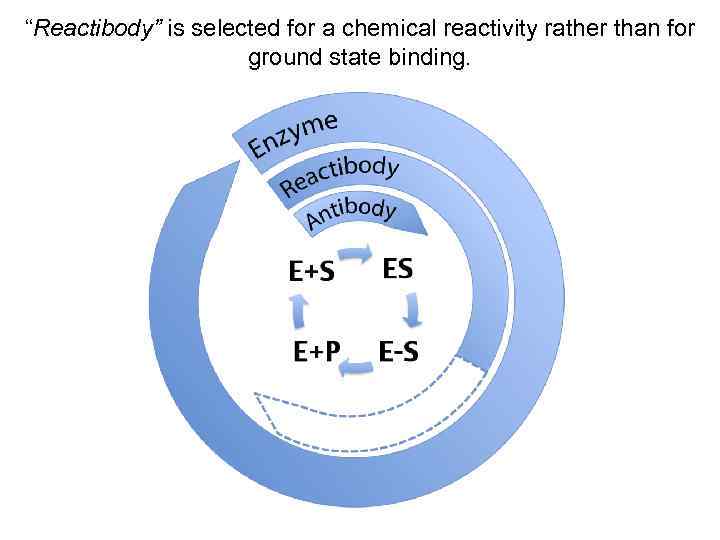
“Reactibody” is selected for a chemical reactivity rather than for ground state binding.
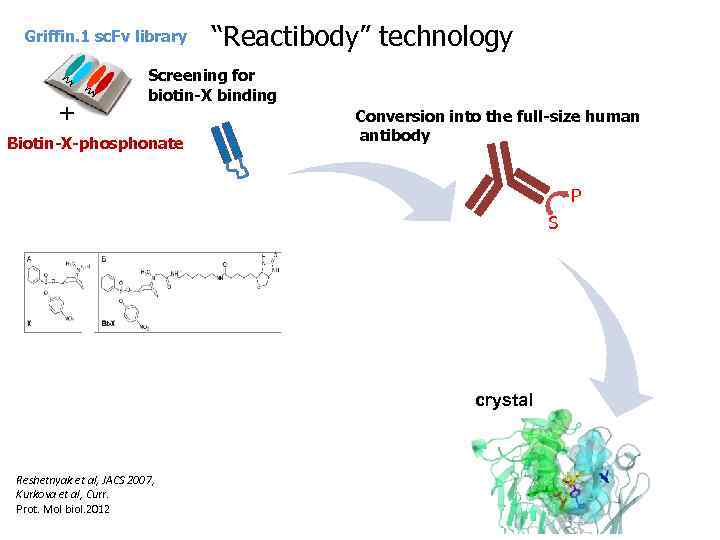
Griffin. 1 sc. Fv library + “Reactibody” technology Screening for biotin-X binding Biotin-X-phosphonate Conversion into the full-size human antibody P S crystal Reshetnyak et al, JACS 2007, Kurkova et al, Curr. Prot. Mol biol. 2012
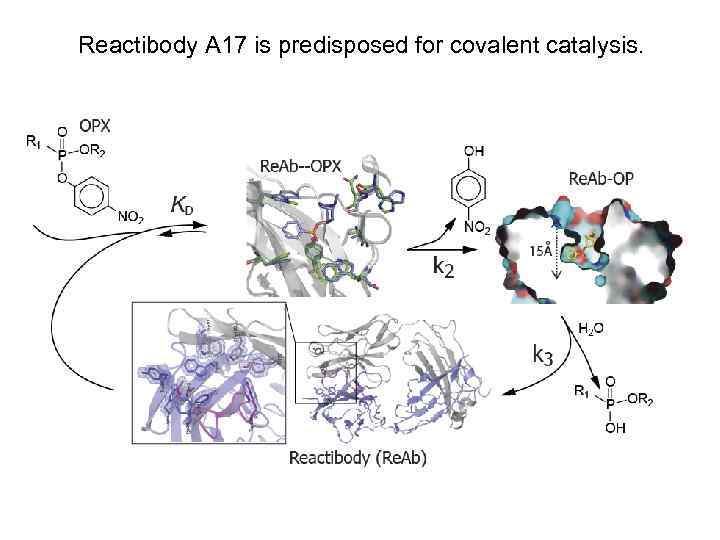
Reactibody A 17 is predisposed for covalent catalysis.
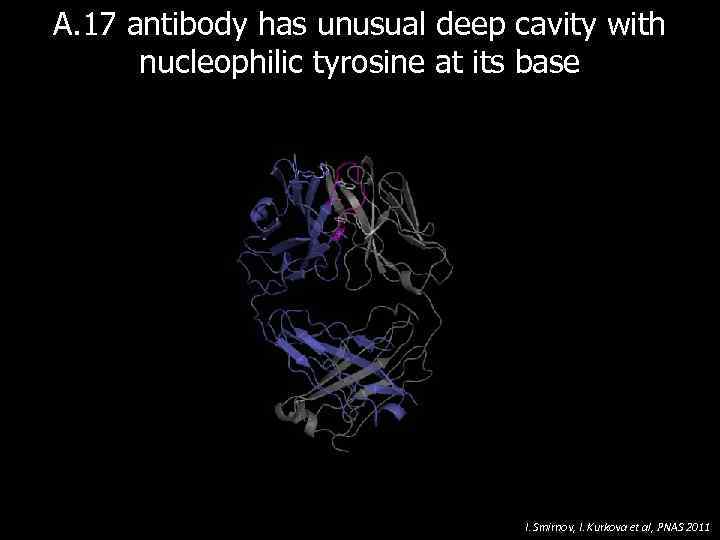
A. 17 antibody has unusual deep cavity with nucleophilic tyrosine at its base I. Smirnov, I. Kurkova et al, PNAS 2011
Crystallization procedures The mother liquor was 0. 1 M ADA buffer p. H 6. 5, 0. 1 M Mg. Cl 2 with 20% (w/V) PEG 6000. The OP-Fab. A 17 conjugate was prepared by soaking crystals of native Fab A 17 for two minutes in 0. 1 M ADA buffer, p. H 6. 5, 0. 1 M Mg. Cl 2 and 20% w/v PEG 6000 containing 1 m. M phosphonyl ester compound. Fab A 17 X-ray Data Collection and Structure Solution Fab A 17 -OP Diffraction data were collected at the European Synchrotron Radiation Facility (ESRF, Grenoble), on ID 14 -eh 4 beam line, using λ= 0. 979 Å wavelength with ADSC Quantum 4 detector.
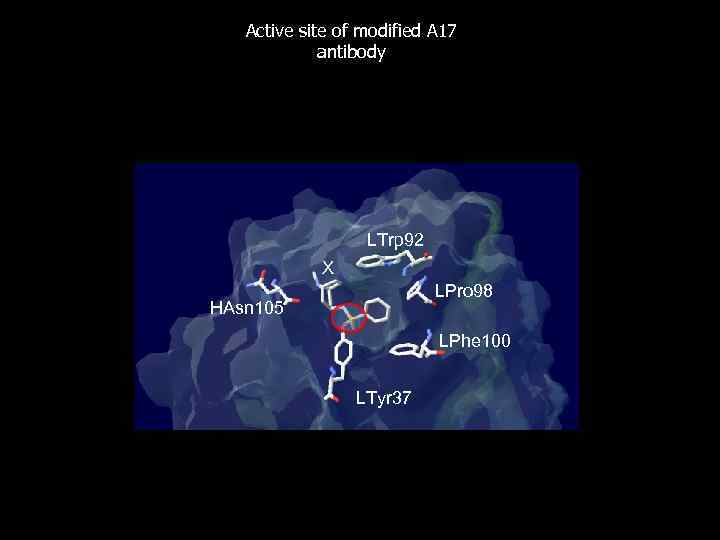
Active site of modified A 17 antibody LTrp 92 X LPro 98 HAsn 105 LPhe 100 LTyr 37
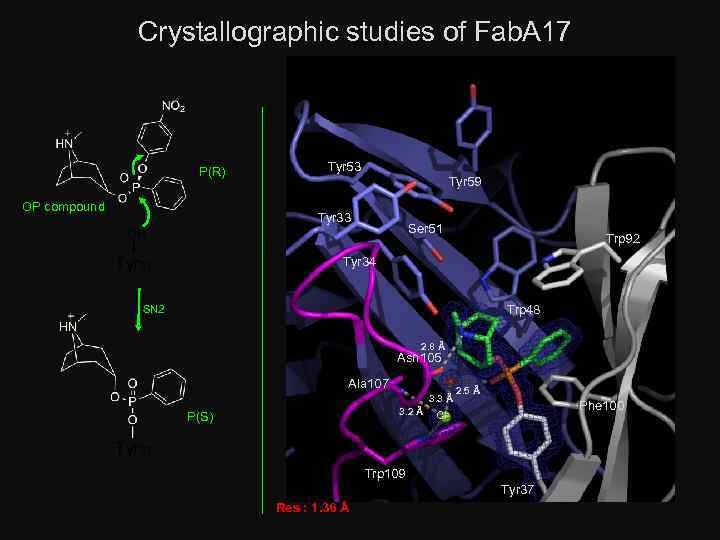
Crystallographic studies of Fab. A 17 P(R) OP compound Tyr 53 Tyr 59 Tyr 33 Ser 51 OH Tyr 37 Trp 92 Tyr 34 Trp 48 SN 2 2. 8 Å Asn 105 Ala 107 3. 3 Å 3. 2 Å P(S) 2. 5 Å Phe 100 Cl- Tyr 37 Trp 109 Tyr 37 Res : 1. 36 Å
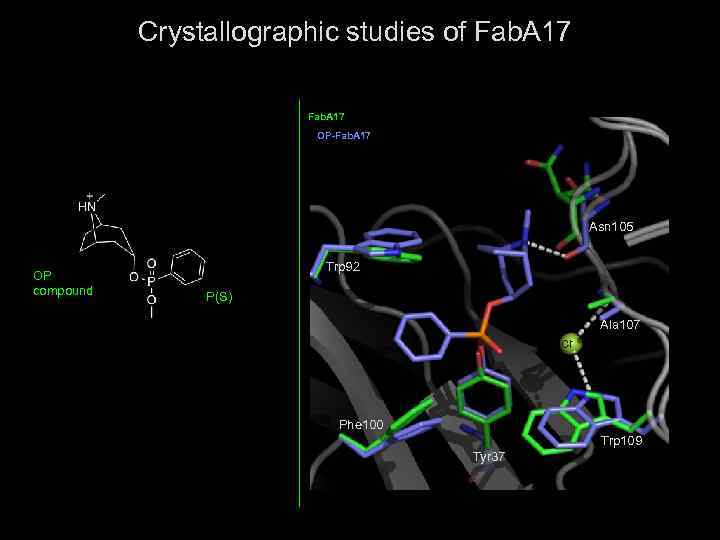
Crystallographic studies of Fab. A 17 OP-Fab. A 17 Asn 105 Trp 92 OP compound P(S) Ala 107 Tyr 37 Cl- Phe 100 Trp 109 Tyr 37
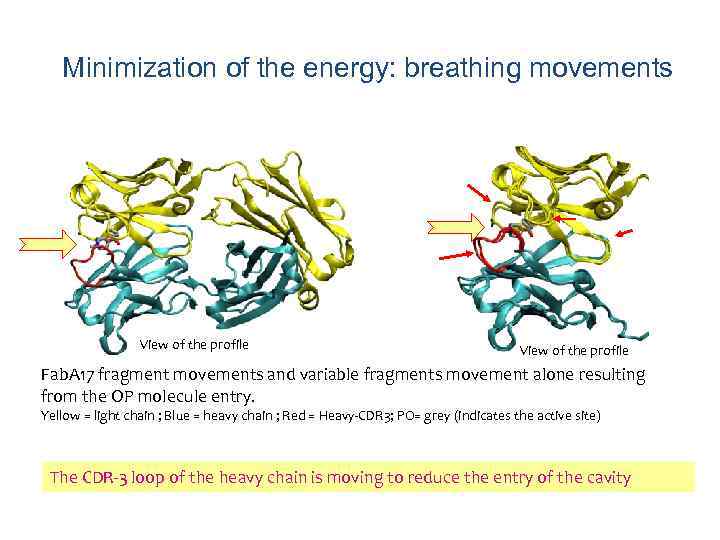
Minimization of the energy: breathing movements View of the profile Fab. A 17 fragment movements and variable fragments movement alone resulting from the OP molecule entry. Yellow = light chain ; Blue = heavy chain ; Red = Heavy-CDR 3; PO= grey (indicates the active site) The CDR-3 loop of the heavy chain is moving to reduce the entry of the cavity
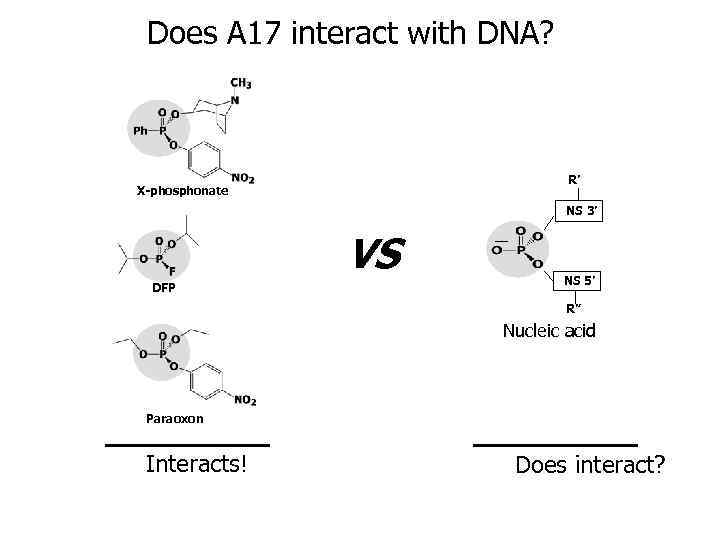
Does A 17 interact with DNA? R’ X-phosphonate NS 3’ VS DFP NS 5’ R” Nucleic acid Paraoxon Interacts! Does interact?
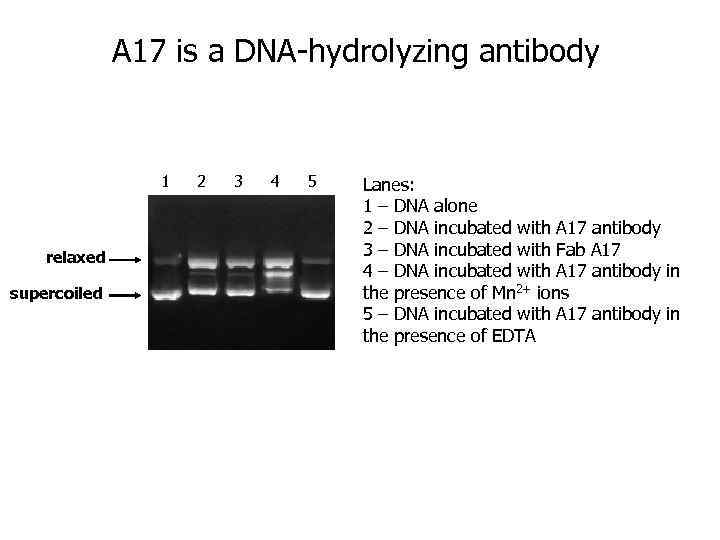
A 17 is a DNA-hydrolyzing antibody 1 relaxed supercoiled 2 3 4 5 Lanes: 1 – DNA alone 2 – DNA incubated with A 17 antibody 3 – DNA incubated with Fab A 17 4 – DNA incubated with A 17 antibody in the presence of Mn 2+ ions 5 – DNA incubated with A 17 antibody in the presence of EDTA
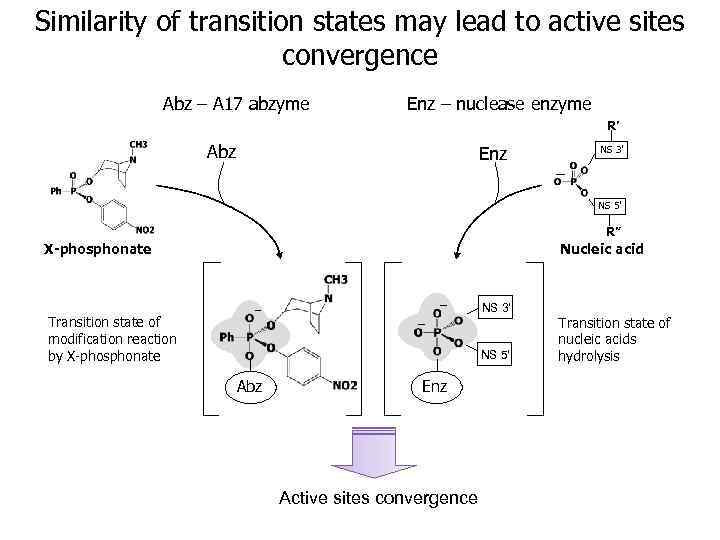
Similarity of transition states may lead to active sites convergence Abz – A 17 abzyme Enz – nuclease enzyme R’ Abz Enz NS 3’ NS 5’ R” X-phosphonate Nucleic acid NS 3’ Transition state of modification reaction by X-phosphonate NS 5’ Abz Enz Active sites convergence Transition state of nucleic acids hydrolysis

