6d51df33b50f6ec880a6d59f7b869577.ppt
- Количество слайдов: 88
 Advances in Maternal Fetal Medicine Mahmoud A. Ismail, MD Professor and Chief Maternal Fetal Medicine University of Chicago Medical Center
Advances in Maternal Fetal Medicine Mahmoud A. Ismail, MD Professor and Chief Maternal Fetal Medicine University of Chicago Medical Center
 Recurrent Early Pregnancy Loss When, Why and How to Evaluate Recurrent Early Pregnancy Loss?
Recurrent Early Pregnancy Loss When, Why and How to Evaluate Recurrent Early Pregnancy Loss?
 Evaluation of Recurrent Early Pregnancy Loss Stephenson, 2008
Evaluation of Recurrent Early Pregnancy Loss Stephenson, 2008
 Stephenson, 2008
Stephenson, 2008
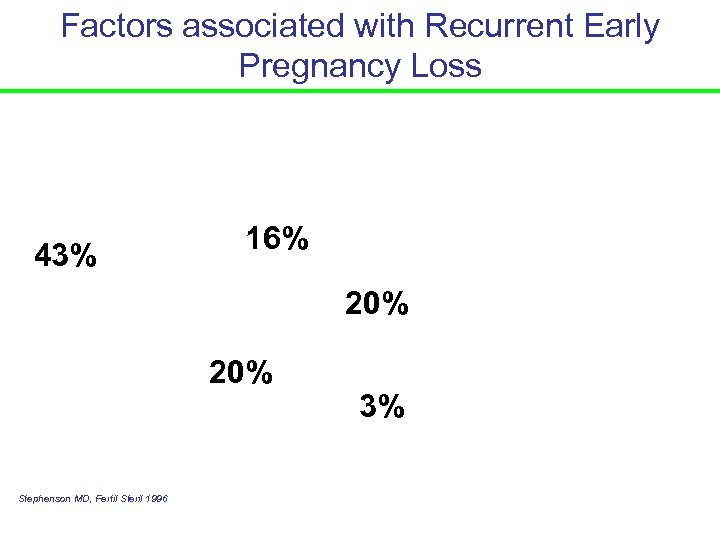 Factors associated with Recurrent Early Pregnancy Loss 43% 16% 20% Stephenson MD, Fertil Steril 1996 3%
Factors associated with Recurrent Early Pregnancy Loss 43% 16% 20% Stephenson MD, Fertil Steril 1996 3%
 APS: Sapporo Criteria (1998) Clinical: • Arterial or venous thrombosis • 3 unexplained miscarriages <10 wks • Unexplained fetal death 10 wks • Premature (<34 wks) birth: severe pre-eclampsia or placental insufficiency Laboratory (at least twice, 6 wks apart): • ACA Ig. G and/or Ig. M: Medium or high titre using a standardized 2 GP 1 -dependent ELISA • Lupus Anticoagulant: International Society of Thrombosis & Hemostasis Guidelines Wilson et al, Arthritis Rheum 1999
APS: Sapporo Criteria (1998) Clinical: • Arterial or venous thrombosis • 3 unexplained miscarriages <10 wks • Unexplained fetal death 10 wks • Premature (<34 wks) birth: severe pre-eclampsia or placental insufficiency Laboratory (at least twice, 6 wks apart): • ACA Ig. G and/or Ig. M: Medium or high titre using a standardized 2 GP 1 -dependent ELISA • Lupus Anticoagulant: International Society of Thrombosis & Hemostasis Guidelines Wilson et al, Arthritis Rheum 1999
 APS: Sydney Criteria (2004) Clinical: Unchanged Laboratory (at least twice, 12 wks apart): • ACA Ig. G and/or Ig. M: Medium or high titre of ≥ 40 GPL or MPL • Anti β 2 glycoprotein-1 Ig. G and/or Ig. M: titer >99 th percentile • Lupus Anticoagulant: International Society on Thrombosis & Hemostasis Guidelines Miyakis et al, J Thromb Haemost 2006
