607c1ce59b9ea10e27d938f0ac4b65f4.ppt
- Количество слайдов: 44
 AIM # 1: What is Chemistry? DO NOW: Answer the following questions 1. What is Chemistry? -is the study of the composition of substances and the changes they undergo - science of matter, an interaction between atoms
AIM # 1: What is Chemistry? DO NOW: Answer the following questions 1. What is Chemistry? -is the study of the composition of substances and the changes they undergo - science of matter, an interaction between atoms
 UNIT 1 INTRODUCTION AND MEASUREMENT • What is Chemistry? • How can problems be solved in a systematic manner? • How do we give meaning and dimension to our descriptions of the world around us? • How can units be used to solve problems? • How do we perform simple calculations during experiments?
UNIT 1 INTRODUCTION AND MEASUREMENT • What is Chemistry? • How can problems be solved in a systematic manner? • How do we give meaning and dimension to our descriptions of the world around us? • How can units be used to solve problems? • How do we perform simple calculations during experiments?
 AIM: What is Chemistry? 2. What are the four main divisions of chemistry? 1. Organic chemistry: the study of substances that contain carbon Example: How gasoline is produces from oil 2. Inorganic chemistry: the study of substances without carbon Example: how table salt reacts with different acids 3. Analytical chemistry: the study of the quantitative composition of substances Examples: how much chlorine is in a sample of tap water 4. Biochemistry: the study of chemistry of living organisms Examples: how sugar in the blood stream of cats affect insulin production
AIM: What is Chemistry? 2. What are the four main divisions of chemistry? 1. Organic chemistry: the study of substances that contain carbon Example: How gasoline is produces from oil 2. Inorganic chemistry: the study of substances without carbon Example: how table salt reacts with different acids 3. Analytical chemistry: the study of the quantitative composition of substances Examples: how much chlorine is in a sample of tap water 4. Biochemistry: the study of chemistry of living organisms Examples: how sugar in the blood stream of cats affect insulin production
 AIM: What is Chemistry? - everyday examples (you do not have to write this down!!!!) 1. Digestion; enzymes promoting chemical reactions that power our bodies. Lifting your arm requires your body to make and burn ATP using oxygen with carbon dioxide as one of the waste gases produced. 2. Cooking is the heating and combination of compounds to make something new. In some cases, like rising bread we have an actual chemical reaction where the yeast changes the food. 3. When concrete dries and hardens the water actually causes a chemical reaction with the cement making a binding action drying concrete isn't just losing water it is undergoing a chemical change and one that creates heat as well (an exothermic reaction).
AIM: What is Chemistry? - everyday examples (you do not have to write this down!!!!) 1. Digestion; enzymes promoting chemical reactions that power our bodies. Lifting your arm requires your body to make and burn ATP using oxygen with carbon dioxide as one of the waste gases produced. 2. Cooking is the heating and combination of compounds to make something new. In some cases, like rising bread we have an actual chemical reaction where the yeast changes the food. 3. When concrete dries and hardens the water actually causes a chemical reaction with the cement making a binding action drying concrete isn't just losing water it is undergoing a chemical change and one that creates heat as well (an exothermic reaction).
 AIM: What is Chemistry? - everyday examples 4. When you write with ink on paper, the ink and paper unite in a chemical reaction so that you can't erase it. Specialized inks allow a short period where you can erase some inks, but most inks dry and can't be erased; they have bound with the paper. This includes your pen and your ink jet printer. 5. Plastics are all about organic chemistry. 6. The sun undergoes fusion and yes that too is chemistry. It creates radiation and photons so we can see. Some of the radiation interacts with oxygen to create ozone and the ozone layer shields us from harmful UV radiation. 7. ANYTHING that burns is undergoing a chemical reaction and almost always creates some form of carbon as waste.
AIM: What is Chemistry? - everyday examples 4. When you write with ink on paper, the ink and paper unite in a chemical reaction so that you can't erase it. Specialized inks allow a short period where you can erase some inks, but most inks dry and can't be erased; they have bound with the paper. This includes your pen and your ink jet printer. 5. Plastics are all about organic chemistry. 6. The sun undergoes fusion and yes that too is chemistry. It creates radiation and photons so we can see. Some of the radiation interacts with oxygen to create ozone and the ozone layer shields us from harmful UV radiation. 7. ANYTHING that burns is undergoing a chemical reaction and almost always creates some form of carbon as waste.
