40bbdc4cdaab9315be62f364bcd245a9.ppt
- Количество слайдов: 21

Airborne Assessment for Aerosols During Manufacturing of Large Molecule Biologics AIHCE – Portland, OR Pharma Forum – Wednesday, May 18, 2011 Dave Pearson – Johnson & Johnson PRD

Discussion Acknowledgements ØMarc Abromovitz, J&J Global Pharma ØBrett Chronister and Steve Nowakowski, J&J Malvern EHS ØDave Pearson, J&J Spring House EHS ØKevin Turner, J&J Radnor EHS ØMark Tartaglia, EHS Impact Solutions, LLC

J&J Historical Background - IH and Large Molecule Compounds ØOnly Qualitative Risk Assessments had been used to evaluate manufacturing processes for large biomolecules § Most large molecule compounds characterized as being low hazard • Not easily absorbed through the skin, digestive system or respiratory tract § Biologics manufacturing processes also characterized as low risk • Primarily wet, closed processes § Consequently, no longer develop OELs or performed air sampling § More recent decision to apply hazard banding instead of developing OELs for large biomolecules ØInterest within EHS to validate these positions

How well do large biomolecules (proteins, monoclonal antibodies, etc. ) get absorbed? ØNot absorbed through the skin ØNot likely absorbed through the upper respiratory tract ØDue to their large molecular weight, those that reach the deep regions of the lungs have low absorption into the bloodstream: § Insulin (MW = 5, 700) approximately 10% absorption § Human Growth Hormone (MW = 30, 000) 5% absorption § Typical Biologic (MW > 100, 000) 10% absorption conservatively assumed

Airborne Exposure to Large Molecule Aerosols vs. Actual Bloodstream Uptake Ø Inhalable Fraction – The fraction of suspended material in ambient air that actually enters the nose or mouth with the volume of air inhaled (typically up to 100 microns in size). Ø Respirable Fraction – The fraction of inhalable aerosols that can reach pulmonary region (up to 10 microns in size) Ø Aerosols greater than 10 microns will often deposit in the nasopharyngeal and tracheobronchial regions. These are trapped in the mucous blanket and removed from the body or swallowed.

What are other influences on biological activity for large molecular weight compounds? ØReadily break down due to exposure to light, low p. H and high temperature § For those compounds that are swallowed, expect compound to be denatured when exposed to acidic environment of the stomach. Therefore, uptake into the bloodstream via the digestive tract is also not expected. § After settling on work surfaces, expect biological activity to be eliminated due to environmental exposure. Therefore, effects of re -suspension in air after drying should be negligible.

Purpose of Monitoring ØIdentify all potential sources of aerosol generation during production of large molecule compounds. ØEvaluate methods for potential measurement of employee exposure to large molecule respirable aerosols and compare with lower end of OEL-2 band (20 ug/m 3). ØMethods considered: § Gravimetric Respirable Aerosol Sampling § Total Protein § Compound-specific IH Method § Real-time Aerosol Monitoring

Monitoring Methods Considered Measuring Total Protein ØAdvantages § More specific than Total Dust § LLD (100 ng/m 3 ) is below the lower end of OEL-2 band ØDisadvantages § Does not differentiate between naturally occurring protein § Historically Total Protein IH sampling has not given an accurate picture of true exposure

Monitoring Methods Considered Specific IH Method ØAdvantages § Most accurate way to determine airborne employee exposure levels § Compound-specific ØDisadvantages § Compound-specific, therefore need new method for each molecule § Delayed time to receive results

Monitoring Methods Considered Real-Time Aerosol Monitoring Ø Advantages § Can provide accurate real time data on respirable aerosols § Can be used at multiple sites for multiple products § Can immediately identify activities and locations of leakage/release Ø Disadvantages § Not validated for real-time measurement of inhalable aerosols >10 microns § Does not differentiate aerosol that is not part of the process. Need to assume that the detected aerosol is 100% product. § Not a personal sample

Monitoring Strategy For Real-Time Aerosol Monitoring Ø Real-time monitoring conducted at two (2) of the J&J Biologics production facilities. § Malvern – GMP, commercial product facility § Spring House – Pilot scale, clinical product facility Ø Area readings taken at workstation operator breathing zones and at the point source for tasks that have the highest potential for generating aerosols. Ø Work area background samples taken during non-processing periods for comparison purposes.