APS: Sydney Criteria (2004) Clinical: Unchanged Laboratory (at least twice, 12 wks apart): • ACA Ig. G and/or Ig. M: Medium or high titre of ≥ 40 GPL or MPL • Anti β 2 glycoprotein-1 Ig. G and/or Ig. M: titer >99 th percentile • Lupus Anticoagulant: International Society on Thrombosis & Hemostasis Guidelines Miyakis et al, J Thromb Haemost 2006
 Treatment of APS: Live Birth Rates • • • No treatment: 10% Level II-3 Rai et al, 1995 Low dose ASA: 42 -44% Level II-1 Kutteh 1996; RCT Rai et al, 1997 ASA and prednisone: ↑ maternal / fetal morbidity RCT Cowchock et al, 1992; RCT Laskin et al, 1997 ASA and unfractionated heparin: 72 -80% Level II-1 Kutteh 1996; RCT Rai et al, 1997 ASA and low molecular weight heparin: 69 -78% RCT Farquharson et al, 2002; RCT Stephenson et al, 2004 Intravenous immunoglobulin: 57% RCT Triolo et al, 2003
Treatment of APS: Live Birth Rates • • • No treatment: 10% Level II-3 Rai et al, 1995 Low dose ASA: 42 -44% Level II-1 Kutteh 1996; RCT Rai et al, 1997 ASA and prednisone: ↑ maternal / fetal morbidity RCT Cowchock et al, 1992; RCT Laskin et al, 1997 ASA and unfractionated heparin: 72 -80% Level II-1 Kutteh 1996; RCT Rai et al, 1997 ASA and low molecular weight heparin: 69 -78% RCT Farquharson et al, 2002; RCT Stephenson et al, 2004 Intravenous immunoglobulin: 57% RCT Triolo et al, 2003
 In Utero Fetal Therapy Twin Transfussion
In Utero Fetal Therapy Twin Transfussion
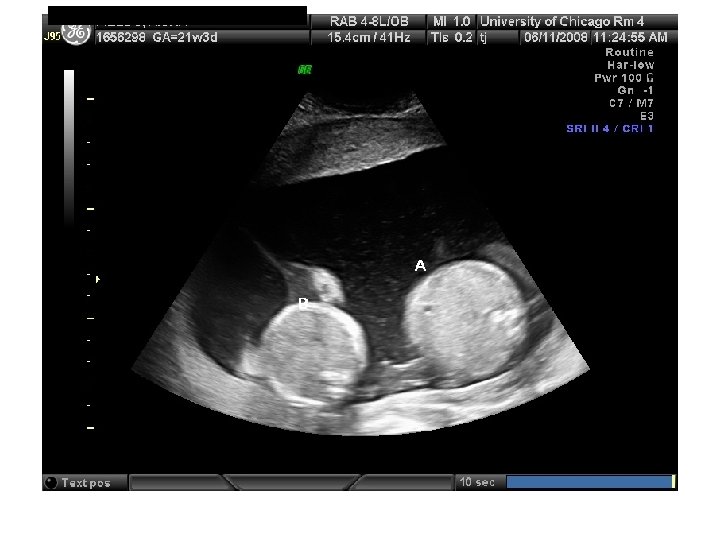
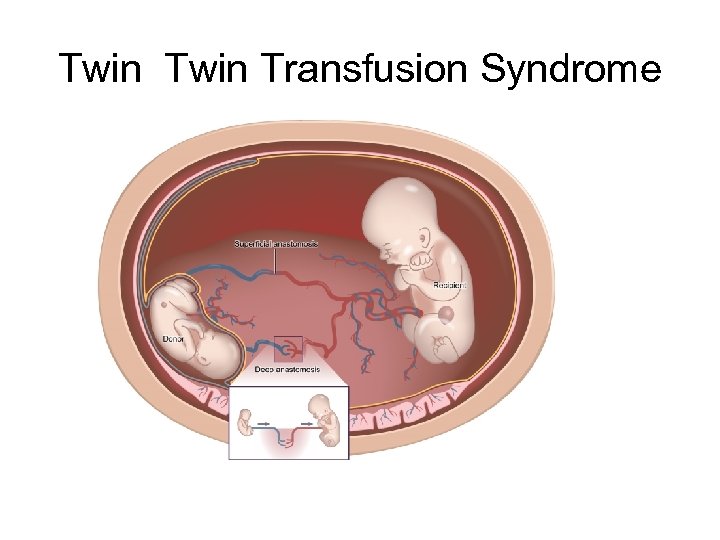 Twin Transfusion Syndrome
Twin Transfusion Syndrome
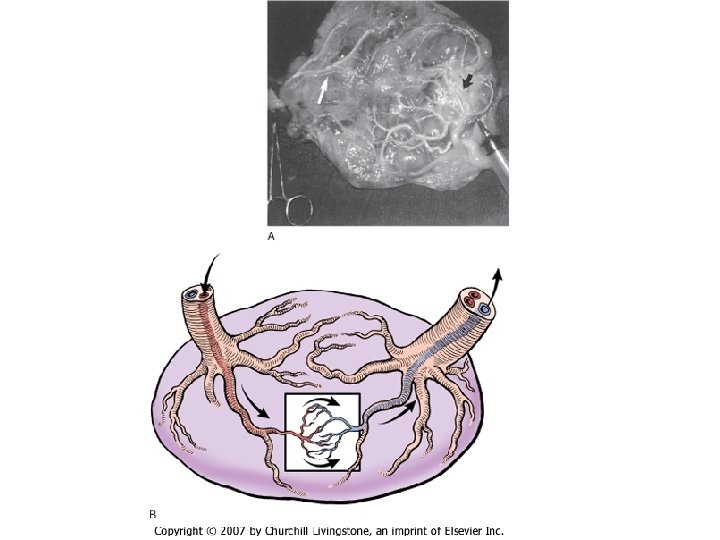
 Requirements for diagnosis of TTTS 1. 2. 3. 4. the presence of a single placenta same-gender fetuses significant weight discordance significant amniotic fluid discordance often with a "stuck twin. "
Requirements for diagnosis of TTTS 1. 2. 3. 4. the presence of a single placenta same-gender fetuses significant weight discordance significant amniotic fluid discordance often with a "stuck twin. "
