4abf30deadd68b1c21f93aa3f9850278.ppt
- Количество слайдов: 26

Advances in drug stability monitoring Stephen E. Zweig, Ph. D. CEO, Clini. Sense Corp. 7/21/06 Clini. Sense Corporation 1

A chain is as strong as the weakest link l Problems can potentially occur at any time during storage, causing “hidden damage” l Manufacturer’s responsibility doesn’t just stop on initial delivery – it continues until the product is actually used! 7/21/06 Clini. Sense Corporation 2

Where hidden problems can occur l Initial shipment: between different storage sites, each with separate temperature recorders l In the health care facility – Receiving – Transport to one or more storage sites – Transport to point of care site – Transport to patient – Time delay before administration – Patient mishandling of drug delivery devices 7/21/06 Clini. Sense Corporation 3

Biotherapeutics are delicate drugs l l Composed of unstable proteins with a precise structure l 7/21/06 Much larger and more complex than traditional pharmaceuticals Easily damaged by unfavorable temperature history during storage Clini. Sense Corporation 4
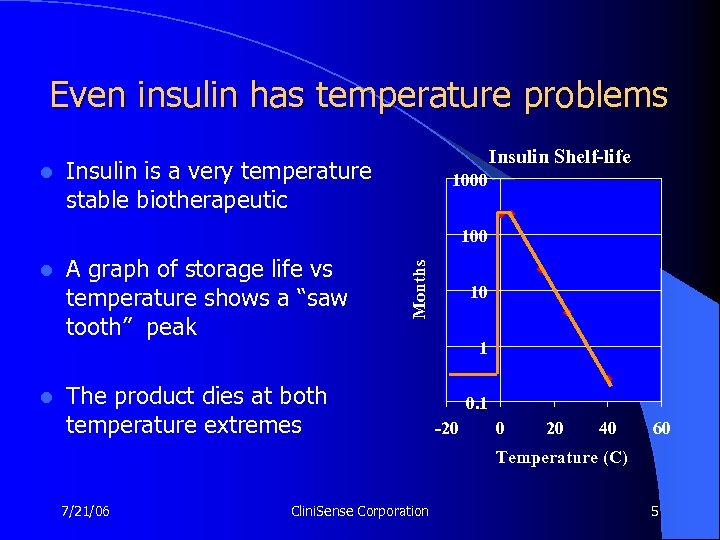
Even insulin has temperature problems l Insulin Shelf-life Insulin is a very temperature stable biotherapeutic 1000 l l A graph of storage life vs temperature shows a “saw tooth” peak Months 100 The product dies at both temperature extremes 10 1 0. 1 -20 0 20 40 60 Temperature (C) 7/21/06 Clini. Sense Corporation 5

Effect of temperature storage extremes < 0 o C l l l > 8 o C Freezing Protein denaturation Formation of aggregates Loss of functional activity Formation of potentially hazardous immunogenic byproducts 7/21/06 l l l Chemical side reactions Protein denaturation Formation of aggregates Loss of functional activity Formation of potentially hazardous immunogenic byproducts Clini. Sense Corporation 6

Smart packaging vs dumb packaging l Smart packaging can guard against human errors in handling, and accidental temperature abuse. l Is traditional dumb packaging simply a relic from an earlier era? Does it pass modern failure modes analysis? 7/21/06 Clini. Sense Corporation 7

An ideal storage monitor: l Accurate and effortless to read l Show remaining life (gas gauge-like display) l Outs thermal history record upon request 7/21/06 Clini. Sense Corporation 8

Previous time-temperature monitors l Chemical indicators are not flexible or accurate l Electronic temperature loggers are accurate, but don’t show shelf-life. l Chemical Electronic Temperature alarms only show extreme “out of bounds” conditions 7/21/06 Clini. Sense Corporation 9

