Protein Gel electrophoresis.pptx
- Количество слайдов: 34

 Агарозный гель Направление движения ДНК Мост из фильтровальной бумаги Kb М 1 2 3 Буферный раствор 10 Электрод 7 6 5 Лунки для образцов ДНК 3 1 Источник тока Рис. 1. Схематическое изображение камеры для электрофореза ДНК в агарозном геле. Справа представлена электрофореграмма фрагментов ДНК, окрашенная этидиум бромидом и помещенная под УФ свет. (М – маркеры, 1, 2, 3 – образцы одной и той же ДНК, разрезанные различными рестриктазами).
Агарозный гель Направление движения ДНК Мост из фильтровальной бумаги Kb М 1 2 3 Буферный раствор 10 Электрод 7 6 5 Лунки для образцов ДНК 3 1 Источник тока Рис. 1. Схематическое изображение камеры для электрофореза ДНК в агарозном геле. Справа представлена электрофореграмма фрагментов ДНК, окрашенная этидиум бромидом и помещенная под УФ свет. (М – маркеры, 1, 2, 3 – образцы одной и той же ДНК, разрезанные различными рестриктазами).
 1. Native PAGE 2. Native Gradient PAGE 3. Urea PAGE 4. SDS PAGE 5. SDS Gradient PAGE 6. IEF 7. 2 D PAGE 8. Western Blot
1. Native PAGE 2. Native Gradient PAGE 3. Urea PAGE 4. SDS PAGE 5. SDS Gradient PAGE 6. IEF 7. 2 D PAGE 8. Western Blot
 Proteins move in the electric field. Their relative speed depends on the charge, size, and shape of the protein
Proteins move in the electric field. Their relative speed depends on the charge, size, and shape of the protein
 Protein gel electrophoresis • used to separate proteins based on a number of characteristics Denaturing gel electrophoresis separate by size Nondenaturing (native) gel electrophoresis separate by size, shape, charge
Protein gel electrophoresis • used to separate proteins based on a number of characteristics Denaturing gel electrophoresis separate by size Nondenaturing (native) gel electrophoresis separate by size, shape, charge