 Quintero Staging (TTTS) • Stage-based treatment of TTTS (Quintero, et al. AJOG May 2003) • I Donor’s bladder visible; dopplers normal II Donor’s bladder not visible x 1 hr; dopplers normal (ish) III Doppler’s critically abnormal in either (i. e. AEDF, RFDV, PUVF) IV Ascites, pericardial/pleural effusion, scalp edema, or overt hydrops V One/both twins dead
Quintero Staging (TTTS) • Stage-based treatment of TTTS (Quintero, et al. AJOG May 2003) • I Donor’s bladder visible; dopplers normal II Donor’s bladder not visible x 1 hr; dopplers normal (ish) III Doppler’s critically abnormal in either (i. e. AEDF, RFDV, PUVF) IV Ascites, pericardial/pleural effusion, scalp edema, or overt hydrops V One/both twins dead
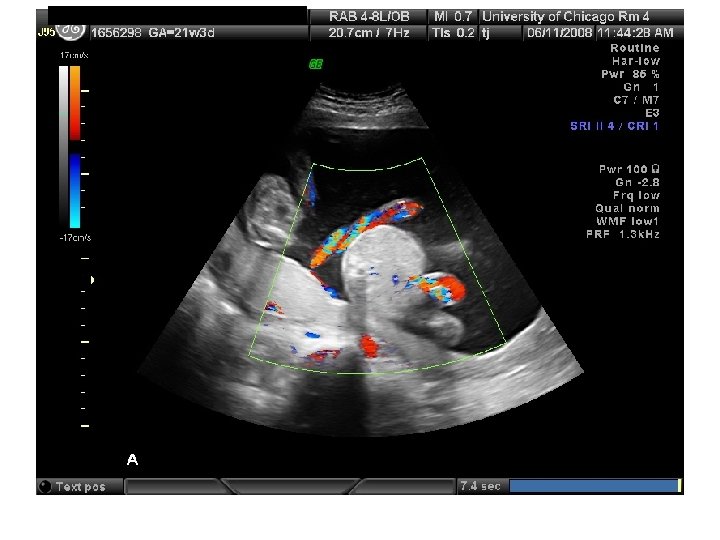
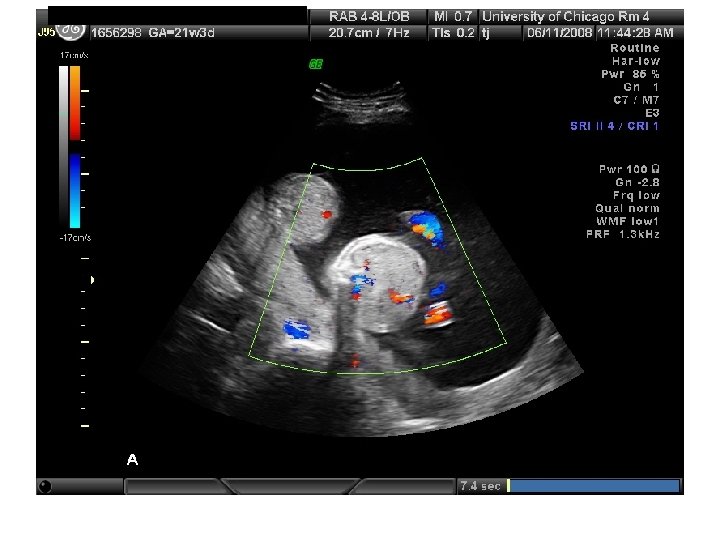
 Management of TTTS • Amnioreduction • Septostomy • Photocoagulation
Management of TTTS • Amnioreduction • Septostomy • Photocoagulation
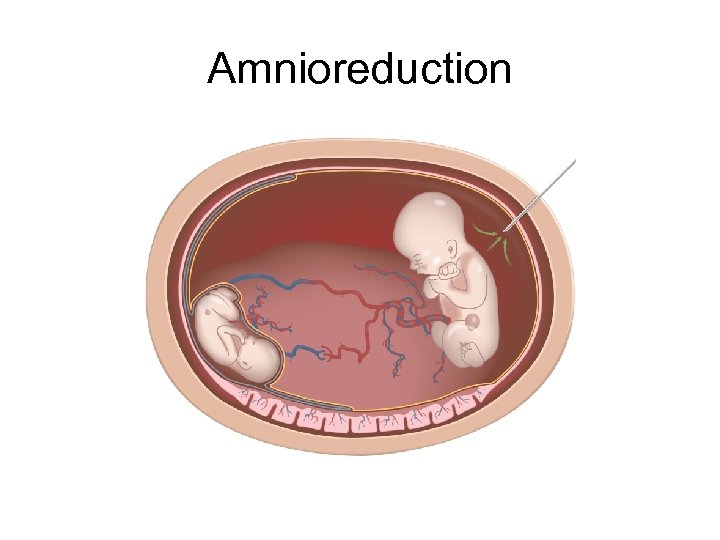 Amnioreduction
Amnioreduction
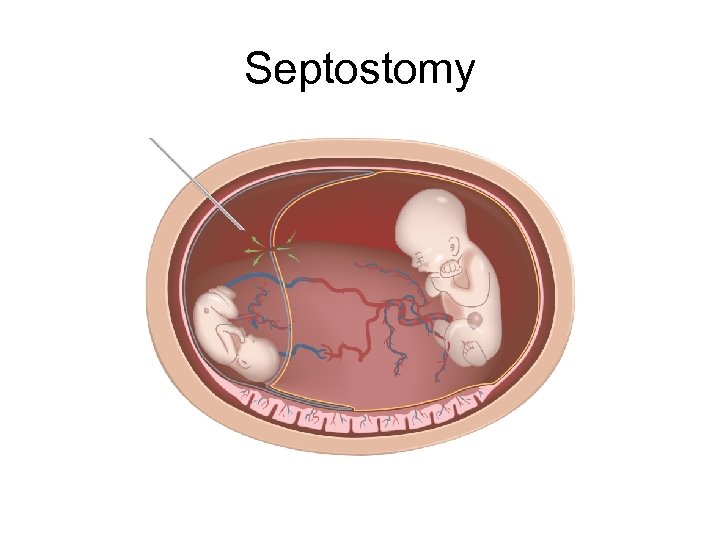 Septostomy
Septostomy
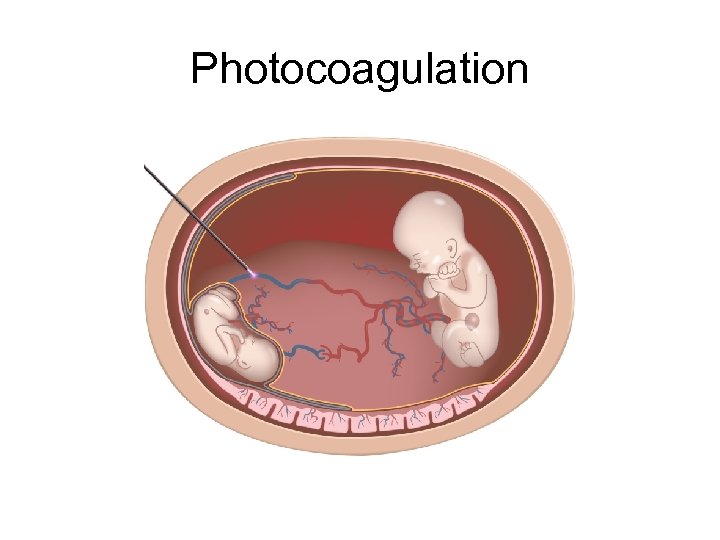 Photocoagulation
Photocoagulation
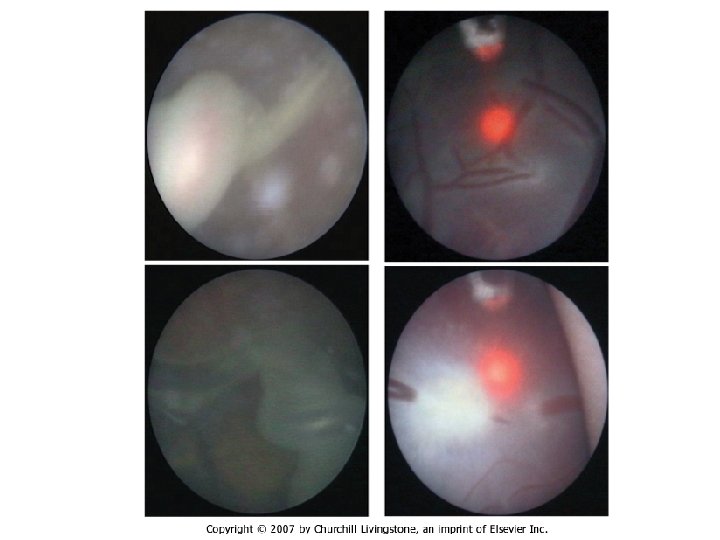
 In Utero Fetal Therapy Meningomyelocele
In Utero Fetal Therapy Meningomyelocele
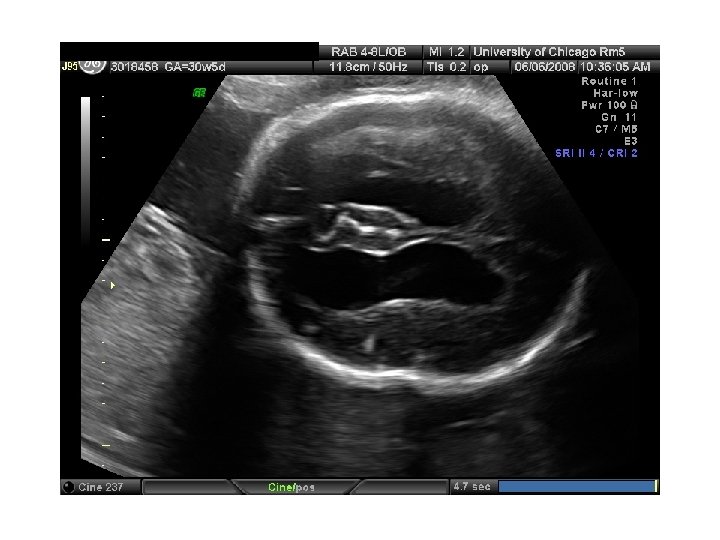
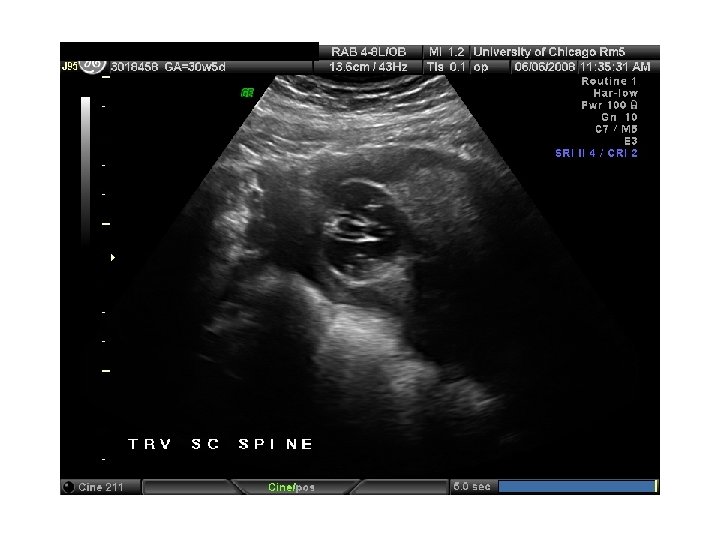
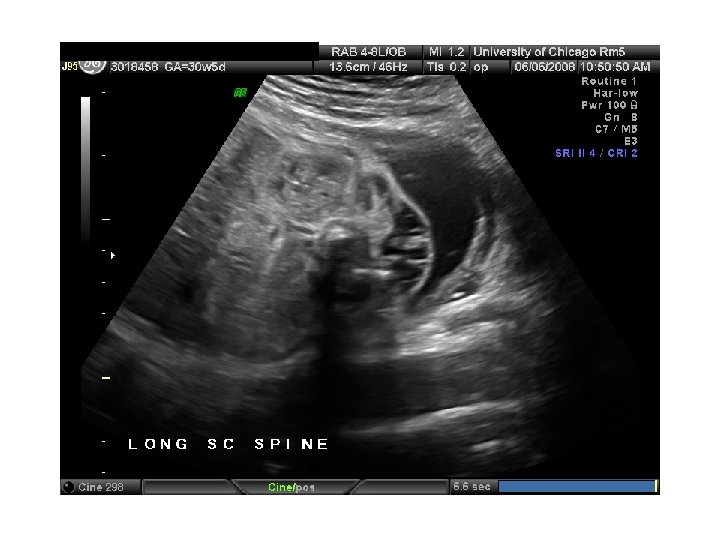
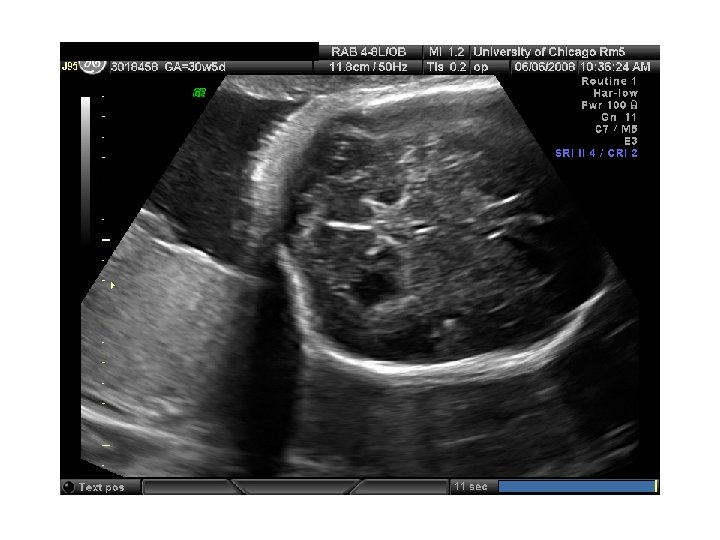
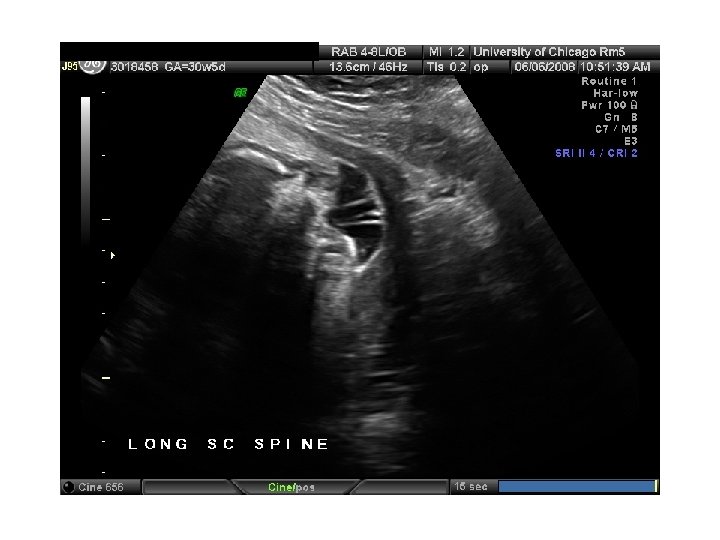
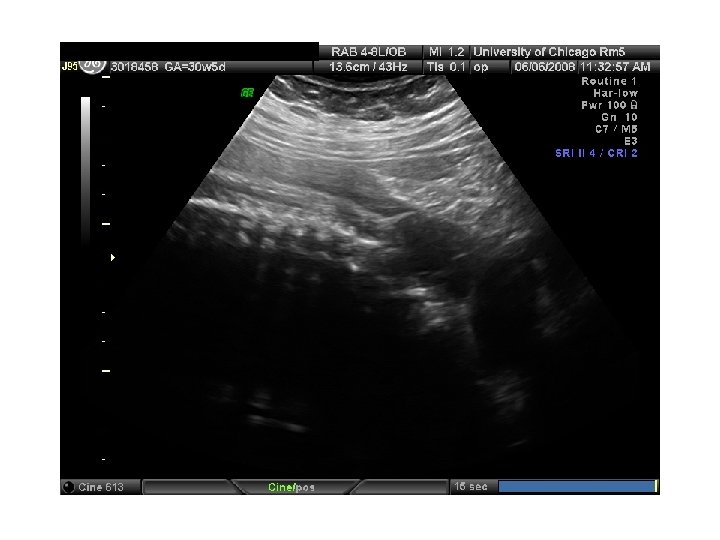
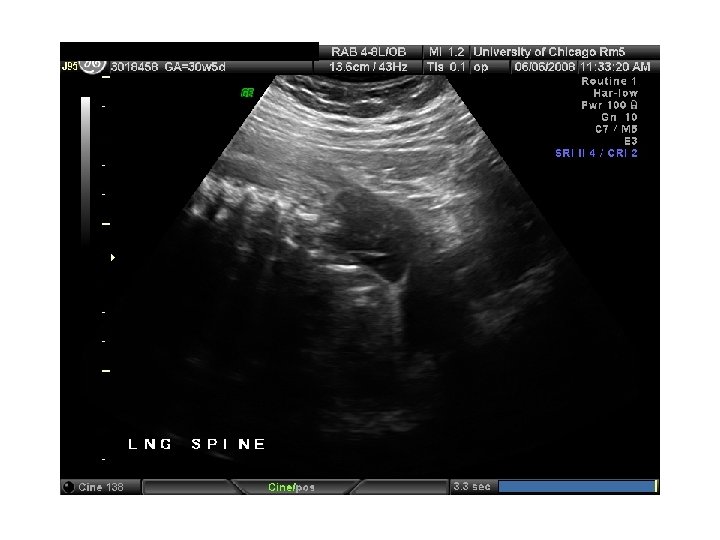
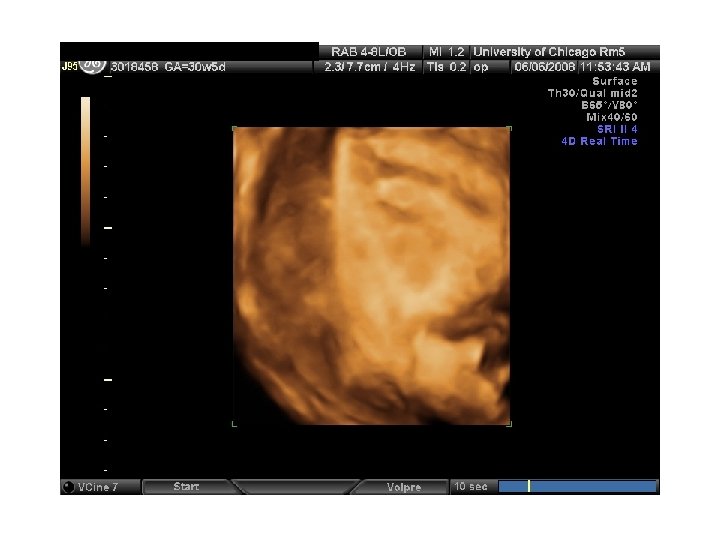
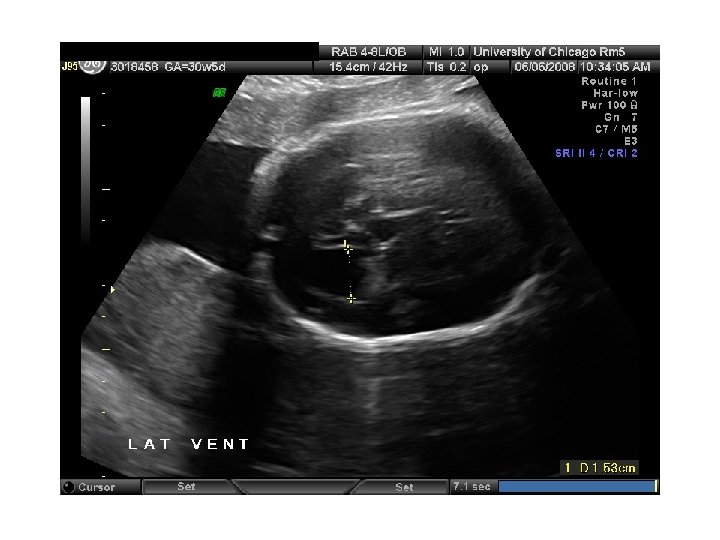
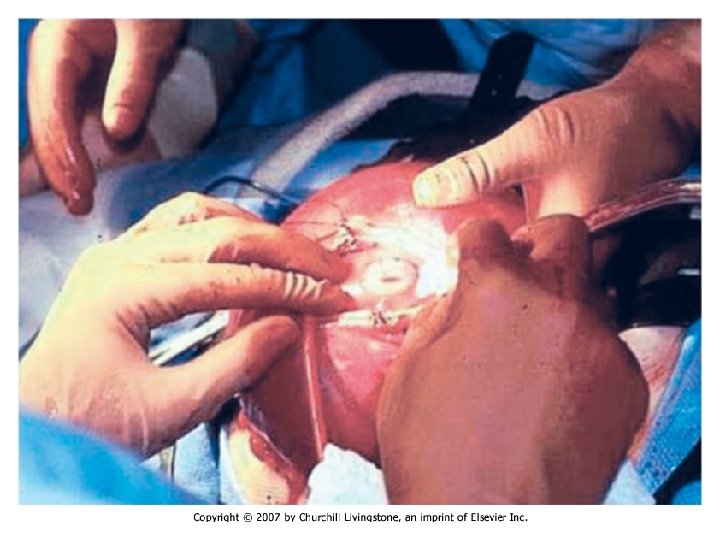
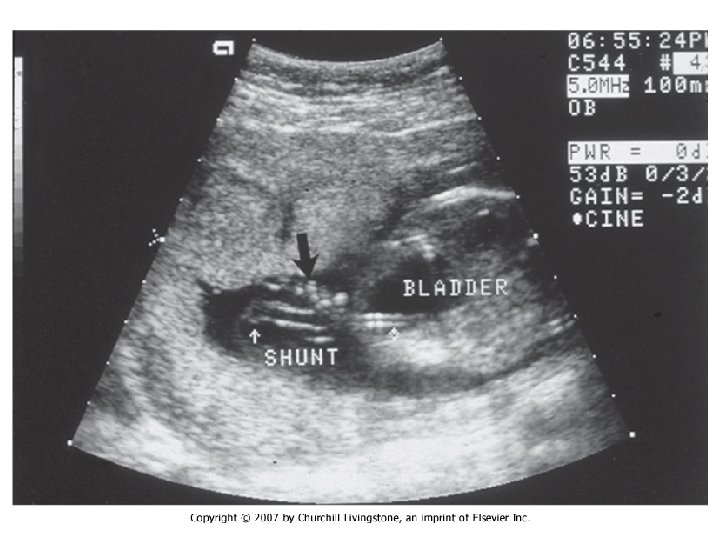
 In Utero Fetal Therapy Bladder Outlet Obstruction
In Utero Fetal Therapy Bladder Outlet Obstruction
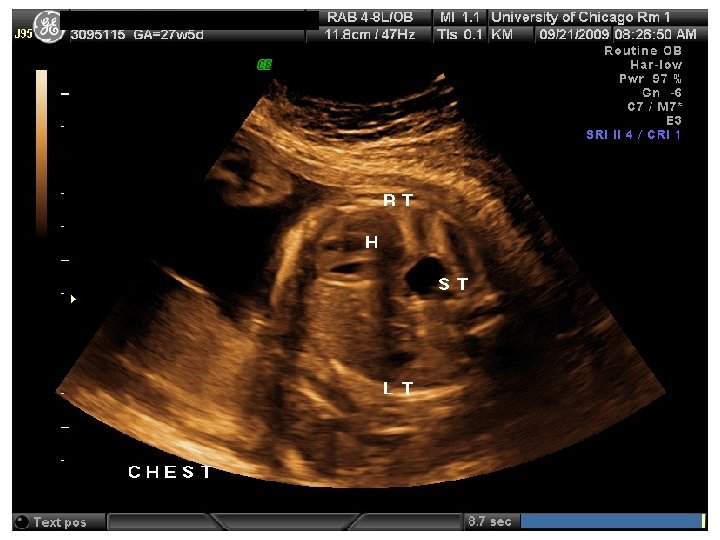
 In Utero Fetal Therapy Congenital Diaphragmatic Hernia
In Utero Fetal Therapy Congenital Diaphragmatic Hernia
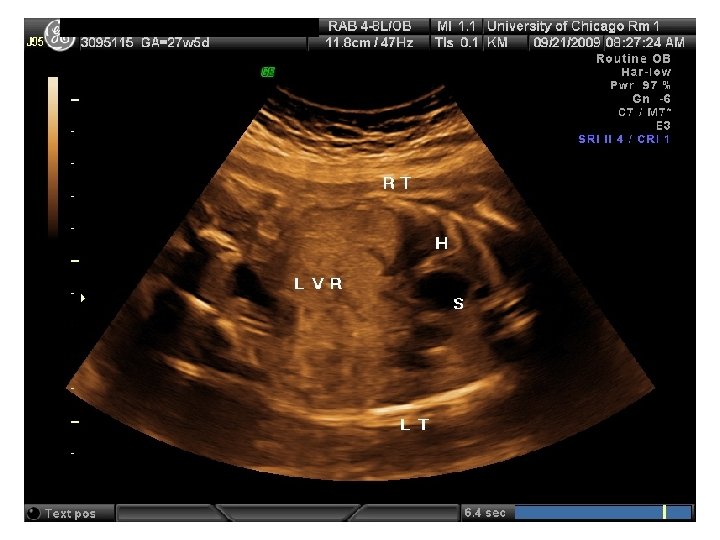
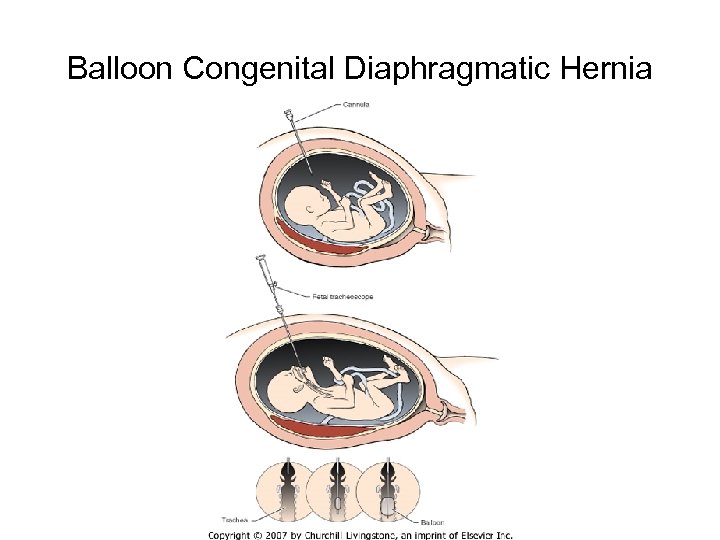 Balloon Congenital Diaphragmatic Hernia
Balloon Congenital Diaphragmatic Hernia
 Exit Procedure
Exit Procedure
 Abdominal Wall Defects
Abdominal Wall Defects
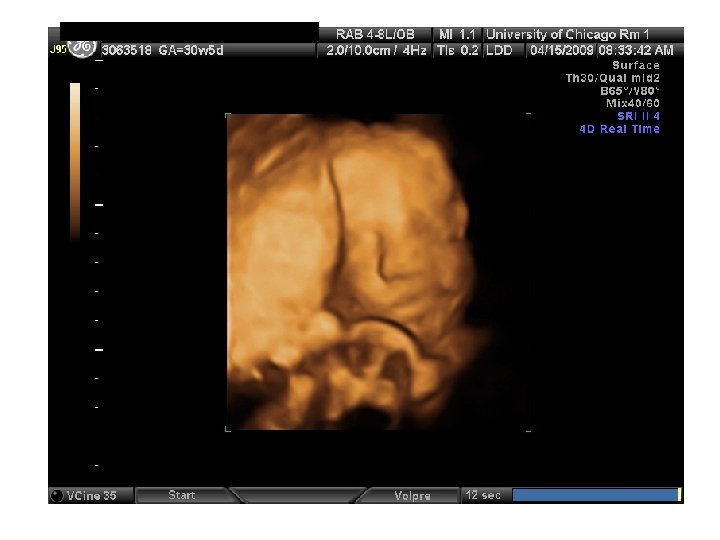
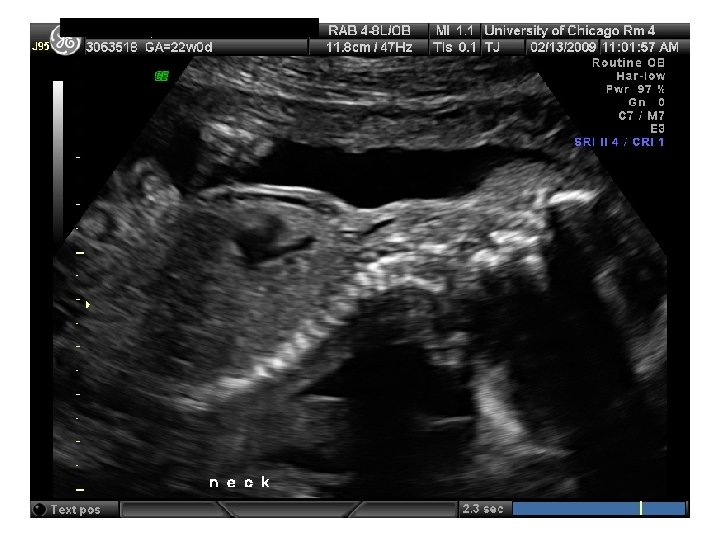