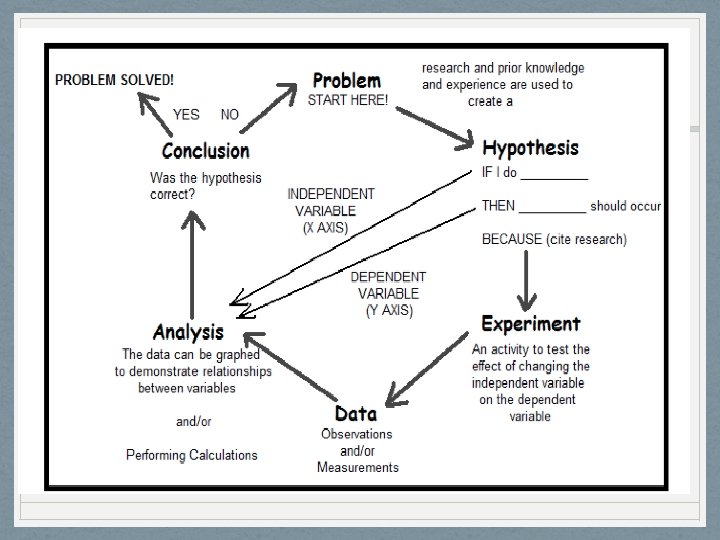
 AIM# 2: How can problems be solved in a systematic manner? • The scientific method is a way to solve a scientific problem. It is an approach to a solution (using mostly common sense)
AIM# 2: How can problems be solved in a systematic manner? • The scientific method is a way to solve a scientific problem. It is an approach to a solution (using mostly common sense)
 AIM: How can problems be solved in a systematic manner? - Steps of the Scientific Method 1. Objective (Problem): statement of purpose 2. Hypothesis (Prediction): Educated guess, in the form: if …. then… 3. Experiment (Test): to test hypothesis, must give reproducible results to be reliable Variable: factor being tested Control: other factors that are held constant
AIM: How can problems be solved in a systematic manner? - Steps of the Scientific Method 1. Objective (Problem): statement of purpose 2. Hypothesis (Prediction): Educated guess, in the form: if …. then… 3. Experiment (Test): to test hypothesis, must give reproducible results to be reliable Variable: factor being tested Control: other factors that are held constant
 AIM: How can problems be solved in a systematic manner? - Steps of the Scientific Method 4. Observations (Data): collect and gather data based on your observations; organize these results to perform analysis in the form of charts, tables or graphs a) Qualitative b) Quantitative 5. Analysis: (Results): organizing data into meaningful groups, tables, graphs, charts
AIM: How can problems be solved in a systematic manner? - Steps of the Scientific Method 4. Observations (Data): collect and gather data based on your observations; organize these results to perform analysis in the form of charts, tables or graphs a) Qualitative b) Quantitative 5. Analysis: (Results): organizing data into meaningful groups, tables, graphs, charts
 6. Conclusions: the determination if your hypothesis was correct, it may be accepted, rejected revised 7. Follow up/application: a repeat with modification is sometimes necessary, and a reevaluation of the results. Also answering one question often leads to new questions. How could you use and communicate the information of your experiment. Why is it important and who could benefit from it?
6. Conclusions: the determination if your hypothesis was correct, it may be accepted, rejected revised 7. Follow up/application: a repeat with modification is sometimes necessary, and a reevaluation of the results. Also answering one question often leads to new questions. How could you use and communicate the information of your experiment. Why is it important and who could benefit from it?