 Acrylamide is a potent neurotoxin and should be handled with care!
Acrylamide is a potent neurotoxin and should be handled with care!
 TEMED - catalyzes free radical formation APS - free radical donor Bisacrylamide - crosslinking agent (19: 1 ratio of acrylamide to bis maximizes crosslinking) Higher % of gel - smaller pores (holes) so smaller fragments can be resolved
TEMED - catalyzes free radical formation APS - free radical donor Bisacrylamide - crosslinking agent (19: 1 ratio of acrylamide to bis maximizes crosslinking) Higher % of gel - smaller pores (holes) so smaller fragments can be resolved
 CATALYST OF POLYMERIZATION Polymerization of acrylamide is initiated by the addition of ammonium persulphate and the base N, N, N’-tetrametyhlenediamine (TEMED) TEMED catalyses the decomposition of the persulphate ion to give a free radical
CATALYST OF POLYMERIZATION Polymerization of acrylamide is initiated by the addition of ammonium persulphate and the base N, N, N’-tetrametyhlenediamine (TEMED) TEMED catalyses the decomposition of the persulphate ion to give a free radical
 POLYMERIZATION OF ACRYLAMIDE n Cross-linked polyacrylamide gels are formed from the polymerisation of acrylamide monomer in the presence of smaller amounts of N, N’methylenebisacrylamide (bis -acrylamide) n Bisacrylamide is the most frequently used cross linking agent for polyacrylamide gels
POLYMERIZATION OF ACRYLAMIDE n Cross-linked polyacrylamide gels are formed from the polymerisation of acrylamide monomer in the presence of smaller amounts of N, N’methylenebisacrylamide (bis -acrylamide) n Bisacrylamide is the most frequently used cross linking agent for polyacrylamide gels