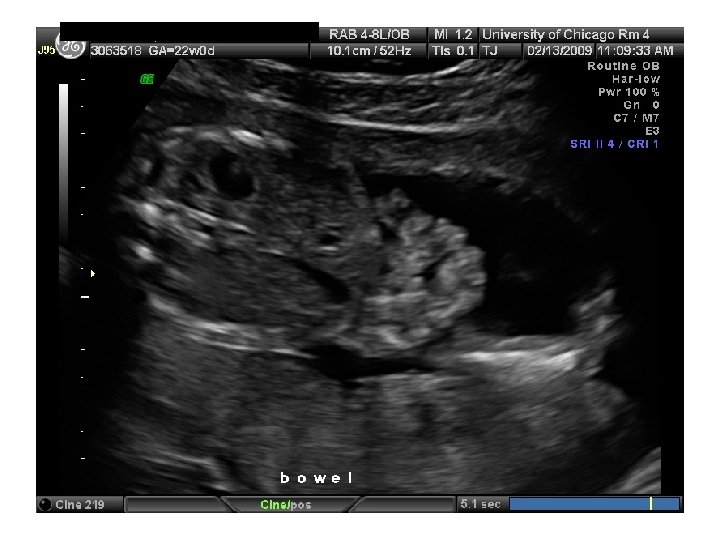
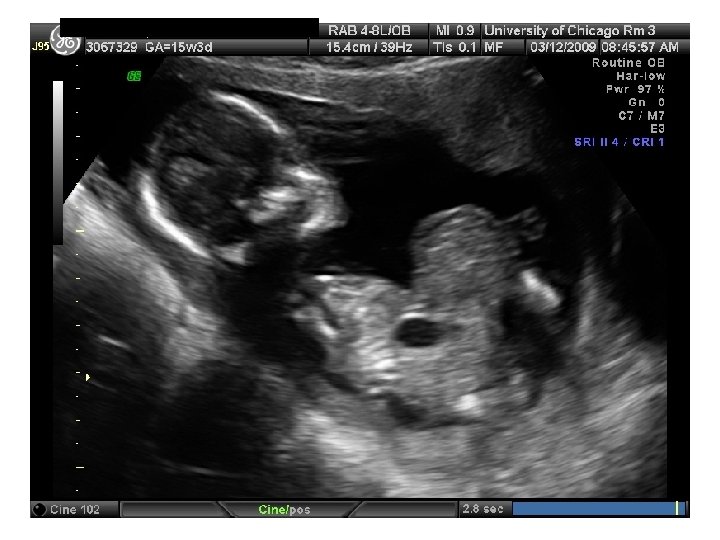
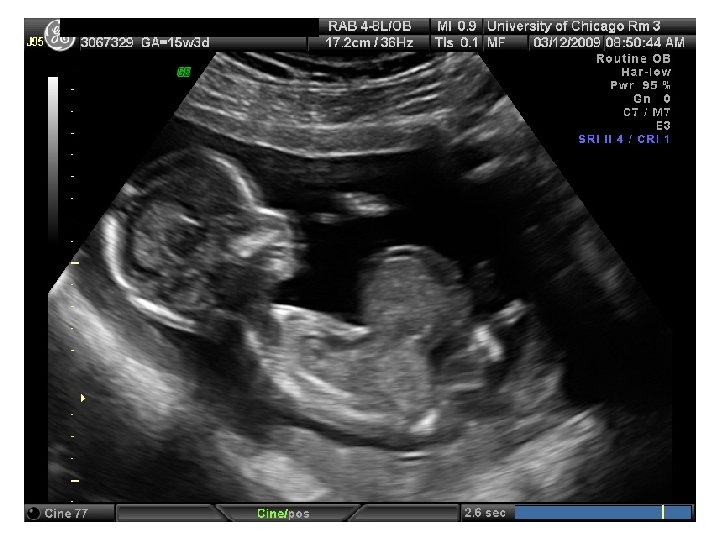
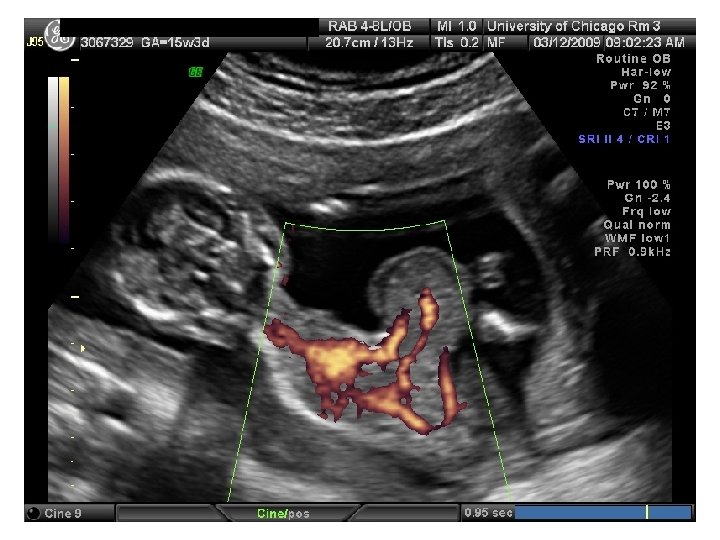
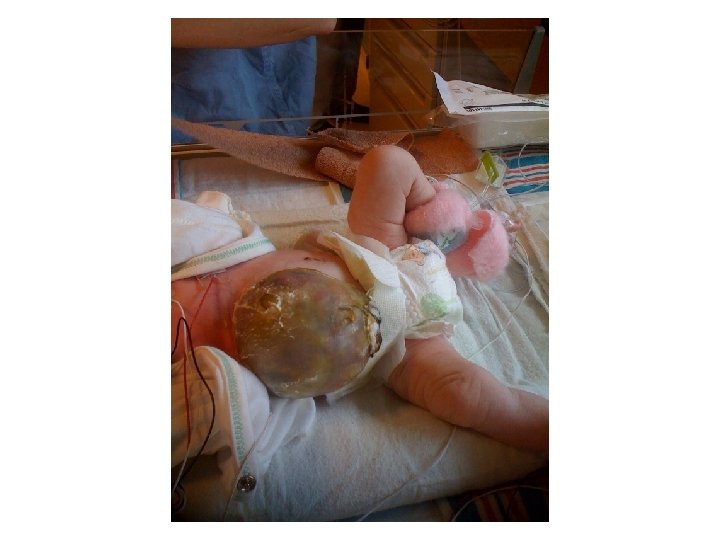
 SR – Fetal Complete Heart Block 31 yo G 6 P 3114 EDC 10/20/09, now 33 wks, presented to HROB clinic at 10 wks for prenatal care. Fetal complete heart block was diagnosed at the routine 2 nd trimester anatomy scan. Pt is referred to The Fetal Center for multidisciplinary planning. PNC: 1. Systemic Lupus Erythematosus 2. Fetal Heart Block 3. H/O preeclampsia at 35 wks 4. Anemia – Presumed iron deficiency 5. Chronic HTN – No meds
SR – Fetal Complete Heart Block 31 yo G 6 P 3114 EDC 10/20/09, now 33 wks, presented to HROB clinic at 10 wks for prenatal care. Fetal complete heart block was diagnosed at the routine 2 nd trimester anatomy scan. Pt is referred to The Fetal Center for multidisciplinary planning. PNC: 1. Systemic Lupus Erythematosus 2. Fetal Heart Block 3. H/O preeclampsia at 35 wks 4. Anemia – Presumed iron deficiency 5. Chronic HTN – No meds
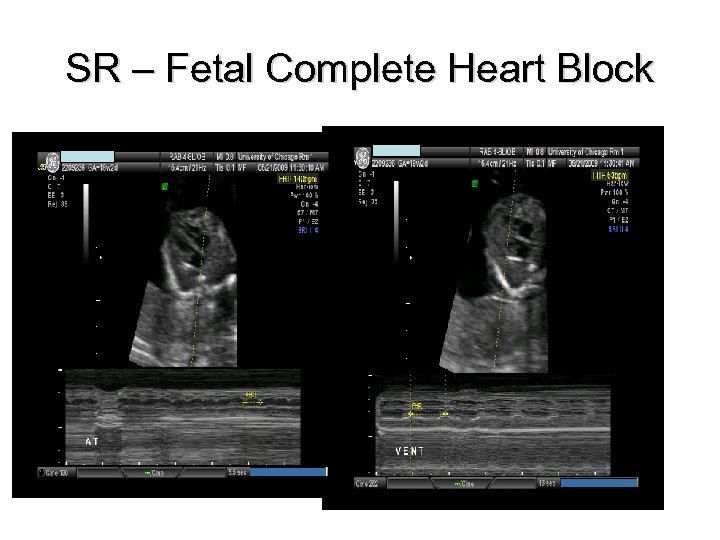 SR – Fetal Complete Heart Block
SR – Fetal Complete Heart Block
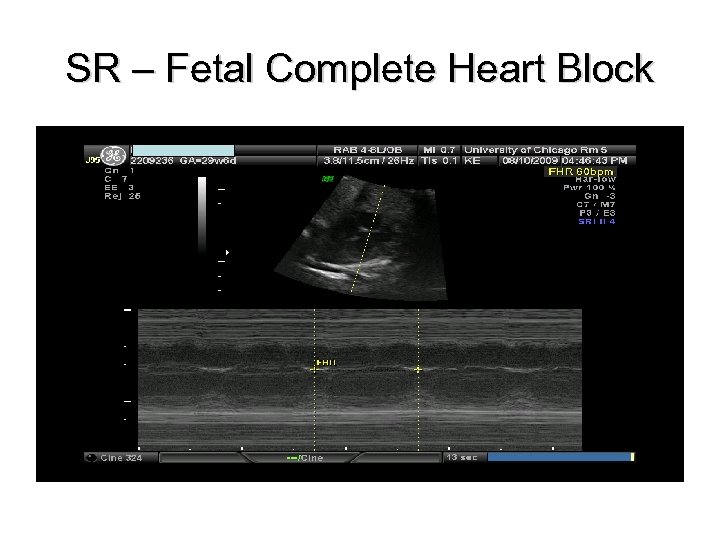 SR – Fetal Complete Heart Block
SR – Fetal Complete Heart Block
 SR – Fetal Complete Heart Block • Antenatal Plan – Weekly BPP with hydrops check • Delivery Plan – Primary Cesarean at 38 -39 wks with desired tubal ligation
SR – Fetal Complete Heart Block • Antenatal Plan – Weekly BPP with hydrops check • Delivery Plan – Primary Cesarean at 38 -39 wks with desired tubal ligation
 Aneuploidy Screening Tests
Aneuploidy Screening Tests
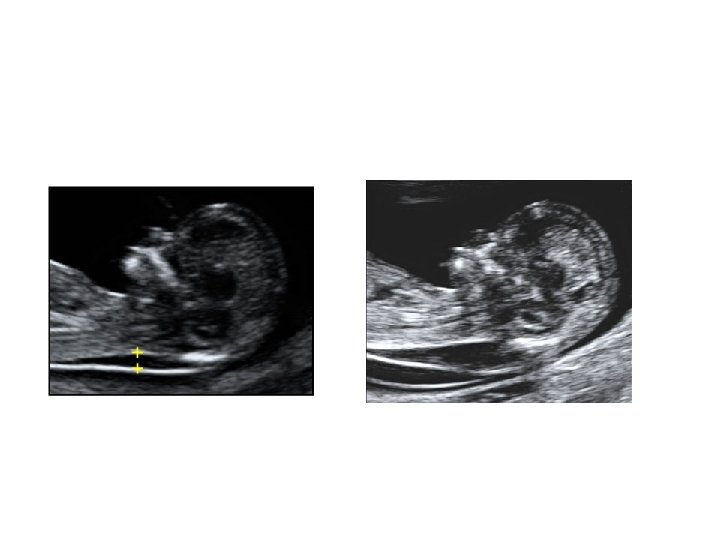
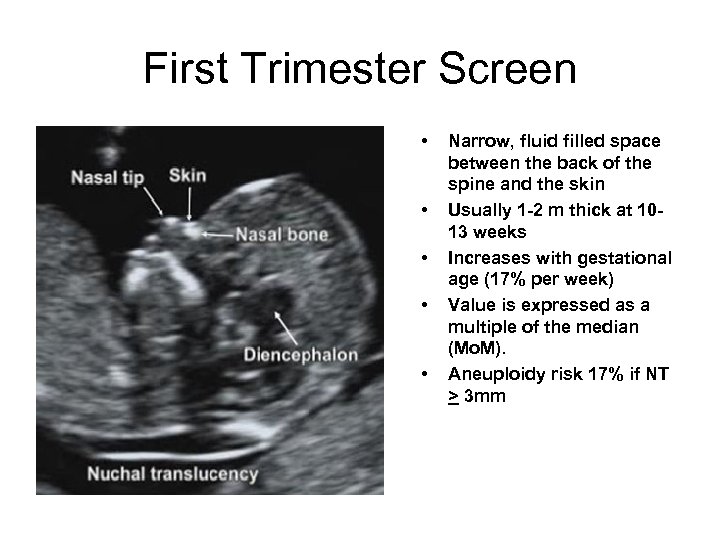 First Trimester Screen • • • Narrow, fluid filled space between the back of the spine and the skin Usually 1 -2 m thick at 1013 weeks Increases with gestational age (17% per week) Value is expressed as a multiple of the median (Mo. M). Aneuploidy risk 17% if NT > 3 mm
First Trimester Screen • • • Narrow, fluid filled space between the back of the spine and the skin Usually 1 -2 m thick at 1013 weeks Increases with gestational age (17% per week) Value is expressed as a multiple of the median (Mo. M). Aneuploidy risk 17% if NT > 3 mm