 AIM: How can problems be solved in a systematic manner? - Law vs. Theory: explains the results of experiments, they can change or be rejected over time because of results from new experiments Law: describes natural phenomena, it tells what happens and does not attempt to explain why the phenomena occurs (that is the purpose of a theory). Laws can often be summarized by a math equation
AIM: How can problems be solved in a systematic manner? - Law vs. Theory: explains the results of experiments, they can change or be rejected over time because of results from new experiments Law: describes natural phenomena, it tells what happens and does not attempt to explain why the phenomena occurs (that is the purpose of a theory). Laws can often be summarized by a math equation
 Precision and Accuracy • Accuracy- closeness of a value to the true value • Hitting the bullseye when you are aiming for it • For most experiments the accuracy means how far the deviation is from the expected value • Precision- is the closeness of measurements to each other
Precision and Accuracy • Accuracy- closeness of a value to the true value • Hitting the bullseye when you are aiming for it • For most experiments the accuracy means how far the deviation is from the expected value • Precision- is the closeness of measurements to each other
 (a) Accurate and precise- Casie (b) Not precise, but one piece of data is accurate- Carmen (c) Precise but not accurate- Cynthia (d) Neither precise nor accurate - Cheryl
(a) Accurate and precise- Casie (b) Not precise, but one piece of data is accurate- Carmen (c) Precise but not accurate- Cynthia (d) Neither precise nor accurate - Cheryl
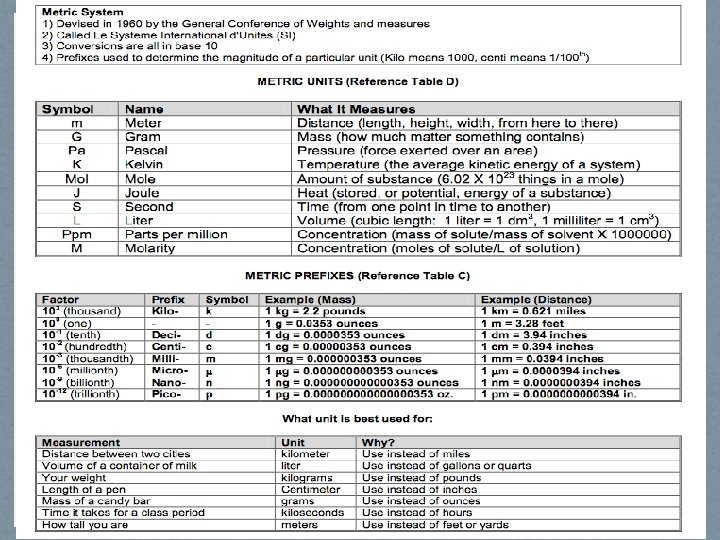
 AIM # 3: How can we give meaning and dimension to our description of the world around us? – Metric System • Measurement gives the universe meaning! How tall are you? How much do you weigh? How old are you? How fast can you run? How much volume do you displace? All of these questions are designed to give us reference to the world around us.
AIM # 3: How can we give meaning and dimension to our description of the world around us? – Metric System • Measurement gives the universe meaning! How tall are you? How much do you weigh? How old are you? How fast can you run? How much volume do you displace? All of these questions are designed to give us reference to the world around us.