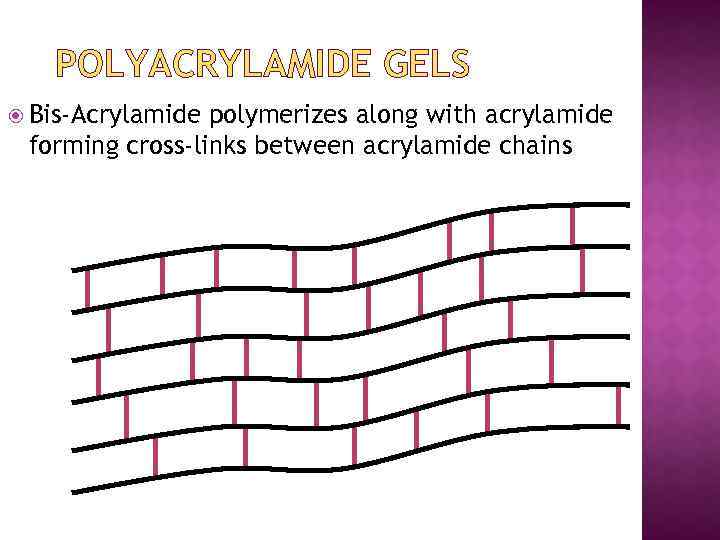 POLYACRYLAMIDE GELS Bis-Acrylamide polymerizes along with acrylamide forming cross-links between acrylamide chains
POLYACRYLAMIDE GELS Bis-Acrylamide polymerizes along with acrylamide forming cross-links between acrylamide chains
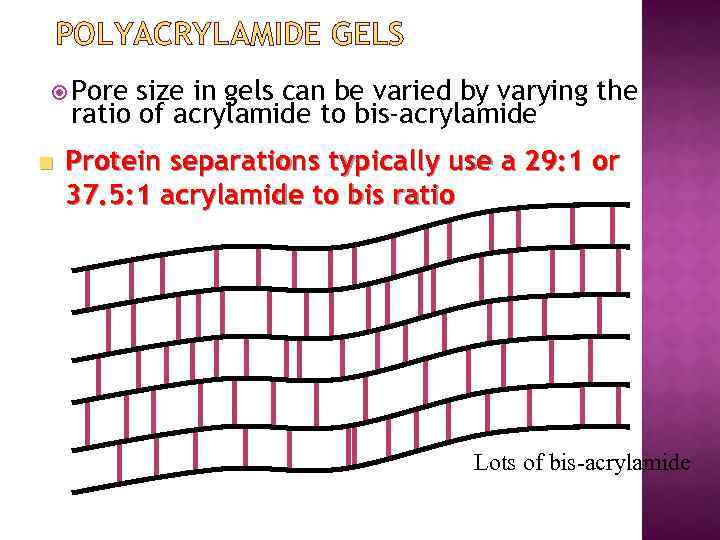 POLYACRYLAMIDE GELS Pore size in gels can be varied by varying the ratio of acrylamide to bis-acrylamide n Protein separations typically use a 29: 1 or 37. 5: 1 acrylamide to bis ratio Lots of bis-acrylamide
POLYACRYLAMIDE GELS Pore size in gels can be varied by varying the ratio of acrylamide to bis-acrylamide n Protein separations typically use a 29: 1 or 37. 5: 1 acrylamide to bis ratio Lots of bis-acrylamide
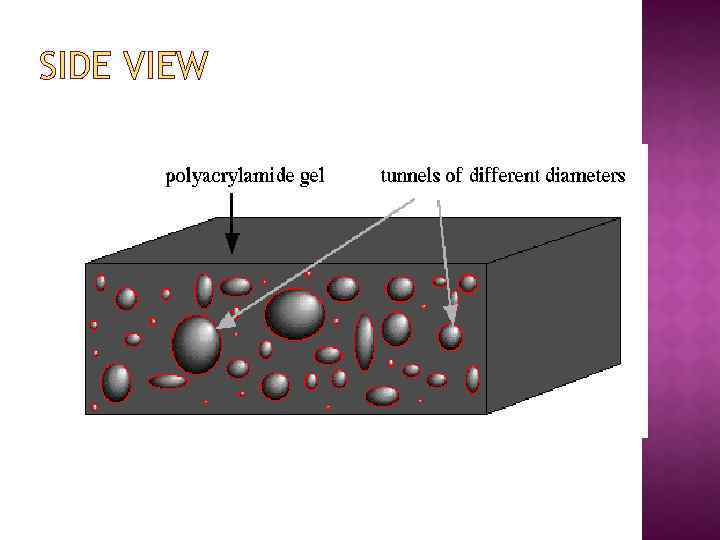 SIDE VIEW
SIDE VIEW
 MOVEMENT OF PROTEINS IN GEL
MOVEMENT OF PROTEINS IN GEL
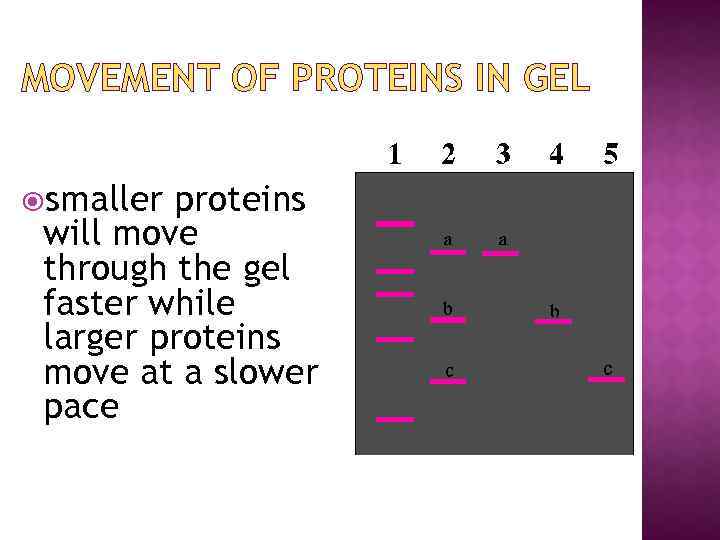 MOVEMENT OF PROTEINS IN GEL smaller proteins will move through the gel faster while larger proteins move at a slower pace
MOVEMENT OF PROTEINS IN GEL smaller proteins will move through the gel faster while larger proteins move at a slower pace
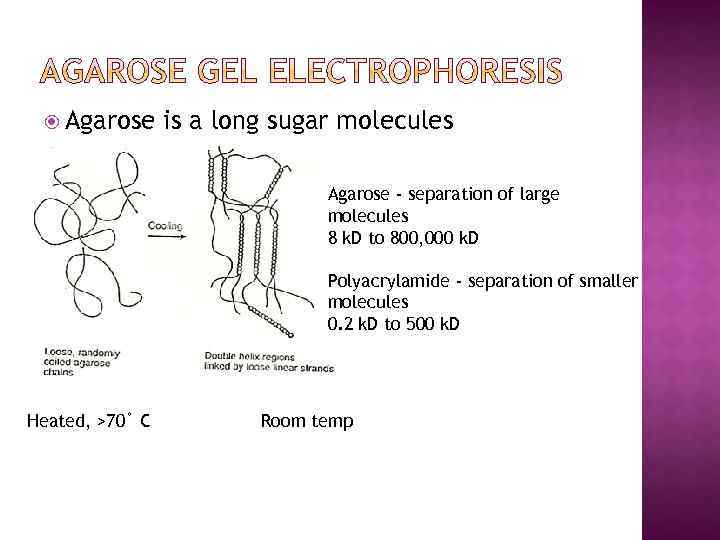 Agarose is a long sugar molecules Agarose - separation of large molecules 8 k. D to 800, 000 k. D Polyacrylamide - separation of smaller molecules 0. 2 k. D to 500 k. D Heated, >70˚ C Room temp
Agarose is a long sugar molecules Agarose - separation of large molecules 8 k. D to 800, 000 k. D Polyacrylamide - separation of smaller molecules 0. 2 k. D to 500 k. D Heated, >70˚ C Room temp
 DNA is negatively charged so it is attracted to the positive end of the gel. The shorter DNA fragments move faster than the longer fragments. DNA is separated on basis of size.