Autoimmune disorders as a source of catalytic antibodies “normal” repertoire is powered in autoimmunity
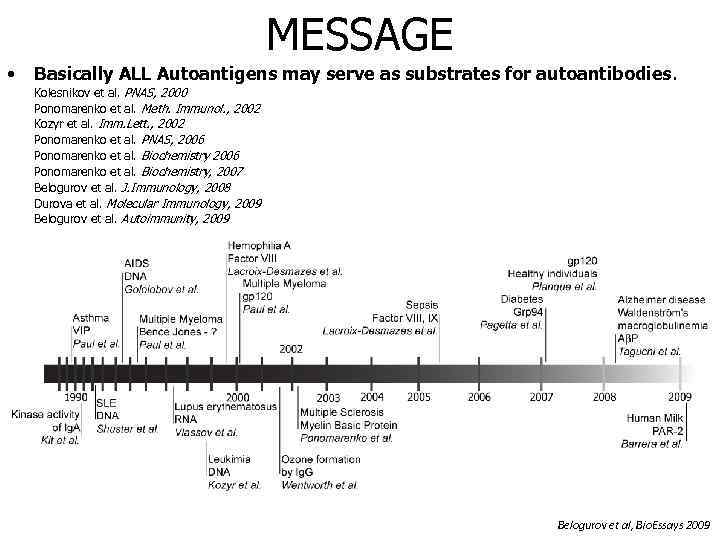
MESSAGE • Basically ALL Autoantigens may serve as substrates for autoantibodies. Kolesnikov et al. PNAS, 2000 Ponomarenko et al. Meth. Immunol. , 2002 Kozyr et al. Imm. Lett. , 2002 Ponomarenko et al. PNAS, 2006 Ponomarenko et al. Biochemistry, 2007 Belogurov et al. J. Immunology, 2008 Durova et al. Molecular Immunology, 2009 Belogurov et al. Autoimmunity, 2009 Belogurov et al, Bio. Essays 2009

