5ac512186c835c7c7ff2281e7a5c2090.ppt
- Количество слайдов: 47

Acute Leukemia and Intracerebral Hemorrhage Tanya Wildes April 22, 2006

Disclosure: Tanya Wildes, M. D. Dr. Tanya Wildes has no relevant financial interests to disclose.

Disclosure: <insert name>, M. D. Tanya Wildes, M. D. has financial interests to disclose. Potential conflicts of interest have been resolved. Research Support / Grants None Stock/Equity (any amount) None Consulting / Employment None Speakers Bureau / Honoraria None Other None 3

Case #1 31 year old female with AML M 5 presents with neutropenic fevers after her first cycle of consolidation chemotherapy. n She also complains of intermittent headaches since she underwent intrathecal chemotherapy two weeks ago. Headaches are aching, bifrontal, worse when supine. n

Case #1 n Physical Exam Temp 38. 6 P 116 n Neuro exam was nonfocal n n Labs: WBC 0. 1 n Hgb 9. 3 n Plt 22 n PT 15. 3 n PTT 36. 0 n
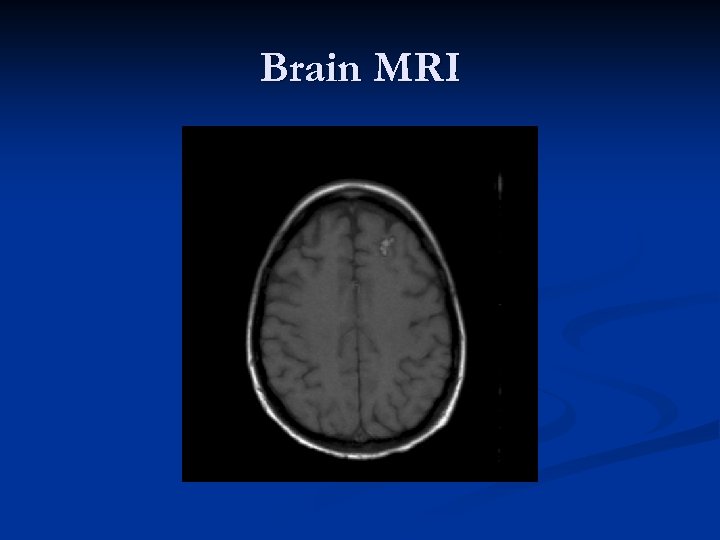
Brain MRI

Case #1 n n n MRI demonstrates parenchymal hemorrhage left frontal lobe that measures 10 x 7 mm. Neurosurgery was consulted; they recommended transfusion of platelets with goal platelet count over 100, 000. Neutropenic fevers were treated with cefepime and supportive care. Follow-up head CT two weeks later showed no increase in the size of hemorrhage. The patient was discharged home once neutropenic fevers resolved.

Case #2 n n n 42 year old female with HTN, DM presented with 1 week of headache. No history of trauma. She was conversant and neurologically intact upon arrival to ED. n Initial labs: n n n WBC 218. 4 Hgb 9. 7 Platelet 20 PT 19. 0 PTT 39. 9
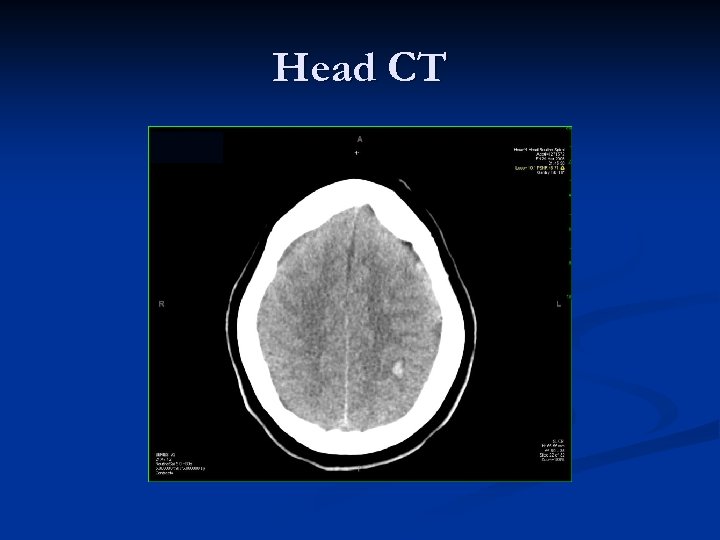
Head CT

