a02f25af63170f08192e8d1952c870dc.ppt
- Количество слайдов: 93
 Advanced Analytical Micro PRO™
Advanced Analytical Micro PRO™
 Overview of Today’s Presentation • • History of Advanced Analytical Technologies, Inc. How the Technology Works The Micro PRO™ System Micro PRO™ Applications and Results • • • Qualitative Analysis – Presence/Absence Quantitative Analysis – Enumeration Customer Presentations
Overview of Today’s Presentation • • History of Advanced Analytical Technologies, Inc. How the Technology Works The Micro PRO™ System Micro PRO™ Applications and Results • • • Qualitative Analysis – Presence/Absence Quantitative Analysis – Enumeration Customer Presentations
 Advanced Analytical Improving Process Empowering Progress OUR MISSION Through innovative technology and collaborative relationships we will: > Deliver superior customer support, services, and solutions > Create sustainable growth > Foster a dynamic and fulfilling work environment
Advanced Analytical Improving Process Empowering Progress OUR MISSION Through innovative technology and collaborative relationships we will: > Deliver superior customer support, services, and solutions > Create sustainable growth > Foster a dynamic and fulfilling work environment
 Advanced Analytical Technologies, Inc. Who are We? • History: Founded 1997, 4 Scientific Co-founders Acquired CE Technology December, 2006 Privately Funded • Business: Rapid Microbial Detection Technology Capillary Electrophoresis Technology • Markets: Pharmaceutical, Personal Care Products, Environmental Fermentation, • Microbiology Solution: Replace current microbial detection methods (requiring 24 h – 5 days) with a technician-friendly, rapid system capable of detection and enumeration • Products: Micro PRO™ Instrument, Micro PRO™ Media Kit, PRO™ Reagent Kit, TVO Kit Micro
Advanced Analytical Technologies, Inc. Who are We? • History: Founded 1997, 4 Scientific Co-founders Acquired CE Technology December, 2006 Privately Funded • Business: Rapid Microbial Detection Technology Capillary Electrophoresis Technology • Markets: Pharmaceutical, Personal Care Products, Environmental Fermentation, • Microbiology Solution: Replace current microbial detection methods (requiring 24 h – 5 days) with a technician-friendly, rapid system capable of detection and enumeration • Products: Micro PRO™ Instrument, Micro PRO™ Media Kit, PRO™ Reagent Kit, TVO Kit Micro
 Basics of Flow Cytometry
Basics of Flow Cytometry
 Basics of Flow Cytometry • Laser-based irradiation of cells • Fluorochromes bound to cells provide information on cell state (e. g. , live, dead, spores, vegetative) • Light scattering provides relative size information • System composed of fluidic, optic and electronic components • Advantages: Rapid and quantitative analysis of individual cells
Basics of Flow Cytometry • Laser-based irradiation of cells • Fluorochromes bound to cells provide information on cell state (e. g. , live, dead, spores, vegetative) • Light scattering provides relative size information • System composed of fluidic, optic and electronic components • Advantages: Rapid and quantitative analysis of individual cells
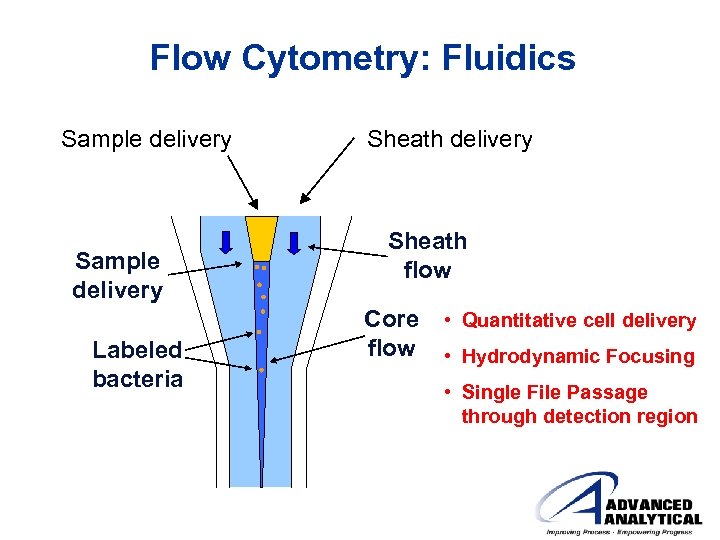 Flow Cytometry: Fluidics Sample delivery Labeled bacteria Sheath delivery Sheath flow Core flow • Quantitative cell delivery • Hydrodynamic Focusing • Single File Passage through detection region
Flow Cytometry: Fluidics Sample delivery Labeled bacteria Sheath delivery Sheath flow Core flow • Quantitative cell delivery • Hydrodynamic Focusing • Single File Passage through detection region
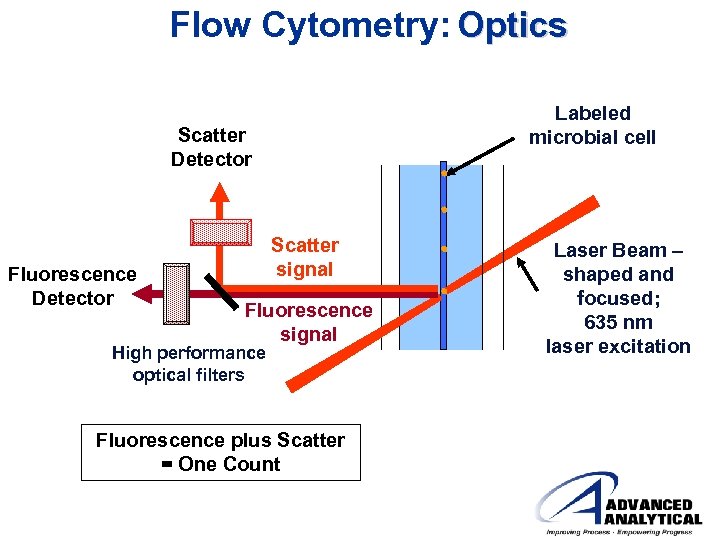 Flow Cytometry: Optics Labeled microbial cell Scatter Detector Fluorescence Detector Scatter signal Fluorescence signal High performance optical filters Fluorescence plus Scatter = One Count Laser Beam – shaped and focused; 635 nm laser excitation
Flow Cytometry: Optics Labeled microbial cell Scatter Detector Fluorescence Detector Scatter signal Fluorescence signal High performance optical filters Fluorescence plus Scatter = One Count Laser Beam – shaped and focused; 635 nm laser excitation
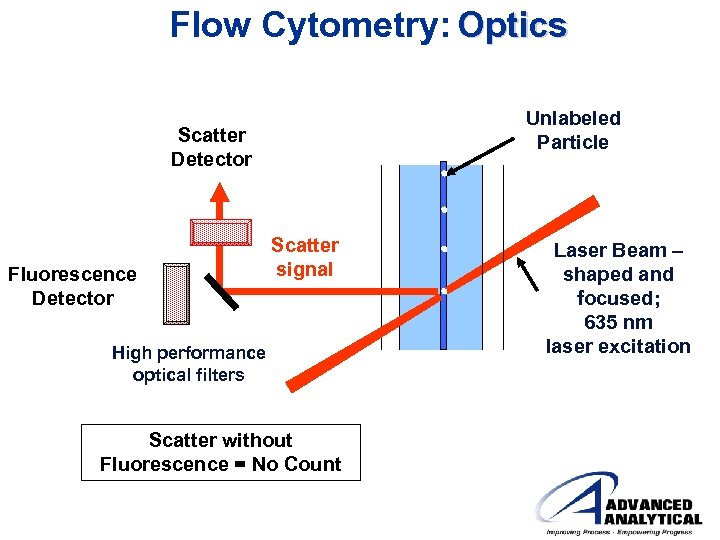 Flow Cytometry: Optics Unlabeled Particle Scatter Detector Fluorescence Detector Scatter signal High performance optical filters Scatter without Fluorescence = No Count Laser Beam – shaped and focused; 635 nm laser excitation
Flow Cytometry: Optics Unlabeled Particle Scatter Detector Fluorescence Detector Scatter signal High performance optical filters Scatter without Fluorescence = No Count Laser Beam – shaped and focused; 635 nm laser excitation
 Electronics • Signal processing component • The Micro PRO™ triggers on fluorescence • Fluorescent event above threshold is processed, along with the corresponding scatter event, and is plotted and recorded as a count Detector output = 1 count = 0 count Time Fluorescence Threshold Level
Electronics • Signal processing component • The Micro PRO™ triggers on fluorescence • Fluorescent event above threshold is processed, along with the corresponding scatter event, and is plotted and recorded as a count Detector output = 1 count = 0 count Time Fluorescence Threshold Level
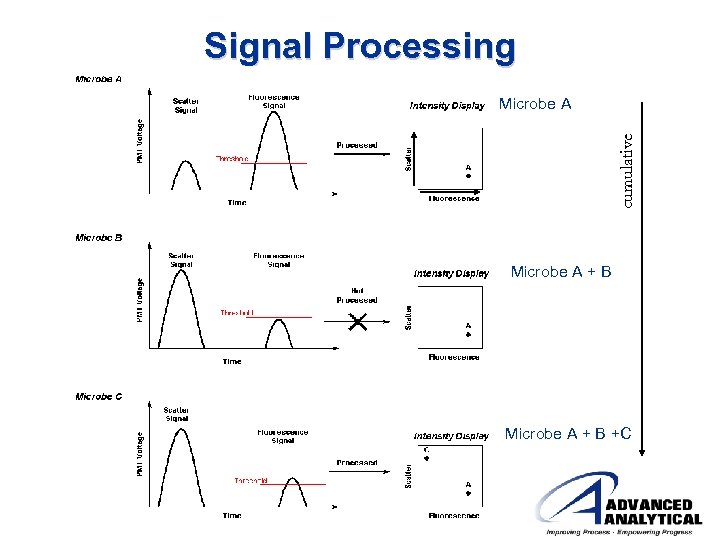 Signal Processing cumulative Microbe A + B +C
Signal Processing cumulative Microbe A + B +C
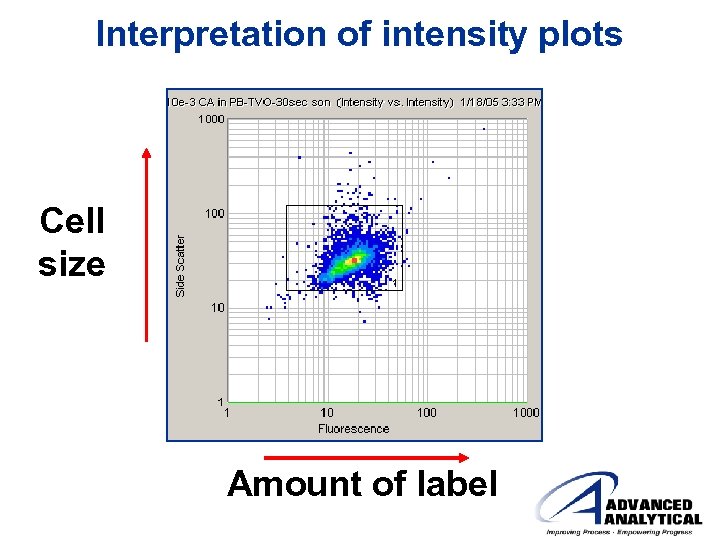 Interpretation of intensity plots Cell size Amount of label
Interpretation of intensity plots Cell size Amount of label
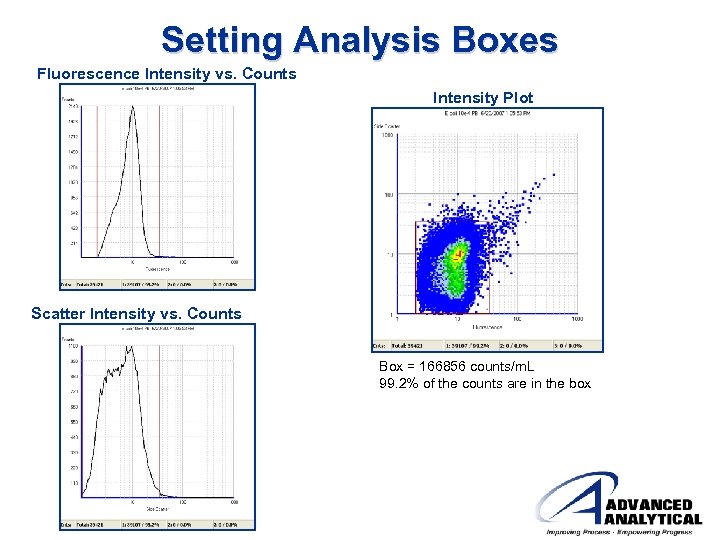 Setting Analysis Boxes Fluorescence Intensity vs. Counts Intensity Plot Scatter Intensity vs. Counts Box = 166856 counts/m. L 99. 2% of the counts are in the box
Setting Analysis Boxes Fluorescence Intensity vs. Counts Intensity Plot Scatter Intensity vs. Counts Box = 166856 counts/m. L 99. 2% of the counts are in the box
 Micro PRO™ INSTRUMENTATION
Micro PRO™ INSTRUMENTATION
 Introducing the Micro PRO™
Introducing the Micro PRO™