Hypotheses ON THE INCREASED ABZYME OCCURRENCE IN AUTOIMMUNITY Expanded repertoire of underevolved antibodies may favor the emergence of abzyme activity by following possible routes: 1. Spontaneous evolution of the existing enzyme plethora. 2. Expanded repertoire can result in the increased production of anti-idiotypes. Biological mechanisms of this phenomenon are still unknown.

Autoimmunity and Antibody Mediated Tissue Destructions Catalytic activity of Bence Jones Proteins 1845 W. Mac. Intire recognized the first case of Multiple myeloma Henry Bence Jones discussed the peculiarities of urinary proteins Gulstonian Lectures on chemical pathology to the Royal College of Physicians in London 1962 Edelman and Gally found that light chain of plasma immunoglobulin from patients with myeloma is identical with Bence Jones urine protein from the same patient. 1974 Erhan and Greller pointed out the statistically significant amino acid identity between immunoglobulin light chain and active cite of serine proteinase 1994 Matsuura et al, 1995 Paul et al. demonstrated catalytic activity of Bence Jones protein Catalytic Activity of Bence Jones Proteins is a possible Cause of Renal Failure

Role of B cells in the pathogenesis of rheumatoid arthritis The rheumatoid factors form large antigen/antibody complexes which may enhance the process of joint destruction. The poor prognosis of rheumatoid factor-positive patients is indicative of the critical role of immunoglobulin complexes in the continuous stimulation of the immune system and thus of the inflammatory processes. Gause A, Berek C. Bio. Drugs 2001; 15(2): 73 -9 Sjogren's syndrome. i) defective apoptosis could lead to lymphoid cell accumulation and chronic inflammation in exocrine glands; ii) increased apoptosis of epithelial cells might explain the loss of secreting epithelium; and iii) orderly destruction of cellular components might induce autoantibody production. Bolstad AI, Jonsson R. Antineutrophil cytoplasmic antibodies induce human monocytes to produce oxygen radicals in vitro. Antineutrophil cytoplasmic antibodies (ANCA) are believed to play a pathogenetic role in necrotizing small-vessel vasculitis. Normal nonspecific Ig. G or their corresponding F(ab')2 fragments induced no release or very little release of oxygen radicals. Preincubation of monocytes with the Fcy receptor type II-blocking monoclonal antibody IV. 3 before addition of ANCA greatly reduced formation of oxygen radicals Weidner S, et al. Arthritis Rheum 2001 Jul; 44(7): 1698 -706 Immunopathogenic and clinical relevance of antibodies against myelin oligodendrocyte glycoprotein (MOG) in Multiple Sclerosis Immunopathogenic role of antibodies against myelin oligodendrocyte glycoprotein (MOG) and other myelin/nonmyelin targets in multiple sclerosis, as well as possible clinical implications for prognosis and therapy in the future. Berger T, Reindl M. J Neural Transm Suppl 2000; (60): 351 -60
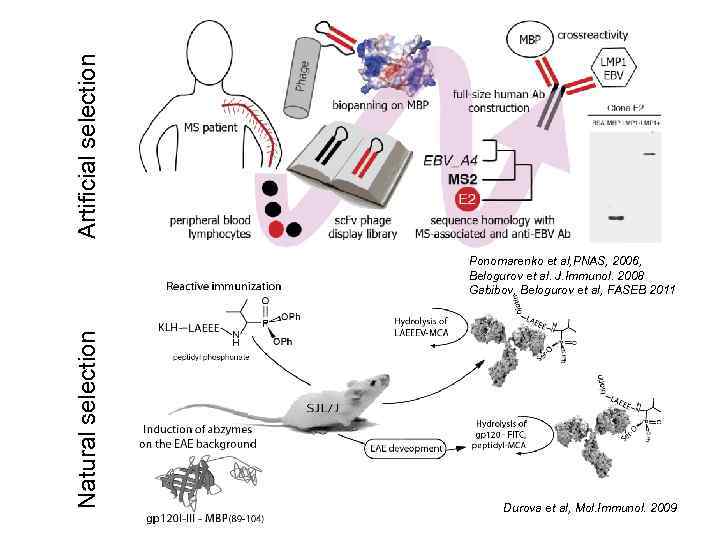
Artificial selection Natural selection Ponomarenko et al, PNAS, 2006, Belogurov et al. J. Immunol. 2008 Gabibov, Belogurov et al, FASEB 2011 Durova et al, Mol. Immunol. 2009