Head CT IMPRESSION: 1. SMALL (10. 7 x 8. 5 mm) PARENCHYMAL HEMORRHAGE AT THE LEFT PARIETAL GREY-WHITE MATTER JUNCTION. 2. SUGGESTION OF VERY MILD EDEMA IN LEFT HEMISPHERE WITH 5 MM MIDLINESHIFT TOWARDS THE RIGHT. n

Case #2 Patient was transferred to BJH BMT service. n On arrival, patient was conversant with normal vital signs. n Peripheral smear revealed abundant blasts, some with bilobed nuclei, minimal granulation, occasional auer rods. n

Case #2 Neurosurgery was consulted. n They recommended correction of her thrombocytopenia and coagulopathy. n Within 3 hours of arrival, the patient’s level of consciousness declined. n She was intubated for airway protection. n A repeat head CT was performed. n
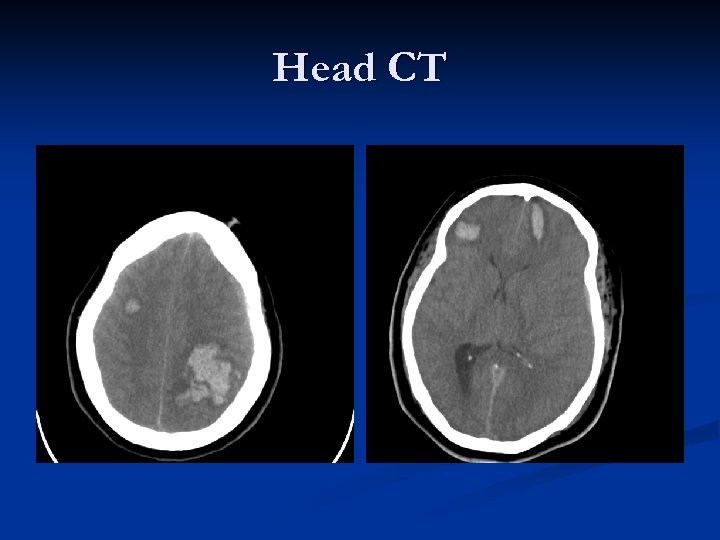
Head CT

Head CT n n FINDINGS: Intraparenchymal hemorrhage in the right parietal lobe has intervally increased in size, measuring approximately 5 cm x 3. 3 cm with extensive surrounding vasogenic edema. Interval development of a new, smaller foci of intraparenchymal hemorrhage within the bilateral frontal lobes and right temporal lobe, as well. Mass effect upon the left ventricle, with near effacement of the temporal and occipital horns. There is slight rightward midline shift.

