17c50992885d60a754d4b3cf549d6ebd.ppt
- Количество слайдов: 48

Adverse Events: An Update Elizabeth Ness, RN, MS Director, Staff Development Center for Cancer Research National Cancer Institute

Objectives • Describe factors that precipitated CTCAE v 3. 0 Revision Project • Define Med. DRA and list major components of Med. DRA • Summarize major AE term issues regarding CTCAE v 3. 0 and Med. DRA • State sources and purpose of CTCAE v 4. 0 AE term definitions • Describe characteristics of general descriptions of CTCAE v 4. 0 Grading Scale • Differentiate between Instrumental ADLs and Self care ADLs • Describe various resources for CTCAE • Discuss Med. DRA versioning related to CTCAE

Background • 1982/1983: Common Toxicity Criteria (CTC) used to aid in the recognition and grading severity of adverse events (AE) of chemotherapy • List of AE terms commonly encountered in oncology and each AE is accompanied by a grading (severity) scale • Fundamentally intended to be an agreed upon terminology for the designation, reporting and grading of AEs that occur in oncology research
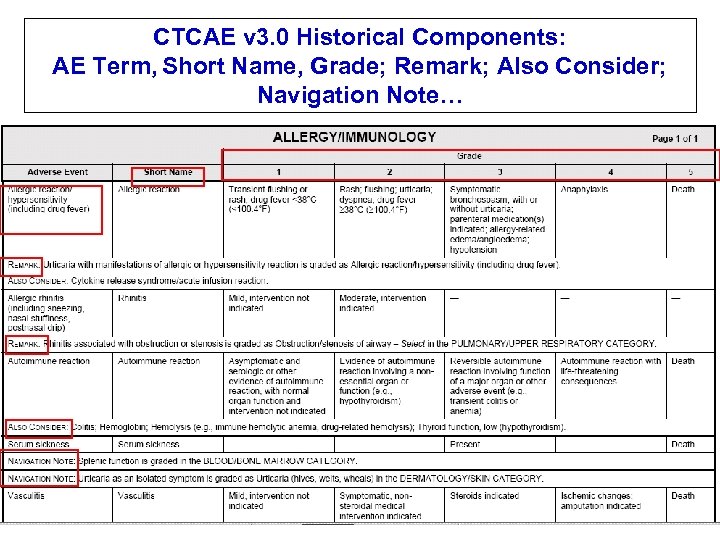
CTCAE v 3. 0 Historical Components: AE Term, Short Name, Grade; Remark; Also Consider; Navigation Note…
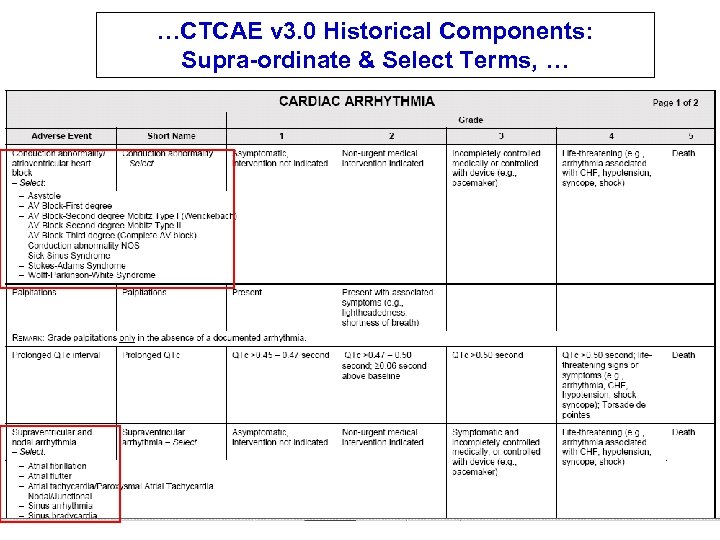
…CTCAE v 3. 0 Historical Components: Supra-ordinate & Select Terms, …
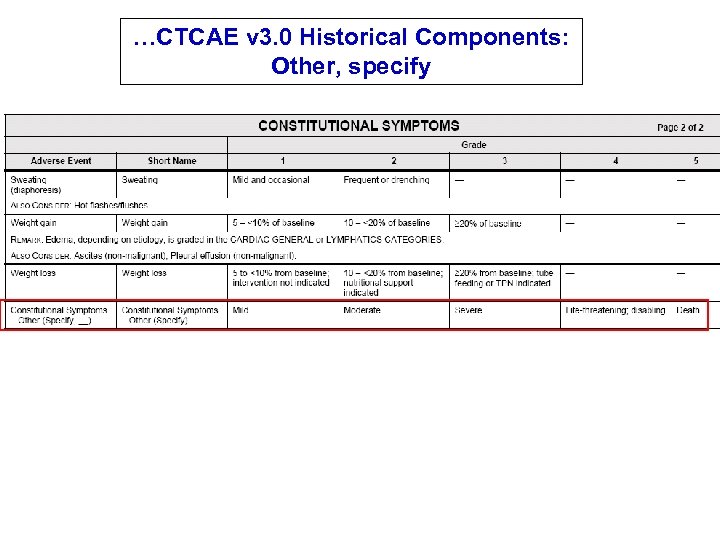
…CTCAE v 3. 0 Historical Components: Other, specify