Why therapy with catalytic antibody is good? • Long life of antibodies in the blood stream • High specificity • The same amount of target may be inactivated by smaller amount of catalytic antibody + Good world experience in therapy with antibody + Good world experience in antibody engineering
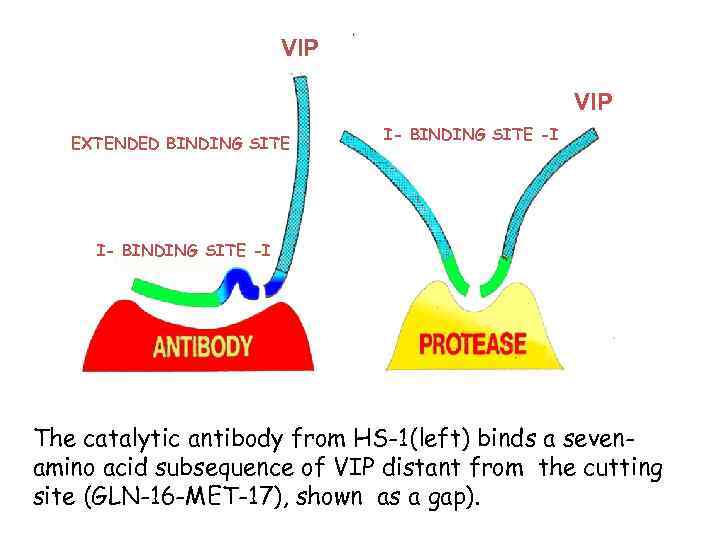
VIP EXTENDED BINDING SITE I- BINDING SITE -I The catalytic antibody from HS-1(left) binds a sevenamino acid subsequence of VIP distant from the cutting site (GLN-16 -MET-17), shown as a gap).

Substates For Proteolytic Abzymes Sca Variant Km kcat k cat /Km vasoactive intestinal peptide (VIP) 0. 2 x 10 -6 M 1. 1 x 10 -2 min -1 5. 5 x 10 4 min -1 M-1 thyroglobulin 3. 9 x 10 -8 M 3. 9 x 10 -3 min -1 1. 0 x 10 5 min -1 M-1 Pro. Phe. Arg. MCA 11. 5 x 10 -6 M 6. 8 x 10 -3 min -1 5. 9 x 10 2 min -1 M-1 ? ? H IV gp 120 ?

Catalitic Activity of Antibodies Against Factor VIII in Patients With Hemohilia A

Hydrolysis of Factor VIII by Ig. G Purified from Plasma of Patients with Severe Hemophilia A Inhibitory activity against factor VIII of IGG from 24 patients with severe hemophilia A and rates of hydrolysis of factor VIII Biotinylated factor VIII (385 n. M) was incubated alone (Control) or in the presence of Ig. G antibody (167 n. M) for 24 hours at 37°C. Immune globulin (IVIg) was used as a source of normal Ig. G and as a negative control. Samples were separated by 10 percent sodium dodecyl sulfate–polyacrylamide-gel electrophoresis. BU denotes Bethesda units, and ND not determined. †Hydrolysis of factor VIII was quantified by scanning of immunoblots. Spontaneous hydrolysis that occurred on incubation of factor VIII in the presence of buffer alone was considered to represent the background level and was subtracted from each analysis. Plus-minus values are means ±SD of two or three experiments. The mean coefficient of variation was 0. 33 (range, 0. 01 to 1. 41). P<0. 05 for the comparison with immune globulin
Autoantigens DNA

Key Questions about pathological functions of DNA- Abzymes • Correlation with the disease development • Involvement in the nuclear processes • How DNA-Abzymes recognize apoptotic cells and involved in the clearance process

Properties of DNA-Abzymes • Crossreactivity with histones, nuclear matrix proteins. • High specificity to the nuclear matrix attachment regions.
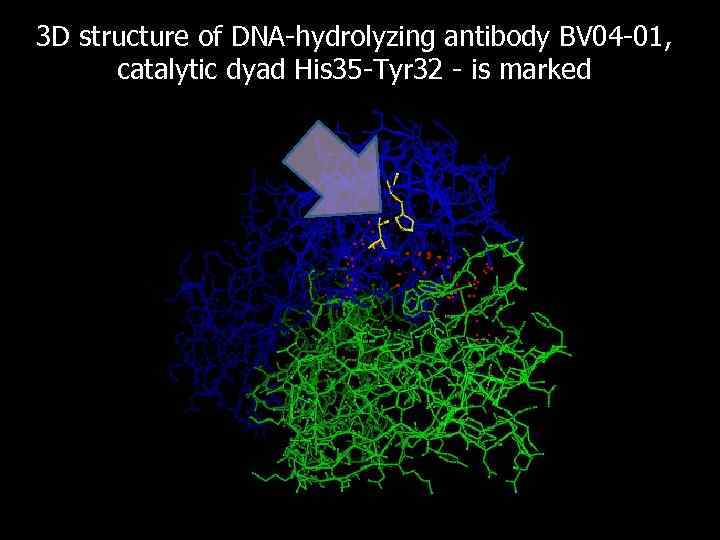
3 D structure of DNA-hydrolyzing antibody BV 04 -01, catalytic dyad His 35 -Tyr 32 - is marked
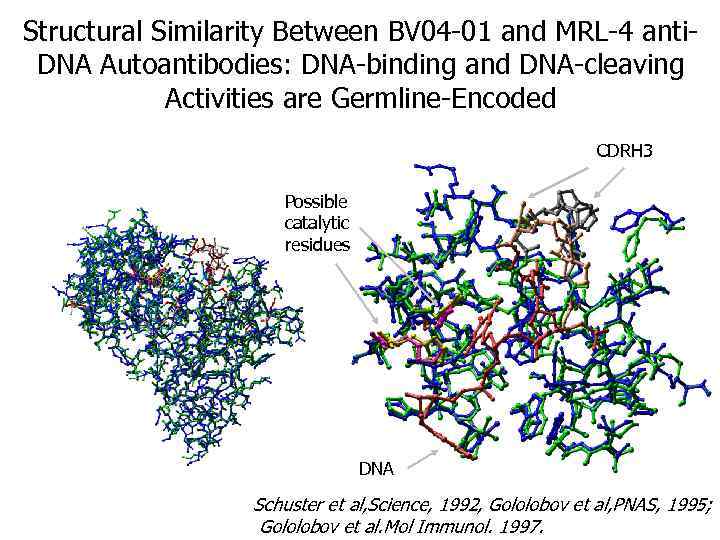
Structural Similarity Between BV 04 -01 and MRL-4 anti. DNA Autoantibodies: DNA-binding and DNA-cleaving Activities are Germline-Encoded CDRH 3 Possible catalytic residues DNA Schuster et al, Science, 1992, Gololobov et al, PNAS, 1995; Gololobov et al. Mol Immunol. 1997.