Case #2 Neurosurgery then recommended recombinant activated factor VII. n Despite aggressive medical intervention, the patient expired within hours of her initial presentation. n
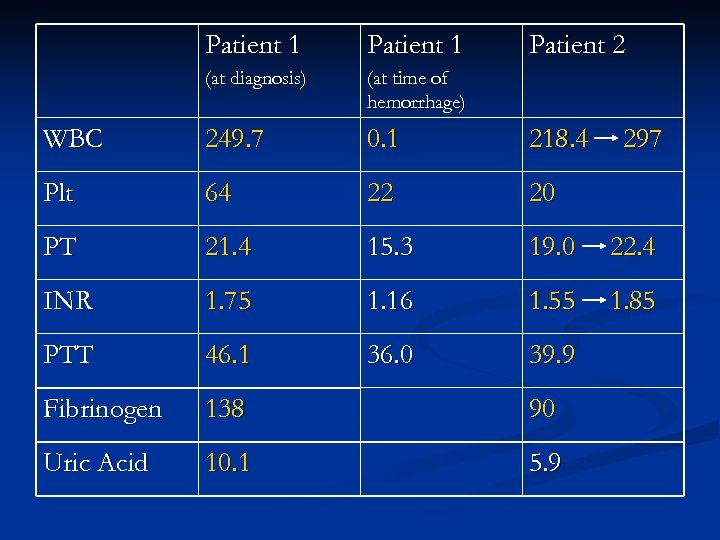
Patient 1 Patient 2 (at diagnosis) (at time of hemorrhage) WBC 249. 7 0. 1 218. 4 Plt 64 22 20 PT 21. 4 15. 3 19. 0 22. 4 INR 1. 75 1. 16 1. 55 1. 85 PTT 46. 1 36. 0 39. 9 Fibrinogen 138 90 Uric Acid 10. 1 5. 9 297

Clinical questions How common is intracranial hemorrhage among leukemia patients? n What clinical factors account for differences in severity of intracranial hemorrhage? n Is there evidence to support the use of recombinant factor VII in our patient population? n

Fatal Intracranial Hemorrhage in Blast Crisis NEJM 1957 n n n Case series N=100 81 deaths 18 deaths due to intracranial hemorrhage (22%) Cause of death WBC >300 WBC<300 ICH Other 9 (69%) 4 9 (13%) 59 18 p<0. 001 63 13 68 81 Fritz, RD, et al. The association of fatal intracranial hemorrhage and “blastic crisis” in patients with acute leukemia. NEJM 1959; 261(2): 59 -64.

Fatal Intracranial Hemorrhage in Blast Crisis NEJM 1957 WBC > 300 WBC <300 >90% blasts 8/9 (89%) 2/9 (22%) P = 0. 015 Platelet Range 8 -205 Range 4 -10 P<0. 01 Other sites of bleeding 4/9 (44%) 9/9 (100%) P = 0. 03 Uric Acid 10. 5 3. 8 Time from diagnosis to hemorrhage 6 weeks (0 -14 months) 11 months (3 -15 months) Autopsy Leukemic nodules associated with intraparenchymal hemorrhages in white matter P<0. 05 Subdural or subarachnoid hemorrhage

ICH in the contemporary era n Autopsy series of 3426 patients with non-CNS cancer 453 patients with leukemia were autopsied n 69/453 (15. 2%) patients with leukemia had ICH n n 71% of ICH were symptomatic n 7% (9/129) of ALL patients had ICH n 55. 5% were petechial or small hemorrhages n 22. 4% (43/192)of AML patients had ICH n 23. 3% were petechial or small hemorrhages Graus F, et al. Cerebrovascular complications in patients with cancer. Medicine 1985; 64(1): 16 -35.