Real Time Aerosol Monitor Dust. Trak™ II Model 8530 Aerosol Monitor (TSI, Inc. ) Desktop Handheld ØDesktop and Handheld models ØBattery operated ØLight scattering LASER photometer ØSimultaneous measurement of mass and size fraction ØData logger

Potential Aerosolization Points in Manufacturing ØReleases during equipment purging or inprocess sampling ØLeaks at connections, hoses, tri-clamps, etc. ØFinal Fill Process (product is dispensed into bulk containers) ØEquipment Disassembly
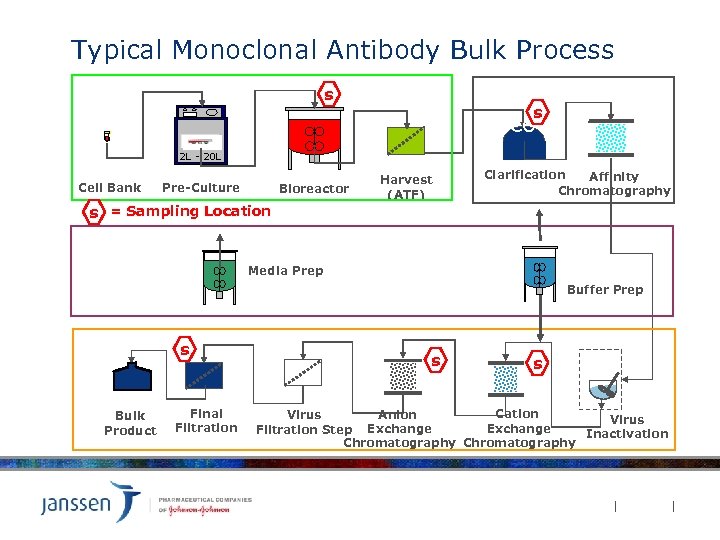
Typical Monoclonal Antibody Bulk Process s s Recovery 2 L - 20 L Cell Bank Pre-Culture Bioreactor Harvest (ATF) Clarification Affinity Chromatography s = Sampling Location Media Prep Buffer Prep s Bulk Product Final Filtration s s Cation Anion Virus Exchange Filtration Step Exchange Inactivation Chromatography