Catalytic activity of mutants of DNA-abzyme Sca Variant Km , M kcat, min-1 Km/kcat L 32 Tyr/L 27 d. His (WT) 2. 5 x 10 -8 1. 3 x 10 -2 5. 2 x 105 L 32 Phe/L 27 d. His 6. 0 x 10 -9 1. 1 x 10 -3 1. 8 x 105 L 32 Ala/L 27 d. His 2. 4 x 10 -7 1. 0 x 10 -1 4. 2 x 105 L 32 Tyr/L 27 d. Ala 4. 1 x 10 -8 4. 9 x 10 -4 1. 2 x 104 Similar Improvement Worsening Site-directed mutagenesis shows that DNA-abzyme participates in the chemical steps of catalysis.
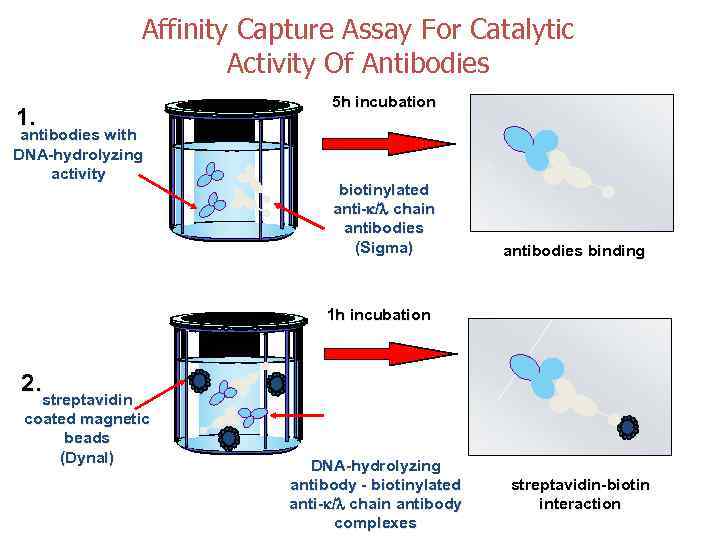
Affinity Capture Assay For Catalytic Activity Of Antibodies 1. antibodies with DNA-hydrolyzing activity 5 h incubation biotinylated anti- / chain antibodies (Sigma) antibodies binding 1 h incubation 2. streptavidin coated magnetic beads (Dynal) DNA-hydrolyzing antibody - biotinylated anti- / chain antibody complexes streptavidin-biotin interaction
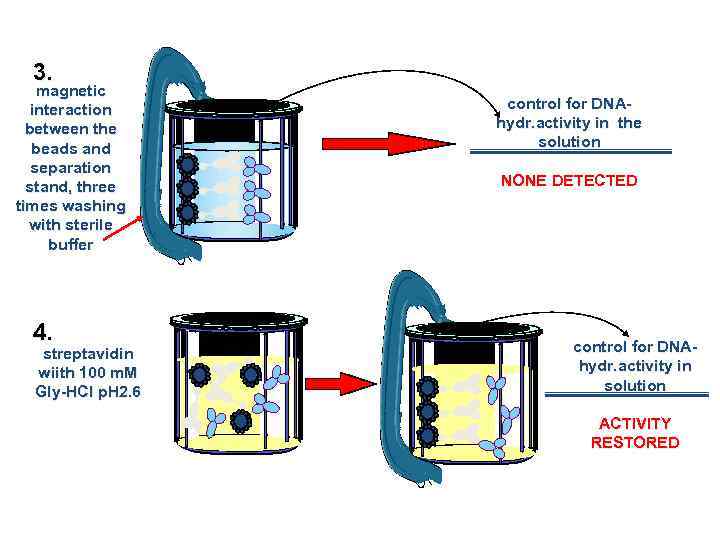
3. magnetic interaction between the beads and separation stand, three times washing with sterile buffer 4. streptavidin wiith 100 m. M Gly-HCl p. H 2. 6 control for DNAhydr. activity in the solution NONE DETECTED control for DNAhydr. activity in solution ACTIVITY RESTORED
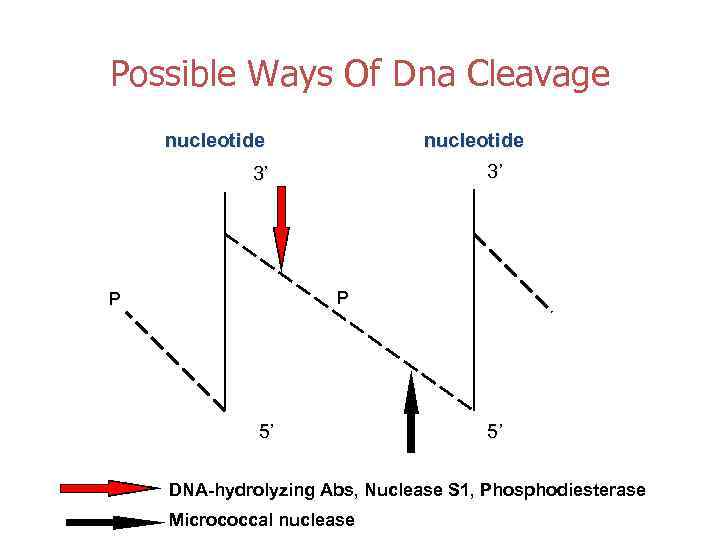
Possible Ways Of Dna Cleavage nucleotide 3’ 3’ P P 5’ 5’ DNA-hydrolyzing Abs, Nuclease S 1, Phosphodiesterase Micrococcal nuclease