DNA is negatively charged so it is attracted to the positive end of the gel. The shorter DNA fragments move faster than the longer fragments. DNA is separated on basis of size.
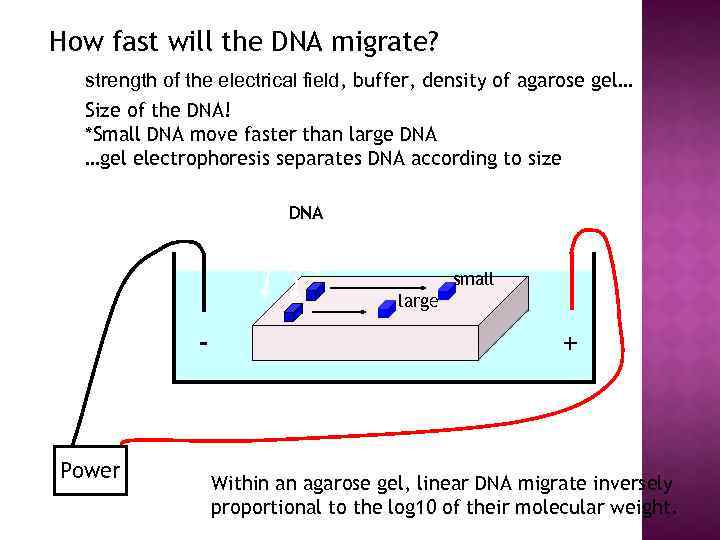 How fast will the DNA migrate? strength of the electrical field, buffer, density of agarose gel… Size of the DNA! *Small DNA move faster than large DNA …gel electrophoresis separates DNA according to size DNA small large - Power + Within an agarose gel, linear DNA migrate inversely proportional to the log 10 of their molecular weight.
How fast will the DNA migrate? strength of the electrical field, buffer, density of agarose gel… Size of the DNA! *Small DNA move faster than large DNA …gel electrophoresis separates DNA according to size DNA small large - Power + Within an agarose gel, linear DNA migrate inversely proportional to the log 10 of their molecular weight.
 Mother Child Man
Mother Child Man
 In 2002 Elizabeth Hurley used DNA profiling to prove that Steve Bing was the father of her child Damien
In 2002 Elizabeth Hurley used DNA profiling to prove that Steve Bing was the father of her child Damien
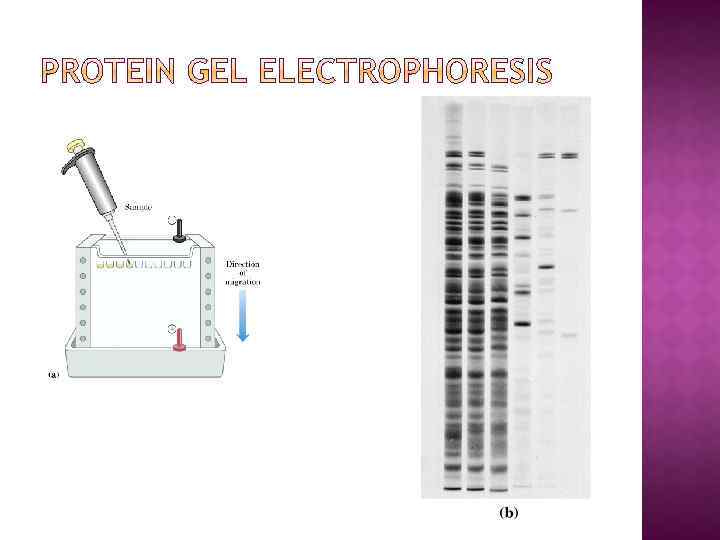
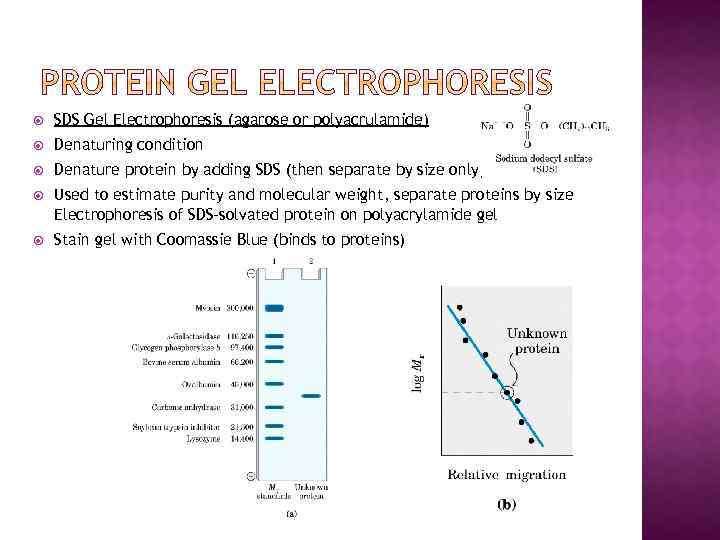 SDS Gel Electrophoresis (agarose or polyacrulamide) Denaturing condition Denature protein by adding SDS (then separate by size only) Used to estimate purity and molecular weight, separate proteins by size Electrophoresis of SDS-solvated protein on polyacrylamide gel Stain gel with Coomassie Blue (binds to proteins)