 AIM # 3 (a): How can we give meaning and dimension to our description of the world around us? – Math Rules for Chem
AIM # 3 (a): How can we give meaning and dimension to our description of the world around us? – Math Rules for Chem

 AIM: How can we give meaning and dimension to our description of the world around us? – Sig Fig Rules • Atlantic and Pacific Rule: • If a decimal point is present (Pacific side) you start counting from left to right with the first non zero number • If a decimal point is absent (Atlantic side) you start count from right to left with the first non zero number
AIM: How can we give meaning and dimension to our description of the world around us? – Sig Fig Rules • Atlantic and Pacific Rule: • If a decimal point is present (Pacific side) you start counting from left to right with the first non zero number • If a decimal point is absent (Atlantic side) you start count from right to left with the first non zero number
 AIM: How can we give meaning and dimension to our description of the world around us? – Sig Fig Rules Examples: 5 1. 23. 285 cm ________ 1 2. 8000 sec ________ 3. 40. L 2 ________ 4. 2300 g 2 ________
AIM: How can we give meaning and dimension to our description of the world around us? – Sig Fig Rules Examples: 5 1. 23. 285 cm ________ 1 2. 8000 sec ________ 3. 40. L 2 ________ 4. 2300 g 2 ________
 Calculating with sig figs Multiplication and Division: want your answer to have the same number of SIG FIGS as the measurement that has the least number of sig figs Examples: 3 SF 1. 3. 1415 x 2. 25 = 7. 07 2 SF 2. 48. 2 cm x 1. 6 cm x 2. 12 cm = 160
Calculating with sig figs Multiplication and Division: want your answer to have the same number of SIG FIGS as the measurement that has the least number of sig figs Examples: 3 SF 1. 3. 1415 x 2. 25 = 7. 07 2 SF 2. 48. 2 cm x 1. 6 cm x 2. 12 cm = 160
 Calculating with sig figs Addition and Subtraction: want your answer to have the same number of DECIMAL PLACES as the measurement that has the least number of DECIMAL PLACES Examples: 1 DP 2 DP 1. 6. 357 - 2. 4 = 4. 0 2. 3. 842 cm + 8. 51 cm + 16. 324 cm = 28. 68 cm 3