 Automated, High-Throughput Analysis • Qualitative analysis (Presence/Absence) 20 samples/hour • Quantitative analysis (Enumeration) 15 samples/hour • Holds up to 42 samples at once • Automatically • adds up to 3 reagents • mixes samples • cleaning and bubble removal
Automated, High-Throughput Analysis • Qualitative analysis (Presence/Absence) 20 samples/hour • Quantitative analysis (Enumeration) 15 samples/hour • Holds up to 42 samples at once • Automatically • adds up to 3 reagents • mixes samples • cleaning and bubble removal
 The Micro PRO™ Additional Advantages • Qualitative Analysis – Presence/Absence • Product Testing – Finished, raw materials, in-process samples • Screen products for bacteria, yeast & mold in 1 test • Next day results for product release • Quantitative Analysis – Enumeration • Purified/process water monitoring • Surface swab analysis • Pure culture enumeration • Validation packages & services available
The Micro PRO™ Additional Advantages • Qualitative Analysis – Presence/Absence • Product Testing – Finished, raw materials, in-process samples • Screen products for bacteria, yeast & mold in 1 test • Next day results for product release • Quantitative Analysis – Enumeration • Purified/process water monitoring • Surface swab analysis • Pure culture enumeration • Validation packages & services available
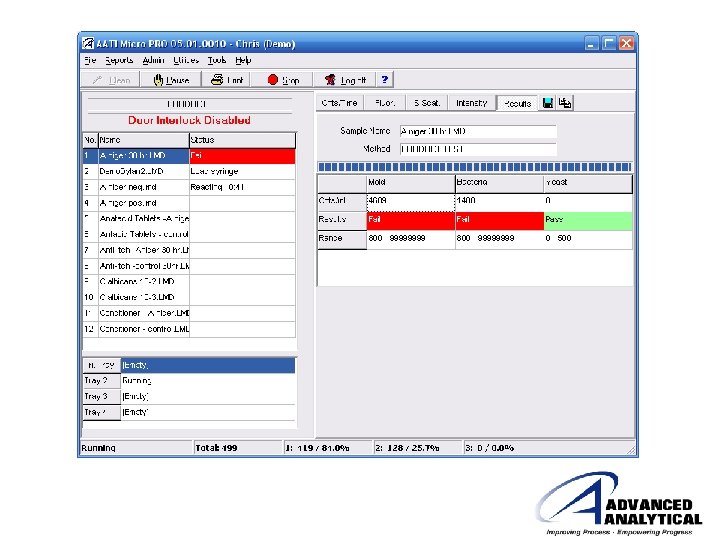
 Analysis on the Micro PRO™ 1 Load sample vials and syringes 3 2 Reagent additions and sample injection performed automatically as defined in the Method Select Tray Setup Micro PRO™ Output: Pass/Fail & Counts/m. L 4
Analysis on the Micro PRO™ 1 Load sample vials and syringes 3 2 Reagent additions and sample injection performed automatically as defined in the Method Select Tray Setup Micro PRO™ Output: Pass/Fail & Counts/m. L 4
 The Micro PRO™ Hardware upgrades • High sample throughput • • Qualitative analysis = 20 samples/hour (3 min/sample) Quantitative analysis = 15 samples/hour (4 min/sample) Preparation of multiple samples at a time Large capacity motors; motor movements streamlined • Sheath flow automatically stops after 30 minutes of inactivity • Cover plate to protect reagent ports from incidental contamination • Sample trays redesigned – lighter, ergonomic, support cap-less syringes • Re-designed vial de-capper • Optical sensor eliminated
The Micro PRO™ Hardware upgrades • High sample throughput • • Qualitative analysis = 20 samples/hour (3 min/sample) Quantitative analysis = 15 samples/hour (4 min/sample) Preparation of multiple samples at a time Large capacity motors; motor movements streamlined • Sheath flow automatically stops after 30 minutes of inactivity • Cover plate to protect reagent ports from incidental contamination • Sample trays redesigned – lighter, ergonomic, support cap-less syringes • Re-designed vial de-capper • Optical sensor eliminated
 The Micro PRO™ Software upgrades • Windows®-based, user-friendly interface • Main page re-design • Instant color-coded pass-fail results • View sample and tray status • • View/download results during sample analysis Reagent level alarm Context-sensitive Help files 21 CFR part 11 compliant
The Micro PRO™ Software upgrades • Windows®-based, user-friendly interface • Main page re-design • Instant color-coded pass-fail results • View sample and tray status • • View/download results during sample analysis Reagent level alarm Context-sensitive Help files 21 CFR part 11 compliant
 Micro PRO™ Qualitative Analysis Product Screening
Micro PRO™ Qualitative Analysis Product Screening
 Traditional methods USP <61> Bacteria 1: 10 dilution of product 1 ml in each of two Petri dishes with Soybean Casein Digest medium melted <45 C Incubate 48 to 72 hours at 30° C Count If zero counts, results are expressed as less than 10 cfu/ml Yeast and Mold 1: 10 dilution of product 1 ml in each of two Petri dishes Saboraud Dextrose Agar Incubate 5 to 7 days at 20 -25° C If zero counts, results are expressed as less than 10 cfu/ml Count
Traditional methods USP <61> Bacteria 1: 10 dilution of product 1 ml in each of two Petri dishes with Soybean Casein Digest medium melted <45 C Incubate 48 to 72 hours at 30° C Count If zero counts, results are expressed as less than 10 cfu/ml Yeast and Mold 1: 10 dilution of product 1 ml in each of two Petri dishes Saboraud Dextrose Agar Incubate 5 to 7 days at 20 -25° C If zero counts, results are expressed as less than 10 cfu/ml Count
 Micro PRO™ Product Screening Protocol 1) Dilute 10 g product in 90 m. L phosphate buffer or media (1: 10 product suspension) 2) Mix to achieve a homogeneous solution 3) Add 1 m. L of the 1: 10 product suspension to Tube A (Media Kit) 4) Incubate the samples at 30 OC for 24 -48 hours at ~200 rpm 5) After enrichment, prepare samples as directed in the Micro PRO™ Media Kit 6) Load samples on the Micro PRO™ for analysis 7) Pass/Fail results in 3 minutes/sample
Micro PRO™ Product Screening Protocol 1) Dilute 10 g product in 90 m. L phosphate buffer or media (1: 10 product suspension) 2) Mix to achieve a homogeneous solution 3) Add 1 m. L of the 1: 10 product suspension to Tube A (Media Kit) 4) Incubate the samples at 30 OC for 24 -48 hours at ~200 rpm 5) After enrichment, prepare samples as directed in the Micro PRO™ Media Kit 6) Load samples on the Micro PRO™ for analysis 7) Pass/Fail results in 3 minutes/sample
 Micro PRO™ Media Kit Microbes 35µm filter Transfer swab from Tube A to B Tube A - GEM Add product, enrich Tube B - PB Add swab, mix Micro PRO™ Tube C - PB Add 0. 1 m. L from Tube B
Micro PRO™ Media Kit Microbes 35µm filter Transfer swab from Tube A to B Tube A - GEM Add product, enrich Tube B - PB Add swab, mix Micro PRO™ Tube C - PB Add 0. 1 m. L from Tube B
 Micro PRO™ Reagent Kit Micro PRO™ Pass/Fail Result Test Sample w/ Microbes All Microbes Stained Viable Microbes Stained Ideal for detecting microorganisms post-enrichment in: Ø Finished Products Ø Raw Materials Ø In-Process Samples
Micro PRO™ Reagent Kit Micro PRO™ Pass/Fail Result Test Sample w/ Microbes All Microbes Stained Viable Microbes Stained Ideal for detecting microorganisms post-enrichment in: Ø Finished Products Ø Raw Materials Ø In-Process Samples