Symptoms n Petechial or small (<2 cm) hemorrhages n n Usually asymptomatic Large (>2 cm) hemorrhages n Single n Acute headache, vomiting, focal deficits, obtundation, transtentorial herniation n Multiple n Sudden lethargy without focal deficits Graus F, et al. Cerebrovascular complications in patients with cancer. Medicine 1985; 64(1): 16 -35.
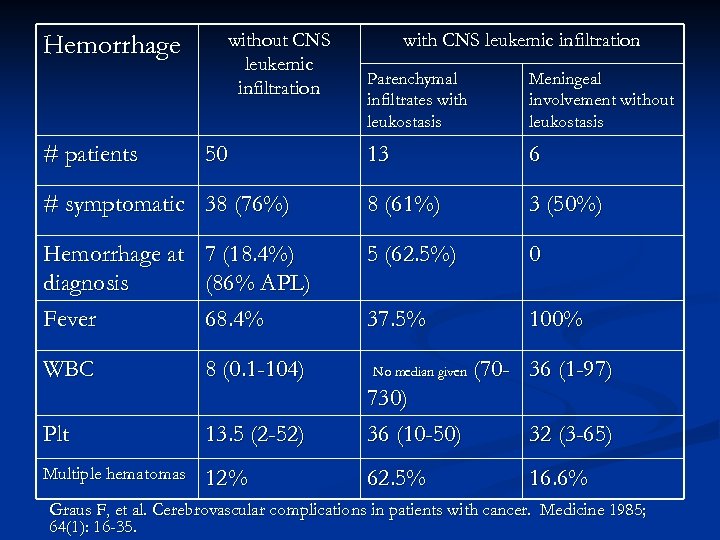
Hemorrhage without CNS leukemic infiltration with CNS leukemic infiltration Parenchymal infiltrates with leukostasis Meningeal involvement without leukostasis 13 6 # symptomatic 38 (76%) 8 (61%) 3 (50%) Hemorrhage at 7 (18. 4%) diagnosis (86% APL) Fever 68. 4% 5 (62. 5%) 0 37. 5% 100% # patients WBC 50 8 (0. 1 -104) No median given Plt 13. 5 (2 -52) 730) 36 (10 -50) Multiple hematomas 12% 62. 5% (70 - 36 (1 -97) 32 (3 -65) 16. 6% Graus F, et al. Cerebrovascular complications in patients with cancer. Medicine 1985; 64(1): 16 -35.

Summary n Patients with high WBC count at diagnosis and hemorrhage tend to have higher platelet counts and multiple hemorrhages. n n Pathophysiology likely related to leukemic infiltration with ischemic, hypoxic vasodilation and vessel rupture. Patients who develop hemorrhage after diagnosis tend to do so in the setting of sepsis, fever and marked thrombocytopenia; they tend to have solitary hemorrhages. n Pathophysiology likely related to multiple abnormalities of coagnulation.

Subdural Hematoma n 25/453 (5. 5%) patients with leukemia suffered subdural hematomas Acute confusion and lethargy were the presenting signs on all patients n Thrombocytopenia with or without DIC and sepsis were present in all patients with leukemia and SDH n None were diagnosed pre-mortem n 3/25 had meningeal leukemic infiltration n Graus F, et al. Cerebrovascular complications in patients with cancer. Medicine 1985; 64(1): 16 -35.

Treatment n Randomized controlled trials: n n n Prospective: n n r. FVIIa for intracranial hemorrhage in patients with normal coagulation parameters r. FVIIa for bleeding following hematopoietic stem cell transplantation Bleeding times after r. FVIIa in thrombocytopenic patients Case reports: n n r. FVIIa for intracranial hemorrhage in patient with refractory ITP r. FVIIa for subdural hemorrhage in AML patient with platelet alloimmunization

Treatment RCT: r. FVIIa in intracranial hemorrhage n n N=399 Inclusion criteria n n Age >18 ICH documented by CT within 3 hours of symptom onset n Exclusion criteria n n n n Thrombocytopenia Coagulopathy or DIC Sepsis Planned surgical evacuation Known AVMs, trauma, aneurysm Use of oral anticoagulants Thrombosis (MI, DVT, CVA) within 30 days Mayer SA, et al. Recombinant activated factor VII for acute intracerebral hemorrhage. NEJM 2005; 352(8): 777 -785.