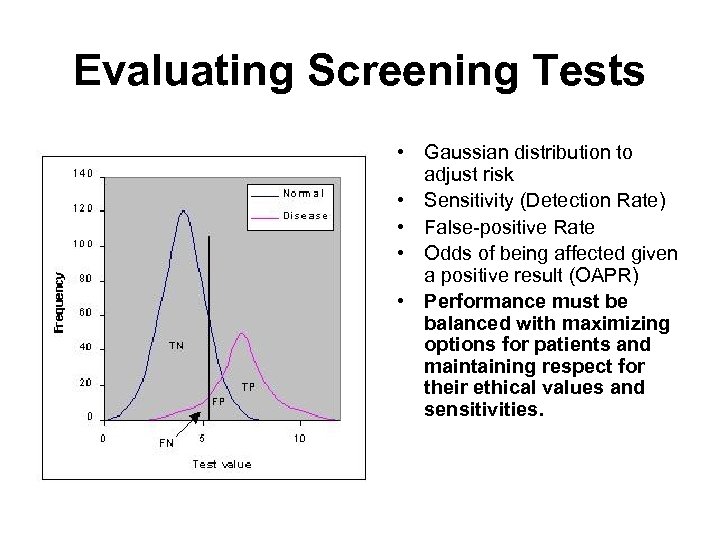 Evaluating Screening Tests • Gaussian distribution to adjust risk • Sensitivity (Detection Rate) • False-positive Rate • Odds of being affected given a positive result (OAPR) • Performance must be balanced with maximizing options for patients and maintaining respect for their ethical values and sensitivities.
Evaluating Screening Tests • Gaussian distribution to adjust risk • Sensitivity (Detection Rate) • False-positive Rate • Odds of being affected given a positive result (OAPR) • Performance must be balanced with maximizing options for patients and maintaining respect for their ethical values and sensitivities.
 Nuchal translucency Combined results of 11 prospective screening studies, 1014 weeks gestation, FMF software, mean maternal age 31: 85% detection rate for trisomy 21 82% for other chromosomal abnormalities 8% screen positive
Nuchal translucency Combined results of 11 prospective screening studies, 1014 weeks gestation, FMF software, mean maternal age 31: 85% detection rate for trisomy 21 82% for other chromosomal abnormalities 8% screen positive
 First Trimester Screen • Offers earlier diagnosis (11 -13 weeks) • Serum markers PAPP-A – 60% lower in DS free ß-h. CG – 80% higher in DS total h. CG -- ok in combination with NT and PAPP-A • Ultrasound nuchal translucency
First Trimester Screen • Offers earlier diagnosis (11 -13 weeks) • Serum markers PAPP-A – 60% lower in DS free ß-h. CG – 80% higher in DS total h. CG -- ok in combination with NT and PAPP-A • Ultrasound nuchal translucency
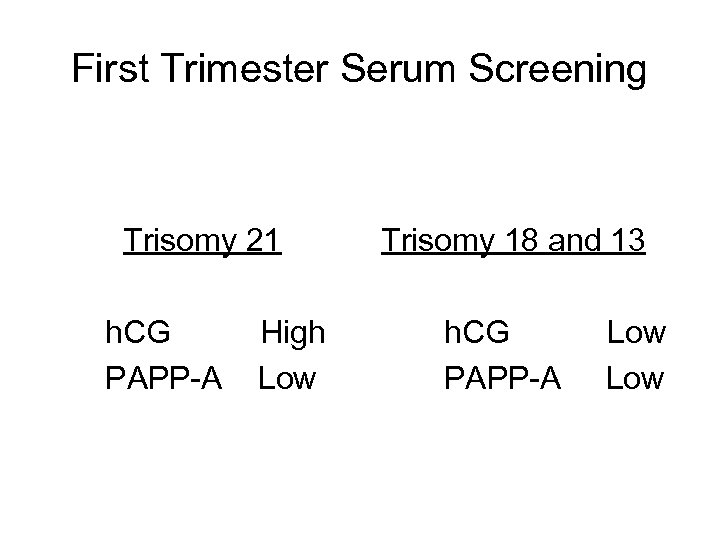 First Trimester Serum Screening Trisomy 21 h. CG PAPP-A High Low Trisomy 18 and 13 h. CG PAPP-A Low
First Trimester Serum Screening Trisomy 21 h. CG PAPP-A High Low Trisomy 18 and 13 h. CG PAPP-A Low
 NT and FIRST TRIMESTER SERUM SCREEN Five prospective first trimester studies, 1013 weeks gestation: 87% detection rate for trisomy 21(5% false positive rate) 90% detection of other chromosome abnormalities (1% false positive rate)