 Interpretation of results • Micro PRO™ picture shows few to no counts within the analysis box (product baseline) • “Pass” result indicates that the sample does not contain microbial contamination Mold Area 1 counts/m. L Bacteria Area 2 counts/m. L Yeast Area 3 counts/m. L Result 1 Result 2 Result 3 Overall Result 32 241 43 Pass
Interpretation of results • Micro PRO™ picture shows few to no counts within the analysis box (product baseline) • “Pass” result indicates that the sample does not contain microbial contamination Mold Area 1 counts/m. L Bacteria Area 2 counts/m. L Yeast Area 3 counts/m. L Result 1 Result 2 Result 3 Overall Result 32 241 43 Pass
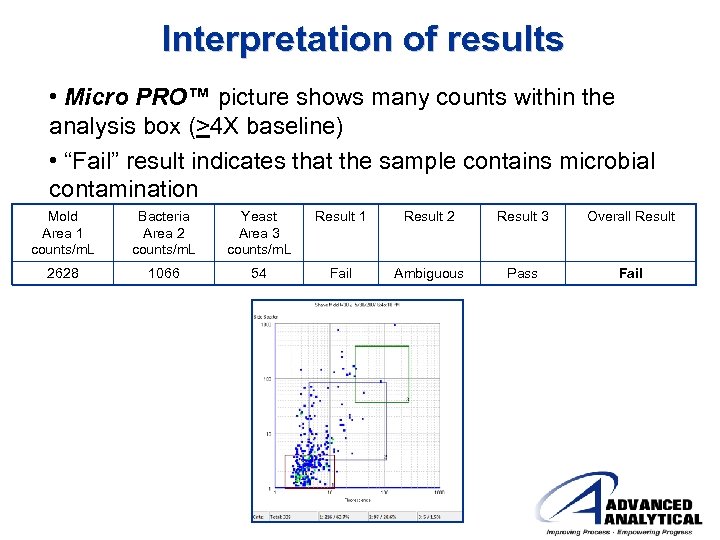 Interpretation of results • Micro PRO™ picture shows many counts within the analysis box (>4 X baseline) • “Fail” result indicates that the sample contains microbial contamination Mold Area 1 counts/m. L Bacteria Area 2 counts/m. L Yeast Area 3 counts/m. L Result 1 Result 2 Result 3 Overall Result 2628 1066 54 Fail Ambiguous Pass Fail
Interpretation of results • Micro PRO™ picture shows many counts within the analysis box (>4 X baseline) • “Fail” result indicates that the sample contains microbial contamination Mold Area 1 counts/m. L Bacteria Area 2 counts/m. L Yeast Area 3 counts/m. L Result 1 Result 2 Result 3 Overall Result 2628 1066 54 Fail Ambiguous Pass Fail
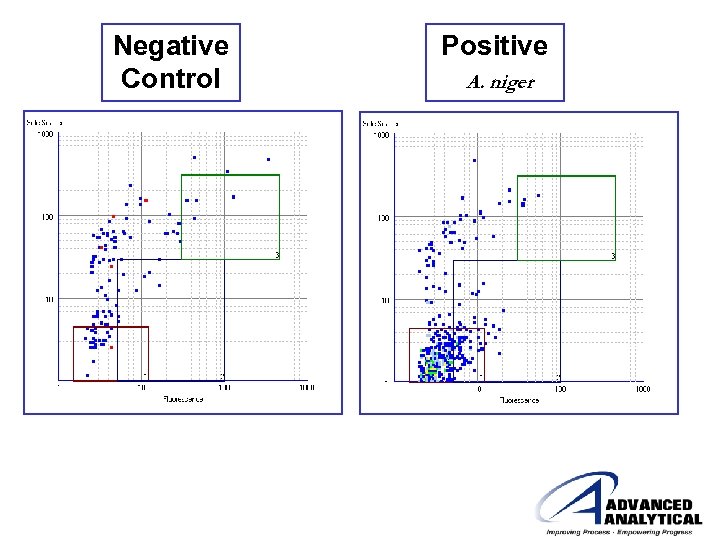 Negative Control Positive A. niger
Negative Control Positive A. niger
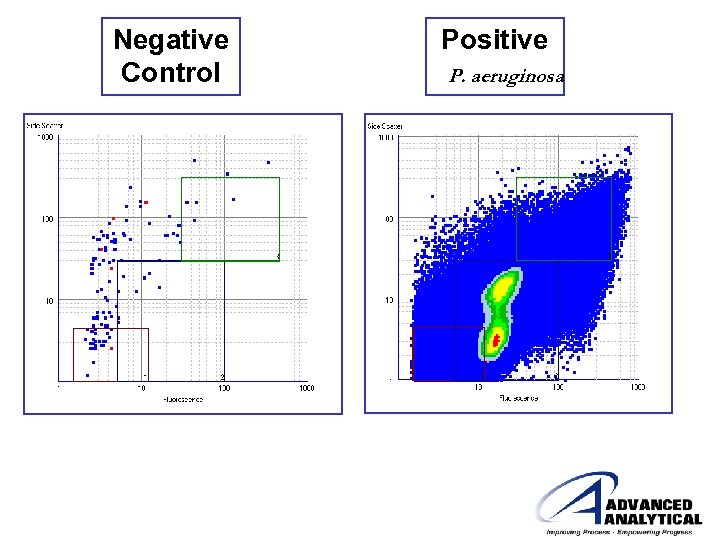 Negative Control Positive P. aeruginosa
Negative Control Positive P. aeruginosa
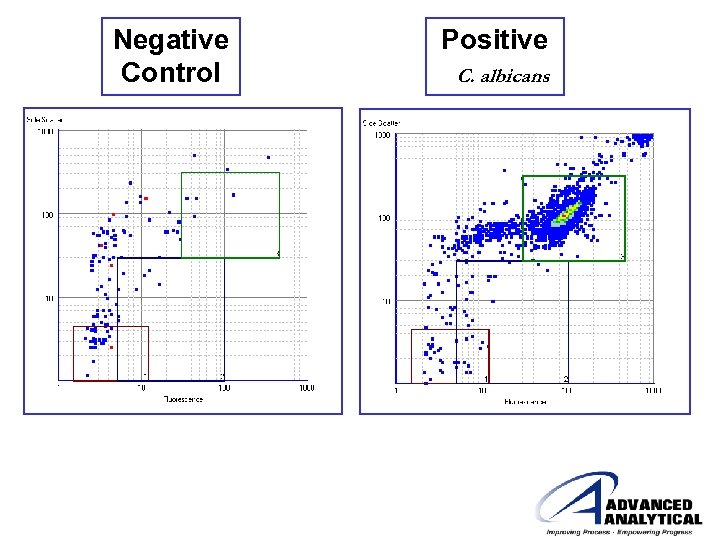 Negative Control Positive C. albicans
Negative Control Positive C. albicans
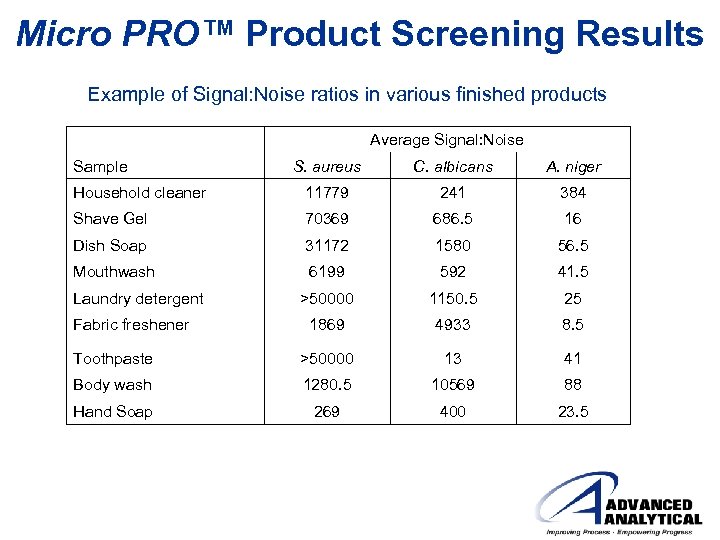 Micro PRO™ Product Screening Results Example of Signal: Noise ratios in various finished products Average Signal: Noise Sample S. aureus C. albicans A. niger Household cleaner 11779 241 384 Shave Gel 70369 686. 5 16 Dish Soap 31172 1580 56. 5 Mouthwash 6199 592 41. 5 >50000 1150. 5 25 1869 4933 8. 5 Toothpaste >50000 13 41 Body wash 1280. 5 10569 88 Hand Soap 269 400 23. 5 Laundry detergent Fabric freshener
Micro PRO™ Product Screening Results Example of Signal: Noise ratios in various finished products Average Signal: Noise Sample S. aureus C. albicans A. niger Household cleaner 11779 241 384 Shave Gel 70369 686. 5 16 Dish Soap 31172 1580 56. 5 Mouthwash 6199 592 41. 5 >50000 1150. 5 25 1869 4933 8. 5 Toothpaste >50000 13 41 Body wash 1280. 5 10569 88 Hand Soap 269 400 23. 5 Laundry detergent Fabric freshener
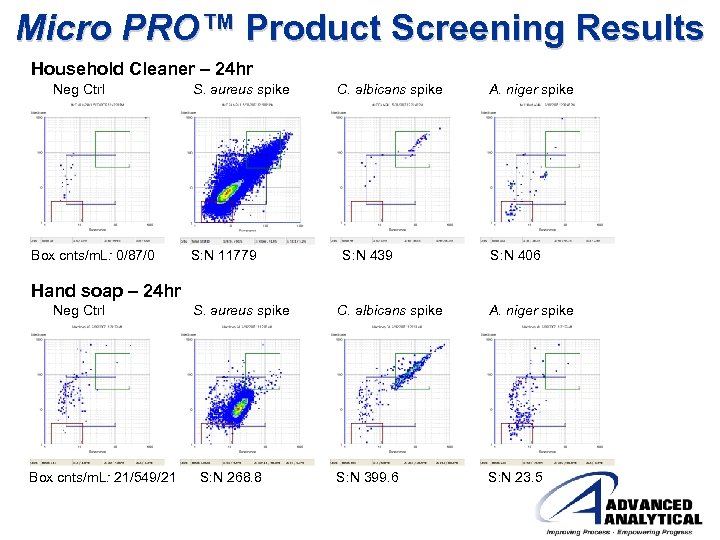 Micro PRO™ Product Screening Results Household Cleaner – 24 hr Neg Ctrl Box cnts/m. L: 0/87/0 S. aureus spike S: N 11779 C. albicans spike S: N 439 A. niger spike S: N 406 Hand soap – 24 hr Neg Ctrl Box cnts/m. L: 21/549/21 S. aureus spike S: N 268. 8 C. albicans spike S: N 399. 6 A. niger spike S: N 23. 5
Micro PRO™ Product Screening Results Household Cleaner – 24 hr Neg Ctrl Box cnts/m. L: 0/87/0 S. aureus spike S: N 11779 C. albicans spike S: N 439 A. niger spike S: N 406 Hand soap – 24 hr Neg Ctrl Box cnts/m. L: 21/549/21 S. aureus spike S: N 268. 8 C. albicans spike S: N 399. 6 A. niger spike S: N 23. 5
 Examples of Products Tested Bacteria-Yeast-Mold detection in 1 assay Personal Care Products OTC Pharma Products Hand soap Shave Gel Antacid Tablets Body wash Shampoo Liquid Antacid Face Scrub Conditioner Anti-itch cream Toothpaste Lotion Cold Syrup Mouthwash Multi-vitamin (chewable) Multi-vitamin (adult) Household Products Nose Drops Dish soap Stomach Relief Laundry detergent Fabric Freshener All-purpose household cleaner
Examples of Products Tested Bacteria-Yeast-Mold detection in 1 assay Personal Care Products OTC Pharma Products Hand soap Shave Gel Antacid Tablets Body wash Shampoo Liquid Antacid Face Scrub Conditioner Anti-itch cream Toothpaste Lotion Cold Syrup Mouthwash Multi-vitamin (chewable) Multi-vitamin (adult) Household Products Nose Drops Dish soap Stomach Relief Laundry detergent Fabric Freshener All-purpose household cleaner
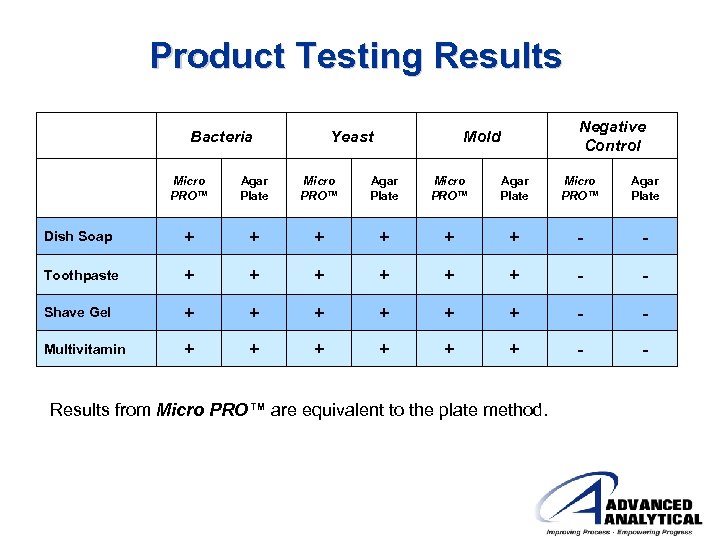 Product Testing Results Bacteria Yeast Negative Control Mold Micro PRO™ Agar Plate Dish Soap + + + - - Toothpaste + + + - - Shave Gel + + + - - Multivitamin + + + - - Results from Micro PRO™ are equivalent to the plate method.
Product Testing Results Bacteria Yeast Negative Control Mold Micro PRO™ Agar Plate Dish Soap + + + - - Toothpaste + + + - - Shave Gel + + + - - Multivitamin + + + - - Results from Micro PRO™ are equivalent to the plate method.
 Micro PRO™ Quantitative Analysis
Micro PRO™ Quantitative Analysis
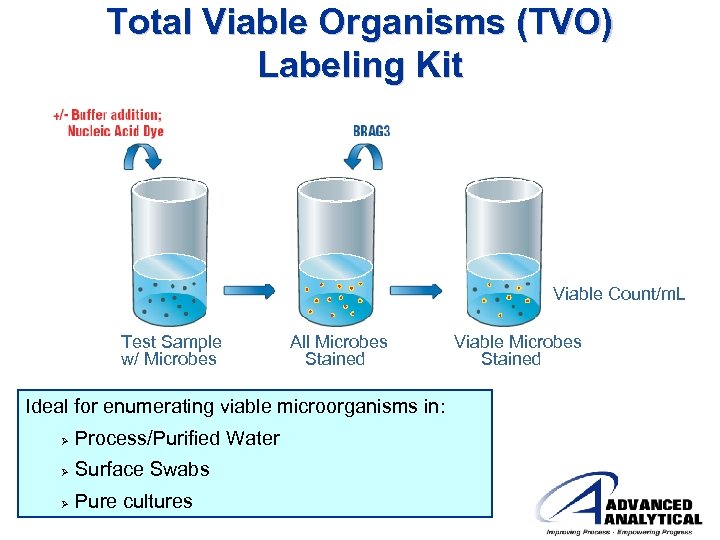 Total Viable Organisms (TVO) Labeling Kit Viable Count/m. L Test Sample w/ Microbes All Microbes Stained Viable Microbes Stained Ideal for enumerating viable microorganisms in: Ø Process/Purified Water Ø Surface Swabs Ø Pure cultures
Total Viable Organisms (TVO) Labeling Kit Viable Count/m. L Test Sample w/ Microbes All Microbes Stained Viable Microbes Stained Ideal for enumerating viable microorganisms in: Ø Process/Purified Water Ø Surface Swabs Ø Pure cultures
 Purified Water Monitoring • Results in less then 4 minutes • Quantitative output in counts/m. L • Significant savings in technician labor
Purified Water Monitoring • Results in less then 4 minutes • Quantitative output in counts/m. L • Significant savings in technician labor
 Implementation Procedure 1) Determine the background range of the Micro PRO™ and TVO kit for accurate enumeration of TVO in purified water. 2) Define the method for purified water samples. Defining the analysis box. 3) Demonstrate that the Micro PRO™ results correlate well with the traditional plate methods. 4) Provides a laboratory with the necessary equipment to detect increases in purified water TVO counts within minutes of sample collection, enabling the scientist to respond proactively.