Calculating with sig figs Addition and Subtraction: want your answer to have the same number of DECIMAL PLACES as the measurement that has the least number of DECIMAL PLACES Examples: 1 DP 2 DP 1. 6. 357 - 2. 4 = 4. 0 2. 3. 842 cm + 8. 51 cm + 16. 324 cm = 28. 68 cm 3
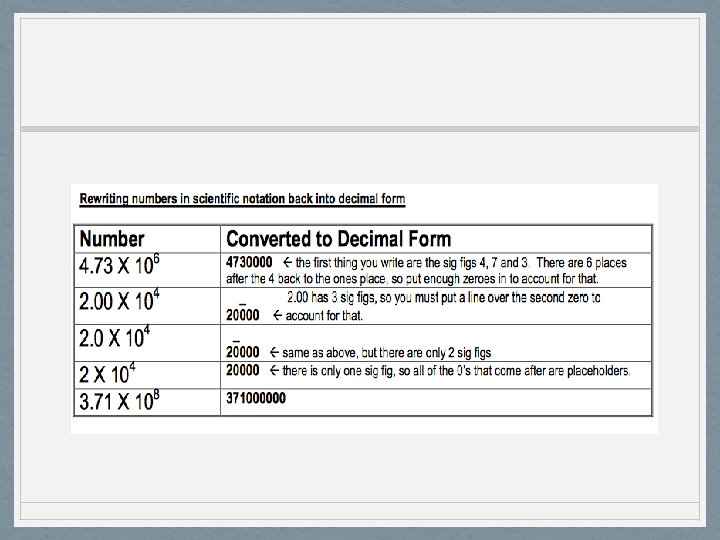
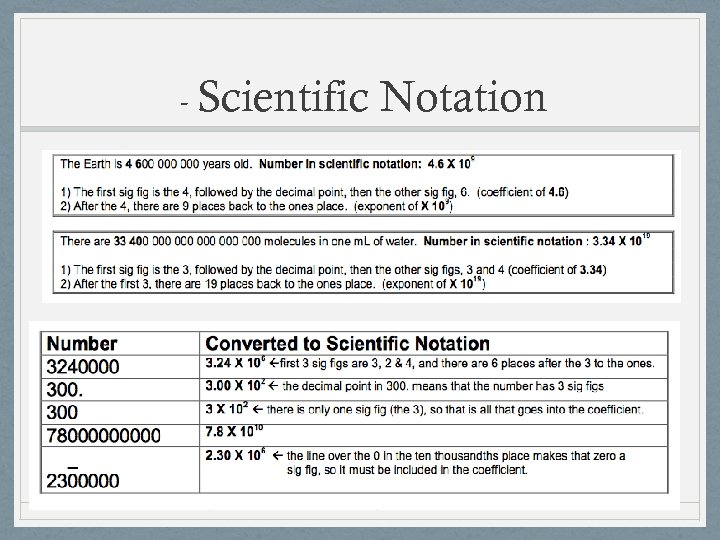 - Scientific Notation
- Scientific Notation
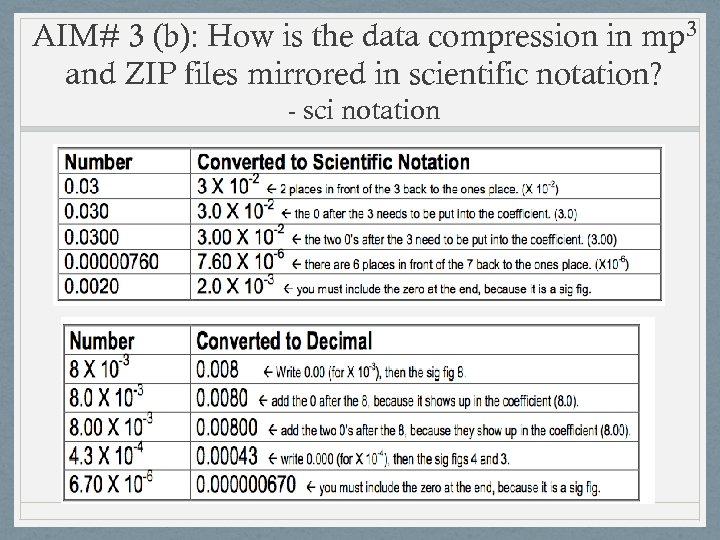 AIM# 3 (b): How is the data compression in mp 3 and ZIP files mirrored in scientific notation? - sci notation
AIM# 3 (b): How is the data compression in mp 3 and ZIP files mirrored in scientific notation? - sci notation
 • Comparing relative magnitudes of two numbers in scientific notation:
• Comparing relative magnitudes of two numbers in scientific notation:
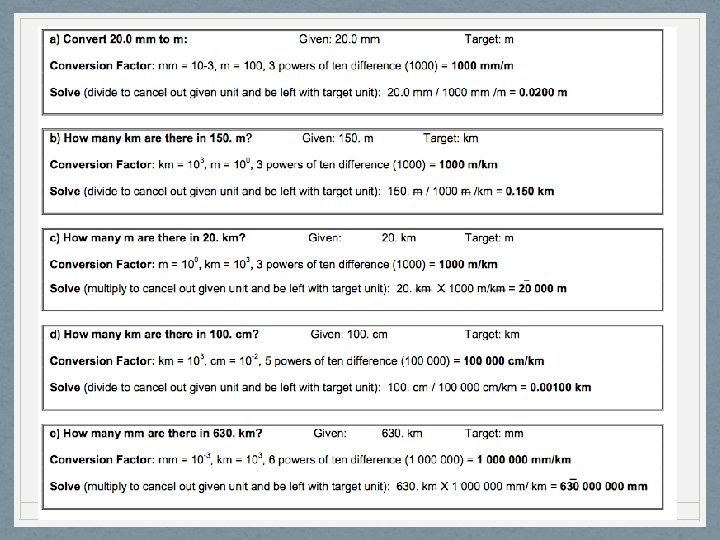
 AIM#4: How can units be used to solve problems? - dimensional analysis • To covert a measurement from one metric unit to another, you must know the difference in magnitude between the two prefixes and use the to create a conversion factor
AIM#4: How can units be used to solve problems? - dimensional analysis • To covert a measurement from one metric unit to another, you must know the difference in magnitude between the two prefixes and use the to create a conversion factor
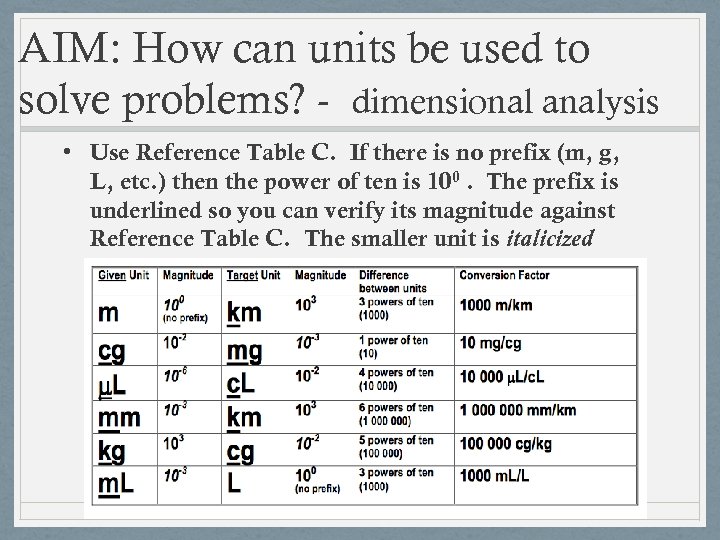 AIM: How can units be used to solve problems? - dimensional analysis • Use Reference Table C. If there is no prefix (m, g, L, etc. ) then the power of ten is 100. The prefix is underlined so you can verify its magnitude against Reference Table C. The smaller unit is italicized
AIM: How can units be used to solve problems? - dimensional analysis • Use Reference Table C. If there is no prefix (m, g, L, etc. ) then the power of ten is 100. The prefix is underlined so you can verify its magnitude against Reference Table C. The smaller unit is italicized
 AIM: How can units be used to solve problems? - dimensional analysis TO USE THE CONVERSION FACTOR: **NOTES: the number of sig figs in your final answer equals the number of sig figs in the number you are converting ** • Given amount multiplied or divided by the conversion = answer • If the given unit is also the numerator unit on the conversion factor, then DIVIDE to cancel it out • If the given unit is also the denominator unit on the conversion factor, then MULTIPLY to cancel it out