Medical Dictionary for Regulatory Activities (Med. DRA) • Clinically validated international medical terminology • Adverse event classification dictionary endorsed by the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) • Used by regulatory authorities and the regulated biopharmaceutical industry throughout the entire regulatory process • from pre-marketing to post-marketing activities • data entry, retrieval, evaluation, and presentation 7

CTCAE v 3. 0 Impetus for Revision • April 2006: Med. DRA MSSO (Maintenance and Support Services Organization) Blue Ribbon Panel determined that a mechanism to “translate” or “convert” CTCAE terms to Med. DRA terms must be established in order to facilitate data exchange within internal databases and between investigators and regulatory authorities for the purpose of Serious Adverse Event (SAE) reporting • CTEP and FDA agreed on a CTCAE redesign to be harmonized with Med. DRA at the AE term level March 22, 2010 http: //evs. nci. nih. gov/ftp 1/CTCAE/Documentation/CTCAE_Governance_2010 -03 -11. doc

Med. DRATM Structure and Content SOC (26) HLGT (>300) HLT (>1, 600) PT (>18, 000) LLT(>86, 000)
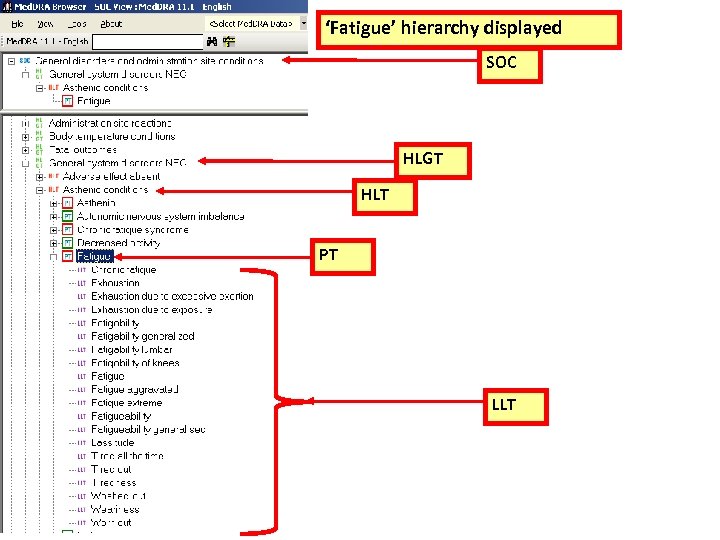
‘Fatigue’ hierarchy displayed SOC HLGT HLT PT LLT

CTCAE Revision Project June 2008 – June 2009 • Organized and funded by CBIIT, NCI • CTEP collaboration • Participants – FDA, Industry, Med. DRA MSSO, Cancer Centers, Others • Working Groups (~140 members) • Content expertise based on Med. DRA SOCs • Steering Committee ( 40 members) • Governance Group (25 members) • Harmonization with Med. DRA • 100% single concept Med. DRA terms • Isolate critical concepts as unique AE terms 11

Comparison Med. DRA to CTCAE Med. DRA List of terms >86, 000 Content: Comprehensive --Multiple translations Hierarchy: 5 levels Medically validated --- CTCAE v 4. 0 List of Med. DRA LLTs 790 Content: Oncology Severity Scale English; Japanese Hierarchy: 2 levels --Includes definitions

CTCAE v 4. 0 Med. DRA SOC Replaces v 3. 0 CATEGORY… CTCAE v 3. 0 CATEGORY (Count 28) CTCAE v 4. 0 SOC (Count 26) AUDITORY / EAR Ear and labyrinth disorders DERMATOLOGY / SKIN Skin and subcutaneous tissue disorders ENDOCRINE Endocrine disorders INFECTION Infections and infestations PULMONARY / UPPER RESPIRATORY Respiratory and thoracic disorders VASCULAR Vascular disorders