Examples of processes with potential for aerosolization
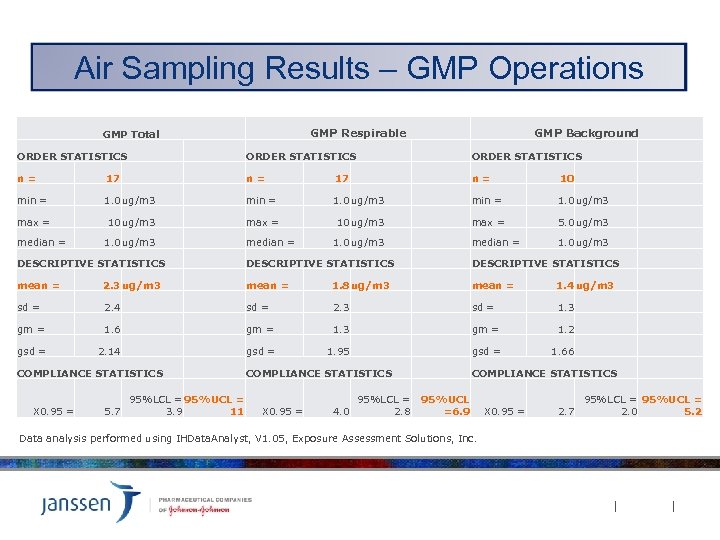
Air Sampling Results – GMP Operations GMP Respirable GMP Total GMP Background ORDER STATISTICS n= 17 n= 10 min = 1. 0 ug/m 3 max = 10 ug/m 3 max = 5. 0 ug/m 3 median = 1. 0 ug/m 3 DESCRIPTIVE STATISTICS mean = 2. 3 ug/m 3 mean = 1. 8 ug/m 3 mean = 1. 4 ug/m 3 sd = 2. 4 sd = 2. 3 sd = 1. 3 gm = 1. 6 gm = 1. 3 gm = 1. 2 gsd = 2. 14 gsd = 1. 95 gsd = 1. 66 COMPLIANCE STATISTICS X 0. 95 = 5. 7 95%LCL = 95%UCL = 3. 9 11 COMPLIANCE STATISTICS X 0. 95 = 4. 0 COMPLIANCE STATISTICS 95%LCL = 95%UCL 2. 8 =6. 9 Data analysis performed using IHData. Analyst, V 1. 05, Exposure Assessment Solutions, Inc. X 0. 95 = 2. 7 95%LCL = 95%UCL = 2. 0 5. 2
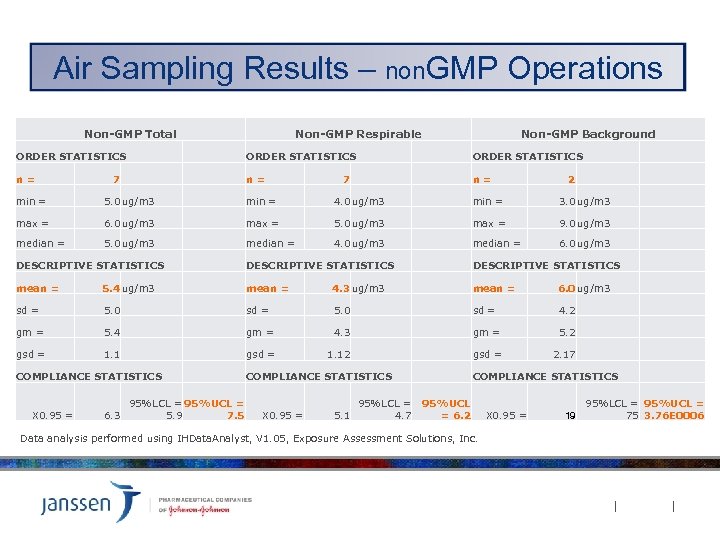
Air Sampling Results – non. GMP Operations Non-GMP Total Non-GMP Respirable Non-GMP Background ORDER STATISTICS n= n= n= 7 7 2 min = 5. 0 ug/m 3 min = 4. 0 ug/m 3 min = 3. 0 ug/m 3 max = 6. 0 ug/m 3 max = 5. 0 ug/m 3 max = 9. 0 ug/m 3 median = 5. 0 ug/m 3 median = 4. 0 ug/m 3 median = 6. 0 ug/m 3 DESCRIPTIVE STATISTICS mean = 5. 4 ug/m 3 mean = 4. 3 ug/m 3 mean = 6. 0 ug/m 3 sd = 5. 0 sd = 4. 2 gm = 5. 4 gm = 4. 3 gm = 5. 2 gsd = 1. 12 gsd = 2. 17 COMPLIANCE STATISTICS X 0. 95 = 6. 3 95%LCL = 95%UCL = 5. 9 7. 5 COMPLIANCE STATISTICS X 0. 95 = 5. 1 COMPLIANCE STATISTICS 95%LCL = 95%UCL 4. 7 = 6. 2 Data analysis performed using IHData. Analyst, V 1. 05, Exposure Assessment Solutions, Inc. X 0. 95 = 19 95%LCL = 95%UCL = 75 3. 76 E 0006

Conclusions Discussion Ø 95% UCL for both Respirable and Total aerosol fraction sampling results were one-half or less lower end of OEL-2 band [20 ug/m 3] without adjusting for % Active Ingredient content. § No operations were identified that require additional engineering controls or use of respirators per J&J criteria. Ø Respirable aerosol sampling results were similar to Background results (substantial overlap in 95% CL band). Ø Respirable aerosol sampling results for GMP operations were similar to non-GMP operations (non-GMP 95% CL band contained within GMP 95% CL band).

Conclusions (cont. ) Discussion Ø Dust. Trak useful as survey instrument to provide real-time concentrations for Respirable fraction during large biomolecule processing. Ø Handheld unit may provide more flexibility for field work. Ø Clean room environment aided in ability to use non-selective instrument to measure target aerosols.

Discussion Next Steps Ø Complete additional comparative statistical analysis of data § Respirable data sets vs. Background sets § GMP data set vs. non-GMP set Ø Collect additional data at PA facilities Ø Collect data at other J&J Biologics facilities § Leiden (The Netherlands) § Cork (Ireland)

? QUESTIONS ?
40bbdc4cdaab9315be62f364bcd245a9.ppt