Treatment RCT: r. FVIIa in intracranial hemorrhage n Intervention n Patients randomized to 40 mcg/kg r. FVIIa, 80 mcg/kg r. FVIIa, 160 mcg/kg r. FVIIa or placebo Dose was given within 1 hour of CT scan and no more than 4 hours after symptom onset Endpoints n n Hematoma size by head CT at 24 hours and 72 hours Clinical Assessment n n n Glasgow Coma Scale Rankin Scale: global outcomes National Institutes of Health Stroke Scale: neurologic impairment Barthel Index: activities of daily living Extended Glasgow Outcomes Scale: ability for self-care and independence Mayer SA, et al. Recombinant activated factor VII for acute intracerebral hemorrhage. NEJM 2005; 352(8): 777 -785.
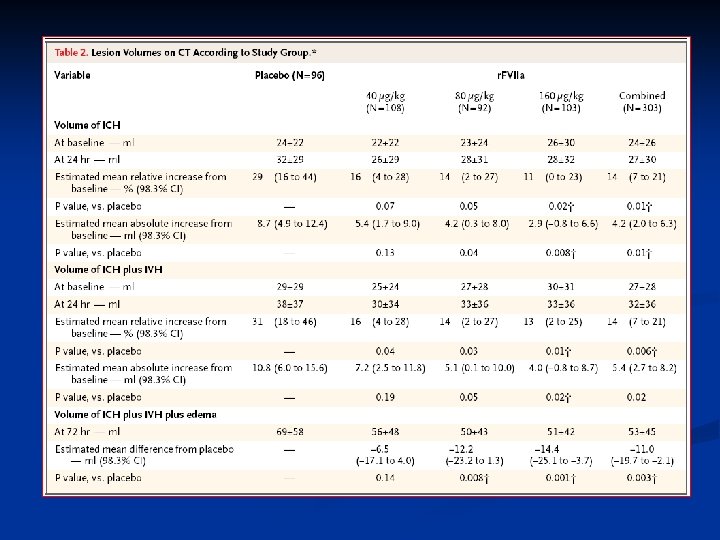
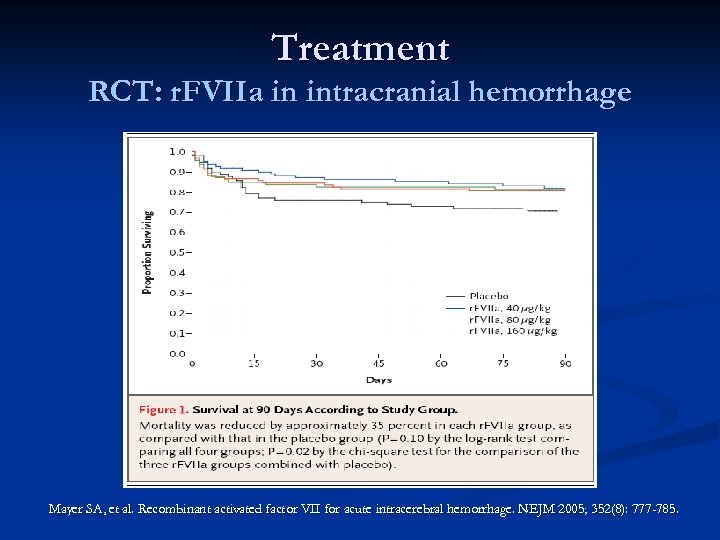
Treatment RCT: r. FVIIa in intracranial hemorrhage Mayer SA, et al. Recombinant activated factor VII for acute intracerebral hemorrhage. NEJM 2005; 352(8): 777 -785.