Life. Track™ Technology l Monitors and analyzes temperature history l Custom programmed to exactly match the sensitivity curve of any pharmaceutical l Life. Track RFID Instantly shows all types of temperature abuse 7/21/06 Life. Track technology demonstrator Clini. Sense Corporation 10
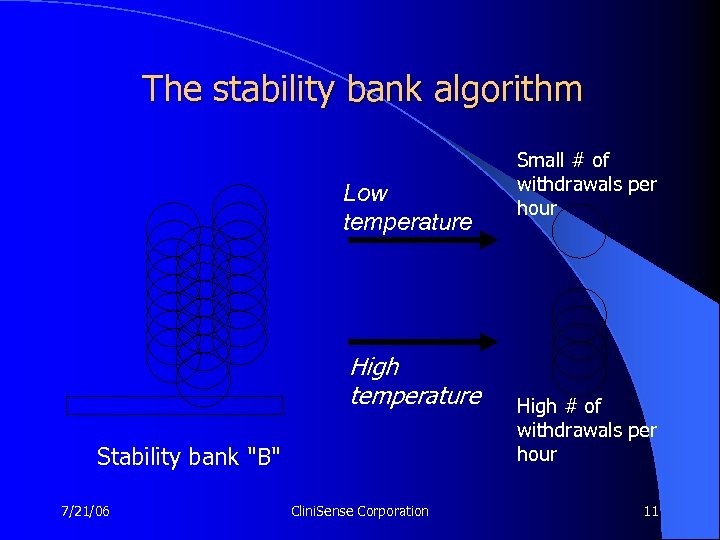
The stability bank algorithm Low temperature High temperature Stability bank "B" 7/21/06 Clini. Sense Corporation Small # of withdrawals per hour High # of withdrawals per hour 11
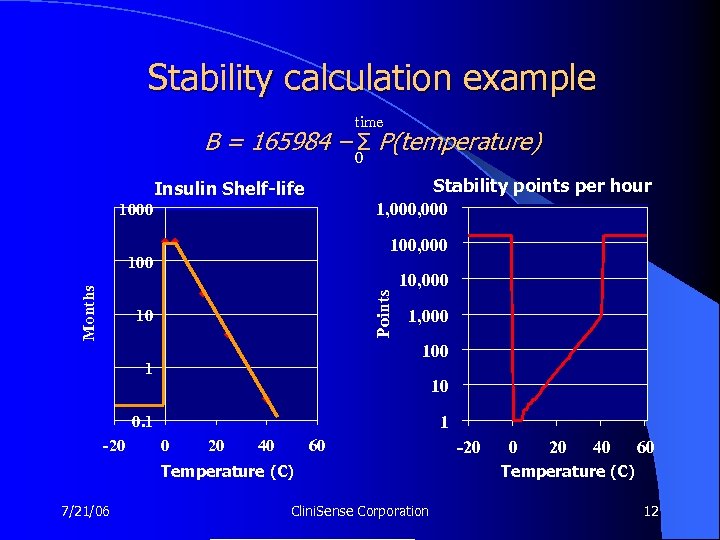
Stability calculation example time B = 165984 – 0 P(temperature) Σ Stability points per hour 1, 000 Insulin Shelf-life 1000 100, 000 Points Months 100 10 10, 000 100 1 10 1 0. 1 -20 0 20 40 60 Temperature (C) 7/21/06 Clini. Sense Corporation -20 0 20 40 60 Temperature (C) 12
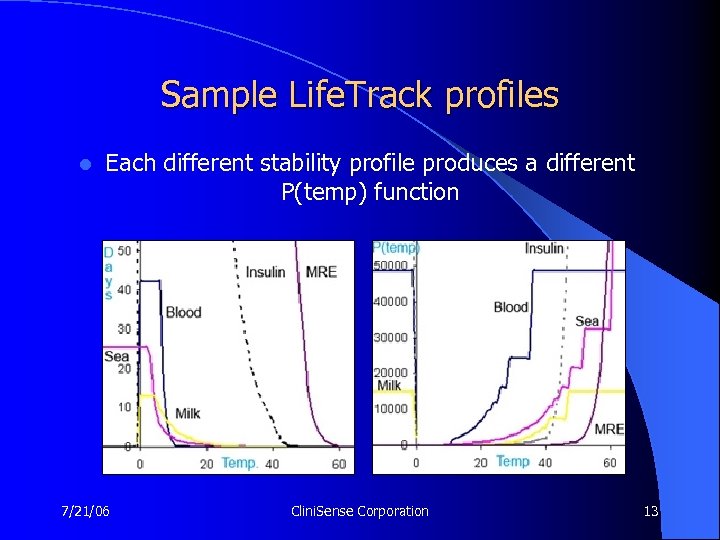
Sample Life. Track profiles l Each different stability profile produces a different P(temp) function 7/21/06 Clini. Sense Corporation 13

Evaluating Life. Track technology l Is our stability data adequate? l Customers like? l FDA requirements? l How can I do a quick feasibility test? 7/21/06 Clini. Sense Corporation 14

Life. Track Technology Demonstrator +/- good bad display Lifetime bar Thermistor Battery door Infrared LED 7/21/06 Programming & expansion Port Clini. Sense Corporation Battery (3 yr life) 15