SDS Gel Electrophoresis (agarose or polyacrulamide) Denaturing condition Denature protein by adding SDS (then separate by size only) Used to estimate purity and molecular weight, separate proteins by size Electrophoresis of SDS-solvated protein on polyacrylamide gel Stain gel with Coomassie Blue (binds to proteins)
 polypeptides retain their higher-order structure and often retain enzymatic activity and interaction with other polypeptides migration of proteins depends on many factors, including size, shape, and native charge. native gels omit the SDS and reducing agent (DTT) do not put SDS or DTT in the sample buffer do not heat the samples prepare the gel and tank buffer solutions without SDS.
polypeptides retain their higher-order structure and often retain enzymatic activity and interaction with other polypeptides migration of proteins depends on many factors, including size, shape, and native charge. native gels omit the SDS and reducing agent (DTT) do not put SDS or DTT in the sample buffer do not heat the samples prepare the gel and tank buffer solutions without SDS.
 Separates folded proteins and protein-protein or protein-ligand complexes by charge, size, and shape Useful for: 1. Examining protein-protein-ligand interactions 2. Detecting protein isoforms/conformers
Separates folded proteins and protein-protein or protein-ligand complexes by charge, size, and shape Useful for: 1. Examining protein-protein-ligand interactions 2. Detecting protein isoforms/conformers
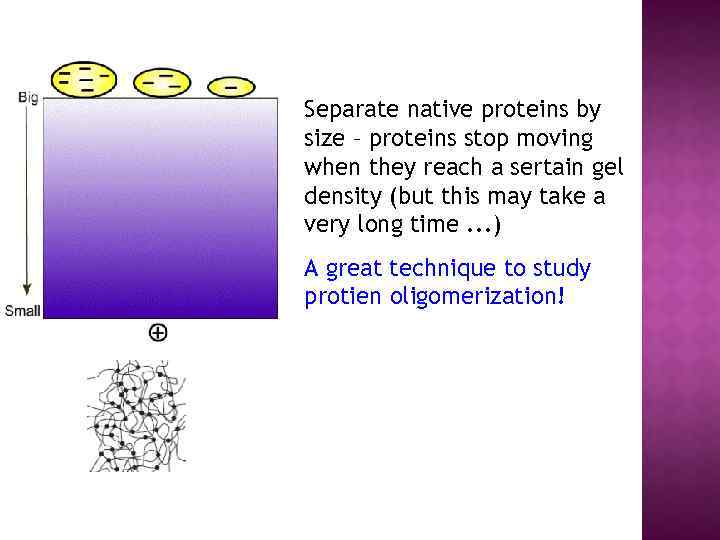 Separate native proteins by size – proteins stop moving when they reach a sertain gel density (but this may take a very long time. . . ) A great technique to study protien oligomerization!