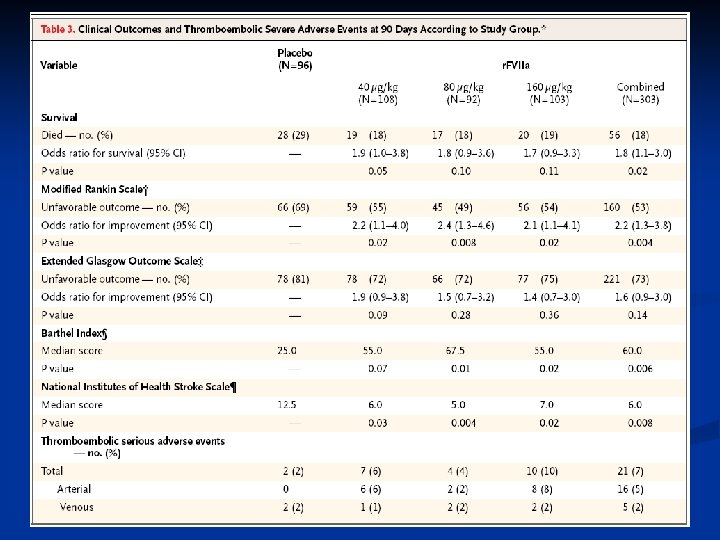
Treatment RCT: r. FVIIa in intracranial hemorrhage n Results: n Clinical Outcomes n Mortality in placebo arm 29% vs 18% in treatment arm n Patients treated with r. FVIIa showed dose-dependent improvement in outcomes on all four outcomes scales (Rankin Scale, National Institutes of Health Stroke Scale, Barthel Index and Extended Glasgow Outcomes Scale). n Thromboembolic events occurred in 2% of placebo treated patients and 7% of r. FVIIa treated patients Mayer SA, et al. Recombinant activated factor VII for acute intracerebral hemorrhage. NEJM 2005; 352(8): 777 -785.

r. FVIIa in thrombocytopenic patients Mayer study of r. FVIIa in intracerebral hemorrhage patients may not be applicable to our patients as they excluded patients with thrombocytopenia, coagulopathy and sepsis. n What evidence is there for efficacy of r. FVIIa in thrombocytopenic patients? n

Treatment r. FVIIa in thrombocytopenia n N=74 Group A: 47 patients with decreased platelet production n Group B: 27 patients with immune destruction n Dose: 50 mcg/kg or 100 mcg/kg n Positive response: Decrease in Bleeding time >2 minutes between 2 hours before and 30 minutes after r. FVIIa n Kristensen, et al. Clinical experience with recombinant factor VIIa in patients with thrombocytopenia. Haemostasis 1996; 26 S 1: 159 -164.

Treatment r. FVIIa in thrombocytopenia Platelet Count Positive Response <20 x 10^9/l 12/37 (32%) 21 -39 x 10^9/l 15/27 (56%) >40 x 10^9/l 28/41 (68%) Overall 55/105 (52%) Kristensen, et al. Clinical experience with recombinant factor VIIa in patients with thrombocytopenia. Haemostasis 1996; 26 S 1: 159 -164.

Treatment r. FVIIa in thrombocytopenia n Median reduction in bleeding time n n n 8 patients had thrombocytopenia and active bleeding n n Decreased platelet production: 14 minutes Increased platelet destruction: 5 minutes Bleeding stopped in 6 patients Theory: n Though thrombocytopenic patients have an intact intrinsic coagulation pathway, exogenous FVIIa ensures that the few platelets available are maximally activated. Kristensen, et al. Clinical experience with recombinant factor VIIa in patients with thrombocytopenia. Haemostasis 1996; 26 S 1: 159 -164.

Treatment r. FVIIa in hematopoietic stem cell transplant patients with bleeding n Prospective, randomized trial of patients undergoing autologous or allogeneic transplant n N=100 n Inclusion: Mild bleeding (score 2) x 3 days or severe to serious bleeding (score 3 or 4) n Exclusion: atherosclerotic disease, stroke or DVT within 3 months, DIC, thrombotic microangiopathy, VOD, active AML M 3, M 4 or M 5 or recent granulocyte infusion. Pihusch M, et al. Recombinant activated factor VII in treatment of bleeding complications following hematopoietic stem cell transplantation. 2005; 3: 1935 -1944.

Treatment r. FVIIa in hematopoietic stem cell transplant patients with bleeding n Treatment: r. FVIIa dose 40, 80 or 160 mcg/kg or placebo IV q 6 hours x 6 doses n “Standard management practices” n n RBC transfusion if Hgb<8 n Platelet transfusion if plt <20 x 10^9 n If diffuse alveolar hemorrhage or hemorrhagic cystitis, platelets were transfused if <75 x 10^9 n Use of antifibrinolytic agents was discouraged Pihusch M, et al. Recombinant activated factor VII in treatment of bleeding complications following hematopoietic stem cell transplantation. 2005; 3: 1935 -1944.