Implementation Procedure 1) Determine the background range of the Micro PRO™ and TVO kit for accurate enumeration of TVO in purified water. 2) Define the method for purified water samples. Defining the analysis box. 3) Demonstrate that the Micro PRO™ results correlate well with the traditional plate methods. 4) Provides a laboratory with the necessary equipment to detect increases in purified water TVO counts within minutes of sample collection, enabling the scientist to respond proactively.
 Determining Background Limits 1) Collect purified water sample 2) 0. 2 mm filter sterilize 3) Background/negative control samples are analyzed on the Micro PRO™ using the Water Method 4) Background is typically less than 26 counts/ml
Determining Background Limits 1) Collect purified water sample 2) 0. 2 mm filter sterilize 3) Background/negative control samples are analyzed on the Micro PRO™ using the Water Method 4) Background is typically less than 26 counts/ml
 Defining the Analysis Box 1) Microbes used: a. Escherichia coli, ATCC 25922 b. Pseudomonas aeruginosa, ATCC 9027 c. Staphylococcus aureus, ATCC 6538 d. Serratia marcescens, ATCC 13880 e. Ralstonia pickettii, ATCC 49129 f. A 6 -isolate mix from in-house water system (unknown species) 2) Grow pure cultures and serially dilute in 0. 2 mm-filtered purified water 3) Analyze on the Micro PRO™ with the TVO kit 4) Set analysis box parameters in Water Method so the box encompasses all microbial populations 5) Use this standard analysis box in the Water Method for all negative control and test samples
Defining the Analysis Box 1) Microbes used: a. Escherichia coli, ATCC 25922 b. Pseudomonas aeruginosa, ATCC 9027 c. Staphylococcus aureus, ATCC 6538 d. Serratia marcescens, ATCC 13880 e. Ralstonia pickettii, ATCC 49129 f. A 6 -isolate mix from in-house water system (unknown species) 2) Grow pure cultures and serially dilute in 0. 2 mm-filtered purified water 3) Analyze on the Micro PRO™ with the TVO kit 4) Set analysis box parameters in Water Method so the box encompasses all microbial populations 5) Use this standard analysis box in the Water Method for all negative control and test samples
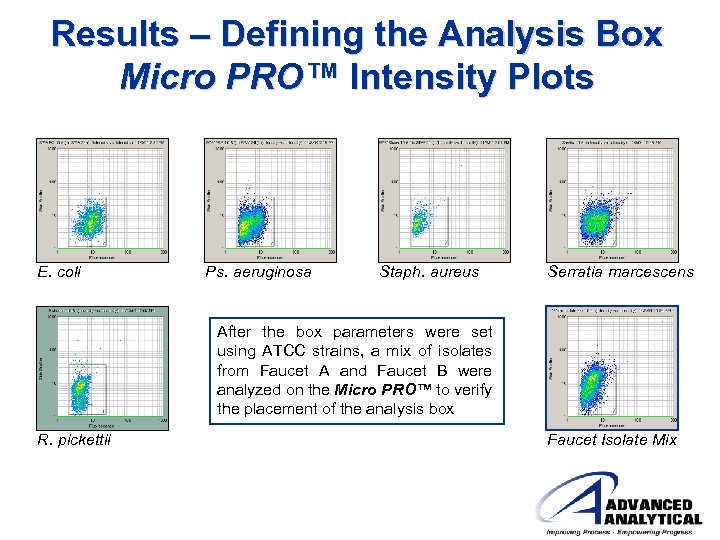 Results – Defining the Analysis Box Micro PRO™ Intensity Plots E. coli Ps. aeruginosa Staph. aureus Serratia marcescens After the box parameters were set using ATCC strains, a mix of isolates from Faucet A and Faucet B were analyzed on the Micro PRO™ to verify the placement of the analysis box R. pickettii Faucet Isolate Mix
Results – Defining the Analysis Box Micro PRO™ Intensity Plots E. coli Ps. aeruginosa Staph. aureus Serratia marcescens After the box parameters were set using ATCC strains, a mix of isolates from Faucet A and Faucet B were analyzed on the Micro PRO™ to verify the placement of the analysis box R. pickettii Faucet Isolate Mix
 Purified Water Monitoring Protocol 1) Sanitize each sampling point if necessary. 2) Flush each sampling point for one minute. 3) Collect a minimum of 10 m. L per sampling point. 4) Dispense 2 x 3 m. L aliquots per sampling point into 5 m. L sample tubes. 5) Prepare 0. 2 -micron filtered water sample 6) Load tray and analyze using “Water Analysis” Tray Setup in the Micro PRO™ software
Purified Water Monitoring Protocol 1) Sanitize each sampling point if necessary. 2) Flush each sampling point for one minute. 3) Collect a minimum of 10 m. L per sampling point. 4) Dispense 2 x 3 m. L aliquots per sampling point into 5 m. L sample tubes. 5) Prepare 0. 2 -micron filtered water sample 6) Load tray and analyze using “Water Analysis” Tray Setup in the Micro PRO™ software
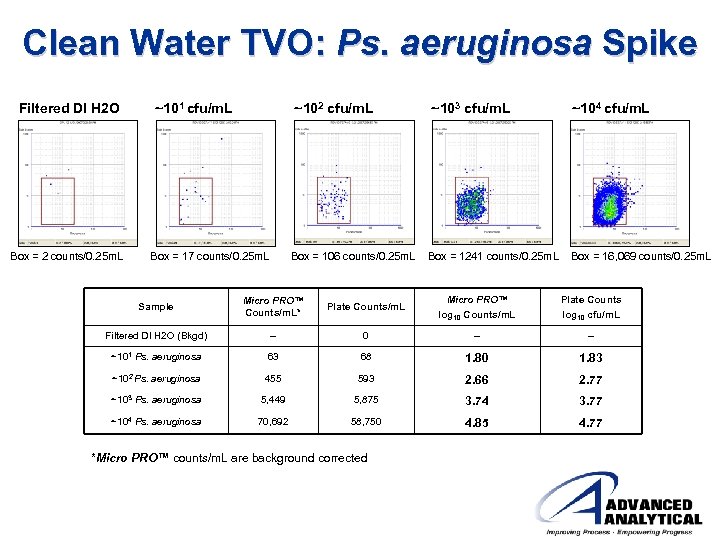 Clean Water TVO: Ps. aeruginosa Spike Filtered DI H 2 O ~101 cfu/m. L ~102 cfu/m. L ~103 cfu/m. L ~104 cfu/m. L Box = 2 counts/0. 25 m. L Box = 17 counts/0. 25 m. L Box = 106 counts/0. 25 m. L Box = 1241 counts/0. 25 m. L Box = 16, 069 counts/0. 25 m. L Sample Micro PRO™ Counts/m. L* Plate Counts/m. L Micro PRO™ log 10 Counts/m. L Plate Counts log 10 cfu/m. L Filtered DI H 2 O (Bkgd) -- 0 -- -- ~101 Ps. aeruginosa 63 68 1. 80 1. 83 ~102 Ps. aeruginosa 455 593 2. 66 2. 77 ~103 Ps. aeruginosa 5, 449 5, 875 3. 74 3. 77 ~104 Ps. aeruginosa 70, 692 58, 750 4. 85 4. 77 *Micro PRO™ counts/m. L are background corrected
Clean Water TVO: Ps. aeruginosa Spike Filtered DI H 2 O ~101 cfu/m. L ~102 cfu/m. L ~103 cfu/m. L ~104 cfu/m. L Box = 2 counts/0. 25 m. L Box = 17 counts/0. 25 m. L Box = 106 counts/0. 25 m. L Box = 1241 counts/0. 25 m. L Box = 16, 069 counts/0. 25 m. L Sample Micro PRO™ Counts/m. L* Plate Counts/m. L Micro PRO™ log 10 Counts/m. L Plate Counts log 10 cfu/m. L Filtered DI H 2 O (Bkgd) -- 0 -- -- ~101 Ps. aeruginosa 63 68 1. 80 1. 83 ~102 Ps. aeruginosa 455 593 2. 66 2. 77 ~103 Ps. aeruginosa 5, 449 5, 875 3. 74 3. 77 ~104 Ps. aeruginosa 70, 692 58, 750 4. 85 4. 77 *Micro PRO™ counts/m. L are background corrected
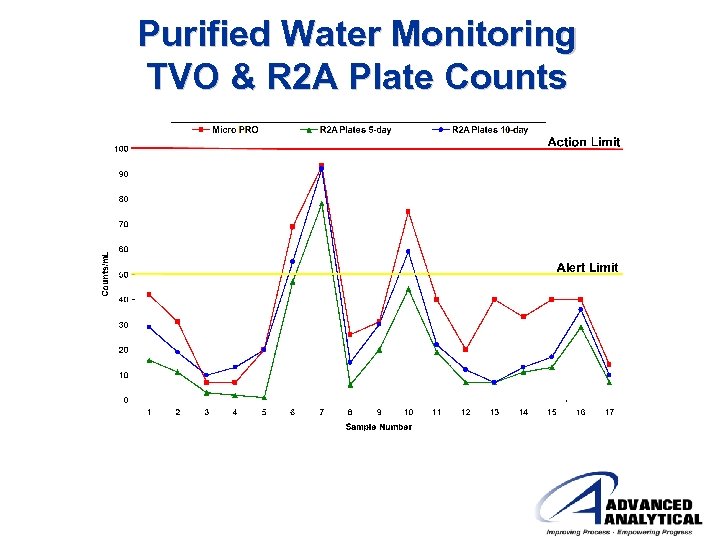 Purified Water Monitoring TVO & R 2 A Plate Counts
Purified Water Monitoring TVO & R 2 A Plate Counts
 Customer Studies
Customer Studies
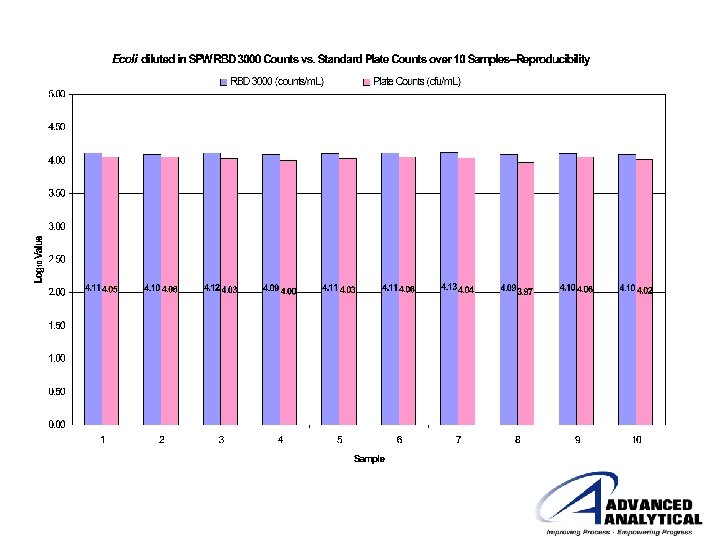
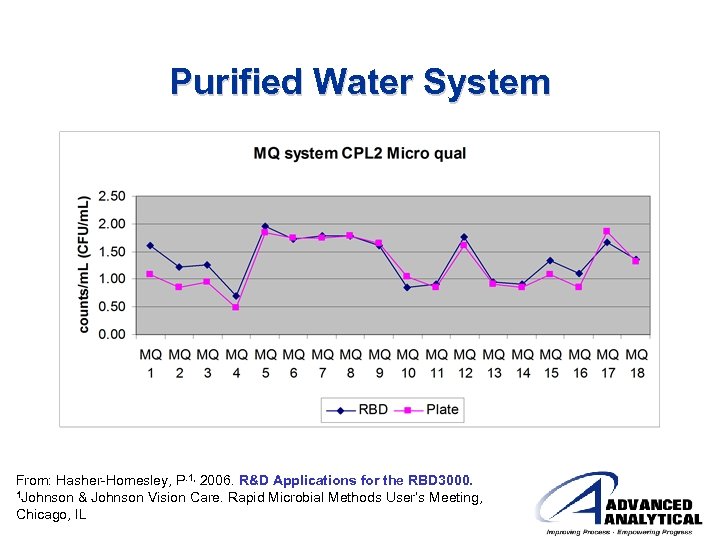 Purified Water System From: Hasher-Homesley, P. 1, 2006. R&D Applications for the RBD 3000. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods User’s Meeting, Chicago, IL
Purified Water System From: Hasher-Homesley, P. 1, 2006. R&D Applications for the RBD 3000. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods User’s Meeting, Chicago, IL
 Surface Swab Analysis/ Environmental Monitoring
Surface Swab Analysis/ Environmental Monitoring
 Environmental Sample Testing • Surface swabs are analyzed on the Micro PRO™ with results within 4 minutes • Data generated by the Micro PRO™ not only provides information about microbial populations but also indicates levels of residual product
Environmental Sample Testing • Surface swabs are analyzed on the Micro PRO™ with results within 4 minutes • Data generated by the Micro PRO™ not only provides information about microbial populations but also indicates levels of residual product