Separate native proteins by size – proteins stop moving when they reach a sertain gel density (but this may take a very long time. . . ) A great technique to study protien oligomerization!
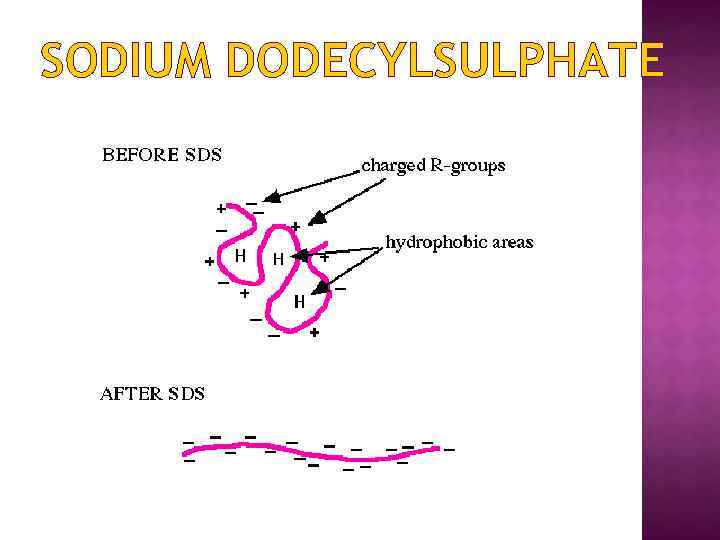
 SODIUM DODECYLSULPHATE SDS (sodium dodecyl sulfate) is a detergent that can dissolve hydrophobic molecules but also has a negative charge (sulfate) attached to it. If SDS is added to proteins, they will be soluablized by the detergent, plus all the proteins will be covered with many negative charges.
SODIUM DODECYLSULPHATE SDS (sodium dodecyl sulfate) is a detergent that can dissolve hydrophobic molecules but also has a negative charge (sulfate) attached to it. If SDS is added to proteins, they will be soluablized by the detergent, plus all the proteins will be covered with many negative charges.
 SODIUM DODECYLSULPHATE** A sample of protein, often freshly isolated and unpurified, is boiled in the detergent sodium dodecyl sulfate and betamercaptoethanol The mercaptoethanol reduces disulfide bonds The detergent disrupts secondary and tertiary structure The end result has two important features: 1. all proteins contain only primary structure and 2. all proteins have a large negative charge which means they will all migrate towards the positive pole when placed in an electric field. They migrate through a gel towards the positive pole at a rate proportional to their linear size Molecular weights with respect to size markers may then be determined