…CTCAE v 4. 0 Med. DRA SOC Replaces v 3. 0 CATEGORY CTCAE v 4. 0 SOC CARDIAC ARRHYTHMIA Cardiac disorders CARDIAC GENERAL Nervous system disorders NEUROLOGY Psychiatric disorders

v 3. 0 CATEGORIES Deleted in v 4. 0… CTCAE v 3. 0 CATEGORY CTCAE v 4. 0 SOC CTCAE v 4. 0 AE Term General disorders and administration site conditions Death NOS Death neonatal Sudden death NOS Pregnancy, puerperium and perinatal conditions Fetal death DEATH GROWTH AND DEVELOPMENT PAIN Musculoskeletal and connective tissue disorders System – specific SOC: Ear and labyrinth disorders; Gastrointestinal disorders; Renal and urinary disorders; Musculoskeletal and connective tissue disorders etc.

…v 3. 0 CATEGORIES Deleted in v 4. 0 CTCAE v 3. 0 CATEGORY CTCAE v 4. 0 SOC Blood and lymphatic system disorders Cardiac disorders Immune system disorders Metabolism and nutrition disorders SYNDROMES Neoplasms benign, malignant and unspecified (incl cysts and polyps) Nervous system disorders Respiratory, thoracic and mediastinal disorders Skin and subcutaneous tissue disorders Vascular disorders

CTCAE v 3. 0 AE Term Issues Related to Med. DRA • CTCAE v 3. 0 AE Terms • Multiple concepts • One/many element of Grade description is critical AE concept • Not all Med. DRA terms • 72% CTCAE = ‘mapped’ to a single Med. DRA term/code • 28% CTCAE = CTEP-only code (leading 9’s with meaning to no one outside CTEP)