 Microorganism Recovery • Recovery was demonstrated by testing pure cultures of Pseudomonas aeruginosa, Staphylococcus aureus, and Candida albicans • 100μL of the pure culture was pipetted onto a sterile Petri dish • A dry swab was used to absorb the sample • Additionally, 100μL of the final solution was spread plated onto Tryptic Soy Agar
Microorganism Recovery • Recovery was demonstrated by testing pure cultures of Pseudomonas aeruginosa, Staphylococcus aureus, and Candida albicans • 100μL of the pure culture was pipetted onto a sterile Petri dish • A dry swab was used to absorb the sample • Additionally, 100μL of the final solution was spread plated onto Tryptic Soy Agar
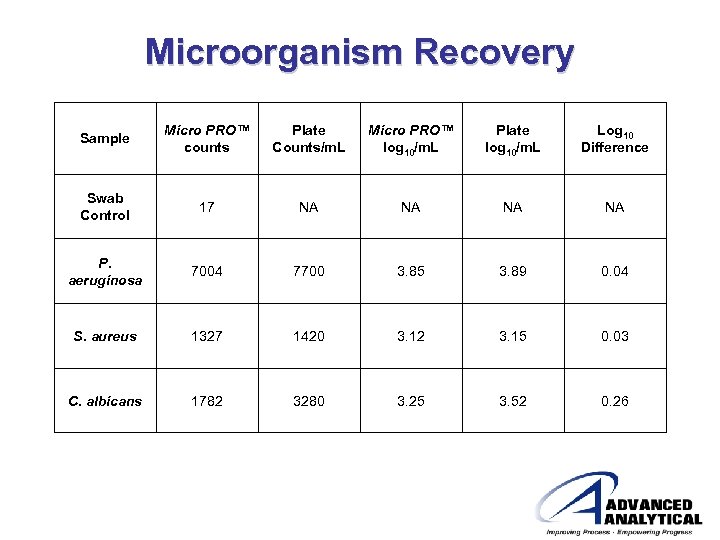 Microorganism Recovery Sample Micro PRO™ counts Plate Counts/m. L Micro PRO™ log 10/m. L Plate log 10/m. L Log 10 Difference Swab Control 17 NA NA P. aeruginosa 7004 7700 3. 85 3. 89 0. 04 S. aureus 1327 1420 3. 12 3. 15 0. 03 C. albicans 1782 3280 3. 25 3. 52 0. 26
Microorganism Recovery Sample Micro PRO™ counts Plate Counts/m. L Micro PRO™ log 10/m. L Plate log 10/m. L Log 10 Difference Swab Control 17 NA NA P. aeruginosa 7004 7700 3. 85 3. 89 0. 04 S. aureus 1327 1420 3. 12 3. 15 0. 03 C. albicans 1782 3280 3. 25 3. 52 0. 26
 Environmental Swab Protocol • • • Place a swab sample in a 5 m. L snap-cap tube containing 1 m. L filtered, sterile PB Break the swab handle over the rim of the tube Replace snap-cap and vortex swab and buffer for 30 seconds Press the swab against the side of the tube to express extra liquid Bring the volume to 3 m. L with filtered, sterile PB Analyze samples and controls on the Micro PRO™
Environmental Swab Protocol • • • Place a swab sample in a 5 m. L snap-cap tube containing 1 m. L filtered, sterile PB Break the swab handle over the rim of the tube Replace snap-cap and vortex swab and buffer for 30 seconds Press the swab against the side of the tube to express extra liquid Bring the volume to 3 m. L with filtered, sterile PB Analyze samples and controls on the Micro PRO™
 1 2 3 4
1 2 3 4
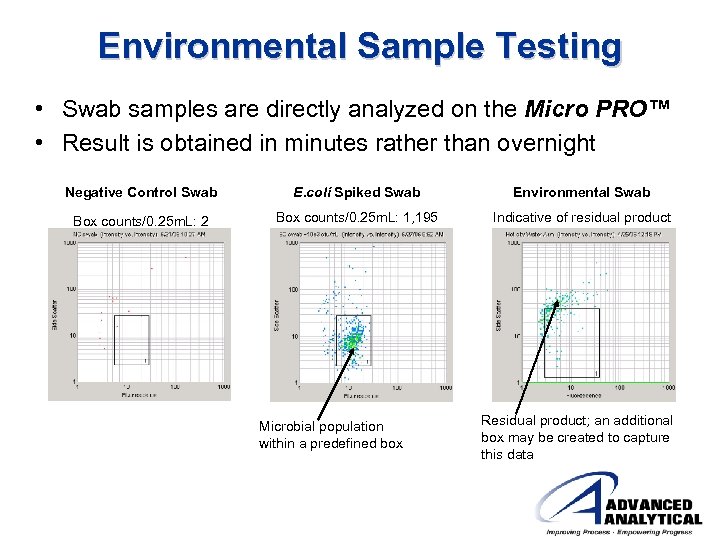 Environmental Sample Testing • Swab samples are directly analyzed on the Micro PRO™ • Result is obtained in minutes rather than overnight Negative Control Swab E. coli Spiked Swab Environmental Swab Box counts/0. 25 m. L: 2 Box counts/0. 25 m. L: 1, 195 Indicative of residual product Microbial population within a predefined box Residual product; an additional box may be created to capture this data
Environmental Sample Testing • Swab samples are directly analyzed on the Micro PRO™ • Result is obtained in minutes rather than overnight Negative Control Swab E. coli Spiked Swab Environmental Swab Box counts/0. 25 m. L: 2 Box counts/0. 25 m. L: 1, 195 Indicative of residual product Microbial population within a predefined box Residual product; an additional box may be created to capture this data
 Pure Culture Enumeration
Pure Culture Enumeration
 Enumeration of Fermentation/Pure Cultures • Collect sample; if necessary dilute to <106 cfu/m. L in PB • Dispense 3 m. L sample into 5 m. L snap-cap tube • Load sample into Micro PRO™ Sample Tray with capped syringe • Select pre-defined (or create new) Analysis/Tray Sequence • Micro PRO™ count result in 4 minutes
Enumeration of Fermentation/Pure Cultures • Collect sample; if necessary dilute to <106 cfu/m. L in PB • Dispense 3 m. L sample into 5 m. L snap-cap tube • Load sample into Micro PRO™ Sample Tray with capped syringe • Select pre-defined (or create new) Analysis/Tray Sequence • Micro PRO™ count result in 4 minutes
 Fermentation Applications Upstream 1. Media Contamination 2. Starter Culture Viability and/or Titer Downstream 1. Number of cells post inactivation 2. Number of cells after processing 3. Cell Concentrations Fermentation 1. Process Monitoring 2. Biomass and/or Viable cell enumerations 3. Determination of when to harvest or induce
Fermentation Applications Upstream 1. Media Contamination 2. Starter Culture Viability and/or Titer Downstream 1. Number of cells post inactivation 2. Number of cells after processing 3. Cell Concentrations Fermentation 1. Process Monitoring 2. Biomass and/or Viable cell enumerations 3. Determination of when to harvest or induce
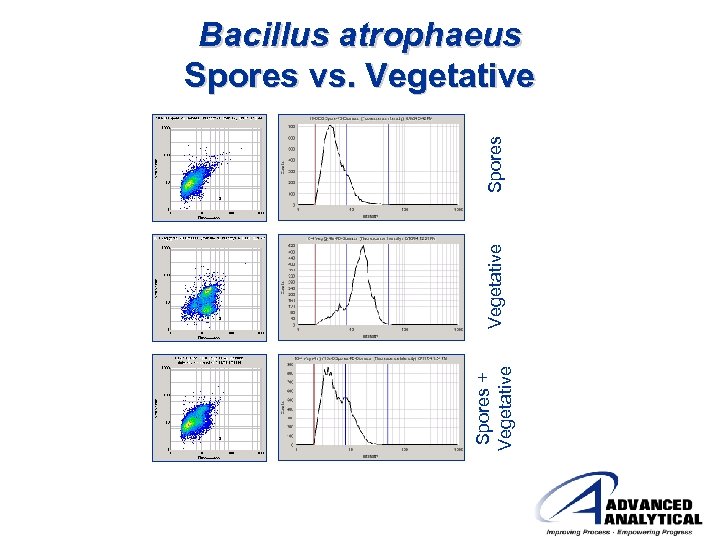 Spores + Vegetative Spores Bacillus atrophaeus Spores vs. Vegetative
Spores + Vegetative Spores Bacillus atrophaeus Spores vs. Vegetative
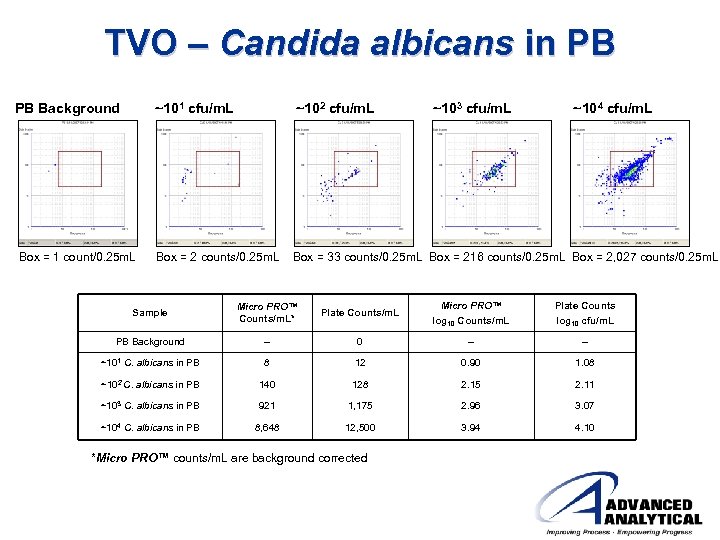 TVO – Candida albicans in PB PB Background ~101 cfu/m. L ~102 cfu/m. L ~103 cfu/m. L ~104 cfu/m. L Box = 1 count/0. 25 m. L Box = 2 counts/0. 25 m. L Box = 33 counts/0. 25 m. L Box = 216 counts/0. 25 m. L Box = 2, 027 counts/0. 25 m. L Sample Micro PRO™ Counts/m. L* Plate Counts/m. L Micro PRO™ log 10 Counts/m. L Plate Counts log 10 cfu/m. L PB Background -- 0 -- -- ~101 C. albicans in PB 8 12 0. 90 1. 08 ~102 C. albicans in PB 140 128 2. 15 2. 11 ~103 C. albicans in PB 921 1, 175 2. 96 3. 07 ~104 C. albicans in PB 8, 648 12, 500 3. 94 4. 10 *Micro PRO™ counts/m. L are background corrected
TVO – Candida albicans in PB PB Background ~101 cfu/m. L ~102 cfu/m. L ~103 cfu/m. L ~104 cfu/m. L Box = 1 count/0. 25 m. L Box = 2 counts/0. 25 m. L Box = 33 counts/0. 25 m. L Box = 216 counts/0. 25 m. L Box = 2, 027 counts/0. 25 m. L Sample Micro PRO™ Counts/m. L* Plate Counts/m. L Micro PRO™ log 10 Counts/m. L Plate Counts log 10 cfu/m. L PB Background -- 0 -- -- ~101 C. albicans in PB 8 12 0. 90 1. 08 ~102 C. albicans in PB 140 128 2. 15 2. 11 ~103 C. albicans in PB 921 1, 175 2. 96 3. 07 ~104 C. albicans in PB 8, 648 12, 500 3. 94 4. 10 *Micro PRO™ counts/m. L are background corrected
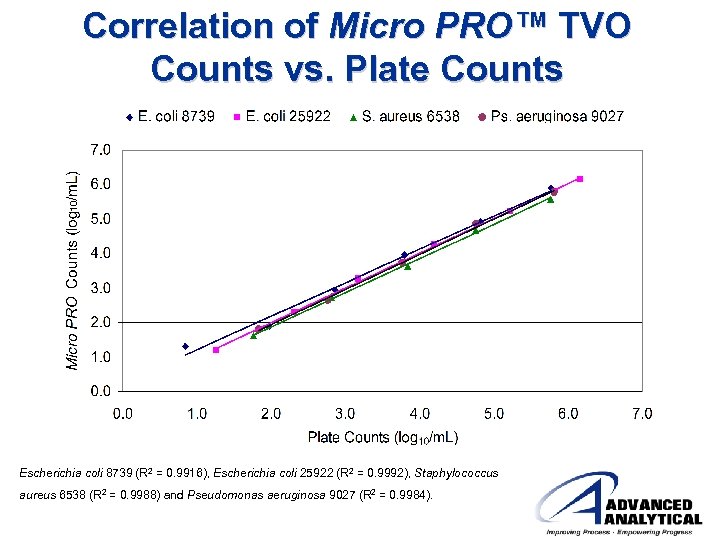 Correlation of Micro PRO™ TVO Counts vs. Plate Counts Escherichia coli 8739 (R 2 = 0. 9916), Escherichia coli 25922 (R 2 = 0. 9992), Staphylococcus aureus 6538 (R 2 = 0. 9988) and Pseudomonas aeruginosa 9027 (R 2 = 0. 9984).