Treatment r. FVIIa in hematopoietic stem cell transplant patients with bleeding n Primary endpoint: change in bleeding score 2 hours after final dose of r. FVIIa n Secondary endpoints: change in bleeding score at 24, 48, 72 and 96 hours after initial dose n RBC, platelet and FFP transfusion requirements during 96 hour follow-up n Pihusch M, et al. Recombinant activated factor VII in treatment of bleeding complications following hematopoietic stem cell transplantation. 2005; 3: 1935 -1944.
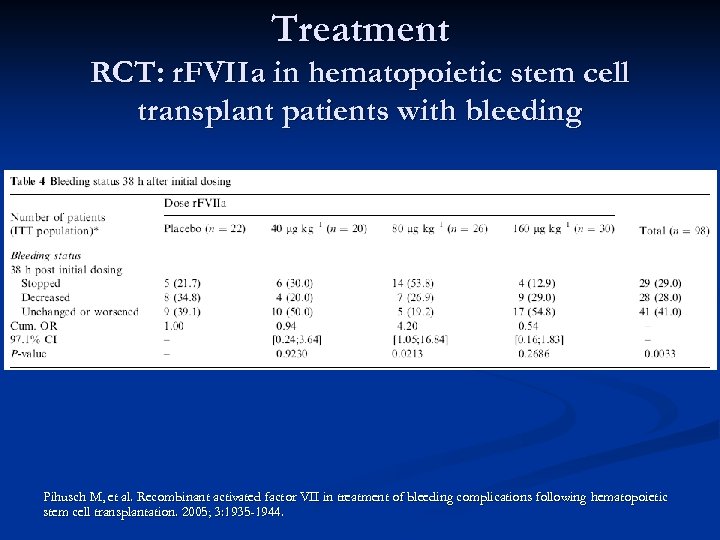
Treatment RCT: r. FVIIa in hematopoietic stem cell transplant patients with bleeding Pihusch M, et al. Recombinant activated factor VII in treatment of bleeding complications following hematopoietic stem cell transplantation. 2005; 3: 1935 -1944.
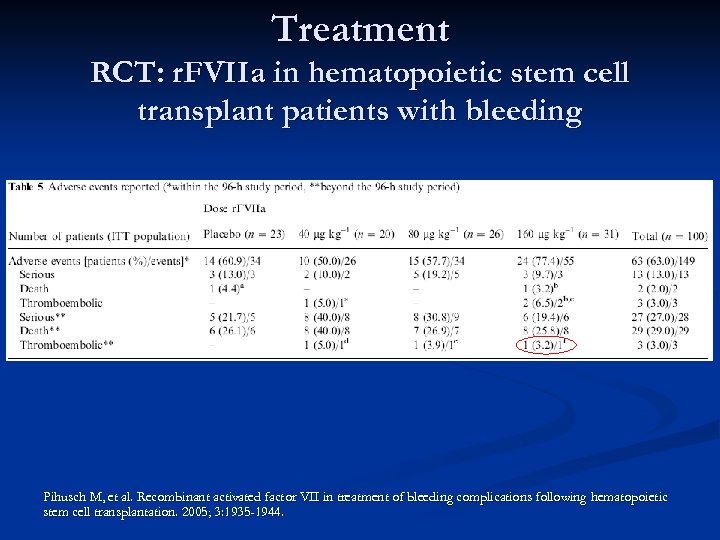
Treatment RCT: r. FVIIa in hematopoietic stem cell transplant patients with bleeding Pihusch M, et al. Recombinant activated factor VII in treatment of bleeding complications following hematopoietic stem cell transplantation. 2005; 3: 1935 -1944.