CTCAE v 3. 0 Multiple Concepts in one AE Term Mapped to Med. DRA: Fatigue 10016256 Fatigue, Asthenia, Lethargy, Malaise • Are unique concepts in Med. DRA (PTs) • Are not –related to a single Preferred Term –synonyms, lexical variants, or quasi-synonyms 18
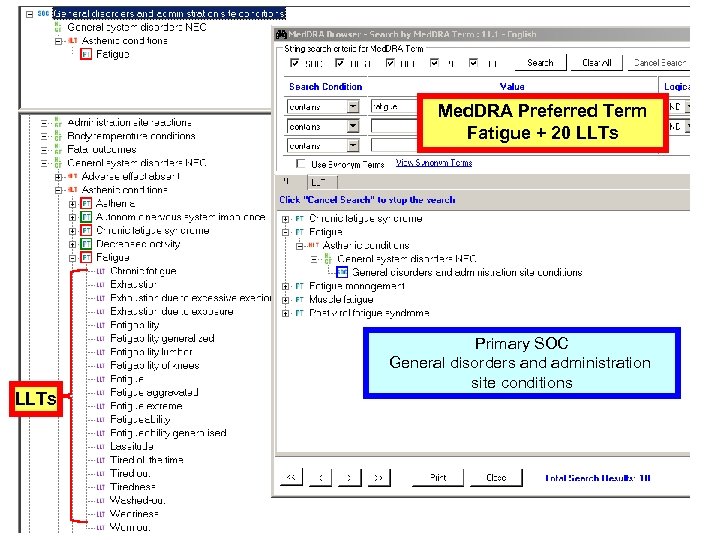
Med. DRA Preferred Term Fatigue + 20 LLTs Primary SOC General disorders and administration site conditions
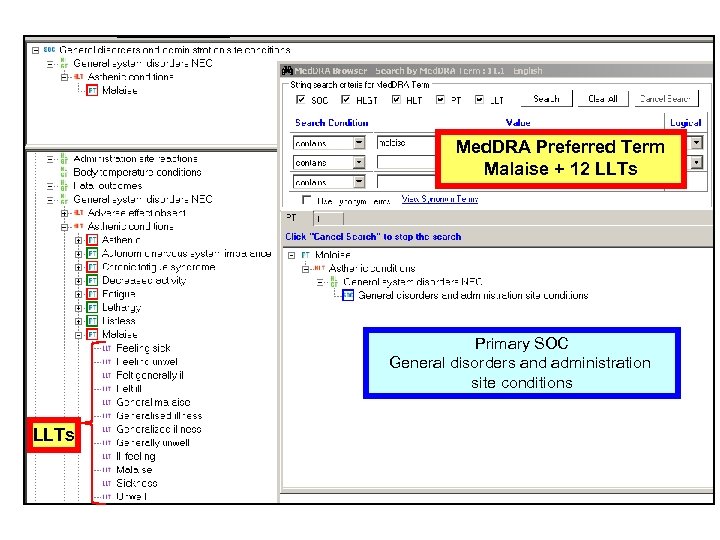
Med. DRA Preferred Term Malaise + 12 LLTs Primary SOC General disorders and administration site conditions LLTs