Correlation of Micro PRO™ TVO Counts vs. Plate Counts Escherichia coli 8739 (R 2 = 0. 9916), Escherichia coli 25922 (R 2 = 0. 9992), Staphylococcus aureus 6538 (R 2 = 0. 9988) and Pseudomonas aeruginosa 9027 (R 2 = 0. 9984).
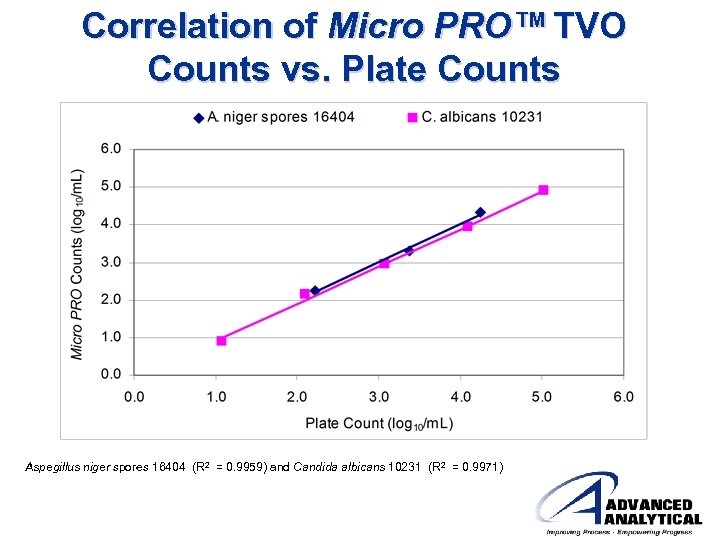 Correlation of Micro PRO™ TVO Counts vs. Plate Counts Aspegillus niger spores 16404 (R 2 = 0. 9959) and Candida albicans 10231 (R 2 = 0. 9971)
Correlation of Micro PRO™ TVO Counts vs. Plate Counts Aspegillus niger spores 16404 (R 2 = 0. 9959) and Candida albicans 10231 (R 2 = 0. 9971)
 Microorganisms Enumerated with AATI Technology • • • • • • Aeromonas caviae Aeromonas hydrophila Aspergillus niger spores Bacillus atrophaeus spores Bacillus pumilus spores Bacillus subtilis spores Bordetella bronchisceptica Brachyspira (Serpulina) hyodysenteriae Burkholderia cepacia Campylobacter jejuni Candida albicans Candida glabrata Citrobacter freundii Clostridium perfringens Cryptococcus spp. Cryptosporidium parvum oocysts Enterobacter aerogenes Enterobacter cloacae Enterococcus casseliflavus Enterococcus durans • • • • • • Enterococcus faecium Enterococcus faecalis Enterococcus gallinarum Enterococcus hirae Enterococcus mundtii Erysipelothrix rhusiopathiae Escherichia coli O 157: H 7 Escherichia coli O 25: HN Escherichia coli O 15: NM Escherichia coli O 1: NM Escherichia coli O 78: NM Escherichia coli ON: H 8 Escherichia coli ON: NM Escherichia coli O 8: HN Geobacillus stearothermophilus spores Giardia lamblia cysts Haemophilus parasuis Haemophilus somnus Halobacterium salinarum • • • • • Klebsiella pneumoniae Lactobacillus acidophilus Lactobacillus casei Lactobacillus delbrueckii Lactobacillus lindneri Lactobacillus plantarum Lactococcus lactis Lawsonia intracellularis Leptospira pomona Listeria grayi Listeria innocua Listeria ivanovii Listeria monocytogenes Listeria seeligeri Listeria welshimeri Micrococcus candicans Micrococcus luteus Moraxella bovis
Microorganisms Enumerated with AATI Technology • • • • • • Aeromonas caviae Aeromonas hydrophila Aspergillus niger spores Bacillus atrophaeus spores Bacillus pumilus spores Bacillus subtilis spores Bordetella bronchisceptica Brachyspira (Serpulina) hyodysenteriae Burkholderia cepacia Campylobacter jejuni Candida albicans Candida glabrata Citrobacter freundii Clostridium perfringens Cryptococcus spp. Cryptosporidium parvum oocysts Enterobacter aerogenes Enterobacter cloacae Enterococcus casseliflavus Enterococcus durans • • • • • • Enterococcus faecium Enterococcus faecalis Enterococcus gallinarum Enterococcus hirae Enterococcus mundtii Erysipelothrix rhusiopathiae Escherichia coli O 157: H 7 Escherichia coli O 25: HN Escherichia coli O 15: NM Escherichia coli O 1: NM Escherichia coli O 78: NM Escherichia coli ON: H 8 Escherichia coli ON: NM Escherichia coli O 8: HN Geobacillus stearothermophilus spores Giardia lamblia cysts Haemophilus parasuis Haemophilus somnus Halobacterium salinarum • • • • • Klebsiella pneumoniae Lactobacillus acidophilus Lactobacillus casei Lactobacillus delbrueckii Lactobacillus lindneri Lactobacillus plantarum Lactococcus lactis Lawsonia intracellularis Leptospira pomona Listeria grayi Listeria innocua Listeria ivanovii Listeria monocytogenes Listeria seeligeri Listeria welshimeri Micrococcus candicans Micrococcus luteus Moraxella bovis
 Microorganisms Enumerated with AATI Technology • • • • • • Mycoplasma bovis Mycoplasma hyopneumoniae Nannocystis exedens Oxalobacter formigenes Pantoea agglomerans Pasteurella multocida Pediococcus acidilactici Pediococcus damnosus Proteus mirabilis Pseudomonas aeruginosa Pseudomonas fluorescens Pseudomonas putida Ralstonia pickettii Raoutella terrigena Saccharomyces cerevisiae Salmonella adelaide Salmonella anatum Salmonella choleraesuis Salmonella dublin Salmonella enteriditis Salmonella hadar Salmonella heidelberg Salmonella iverness • • • Salmonella schalwijk Salmonella typhimurium Salmonella worthington Serratia marcescens Shigella boydii Staphylococcus aureus Staphylococcus epidermidis Staphylococcus saprophyticus Stenotrophomonas maltophila Streptococcus bovis Streptococcus equinus Streptococcus pyogenes
Microorganisms Enumerated with AATI Technology • • • • • • Mycoplasma bovis Mycoplasma hyopneumoniae Nannocystis exedens Oxalobacter formigenes Pantoea agglomerans Pasteurella multocida Pediococcus acidilactici Pediococcus damnosus Proteus mirabilis Pseudomonas aeruginosa Pseudomonas fluorescens Pseudomonas putida Ralstonia pickettii Raoutella terrigena Saccharomyces cerevisiae Salmonella adelaide Salmonella anatum Salmonella choleraesuis Salmonella dublin Salmonella enteriditis Salmonella hadar Salmonella heidelberg Salmonella iverness • • • Salmonella schalwijk Salmonella typhimurium Salmonella worthington Serratia marcescens Shigella boydii Staphylococcus aureus Staphylococcus epidermidis Staphylococcus saprophyticus Stenotrophomonas maltophila Streptococcus bovis Streptococcus equinus Streptococcus pyogenes
 Installation, Training, Validation & Support for the Micro PRO™ Pre-Installation • Feasibility is demonstrated by providing information on similar applications or performing a feasibility study • Potential clients can contact current users of the Micro PRO™ technology • Potential clients are welcome to visit the Ames, IA facility for a hands-on demonstration Training • Training for up to two employees at the Ames, IA facility is included in the purchase of the Micro PRO™ system Installation • Advanced Analytical issues an Installation Checklist to ensure the client’s lab is equipped with the materials and environment necessary to operate the Micro PRO™ • Micro PRO™ is installed by Advanced Analytical personnel using Installation and Operation Qualification (IQ/OQ) documents
Installation, Training, Validation & Support for the Micro PRO™ Pre-Installation • Feasibility is demonstrated by providing information on similar applications or performing a feasibility study • Potential clients can contact current users of the Micro PRO™ technology • Potential clients are welcome to visit the Ames, IA facility for a hands-on demonstration Training • Training for up to two employees at the Ames, IA facility is included in the purchase of the Micro PRO™ system Installation • Advanced Analytical issues an Installation Checklist to ensure the client’s lab is equipped with the materials and environment necessary to operate the Micro PRO™ • Micro PRO™ is installed by Advanced Analytical personnel using Installation and Operation Qualification (IQ/OQ) documents
 Installation, Training, Validation & Support for the Micro PRO™ Validation • Advanced Analytical provides a validation guide and protocols for several applications to assist in the implementation process • Provide onsite validation support and testing • Conduct customer validation testing at our Ames, IA facility On-going Support • Comprehensive support in the US is provided by Advanced Analytical’s team of microbiologists and service technicians • International equipment and application support is provided by Advanced Analytical and our extensive network of distributors • Preventive maintenance program • 21 CFR Part 11 compliant software
Installation, Training, Validation & Support for the Micro PRO™ Validation • Advanced Analytical provides a validation guide and protocols for several applications to assist in the implementation process • Provide onsite validation support and testing • Conduct customer validation testing at our Ames, IA facility On-going Support • Comprehensive support in the US is provided by Advanced Analytical’s team of microbiologists and service technicians • International equipment and application support is provided by Advanced Analytical and our extensive network of distributors • Preventive maintenance program • 21 CFR Part 11 compliant software