SODIUM DODECYLSULPHATE** A sample of protein, often freshly isolated and unpurified, is boiled in the detergent sodium dodecyl sulfate and betamercaptoethanol The mercaptoethanol reduces disulfide bonds The detergent disrupts secondary and tertiary structure The end result has two important features: 1. all proteins contain only primary structure and 2. all proteins have a large negative charge which means they will all migrate towards the positive pole when placed in an electric field. They migrate through a gel towards the positive pole at a rate proportional to their linear size Molecular weights with respect to size markers may then be determined
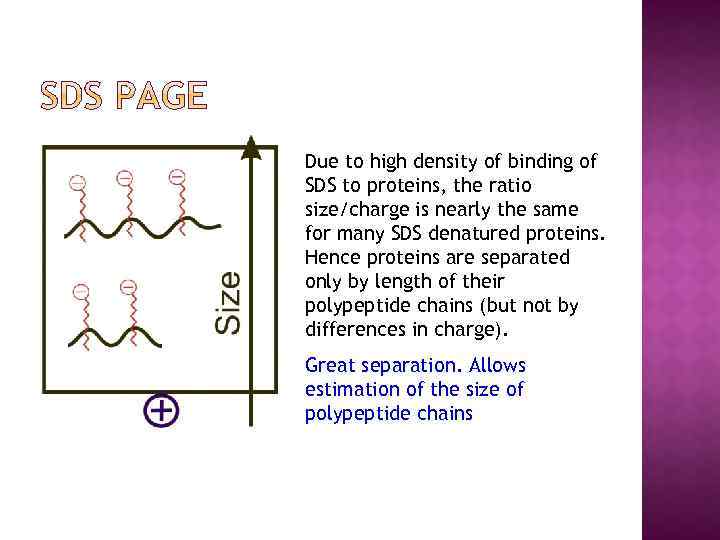 Due to high density of binding of SDS to proteins, the ratio size/charge is nearly the same for many SDS denatured proteins. Hence proteins are separated only by length of their polypeptide chains (but not by differences in charge). Great separation. Allows estimation of the size of polypeptide chains
Due to high density of binding of SDS to proteins, the ratio size/charge is nearly the same for many SDS denatured proteins. Hence proteins are separated only by length of their polypeptide chains (but not by differences in charge). Great separation. Allows estimation of the size of polypeptide chains
 SODIUM DODECYLSULPHATE
SODIUM DODECYLSULPHATE
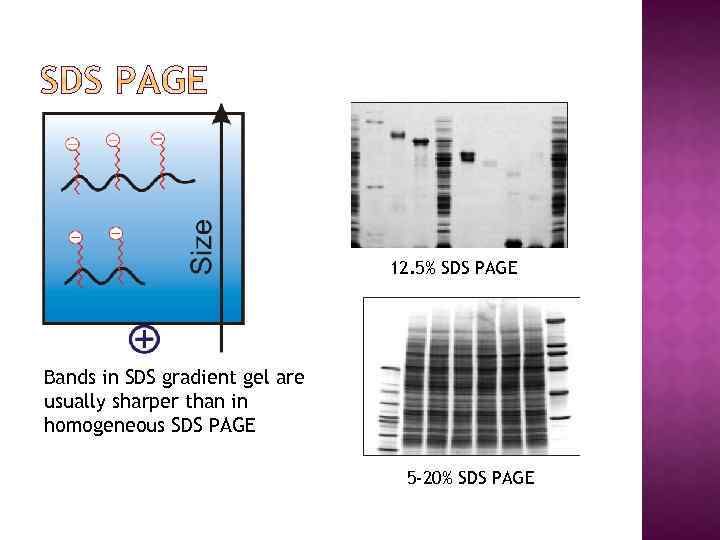 12. 5% SDS PAGE Bands in SDS gradient gel are usually sharper than in homogeneous SDS PAGE 5 -20% SDS PAGE