Evidence for anti-DNA antibody-mediated complement independent cytotoxicity 1. Antibodies are cytotoxic for cells cultured in absence of fetal calf serum 2. Cytotoxicity is mediated by antibody Fab-fragment only 3. Antibodies induce apoptosis in cultured cells 4. Cytotoxic effect of antibodies remains after gelfiltration of antibodies in 1 M acetic acid, 0. 1 M Na. Cl (“acid shock”)
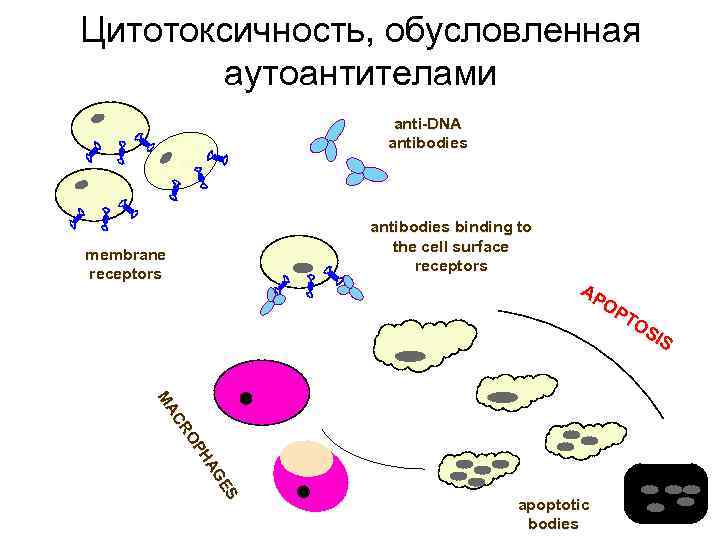
Цитотоксичность, обусловленная аутоантителами anti-DNA antibodies membrane receptors antibodies binding to the cell surface receptors AP OP TO SI S S S GE GE HA HA OP RO CR MA MA apoptotic bodies
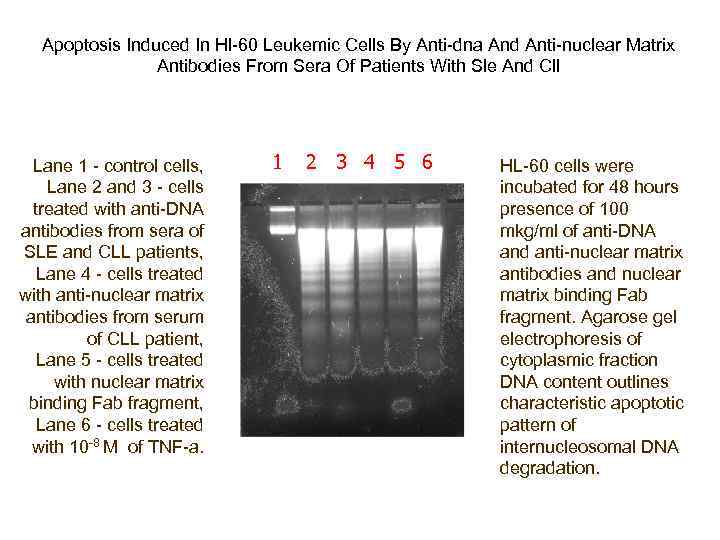
Apoptosis Induced In Hl-60 Leukemic Cells By Anti-dna And Anti-nuclear Matrix Antibodies From Sera Of Patients With Sle And Cll Lane 1 - control cells, Lane 2 and 3 - cells treated with anti-DNA antibodies from sera of SLE and CLL patients, Lane 4 - cells treated with anti-nuclear matrix antibodies from serum of CLL patient, Lane 5 - cells treated with nuclear matrix binding Fab fragment, Lane 6 - cells treated with 10 -8 M of TNF-a. 1 2 3 4 5 6 HL-60 cells were incubated for 48 hours presence of 100 mkg/ml of anti-DNA and anti-nuclear matrix antibodies and nuclear matrix binding Fab fragment. Agarose gel electrophoresis of cytoplasmic fraction DNA content outlines characteristic apoptotic pattern of internucleosomal DNA degradation.
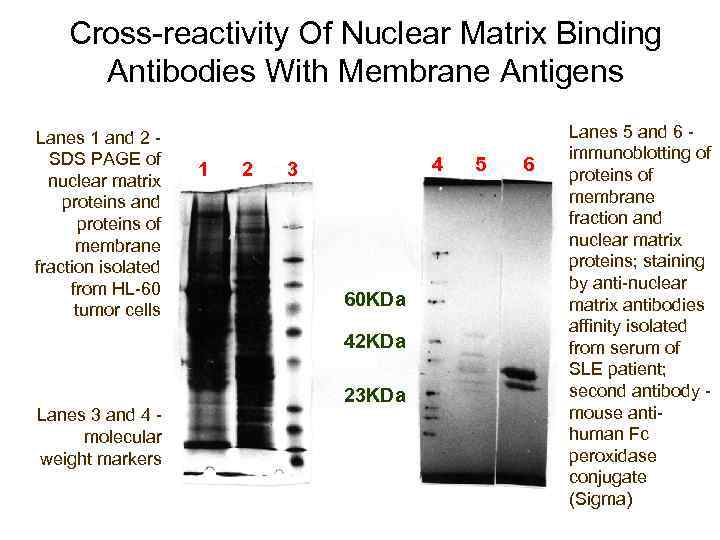
Cross-reactivity Of Nuclear Matrix Binding Antibodies With Membrane Antigens Lanes 1 and 2 - SDS PAGE of nuclear matrix proteins and proteins of membrane fraction isolated from HL-60 tumor cells 1 2 4 3 60 KDa 42 KDa Lanes 3 and 4 - molecular weight markers 23 KDa 5 6 Lanes 5 and 6 - immunoblotting of proteins of membrane fraction and nuclear matrix proteins; staining by anti-nuclear matrix antibodies affinity isolated from serum of SLE patient; second antibody - mouse antihuman Fc peroxidase conjugate (Sigma)
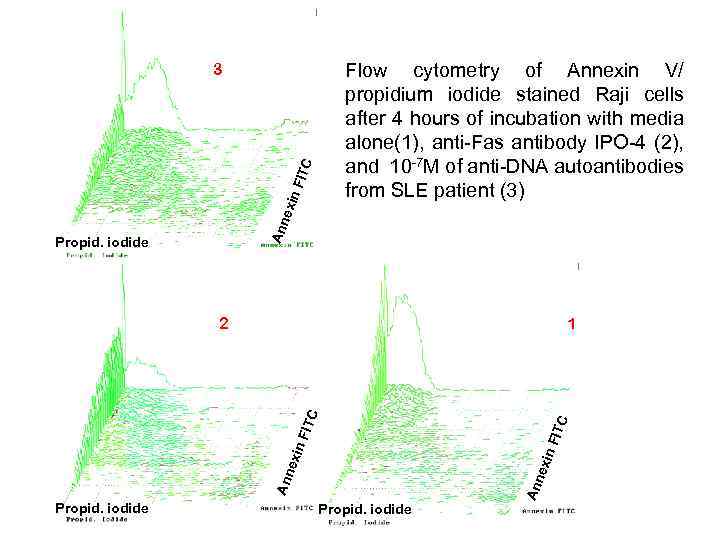
Flow cytometry of Annexin V/ propidium iodide stained Raji cells after 4 hours of incubation with media alone(1), anti-Fas antibody IPO-4 (2), and 10 -7 M of anti-DNA autoantibodies from SLE patient (3) Ann exi n. F ITC 3 Propid. iodide 1 Propid. iodide Ann exi n FIT C C 2
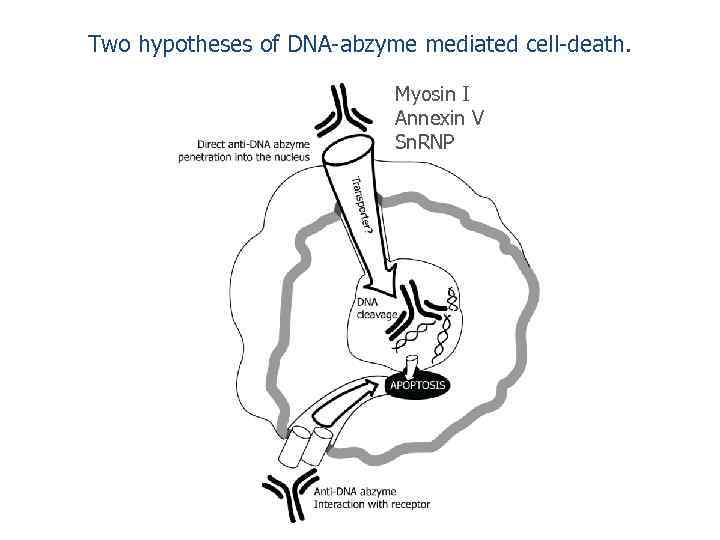
Two hypotheses of DNA-abzyme mediated cell-death. Myosin I Annexin V Sn. RNP
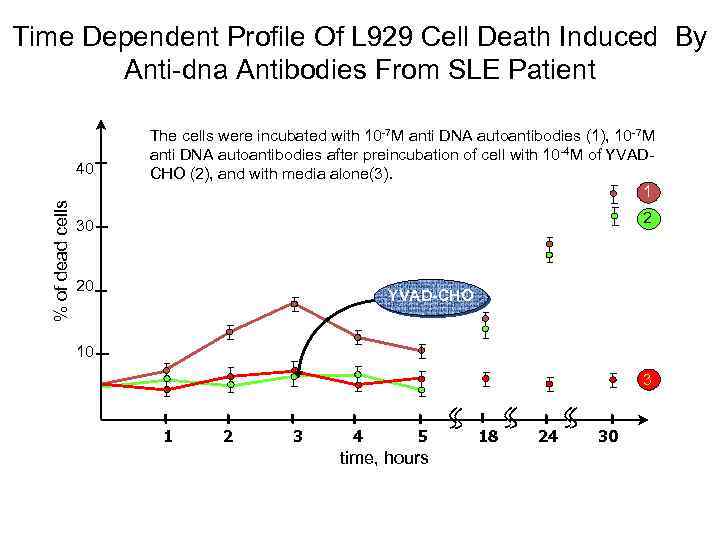
Time Dependent Profile Of L 929 Cell Death Induced By Anti-dna Antibodies From SLE Patient % of dead cells 40 The cells were incubated with 10 -7 M anti DNA autoantibodies (1), 10 -7 M anti DNA autoantibodies after preincubation of cell with 10 -4 M of YVADCHO (2), and with media alone(3). 1 2 30 20 YVAD-CHO 10 3 1 2 3 4 5 time, hours 18 24 30
Multiple Sclerosis Do Myelin-Directed Antibodies Predict Multiple Sclerosis? N EJM, 2003 The B-Cell – Old Player, New Position on the Team NEJM, 2008 B-Cells as one of the key “Players” in the MS
Autoantigens Multiple Sclerosis
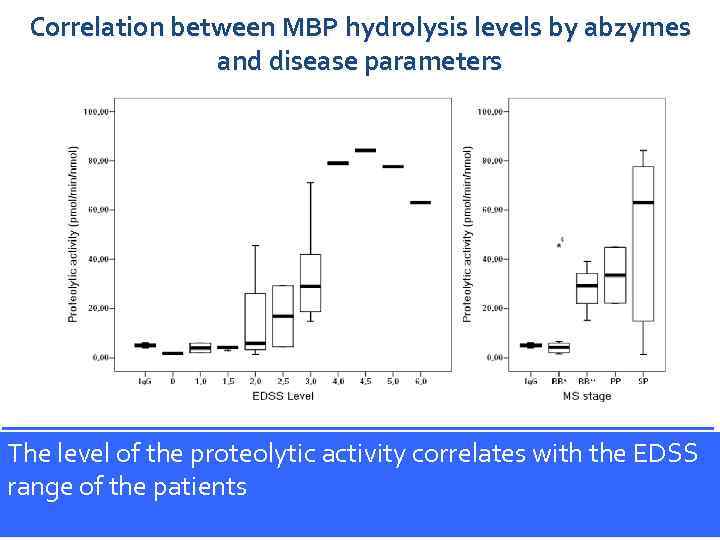
Correlation between MBP hydrolysis levels by abzymes and disease parameters Proteolytic activity the proteolytic activity correlates expanded disability status The (EDSS) levelsof anti-MBP antibodies. Median in MS patients is shown by bold line, quartiles are level of (A) and MS stage (B). from the each group with different with the EDSS scale displayed by bars, error bars represent confidence interval. Ig. G–control donors, of the patients the stage of remission relapsing–remitting the stage range. RR*, relapsing–remitting inprimary and secondary (n = 10); RR**, immunoglobulin frominhealthy of exacerbation (n = 3); PP and SP, progression (n = 2 and 9, respectively). Immunol Lett. 2006; 103(1): 4