Inside the unit Low-cost electronics l Simple construction l Designed for high production volumes l Digital watch example l – Modules cost < $1. 00 – Ultimate costs in $2. 00 to $3. 00 range 7/21/06 Clini. Sense Corporation 16

Life. Track Demonstrator Display l Shows “+” when product is good, “-” when product has expired l Lifetime indicator bars decrease as the lifetime is used up 7/21/06 Clini. Sense Corporation 17

Programming a unit l Enter the stability data into an Excel spreadsheet model l Transfer parameters to Life. Track program l Load Life. Track program into Life. Track device 7/21/06 Clini. Sense Corporation 18

Demonstrator Data Output l Downloads temperature statistical data via IR cable to an RS 232 port l Only one bit needed to tell good/not good, additional bits tell you how and why l Highly compatible with the limited data transmitted by RFID tags 7/21/06 Clini. Sense Corporation 19
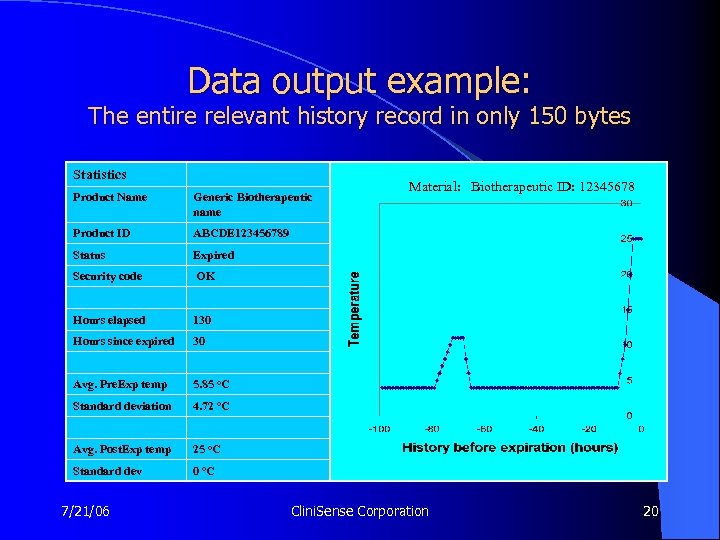
Data output example: The entire relevant history record in only 150 bytes Statistics Product Name Generic Biotherapeutic name Product ID ABCDE 123456789 Status Expired Security code OK Hours elapsed 130 Hours since expired 30 Avg. Pre. Exp temp 5. 85 o. C Standard deviation 4. 72 o. C Avg. Post. Exp temp 25 o. C Standard dev Material: Biotherapeutic ID: 12345678 0 o. C 7/21/06 Clini. Sense Corporation 20

Using Life. Track Technology • Provide biotherapeutic stability data for multiple temperatures (e. g < 0, 4 -8 o C, 25 o C, etc. ) • Clini. Sense will generate one or more provisional algorithms, request approval, and return flash programmed Life. Track technology demonstrator units for evaluation within a few days • Clini. Sense will work with you to embed this technology into your preferred drug delivery device or other type platform, license as needed 7/21/06 Clini. Sense Corporation 21

Smart biotherapeutic packaging Drug delivery devices l Life. Track detects deteriorated product l Can be embedded into drug delivery devices l Accurate and easily customized 7/21/06 Life. Track RFID Life. Track technology demonstrator Clini. Sense Corporation 22

Life. Track. TM Technology Injection pens Life. Track RFID Life. Track technology demonstrator Detects all types of improper storage 1/7/04 Clini. Sense Corporation

Recent publications Zweig SE: Advances in vaccine stability monitoring technology, Vaccine, 2006 (in press) l Zweig SE: 21 st century electronics for 21 st century drugs, Drug Delivery Reports, Spring/Summer 2006 l Zweig SE: From Smart Tags to Brilliant Tags: Advances in Drug Stability Monitoring, Bio. Pharm International, November 2005, 36 -44. l Zweig SE: Technologies for monitoring IVD stability, IVD Technology 10(5) 59 -63, June 2004 l 7/21/06 Clini. Sense Corporation 24

Recent patents l Patent 6, 950, 028, Electronic Time-Temperature Indicator l Patent 7, 096, 090, Electronic time-temperature indicator and logger (basis for PCT filings) l Patent application 10/747, 926 (pending) Method and device to reduce therapeutic protein immunogenicity 7/21/06 Clini. Sense Corporation 25

For more information l Clini. Sense website (www. clinisense. com) has many examples l Demo units available l Feel free to call (408) 348 -1495 and ask questions 7/21/06 Clini. Sense Corporation 26
4abf30deadd68b1c21f93aa3f9850278.ppt