 The Micro PRO™ Delivers on the Promise of Rapid Microbiology • Versatile system covering the many aspects of microbial testing (qualitative and quantitative modes of operation) • Automated, high throughput processing (up to 20 samples/hour) with unattended analysis • Reagents stable at room temperature for 7 days • Easy to use and maintain Media kit containing all necessary components Intuitive, 21 CFR compliant software Preventive maintenance agreements available • Excellent technical and validation support
The Micro PRO™ Delivers on the Promise of Rapid Microbiology • Versatile system covering the many aspects of microbial testing (qualitative and quantitative modes of operation) • Automated, high throughput processing (up to 20 samples/hour) with unattended analysis • Reagents stable at room temperature for 7 days • Easy to use and maintain Media kit containing all necessary components Intuitive, 21 CFR compliant software Preventive maintenance agreements available • Excellent technical and validation support
 THANK YOU www. aati-us. com
THANK YOU www. aati-us. com
 RBD 3000 Customer Case Studies
RBD 3000 Customer Case Studies
 VISTAKON (J&J)
VISTAKON (J&J)
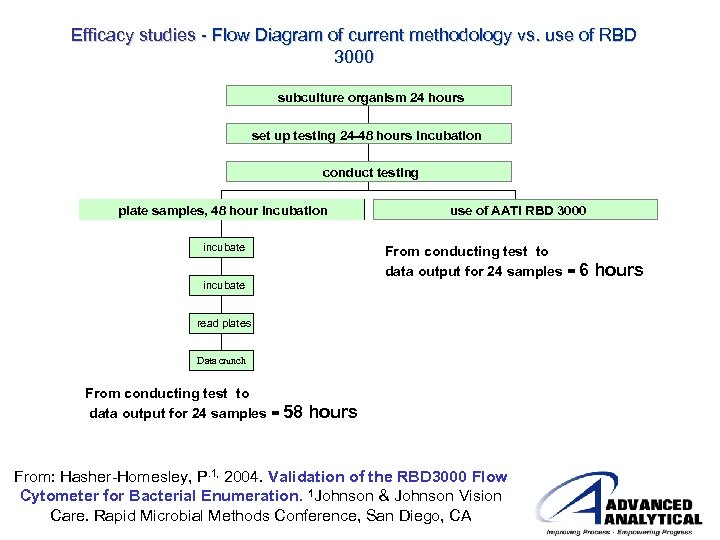 Efficacy studies - Flow Diagram of current methodology vs. use of RBD 3000 subculture organism 24 hours set up testing 24 -48 hours incubation conduct testing plate samples, 48 hour incubation incubate use of AATI RBD 3000 From conducting test to data output for 24 samples = 6 incubate read plates Data crunch data From conducting test to data output for 24 samples = 58 hours From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA hours
Efficacy studies - Flow Diagram of current methodology vs. use of RBD 3000 subculture organism 24 hours set up testing 24 -48 hours incubation conduct testing plate samples, 48 hour incubation incubate use of AATI RBD 3000 From conducting test to data output for 24 samples = 6 incubate read plates Data crunch data From conducting test to data output for 24 samples = 58 hours From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA hours
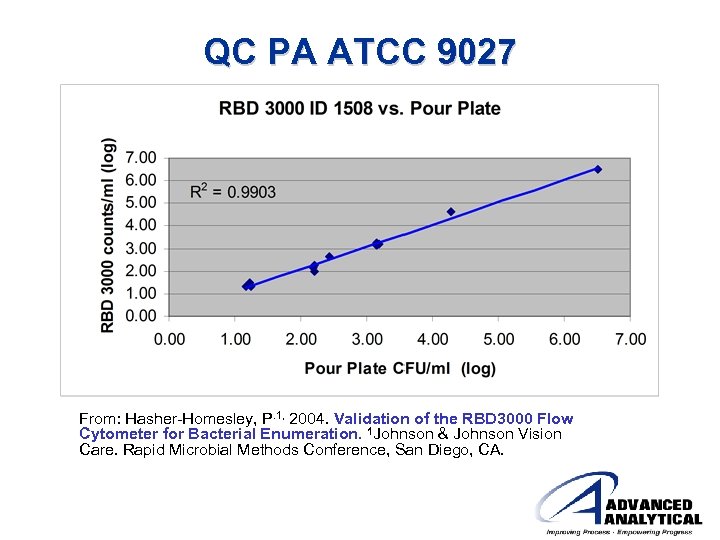 QC PA ATCC 9027 From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
QC PA ATCC 9027 From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
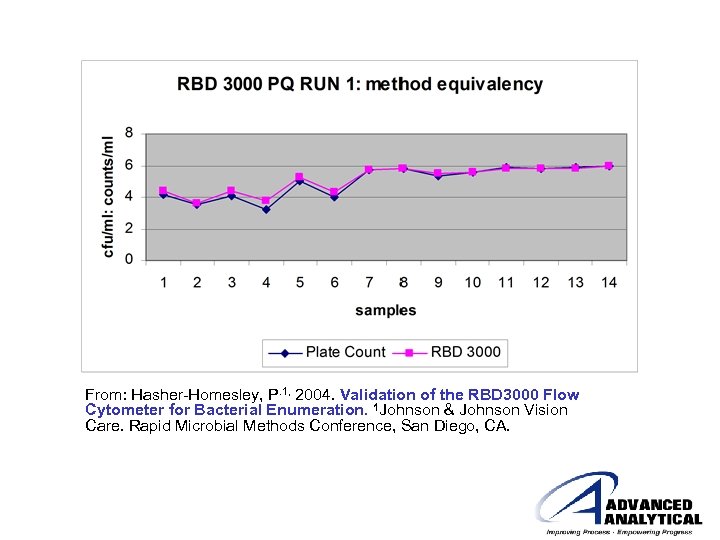 From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
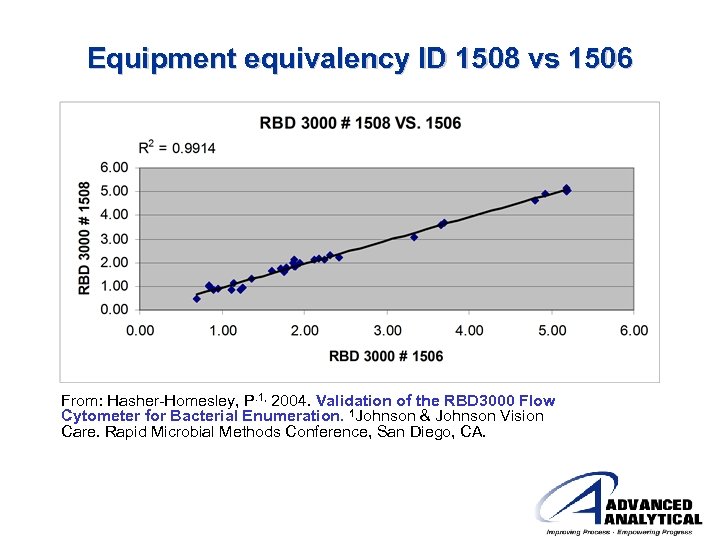 Equipment equivalency ID 1508 vs 1506 From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
Equipment equivalency ID 1508 vs 1506 From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
 Annual Cost for Efficacy studies Current method vs. RBD 3000 system • Current method Estimated annual expenditure for Efficacy studies • RBD 3000 Estimated 1 st year expenditure for Efficacy studies using the RBD 3000 system $126, 640 $219, 960 Estimated 1 st year savings of $93, 320 After 1 year warranty maintenance contract $9600 Estimated thereafter savings of $172, 320 Estimated annual expenditure for Efficacy studies $47, 640 From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
Annual Cost for Efficacy studies Current method vs. RBD 3000 system • Current method Estimated annual expenditure for Efficacy studies • RBD 3000 Estimated 1 st year expenditure for Efficacy studies using the RBD 3000 system $126, 640 $219, 960 Estimated 1 st year savings of $93, 320 After 1 year warranty maintenance contract $9600 Estimated thereafter savings of $172, 320 Estimated annual expenditure for Efficacy studies $47, 640 From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
 Applications • BI Enumerations • In-Process and End-Use solution testing • Label claim enumerations from outside vendor sources i. e. quanti-cults® and spore preparations • Routine finished goods Bioburden Testing • Microbial adhesion assays • Sample purity check From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
Applications • BI Enumerations • In-Process and End-Use solution testing • Label claim enumerations from outside vendor sources i. e. quanti-cults® and spore preparations • Routine finished goods Bioburden Testing • Microbial adhesion assays • Sample purity check From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
 Advantages of RBD 3000 Rapid bacteria detection • Counting organisms in real time within a defined volume Total bacterial testing or specific pathogen testing • Non-specific labeling with fluorescent probes • Organism specific labeling with selective fluorescent antibodies or gene probes Automated sample preparation and testing • Real time validation • 42 -position auto sampler for high sample throughput Fast, accurate and easy to use • Accurate detection 101 -106 cfu/ml and reproducibility shown at 2 -3 x 101 • Detection 0. 1 micron and larger • Red laser excitation for reduction in background signal of non-bacterial origin Standardized testing protocols • In house method development in the form of application notes providing customers with working protocols On system reagents for unattended operation • Reducing chances of cross contamination and human error 21 CFR Part 11 Compliant From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
Advantages of RBD 3000 Rapid bacteria detection • Counting organisms in real time within a defined volume Total bacterial testing or specific pathogen testing • Non-specific labeling with fluorescent probes • Organism specific labeling with selective fluorescent antibodies or gene probes Automated sample preparation and testing • Real time validation • 42 -position auto sampler for high sample throughput Fast, accurate and easy to use • Accurate detection 101 -106 cfu/ml and reproducibility shown at 2 -3 x 101 • Detection 0. 1 micron and larger • Red laser excitation for reduction in background signal of non-bacterial origin Standardized testing protocols • In house method development in the form of application notes providing customers with working protocols On system reagents for unattended operation • Reducing chances of cross contamination and human error 21 CFR Part 11 Compliant From: Hasher-Homesley, P. 1, 2004. Validation of the RBD 3000 Flow Cytometer for Bacterial Enumeration. 1 Johnson & Johnson Vision Care. Rapid Microbial Methods Conference, San Diego, CA.
 Personal Care Products Company
Personal Care Products Company
 Evaluation performed : - Presence/Absence of bacteria in the final product - Bacterial water testing - Environmental surface monitoring on the production line
Evaluation performed : - Presence/Absence of bacteria in the final product - Bacterial water testing - Environmental surface monitoring on the production line
 Presence/Absence 1. Dilute 1 m. L product in 9 m. L Enrichment Broth 2. Place samples at 32 o. C and rock for 18 -24 hours 3. Dilute enriched samples 10 -3 in 10 m. M PB, 40µm filter if necessary 4. Dispense 3 m. L sample into a 5 m. L snap cap tube 5. Analyze on the RBD 3000 for TVO Interpretation of Results: Enriched samples are considered positive if they are 10 x the background
Presence/Absence 1. Dilute 1 m. L product in 9 m. L Enrichment Broth 2. Place samples at 32 o. C and rock for 18 -24 hours 3. Dilute enriched samples 10 -3 in 10 m. M PB, 40µm filter if necessary 4. Dispense 3 m. L sample into a 5 m. L snap cap tube 5. Analyze on the RBD 3000 for TVO Interpretation of Results: Enriched samples are considered positive if they are 10 x the background
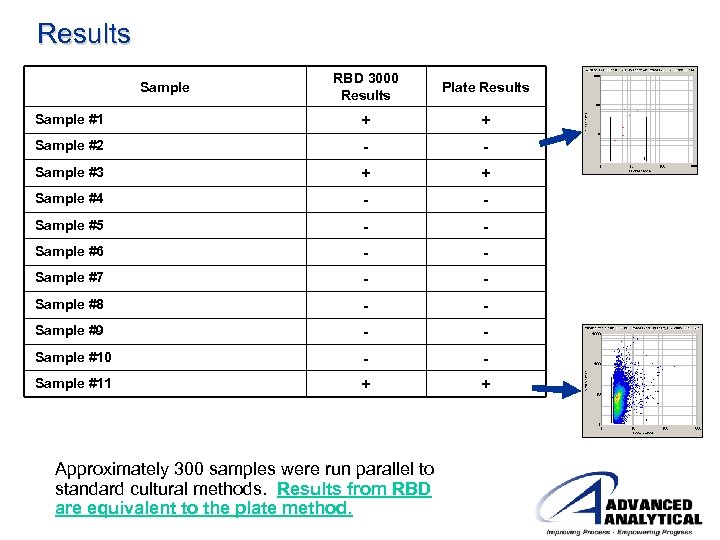 Results RBD 3000 Results Plate Results Sample #1 + + Sample #2 - - Sample #3 + + Sample #4 - - Sample #5 - - Sample #6 - - Sample #7 - - Sample #8 - - Sample #9 - - Sample #10 - - Sample #11 + + Sample Approximately 300 samples were run parallel to standard cultural methods. Results from RBD are equivalent to the plate method.
Results RBD 3000 Results Plate Results Sample #1 + + Sample #2 - - Sample #3 + + Sample #4 - - Sample #5 - - Sample #6 - - Sample #7 - - Sample #8 - - Sample #9 - - Sample #10 - - Sample #11 + + Sample Approximately 300 samples were run parallel to standard cultural methods. Results from RBD are equivalent to the plate method.