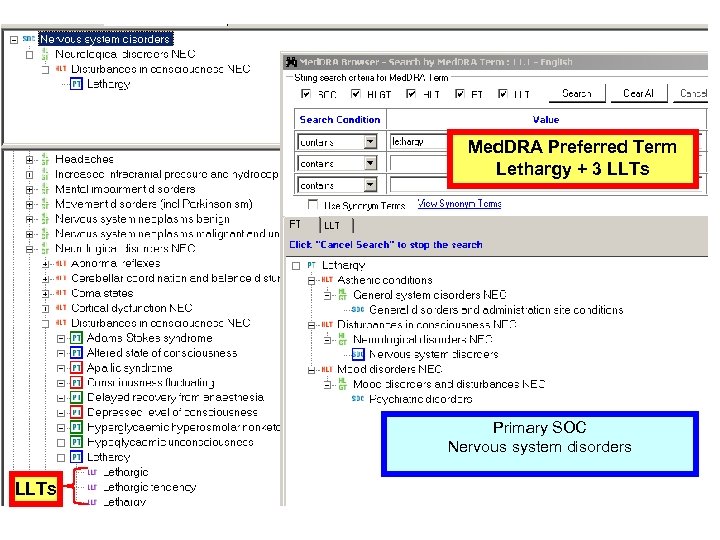
Med. DRA Preferred Term Lethargy + 3 LLTs Primary SOC Nervous system disorders LLTs
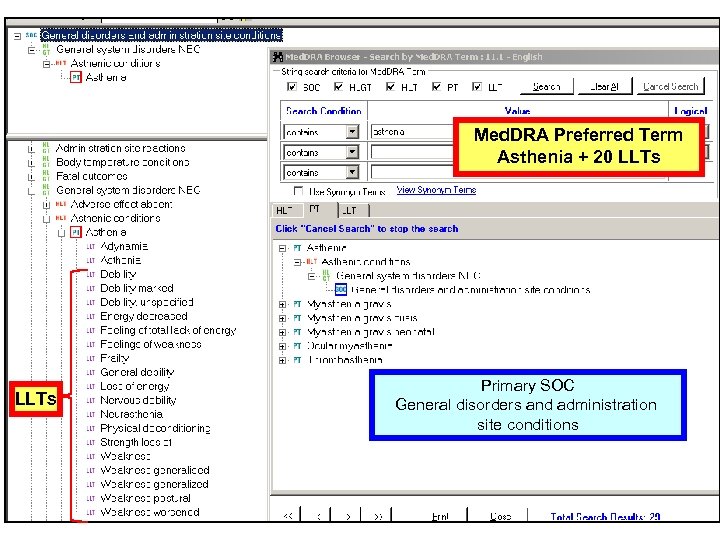
Med. DRA Preferred Term Asthenia + 20 LLTs Primary SOC General disorders and administration site conditions
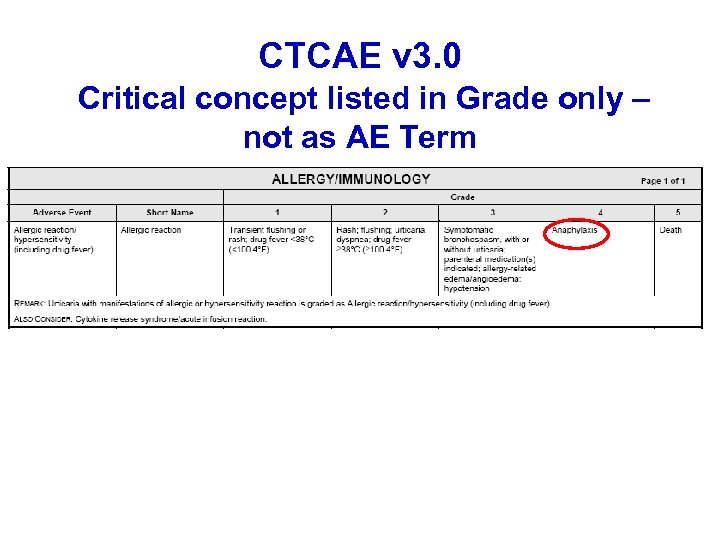
CTCAE v 3. 0 Critical concept listed in Grade only – not as AE Term
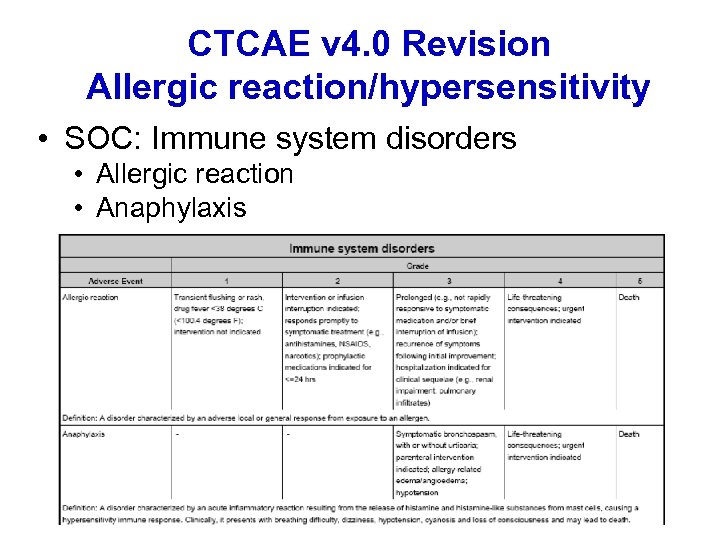
CTCAE v 4. 0 Revision Allergic reaction/hypersensitivity • SOC: Immune system disorders • Allergic reaction • Anaphylaxis