NT and FIRST TRIMESTER SERUM SCREEN Five prospective first trimester studies, 1013 weeks gestation: 87% detection rate for trisomy 21(5% false positive rate) 90% detection of other chromosome abnormalities (1% false positive rate)
 SCREENING OF MULTIPLE GESTATIONS FOR TRISOMY 21 • Maternal age: Detection same as singletons • Second-trimester serum: 45% detection in twins • NT: Detection same as singletons if dichorionic; false positive rate higher if monochorionic • NT+serum: 75% detection in twins with 9% false positives • Higher-order pregnancies: Only maternal age and NT are reliable
SCREENING OF MULTIPLE GESTATIONS FOR TRISOMY 21 • Maternal age: Detection same as singletons • Second-trimester serum: 45% detection in twins • NT: Detection same as singletons if dichorionic; false positive rate higher if monochorionic • NT+serum: 75% detection in twins with 9% false positives • Higher-order pregnancies: Only maternal age and NT are reliable
 Thrombophilias in Pregnancy
Thrombophilias in Pregnancy
 The inherited thrombophilias • • • Factor V Leiden mutation Prothrombin gene mutation Antithrombin (III) deficiency Protein C and Protein S deficiencies Hyperhomocysteinemia (cystathione β-synthase or MTHFR)
The inherited thrombophilias • • • Factor V Leiden mutation Prothrombin gene mutation Antithrombin (III) deficiency Protein C and Protein S deficiencies Hyperhomocysteinemia (cystathione β-synthase or MTHFR)
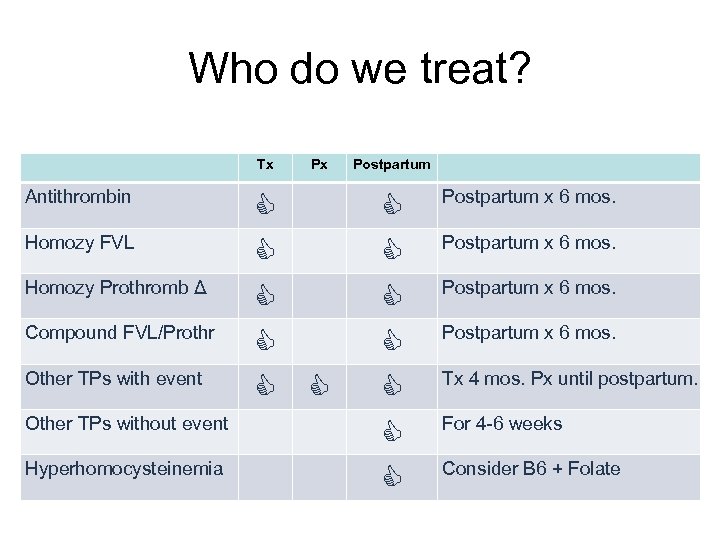 Who do we treat? Tx Antithrombin Homozy FVL Homozy Prothromb Δ Compound FVL/Prothr Other TPs with event Other TPs without event Hyperhomocysteinemia Px Postpartum x 6 mos. Tx 4 mos. Px until postpartum. For 4 -6 weeks Consider B 6 + Folate
Who do we treat? Tx Antithrombin Homozy FVL Homozy Prothromb Δ Compound FVL/Prothr Other TPs with event Other TPs without event Hyperhomocysteinemia Px Postpartum x 6 mos. Tx 4 mos. Px until postpartum. For 4 -6 weeks Consider B 6 + Folate
 How should we treat? • Antepartum treatment LMWH at 1 mg/kg bid Check LMWH levels and titrate to therapeutic (0. 6 -1 U/m. L) Check platelets q. Wk x 3 wks, then q. Month • Antepartum prophylaxis LMWH at 40 mg daily Levels not necessary. Check platelets q. Wk x 3 wks, then q. Month • Postpartum treatment LMWH 4 -6 hours post NSVD or 6 -12 hours post C/S Start Coumadin concurrently. D/C LMWH when Coumadin therapeutic and continue x 4 -6 wks