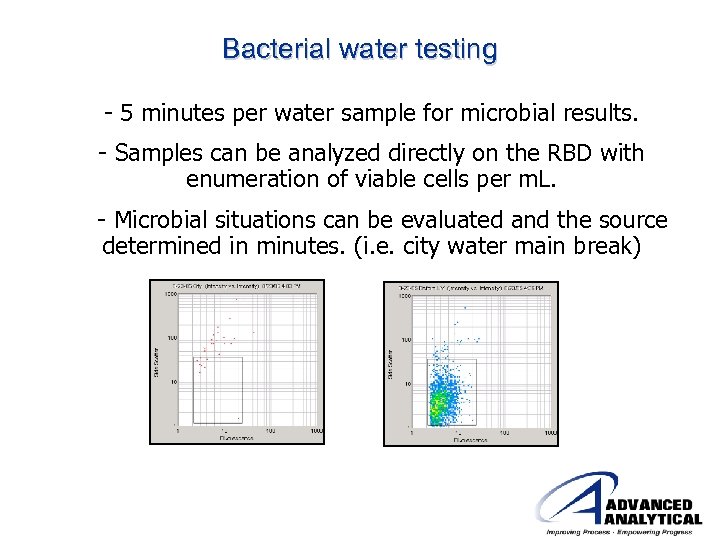 Bacterial water testing - 5 minutes per water sample for microbial results. - Samples can be analyzed directly on the RBD with enumeration of viable cells per m. L. - Microbial situations can be evaluated and the source determined in minutes. (i. e. city water main break)
Bacterial water testing - 5 minutes per water sample for microbial results. - Samples can be analyzed directly on the RBD with enumeration of viable cells per m. L. - Microbial situations can be evaluated and the source determined in minutes. (i. e. city water main break)
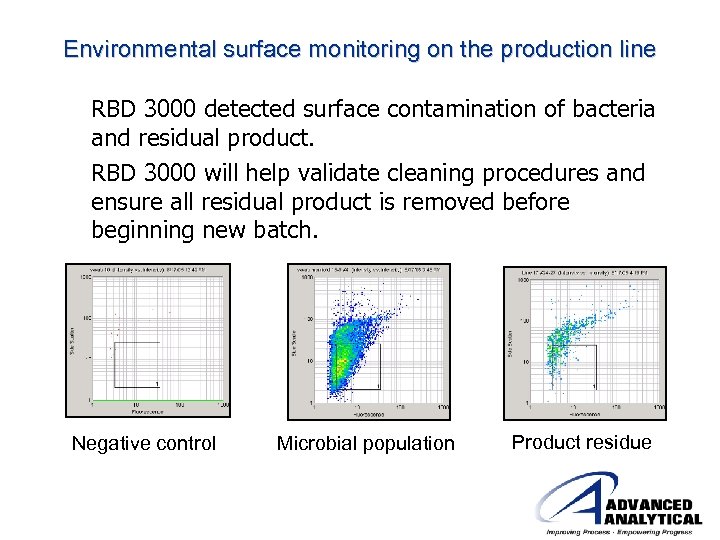 Environmental surface monitoring on the production line RBD 3000 detected surface contamination of bacteria and residual product. RBD 3000 will help validate cleaning procedures and ensure all residual product is removed before beginning new batch. Negative control Microbial population Product residue
Environmental surface monitoring on the production line RBD 3000 detected surface contamination of bacteria and residual product. RBD 3000 will help validate cleaning procedures and ensure all residual product is removed before beginning new batch. Negative control Microbial population Product residue
 Conclusions • Capable of detecting bacterial contamination using Presence/Absence test in finished goods • Capable to enumerate bacterial contamination in DI water. Results were obtained within hours after pulling samples • Environmental surface monitoring tests on the RBD 3000 detected microbial contaminants. • Unexpected result showed residual product could also be seen after cleaning. • Pass/Fail criteria were able to be set in RBD 3000 to automatically tell user if samples are good/bad
Conclusions • Capable of detecting bacterial contamination using Presence/Absence test in finished goods • Capable to enumerate bacterial contamination in DI water. Results were obtained within hours after pulling samples • Environmental surface monitoring tests on the RBD 3000 detected microbial contaminants. • Unexpected result showed residual product could also be seen after cleaning. • Pass/Fail criteria were able to be set in RBD 3000 to automatically tell user if samples are good/bad
 Procter & Gamble
Procter & Gamble
 Loose Adsorbents study • As a screener, loose adsorbents are placed in water to test their effectiveness on bacterial removal • Enumerates the removal efficiency and potential capacity for microbial adsorption • Prior to counting, the sample is passed through a 5 μm syringe filter to remove adsorbent • Background run to ensure filtered adsorbents had no adverse effect on RBD • For correlation, the RBD counts were initially compared to other methods (Pour plating and Colilert. TM) From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
Loose Adsorbents study • As a screener, loose adsorbents are placed in water to test their effectiveness on bacterial removal • Enumerates the removal efficiency and potential capacity for microbial adsorption • Prior to counting, the sample is passed through a 5 μm syringe filter to remove adsorbent • Background run to ensure filtered adsorbents had no adverse effect on RBD • For correlation, the RBD counts were initially compared to other methods (Pour plating and Colilert. TM) From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
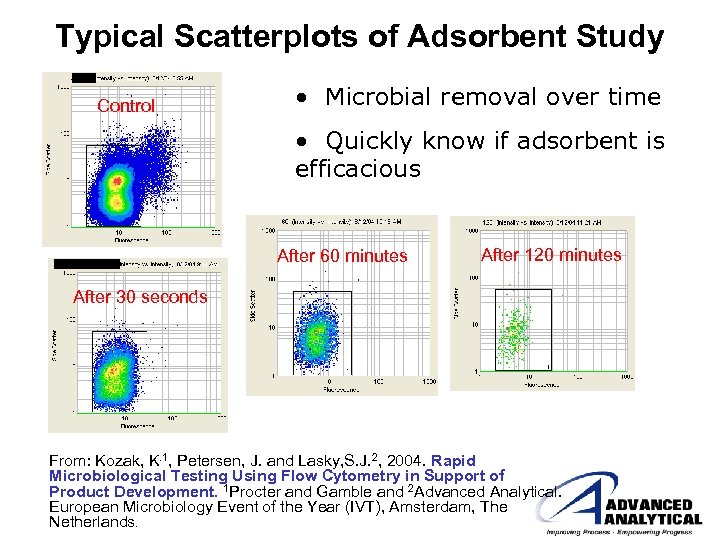 Typical Scatterplots of Adsorbent Study Control • Microbial removal over time • Quickly know if adsorbent is efficacious After 60 minutes After 120 minutes After 30 seconds From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
Typical Scatterplots of Adsorbent Study Control • Microbial removal over time • Quickly know if adsorbent is efficacious After 60 minutes After 120 minutes After 30 seconds From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
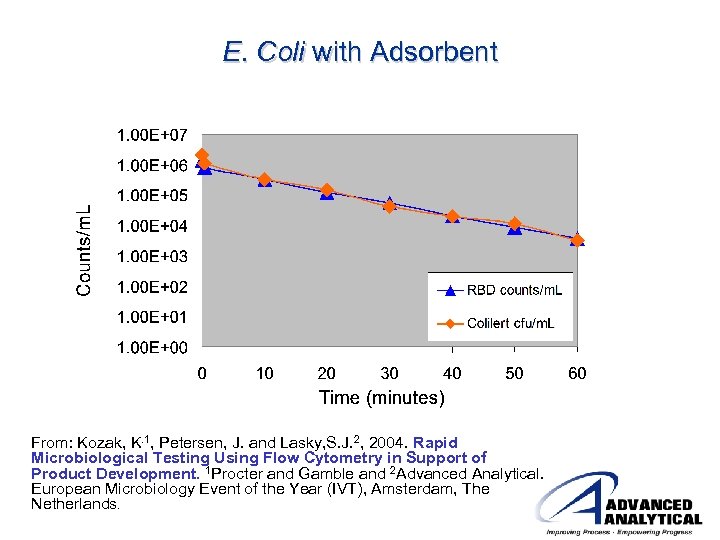 E. Coli with Adsorbent From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
E. Coli with Adsorbent From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
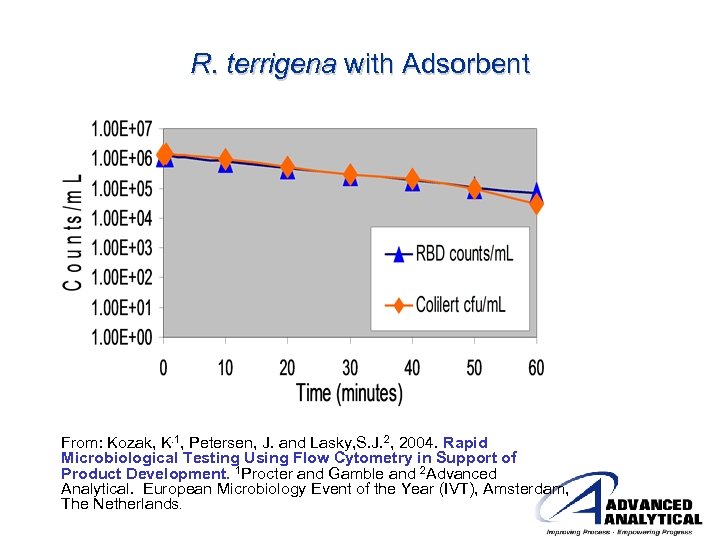 R. terrigena with Adsorbent From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
R. terrigena with Adsorbent From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
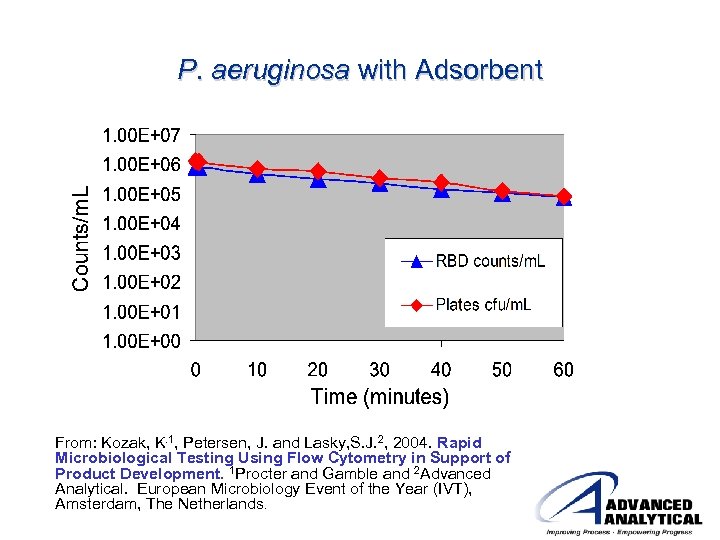 P. aeruginosa with Adsorbent From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
P. aeruginosa with Adsorbent From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
 Loose Adsorbent Screening • How is this useful to R&D? • Good correlation between traditional methods and the RBD 3000 • Rapid screener for potential adsorbents • Pretreatment (5 um filter) allows for bacterial enumeration in a complex media • Noticed differences in adsorption of microbes From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
Loose Adsorbent Screening • How is this useful to R&D? • Good correlation between traditional methods and the RBD 3000 • Rapid screener for potential adsorbents • Pretreatment (5 um filter) allows for bacterial enumeration in a complex media • Noticed differences in adsorption of microbes From: Kozak, K. 1, Petersen, J. and Lasky, S. J. 2, 2004. Rapid Microbiological Testing Using Flow Cytometry in Support of Product Development. 1 Procter and Gamble and 2 Advanced Analytical. European Microbiology Event of the Year (IVT), Amsterdam, The Netherlands.
 RBD 3000 Reference Miller, M. J. , Encyclopedia of Rapid Microbiological Methods, Volume 2, DHI Publishing, River Grove, IL, USA. 2005. Chapter 16: Steger, A. M. “Rapid enumeration of microorganisms using Advanced Analytical’s RBD 3000. ” Encyclopedia of Rapid Microbiological Methods, Volume 2. Ed. M. J. Miller. River Grove, IL, USA: DHI Publishing, LLC, 2005. (AATI) Chapter 17: Kozak, K. C. and D. E. Langworthy. “Rapid Microbial Counting by Flow Cytometry: Validation and Implementation for Research and Development (R&D) Applications. ” Encyclopedia of Rapid Microbiological Methods, Volume 2. Ed. M. J. Miller. River Grove, IL, USA: DHI Publishing, LLC, 2005. (P&G) Chapter 18: Homesley, P. H. “The RBD 3000 Rapid Bacterial Enumeration System as an Alternative to Traditional Pour Plate Enumeration. ” Encyclopedia of Rapid Microbiological Methods, Volume 2. Ed. M. J. Miller. River Grove, IL, USA: DHI Publishing, LLC, 2005. (J&J)
RBD 3000 Reference Miller, M. J. , Encyclopedia of Rapid Microbiological Methods, Volume 2, DHI Publishing, River Grove, IL, USA. 2005. Chapter 16: Steger, A. M. “Rapid enumeration of microorganisms using Advanced Analytical’s RBD 3000. ” Encyclopedia of Rapid Microbiological Methods, Volume 2. Ed. M. J. Miller. River Grove, IL, USA: DHI Publishing, LLC, 2005. (AATI) Chapter 17: Kozak, K. C. and D. E. Langworthy. “Rapid Microbial Counting by Flow Cytometry: Validation and Implementation for Research and Development (R&D) Applications. ” Encyclopedia of Rapid Microbiological Methods, Volume 2. Ed. M. J. Miller. River Grove, IL, USA: DHI Publishing, LLC, 2005. (P&G) Chapter 18: Homesley, P. H. “The RBD 3000 Rapid Bacterial Enumeration System as an Alternative to Traditional Pour Plate Enumeration. ” Encyclopedia of Rapid Microbiological Methods, Volume 2. Ed. M. J. Miller. River Grove, IL, USA: DHI Publishing, LLC, 2005. (J&J)