CTCAE v 3. 0 Issue Non-Med. DRA Terms + CTEP-Only Codes INFECTION CATEGORY

CTCAE v 3. 0 Count of 77 INFECTION terms 99% map to Med. DRA

CTCAE v 3. 0 INFECTION CATEGORY • Infection with unknown ANC – Conjunctiva • Med. DRA: Conjunctivitis infective -10010742 • Infection (documented clinically or microbiologically) with Grade 3 or 4 neutrophils (ANC <1. 0 x 10 e 9/L) – Conjunctiva 9 codes • Infection with normal ANC or Grade 1 or 2 neutrophils - Conjunctiva
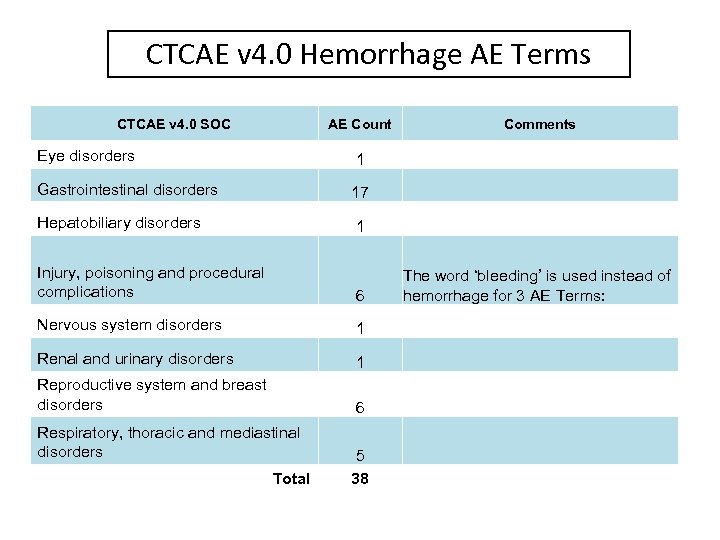
CTCAE v 4. 0 Hemorrhage AE Terms CTCAE v 4. 0 SOC AE Count Eye disorders 1 Gastrointestinal disorders 17 Hepatobiliary disorders Comments 1 Injury, poisoning and procedural complications 6 Nervous system disorders 1 Renal and urinary disorders 1 Reproductive system and breast disorders 6 Respiratory, thoracic and mediastinal disorders Total 5 38 The word ‘bleeding’ is used instead of hemorrhage for 3 AE Terms:
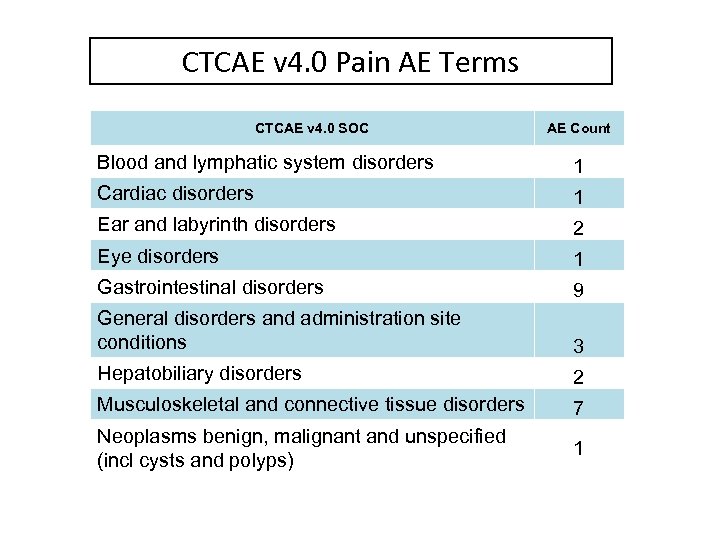
CTCAE v 4. 0 Pain AE Terms CTCAE v 4. 0 SOC AE Count Blood and lymphatic system disorders 1 Cardiac disorders 1 Ear and labyrinth disorders 2 Eye disorders 1 Gastrointestinal disorders 9 General disorders and administration site conditions 3 Hepatobiliary disorders 2 Musculoskeletal and connective tissue disorders 7 Neoplasms benign, malignant and unspecified (incl cysts and polyps) 1