MBP Abzymatic Sitespecific MBP hydrolysis Musse et al, PNAS 2006 Analysis of major MBP cleavage products. Reverse-phase HPLC-MS analysis of major MBP cleavage products. Column eluate fractions, corresponding to dominant chromatography peaks (A), were collected, freeze dried, redissolved, and applied to SELDI H 4 chip and tricine-SDSPAGE ( B). Gel was stained by Sypro Orange. Peptides, unambiguously identified by SELDI and clearly seen in corresponding gel lanes, are indicated. ( C) Schematic description of the preferential antibody cleavage sites in the MBP sequence. Sequence fragments identical to the immunodominant MBP-derived peptides (12– 31, 82– 98, 110– 128, and 144– 169) are shown in yellow rectangles, and the encephalitogenic peptide region (86– 98) is marked by the red box. ( D) 32 P-MBP degradation by autoantibodies. Autoradiography of 32 P-phosphorylated MBP hydrolysis by proteolytic mouse (EAE SJL) and human (MS) Ig. G. Line M, molecular mass markers (range 2. 5– 16. 9 k. Da, Amersham Pharmacia). The cleavage sites are localized inside the encephalitogenic epitopes
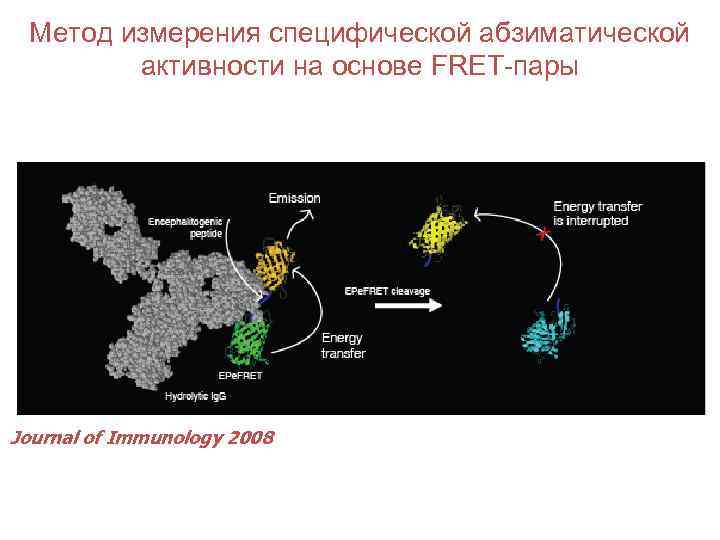
Метод измерения специфической абзиматической активности на основе FRET-пары Journal of Immunology 2008
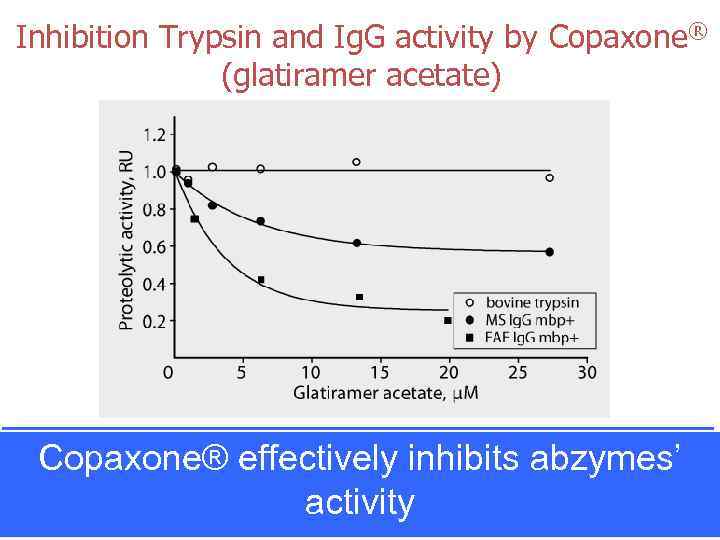
Inhibition Trypsin and Ig. G activity by Copaxone® (glatiramer acetate) Copaxone® effectively inhibits abzymes’ activity Ponomarenko et al, PNAS 2006 Inhibition of MBP degradation, catalyzed by human (MS 1) and mouse (EAE SJL) Ig. G by glatiramer acetate (Copaxone®). MBP trypsinolysis is not effected by glatiramer acetate.