How should we treat? • Antepartum treatment LMWH at 1 mg/kg bid Check LMWH levels and titrate to therapeutic (0. 6 -1 U/m. L) Check platelets q. Wk x 3 wks, then q. Month • Antepartum prophylaxis LMWH at 40 mg daily Levels not necessary. Check platelets q. Wk x 3 wks, then q. Month • Postpartum treatment LMWH 4 -6 hours post NSVD or 6 -12 hours post C/S Start Coumadin concurrently. D/C LMWH when Coumadin therapeutic and continue x 4 -6 wks
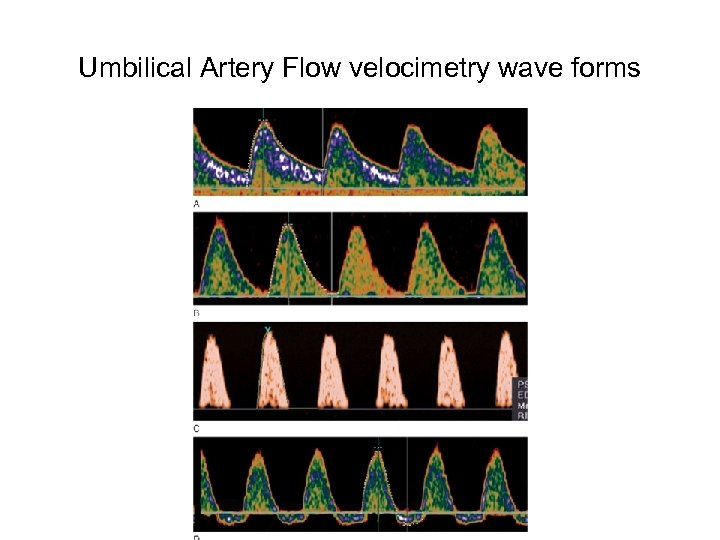 Umbilical Artery Flow velocimetry wave forms
Umbilical Artery Flow velocimetry wave forms
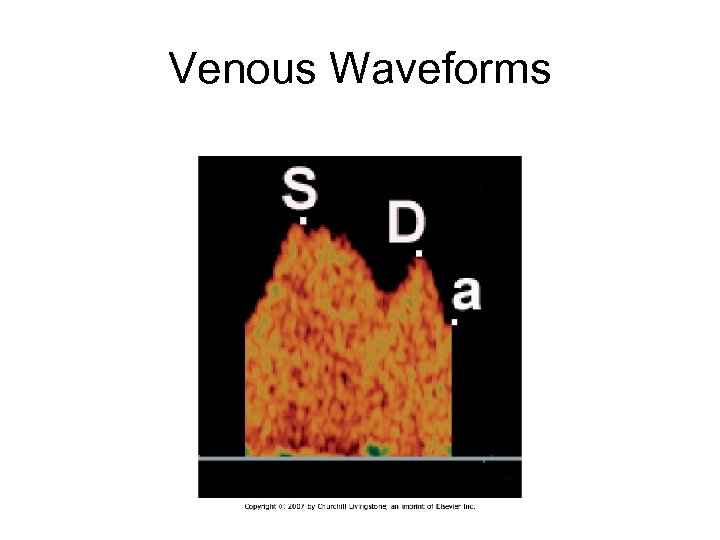 Venous Waveforms
Venous Waveforms
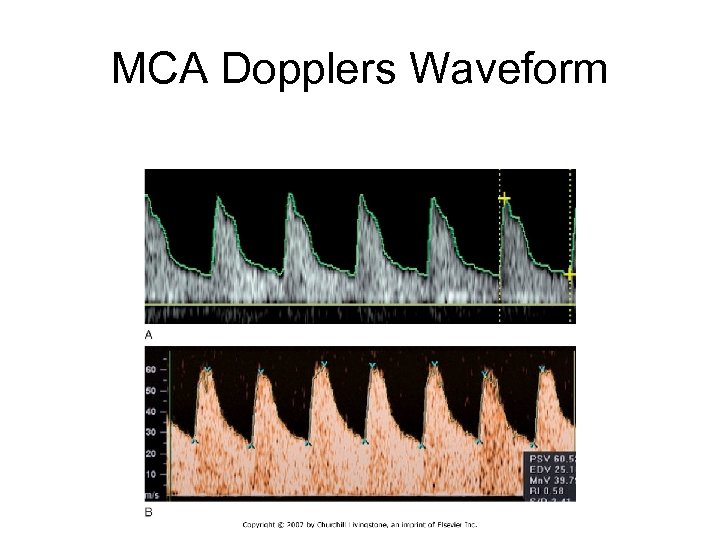 MCA Dopplers Waveform
MCA Dopplers Waveform
 IMPACT OF A ‘RESCUE COURSE’ OF ANTENATAL CORTICOSTEROIDS: A MULTICENTER RANDOMIZED PLACEBOCONTROLLED TRIAL THOMAS J. GARITE, MD; JAMES KURTZMAN, MD; KIMBERLY MAUREL, MSN; REESE CLARK, MD; FOR THE OBSTETRIX COLLABORATIVE RESEARCH NETWORK
IMPACT OF A ‘RESCUE COURSE’ OF ANTENATAL CORTICOSTEROIDS: A MULTICENTER RANDOMIZED PLACEBOCONTROLLED TRIAL THOMAS J. GARITE, MD; JAMES KURTZMAN, MD; KIMBERLY MAUREL, MSN; REESE CLARK, MD; FOR THE OBSTETRIX COLLABORATIVE RESEARCH NETWORK
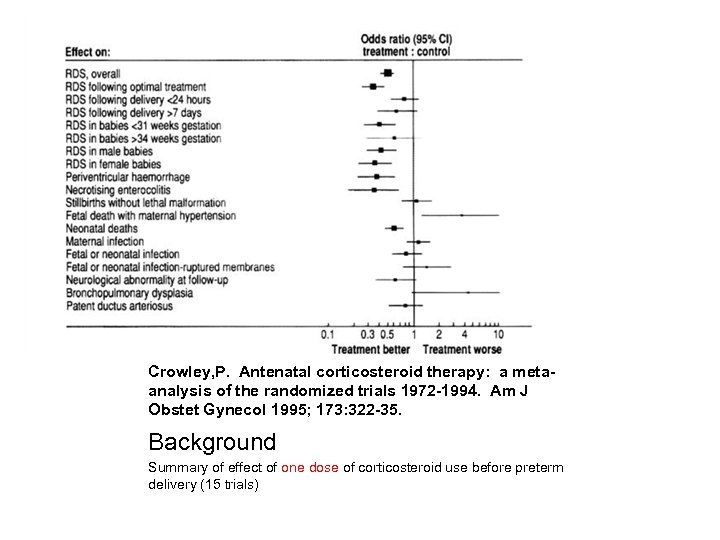 Crowley, P. Antenatal corticosteroid therapy: a metaanalysis of the randomized trials 1972 -1994. Am J Obstet Gynecol 1995; 173: 322 -35. Background Summary of effect of one dose of corticosteroid use before preterm delivery (15 trials)
Crowley, P. Antenatal corticosteroid therapy: a metaanalysis of the randomized trials 1972 -1994. Am J Obstet Gynecol 1995; 173: 322 -35. Background Summary of effect of one dose of corticosteroid use before preterm delivery (15 trials)
 Background • ACOG Committee Opinion, March 2008 – Single course of ACS between 24 and 34 weeks with a risk of preterm delivery in the next 7 days. – Single course of ACS before 32 weeks in PPROM. – “Rescue therapy” should not be routinely used due to insufficient scientific evidence.