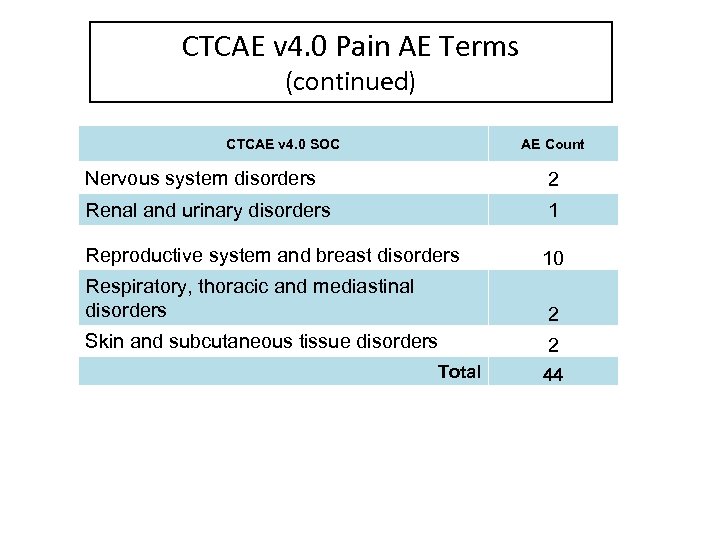
CTCAE v 4. 0 Pain AE Terms (continued) CTCAE v 4. 0 SOC AE Count Nervous system disorders 2 Renal and urinary disorders 1 Reproductive system and breast disorders 10 Respiratory, thoracic and mediastinal disorders 2 Skin and subcutaneous tissue disorders 2 Total 44
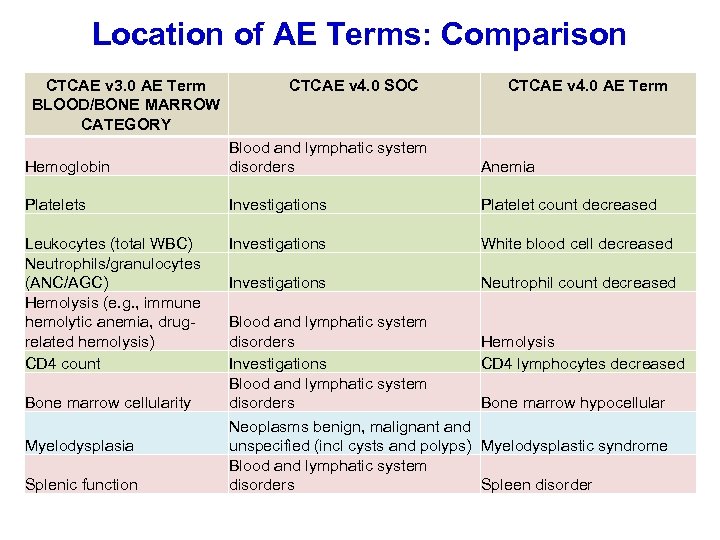
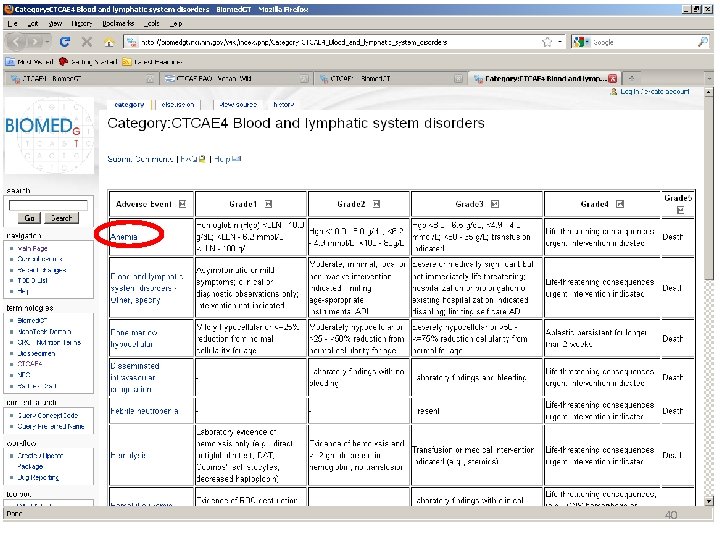
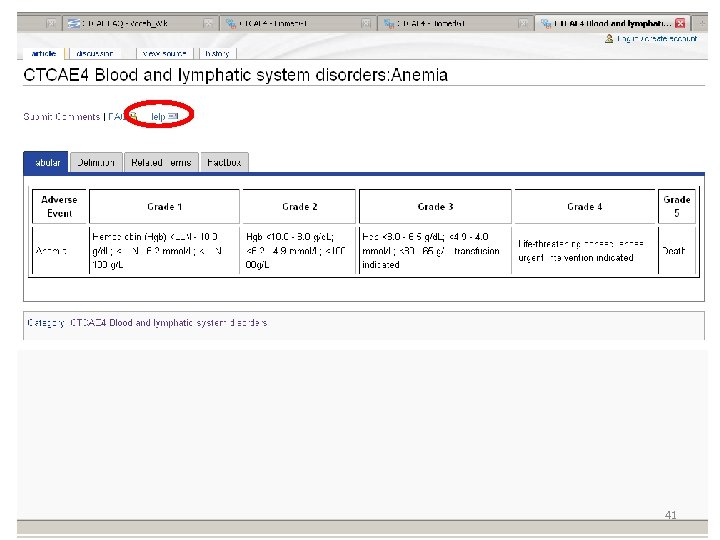
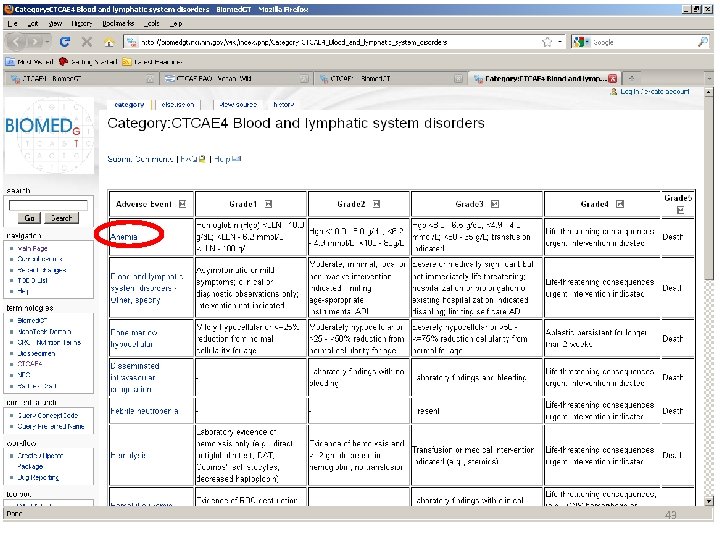
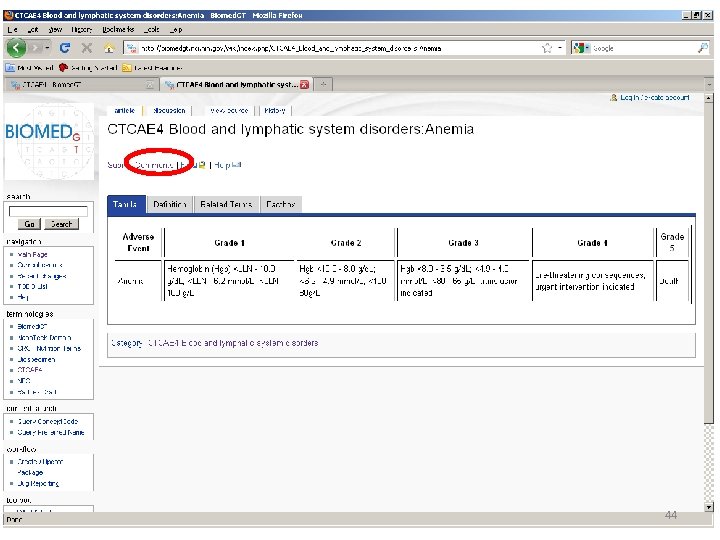
Location of AE Terms: Comparison CTCAE v 3. 0 AE Term BLOOD/BONE MARROW CATEGORY CTCAE v 4. 0 SOC CTCAE v 4. 0 AE Term Hemoglobin Blood and lymphatic system disorders Anemia Platelets Investigations Platelet count decreased Leukocytes (total WBC) Neutrophils/granulocytes (ANC/AGC) Hemolysis (e. g. , immune hemolytic anemia, drugrelated hemolysis) CD 4 count Investigations White blood cell decreased Investigations Neutrophil count decreased Bone marrow cellularity Myelodysplasia Splenic function Blood and lymphatic system disorders Investigations Blood and lymphatic system disorders Hemolysis CD 4 lymphocytes decreased Bone marrow hypocellular Neoplasms benign, malignant and unspecified (incl cysts and polyps) Myelodysplastic syndrome Blood and lymphatic system disorders Spleen disorder