12. 5% SDS PAGE Bands in SDS gradient gel are usually sharper than in homogeneous SDS PAGE 5 -20% SDS PAGE
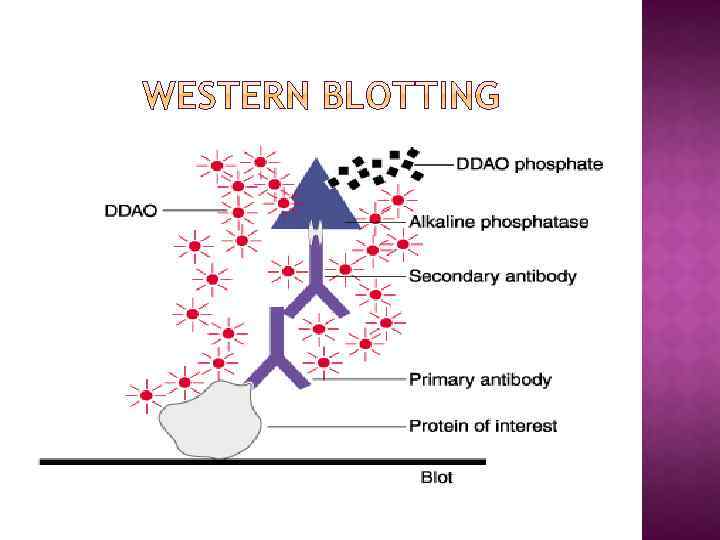
 WESTERN BLOTTING (WB) WB is a protein detection technique that combines the separation power of SDS PAGE together with high recognition specificity of antibodies An antibody against the target protein could be purified from serum of animals (mice, rabbits, goats) immunized with this protein Alternatively, if protein contains a commonly used tag or epitope, an antibody against the tag/epitope could be purchase from a commercial source (e. g. anti-6 His antibody)
WESTERN BLOTTING (WB) WB is a protein detection technique that combines the separation power of SDS PAGE together with high recognition specificity of antibodies An antibody against the target protein could be purified from serum of animals (mice, rabbits, goats) immunized with this protein Alternatively, if protein contains a commonly used tag or epitope, an antibody against the tag/epitope could be purchase from a commercial source (e. g. anti-6 His antibody)
 1. Separation of proteins using SDS PAGE 2. Transfer of the proteins onto e. g. a nitrocellulose membrane (blotting) 3. Immune reactions 4. Visualization
1. Separation of proteins using SDS PAGE 2. Transfer of the proteins onto e. g. a nitrocellulose membrane (blotting) 3. Immune reactions 4. Visualization
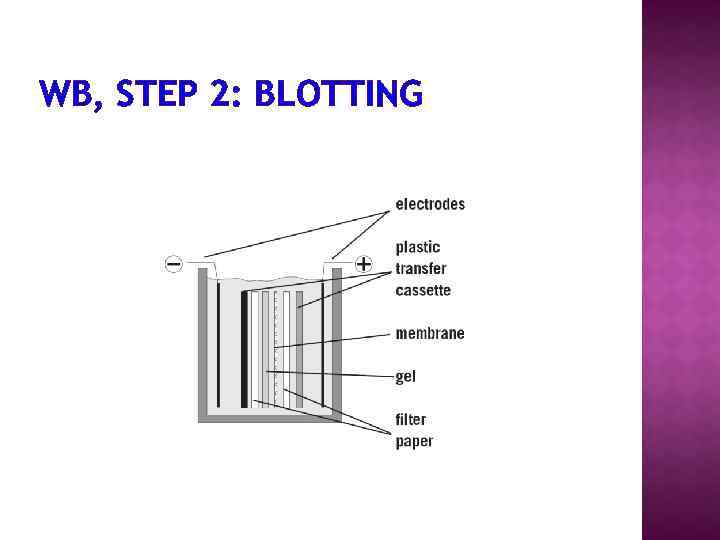 WB, STEP 2: BLOTTING
WB, STEP 2: BLOTTING
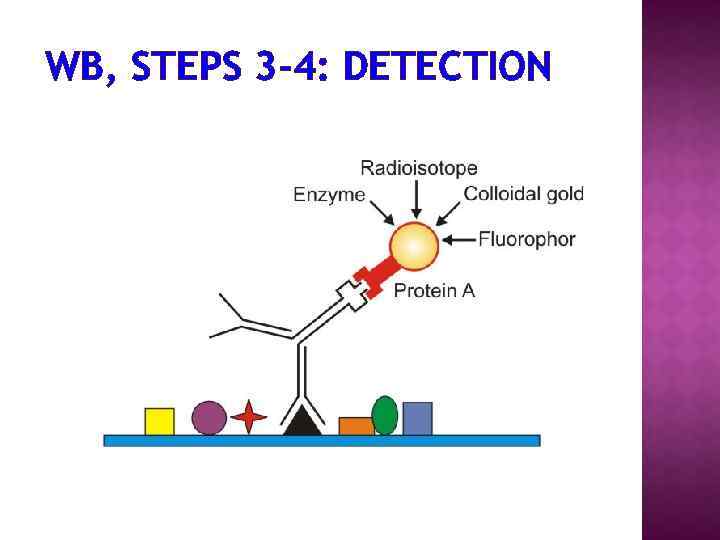 WB, STEPS 3 -4: DETECTION
WB, STEPS 3 -4: DETECTION