Background • ACOG Committee Opinion, March 2008 – Single course of ACS between 24 and 34 weeks with a risk of preterm delivery in the next 7 days. – Single course of ACS before 32 weeks in PPROM. – “Rescue therapy” should not be routinely used due to insufficient scientific evidence.
 Hypothesis • Administration of a second rescue course of antenatal corticosteroids, compared with one course, would show a 40% reduction in the incidence of composite neonatal outcome in patients delivering before 34 weeks’ gestation.
Hypothesis • Administration of a second rescue course of antenatal corticosteroids, compared with one course, would show a 40% reduction in the incidence of composite neonatal outcome in patients delivering before 34 weeks’ gestation.
 Author’s Conclusions • A rescue course of ACS in pregnant women treated initially >2 weeks prior and who are judged by the clinician to be likely to deliver within the next week and before 34 weeks of gestation, is proven beneficial by significantly decreasing respiratory complications of prematurity and is without apparent immediate or short-term adverse effects to the mother or baby.
Author’s Conclusions • A rescue course of ACS in pregnant women treated initially >2 weeks prior and who are judged by the clinician to be likely to deliver within the next week and before 34 weeks of gestation, is proven beneficial by significantly decreasing respiratory complications of prematurity and is without apparent immediate or short-term adverse effects to the mother or baby.
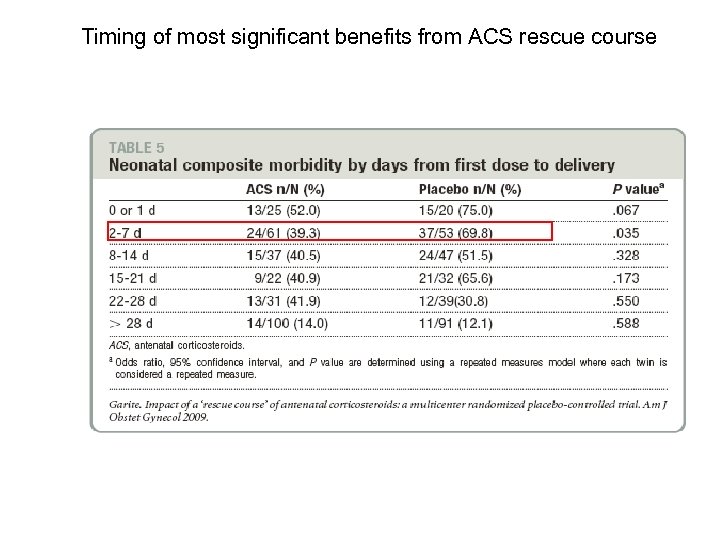 Timing of most significant benefits from ACS rescue course
Timing of most significant benefits from ACS rescue course
 QUIZ What is Your Diagnosis? Case: Patient J. F. 30 years old, at 32 week gestation. Presented with the following pictures.
QUIZ What is Your Diagnosis? Case: Patient J. F. 30 years old, at 32 week gestation. Presented with the following pictures.
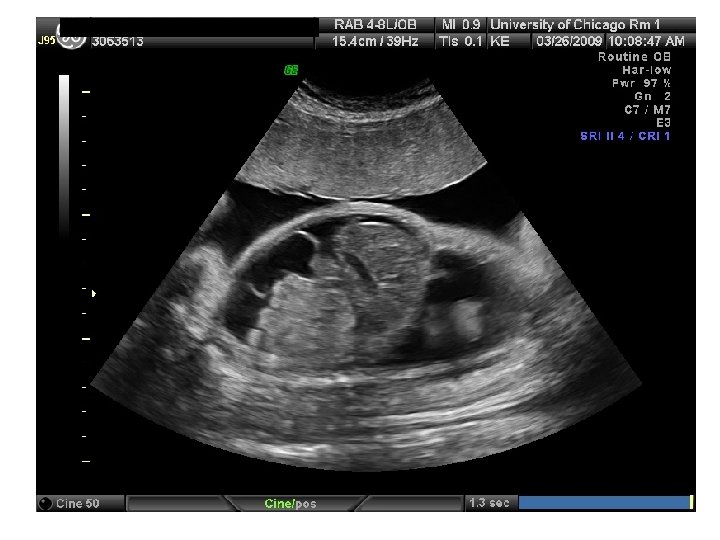
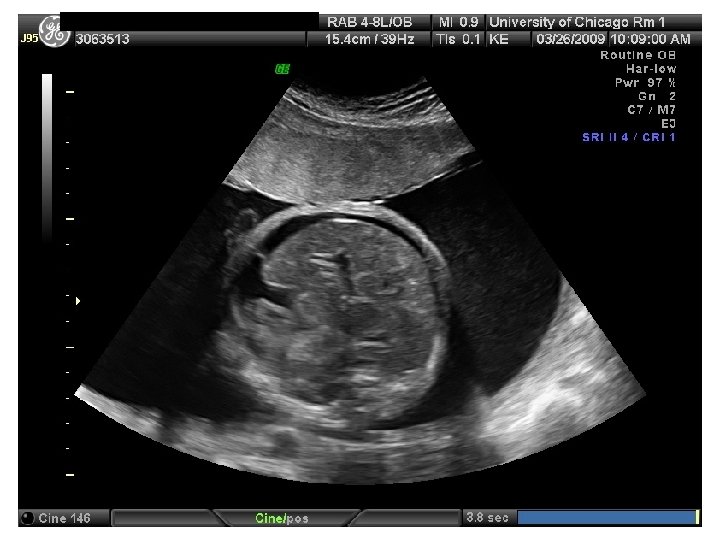
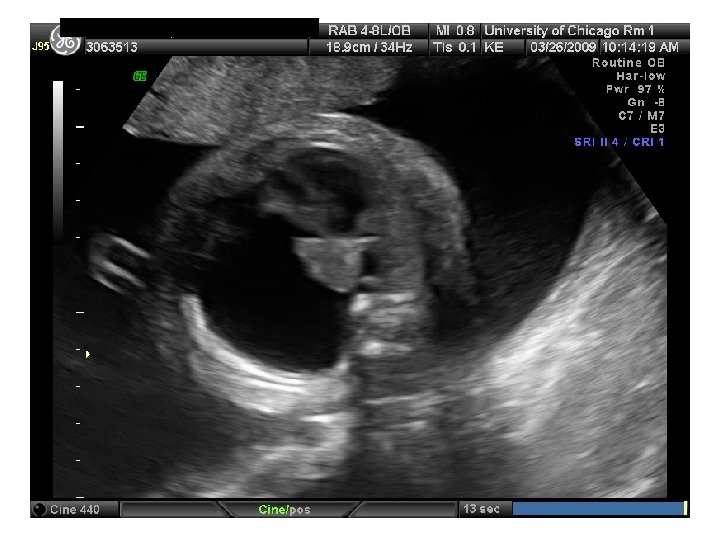
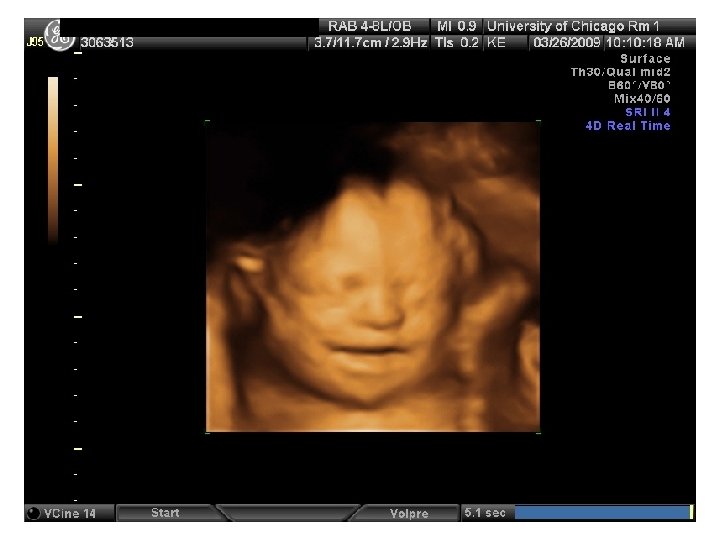
 Answer: a) Trisomy 21 b) Non-immune Hydops c) Paravo virus Infection
Answer: a) Trisomy 21 b) Non-immune Hydops c) Paravo virus Infection
 Answer: a) Trisomy 21 b) Non-immune Hydops c) Paravo virus Infection
Answer: a) Trisomy 21 b) Non-immune Hydops c) Paravo virus Infection
 Thanks to my children Lena, Sami, Nader and Nadia for technical support, and Jennifer Mc. Namara (mfm fellow) and Melissa Wong (ob/gyn resident) for research assistance.
Thanks to my children Lena, Sami, Nader and Nadia for technical support, and Jennifer Mc. Namara (mfm fellow) and Melissa Wong (ob/gyn resident) for research assistance.