AIM: How can units be used to solve problems? - dimensional analysis TO USE THE CONVERSION FACTOR: **NOTES: the number of sig figs in your final answer equals the number of sig figs in the number you are converting ** • Given amount multiplied or divided by the conversion = answer • If the given unit is also the numerator unit on the conversion factor, then DIVIDE to cancel it out • If the given unit is also the denominator unit on the conversion factor, then MULTIPLY to cancel it out
 DIMENSIONAL ANALYSIS EXAMPLES
DIMENSIONAL ANALYSIS EXAMPLES
 Ex 1) A pin measuring 2. 85 cm in length. What is its length in inches? • Need an equivalence statement 2. 54 cm = 1 in • Divide both sides by 2. 54 cm • Unit Factor • Multiply any expression by this unit factor and it will not change its value
Ex 1) A pin measuring 2. 85 cm in length. What is its length in inches? • Need an equivalence statement 2. 54 cm = 1 in • Divide both sides by 2. 54 cm • Unit Factor • Multiply any expression by this unit factor and it will not change its value
 Ex 1) A pin measuring 2. 85 cm in length. What is its length in inches? •
Ex 1) A pin measuring 2. 85 cm in length. What is its length in inches? •
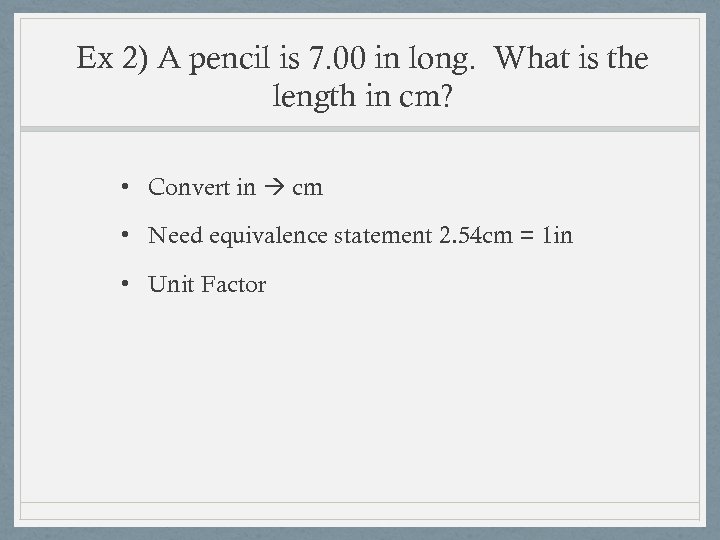 Ex 2) A pencil is 7. 00 in long. What is the length in cm? • Convert in cm • Need equivalence statement 2. 54 cm = 1 in • Unit Factor
Ex 2) A pencil is 7. 00 in long. What is the length in cm? • Convert in cm • Need equivalence statement 2. 54 cm = 1 in • Unit Factor
 DIMENSIONAL ANALYSIS •
DIMENSIONAL ANALYSIS •
 DIMENSIONAL ANALYSIS and • How to choose – look at direction of required change • in cm (need to cancel in – goes in denominator) • cm in (need to cancel cm – goes in denominator)
DIMENSIONAL ANALYSIS and • How to choose – look at direction of required change • in cm (need to cancel in – goes in denominator) • cm in (need to cancel cm – goes in denominator)
 Ex 3) You want to order a bicycle with a 25. 5 in frame, but the sizes in the catalog are given only in cm. What size should you order?
Ex 3) You want to order a bicycle with a 25. 5 in frame, but the sizes in the catalog are given only in cm. What size should you order?
 Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • km mi • Equivalence statement 1 m = 1. 094 yd • Strategy first km m yards mi • Equivalence statements: 1 km = 1000 m 1 m = 1. 094 yd 1760 yd = 1 mi
Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • km mi • Equivalence statement 1 m = 1. 094 yd • Strategy first km m yards mi • Equivalence statements: 1 km = 1000 m 1 m = 1. 094 yd 1760 yd = 1 mi
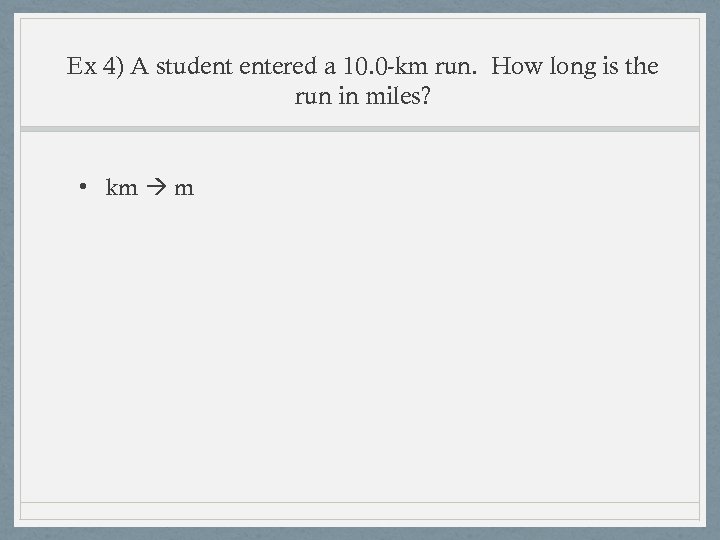 Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • km m
Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • km m
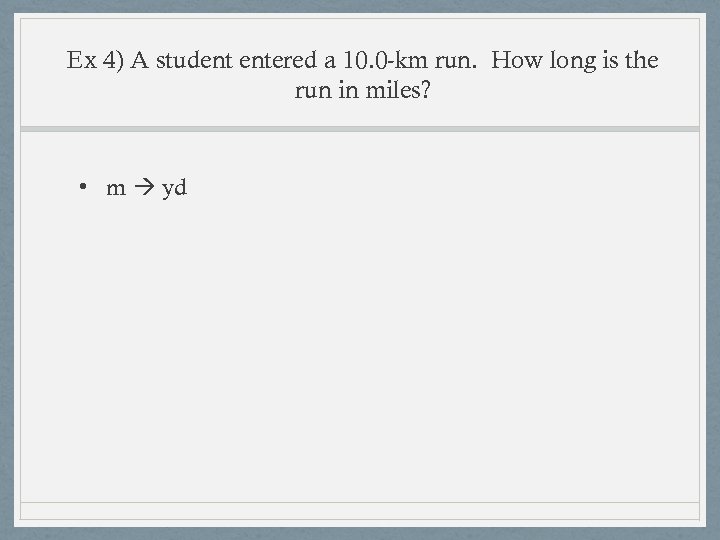 Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • m yd
Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • m yd
 Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • yd mi • Original 10. 0 which has 3 sig figs so you want 3 sig figs in your answer
Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • yd mi • Original 10. 0 which has 3 sig figs so you want 3 sig figs in your answer
 Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • Can combine all conversions into one step
Ex 4) A student entered a 10. 0 -km run. How long is the run in miles? • Can combine all conversions into one step
 AIM # 5: How do we perform simple calculations during experiments Percent Error- the measurement of the percent that the measured value is “off” from the accepted value • Measured value • Accepted value Percent error % error= measured value- accepted value x 100 accepted value (found on table T)
AIM # 5: How do we perform simple calculations during experiments Percent Error- the measurement of the percent that the measured value is “off” from the accepted value • Measured value • Accepted value Percent error % error= measured value- accepted value x 100 accepted value (found on table T)
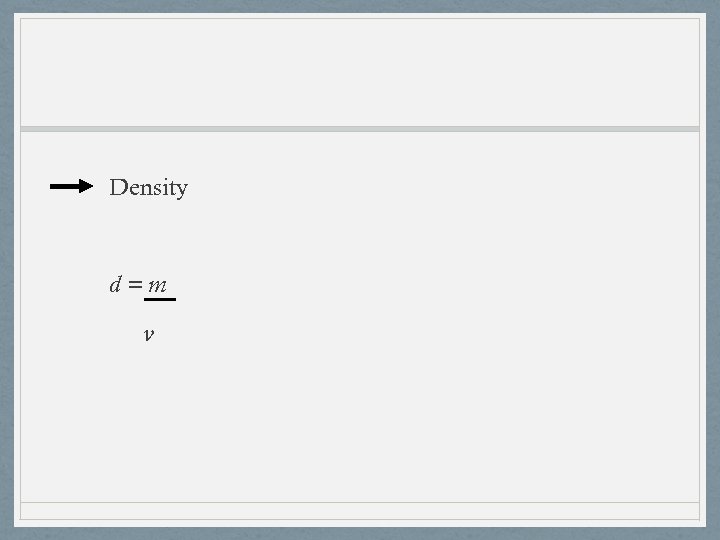 Density d=m v
Density d=m v