Treatment Case report: r. FVIIa in ITP patient with ICH n n n n 16 y. o. F with ITP refractory to IVIg, steroids, cyclophosphamide anti-CD 20 and anti-TNFalpha monoclonal antibiodies Presented with severe headache, N/V x 36 hours; no trauma. Platelet count - 4 x 10^9 HCT demonstrated large intraparenchymal hemorrhage. Treated with platelet transfusion, FFP, IV tranexamic acid. Started on r. FVIIa 122 mcg/kg q 2 hours, weaned to q 8 hours, then q day x 5 days. Despite transfusion of 98 u of plt, highest platelet count was 35. Serial neuro-imaging demonstrated no further hemorrhage. She was discharged after 3 weeks with no residual neurologic deficits. Barnes, C. Recombinant FVIIa in the management of intracerebral haemorrhage in severe thrombocytopenia unresponsive to platelet-enhancing treatment. Transfusion Medicine 2005; 15: 145 -150.

Treatment Case report: r. FVIIa in platelet refractory AML patient with ICH n 27 y. o. F with MDS evolved into AML. She was refractory to platelets at time of induction chemotherapy n Day +7: Subdural Hemorrhage n Treated with tranexamic acid and platelet transfusion Day +21: Head CT with stable hematoma n Day +23: hemoptysis, periorbital hematoma n Day +27: left hemiparesis; head CT demonstrated progression of SDH n Vidarsson B. Recombinant Factor VIIa for bleeding in refractory thrombocytopenia. Thromb Haemost 2001; 83: 634 -5.

Treatment Case report: r. FVIIa in platelet refractory AML patient with ICH Day +32 -33: r. FVIIa 100 mcg/kg q 2 hours x 5 doses, then q 4 hours x 6 doses. n Day +33: headache resolved, periorbital hematomas stable, no left sided weakness n Patient had no further bleeding for remainder of her course. n Patient died on Day +81 of persistent disease. n Vidarsson B. Recombinant Factor VIIa for bleeding in refractory thrombocytopenia. Thromb Haemost 2001; 83: 634 -5.

Summary n Intracerebral hemorrhage is common in acute leukemia Blast crisis – related to leukostasis n Coagulopathy n Mainstay of treatment is supportive therapy n Further study needed to determine the role of r. FVIIa in patients with leukemia and intracerebral hemorrhage n

References n n n n Barnes, C. Recombinant FVIIa in the management of intracerebral haemorrhage in severe thrombocytopenia unresponsive to platelet-enhancing treatment. Transfusion Medicine 2005; 15: 145 -150. Fritz, RD, et al. The association of fatal intracranial hemorrhage and “blastic crisis” in patients with acute leukemia. NEJM 1959; 261(2): 59 -64. Graus F, et al. Cerebrovascular complications in patients with cancer. Medicine 1985; 64(1): 16 -35. Kristensen, et al. Clinical experience with recombinant factor VIIa in patients with thrombocytopenia. Haemostasis 1996; 26 S 1: 159 -164. Mayer SA, et al. Recombinant activated factor VII for acute intracerebral hemorrhage. NEJM 2005; 352(8): 777 -785. Pihusch M, et al. Recombinant activated factor VII in treatment of bleeding complications following hematopoietic stem cell transplantation. 2005; 3: 1935 -1944. Quinones-Hinojosa, et al. Spontaneous intracerebral hemorrhage due to coagulation disorders. Neurosurg Focus 2003; 15(4): 1 -17. Vidarsson B. Recombinant Factor VIIa for bleeding in refractory thrombocytopenia. Thromb Haemost 2001; 83: 634 -5.


Fatal Intracranial Hemorrhage in Blast Crisis NEJM 1957 n Of those with WBC>300, there were 2 distinct subgroups: n n Type Subgroup A: WBC exceeded 300 >8 days before ICH, peaked at 450 -850 Subgroup B: WBC rose abruptly <2 days before ICH. A (N=5) 5/5 ALL B (N=4) 3/4 AML Plt 8 -17 20 -200 No difference in uric acid or survival. P<0. 05
5ac512186c835c7c7ff2281e7a5c2090.ppt