CTCAE v 4. 0: Highlights of What Else is New • AE term definitions • Initial definitions for the CTCAE terms were provided by the NCI’s Enterprise Vocabulary Services (EVS) • ADLs • Grade 2: limiting instrumental ADL • e. g. , shopping; laundry; transportation; ability to conduct finances • Grade 3: limiting self care ADL • e. g. , getting in and out of bed; dressing; eating; getting around inside; bathing; using the toilet • General Guidelines for grade descriptions

CTCAE v 4. 0 General Guidelines for Grade Descriptions… Grade 1 Mild AE - minor; - no specific medical intervention - asymptomatic laboratory finding only; - radiographic finding only; - marginal clinical relevance; - mild symptoms and intervention not indicated; - non-prescription intervention indicated Grade 2 Moderate AE - intervention indicated; - minimal, local, noninvasive intervention (e. g. packing, cautery); - limiting instrumental ADL (e. g. , shopping; laundry; transportation; ability to conduct finances) General Guidelines for Grades are Foundation for AE-Specific Severity Scales

…CTCAE v 4. 0 General Guidelines for Grade Descriptions Grade 3 Severe AE - medically significant but not life-threatening; - inpatient or prolongation of hospitalization indicated; - important medical events that do not result in hospitalization 1 but may jeopardize the patient or may require intervention either to prevent hospitalization or to prevent the AE from becoming life-threatening or potentially resulting in death; - limiting self care ADL (e. g. , getting in and out of bed; dressing; eating; getting around inside; bathing; using the toilet) - disabling 2; - results in persistent or significant disability or incapacity 1 Exception: Grade 4 Grade 5 Life-threatening AE - life-threatening consequences - urgent intervention indicated - urgent operative intervention indicated - patient is at risk of death at the time of the event if immediate intervention is not undertaken SOC Psychiatric disorders Gr 4 = hospitalization 2 Exception: Blindness Gr 4 & Hearing impaired Gr 4 = disabling Death
CTCAE v 4. 0 Resources http: //evs. nci. nih. gov/ftp 1/CTCAE 35

CTCAE v 4. 0 Resources Bio. Med. GT Wiki http: //biomedgt. nci. nih. gov/wiki/index. php/CTCAE 4
37
38
39
40
41
42
43
44
45
46

Med. DRA Versioning • CTCAE will continue to be harmonized with Med. DRA • Med. DRA versions: • March: major release • September: minor release • Estimated that a new version of CTCAE will occur every 2 years in March • CTCAE/Med. DRA LLTs will not be deleted • Governance/oversight http: //ctep. info. nih. gov/protocol. Developme nt/electronic_applications/ctc. htm 47

Questions Liz Ness nesse@mail. nih. gov 301 -451 -2179
17c50992885d60a754d4b3cf549d6ebd.ppt