How antigen degradation machinery may be used for Biomedical purposes?

Induction of a protein-specific catalytic response in autoimmune prone mice: a novel strategy for design of a gp 120 -targeted “catalytic vaccine”
The same amount of target may be inactivated by smaller amount of catalytic antibody Binding antibody Catalytic antibody
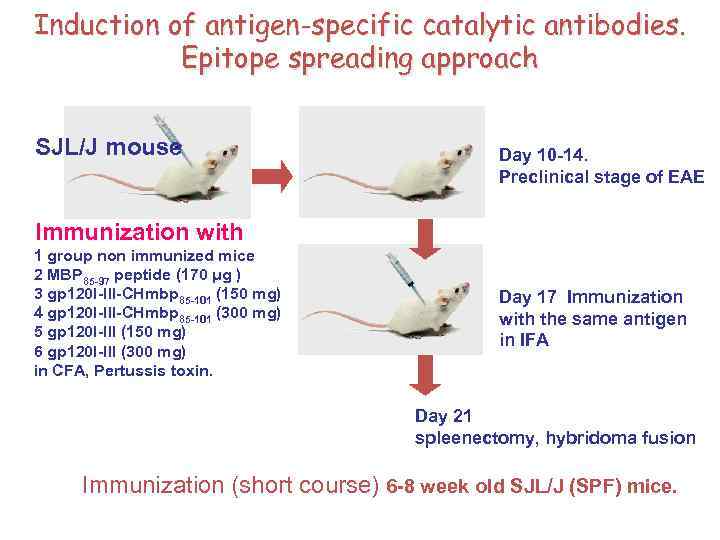
Induction of antigen-specific catalytic antibodies. Epitope spreading approach SJL/J mouse Day 10 -14. Preclinical stage of EAE Immunization with 1 group non immunized mice 2 MBP 85 -97 peptide (170 μg ) 3 gp 120 I-III-CHmbp 85 -101 (150 mg) 4 gp 120 I-III-CHmbp 85 -101 (300 mg) 5 gp 120 I-III (150 mg) 6 gp 120 I-III (300 mg) in CFA, Pertussis toxin. Day 17 Immunization with the same antigen in IFA Day 21 spleenectomy, hybridoma fusion Immunization (short course) 6 -8 week old SJL/J (SPF) mice.
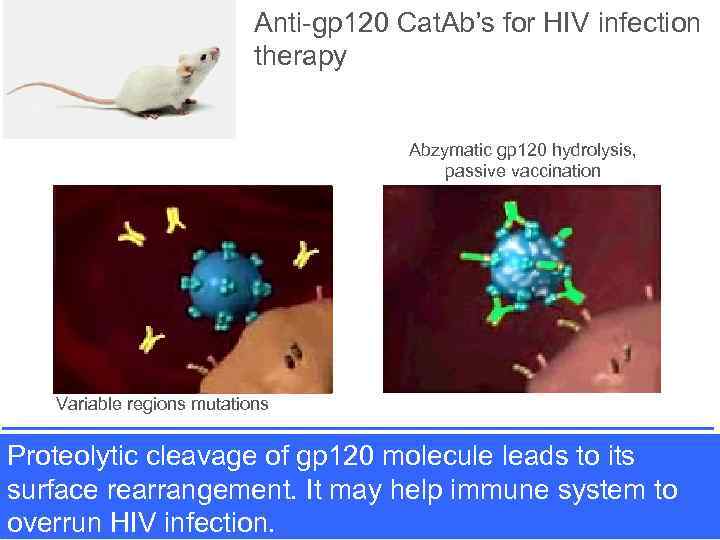
Anti-gp 120 Cat. Ab’s for HIV infection therapy Abzymatic gp 120 hydrolysis, passive vaccination Variable regions mutations Proteolytic cleavage of gp 120 molecule leads to its surface rearrangement. It may help immune system to Ponomarenko et al, Biochemistry 2006 overrun HIV infection.
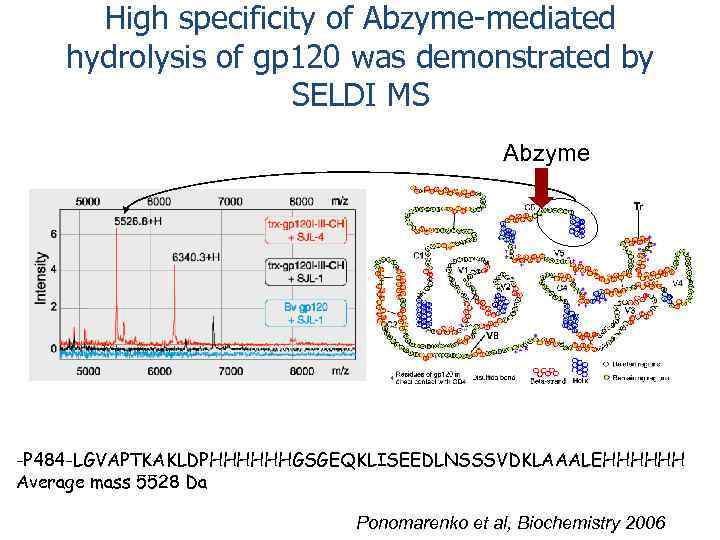
High specificity of Abzyme-mediated hydrolysis of gp 120 was demonstrated by SELDI MS Abzyme -P 484 -LGVAPTKAKLDPHHHHHHGSGEQKLISEEDLNSSSVDKLAAALEHHHHHH Average mass 5528 Da Ponomarenko et al, Biochemistry 2006
Perspectives: generation of artificial biocatalysts in vivo IL 6 TNFa Cocaine Prodrugs Abzymes gp 120 Ab. P OPC ? ? ?
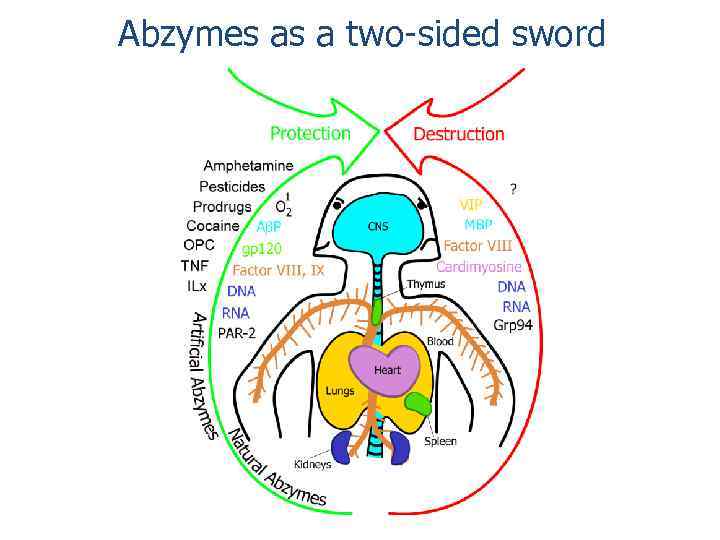
Abzymes as a two-sided sword
введение в иммулогию(25ноя2014).pptx