cc49cc34f7b2844bf59e161a600b87f8.ppt
- Количество слайдов: 19

ACRIN Annual Meeting OVERVIEW OF ECOG Robert L. Comis, M. D. Chair Eastern Cooperative Oncology Group September 22, 2011 eastern cooperative oncology group

ECOG Constitution Mission Statement The Eastern Cooperative Oncology Group (ECOG) is a multidisciplinary organization devoted to the prevention, treatment and study of neoplastic disease that will eventually lead to the control and cure of cancer. eastern cooperative oncology group 1
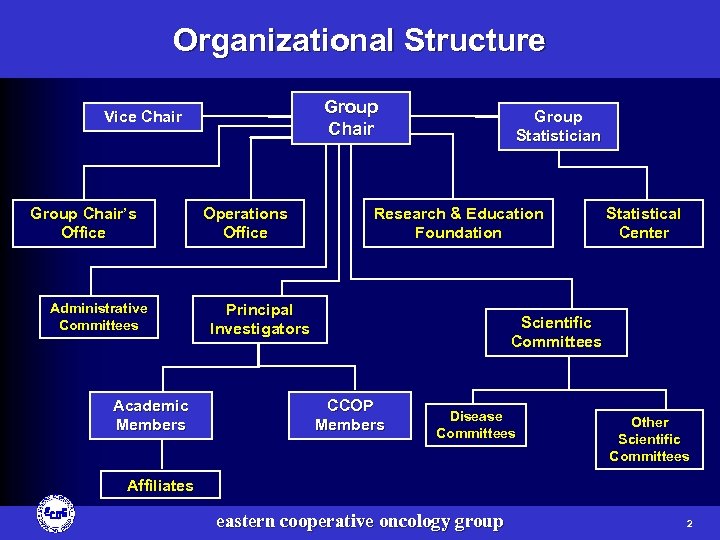
Organizational Structure Group Chair Vice Chair Group Chair’s Office Administrative Committees Academic Members Operations Office Group Statistician Research & Education Foundation Principal Investigators Statistical Center Scientific Committees CCOP Members Academic Disease Members Committees Other Scientific Committees Affiliates eastern cooperative oncology group 2

Scientific and Administrative Senior Leadership ¡ Chair – Dr. Robert Comis ¡ Vice Chair – Dr. Peter O’Dwyer ¡ Group Statistician – Dr. Robert Gray ¡ Executive Officer – Dr. Bruce Giantonio ¡ Associate Chair – Dr. Joseph Sparano ¡ Coordinating Center Co-Directors – Ms. Jean Mac. Donald and Ms. Mary Steele ¡ Director of Operations – Ms. Donna Marinucci ¡ Regulatory Officer – Dr. Robert Catalano eastern cooperative oncology group 3

Main Academic Member Institutions U 10 Funded Non-U 10 Main Members ¡ Albert Einstein Cancer Center ¡ Beth Israel Deaconess ¡ Emory University School of Medicine Medical Center Neoplasicas (Lima, Peru) ¡ Cancer Institute of New Jersey ¡ Case Western-Metro. Health Medical Center ¡ ¡ ¡ ¡ ¡ Instituto de Enfermedades Fox Chase Cancer Center Indiana University Medical Center Johns Hopkins University Mayo Clinic ¡ Ireland Cooperative Oncology Research Group ¡ New York University Medical Center ¡ Stanford University ¡ The Penn State Cancer Institute ¡ Tufts Medical Center Northwestern University ¡ University of Alabama University of Pennsylvania ¡ University of Miami University of Pittsburgh University of Wisconsin Vanderbilt University eastern cooperative oncology group 4

Primary CCOPs ¡ ¡ ¡ ¡ Colorado Cancer Research Program CCOP Cook County MBCCOP* Evanston Hospital CCOP Kalamazoo CCOP Main Line Health CCOP Marshfield Clinic CCOP Medical College of Georgia MBCCOP* Meharry Medical College MBCCOP* Metro-Minnesota CCOP Ochsner CCOP Oklahoma CCOP *Minority Based CCOP San Juan MBCCOP* SUNY Downstate MBCCOP* St Vincent Hospital Regional Center CCOP eastern cooperative oncology group 5

Administrative and Scientific Organizational Structure Administrative Committees - Audit - CRA - Data Monitoring - Executive Review - Nominating - Oncology Nursing - Patient Representative - Pharmacy - Radiation Oncology - Surgery Scientific Committees Disease Committees Other Scientific Committees - Breast - Laboratory Science - Gastrointestinal and Pathology - Genitourinary - Developmental Therapeutics - Head and Neck - Pathology Coordinating - Leukemia Office/Reference Laboratory - Lymphoma - Central Immunologic - Melanoma Monitoring Laboratory - Myeloma - Leukemia Laboratory - Thoracic - Cytogenetics DCP Funded Committees: - Patient Outcomes, Survivorship and Underserved - Prevention - Symptom Management eastern cooperative oncology group 6
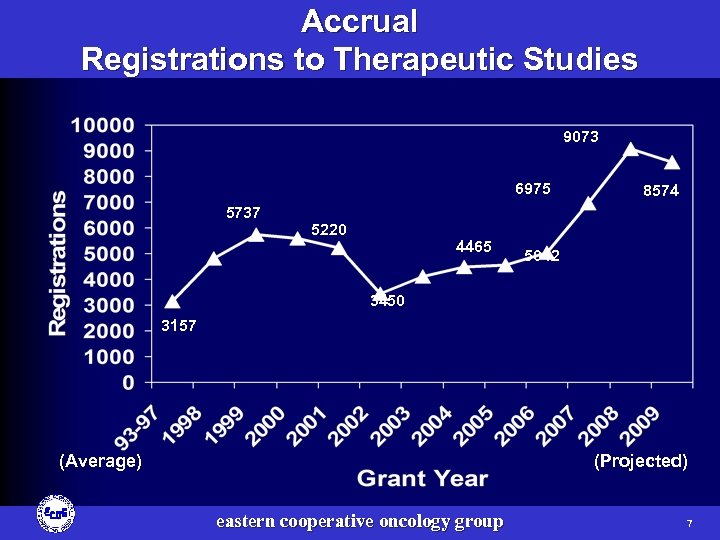
Accrual Registrations to Therapeutic Studies 9073 6975 5737 5220 4465 8574 5042 3450 3157 (Average) (Projected) eastern cooperative oncology group 7

CTSU Participation 2004 -2009 ECOG Participation in CTSU Menu Studies ECOG studies on the CTSU menu 31 CTSU studies “endorsed” by ECOG 44 CTSU patients accrued to ECOG studies 11, 417 ECOG patients accrued to CTSU studies 6, 618 ECOG patients accrued to other intergroup studies 1, 714 eastern cooperative oncology group 8

Major Accomplishments ¡ Six studies led to new standard of care and FDA indications w Bevacizumab in metastatic colon (E 3200), lung (E 4599), breast (E 2100) w Rituximab in indolent (E 1496) and aggressive lymphoma (E 4494) w Thalidomide in multiple myeloma (E 1 A 00) ¡ Two phase III adjuvant trials: w E 5103 – bevacizumab + chemotherapy in HER 2 -negative breast cancer w E 1505 – bevacizumab + chemotherapy in stage IB – IIIA NSCLC eastern cooperative oncology group 9

Major Accomplishments ¡ Head and Neck cancer: defined prognostic and predictive roles of p 53 mutations and HPV+ ¡ AML: established superiority of high dose daunorubicin induction ¡ Adult ALL: defined new adverse cytogenetic parameters ¡ Breast cancer: described VEGF SNPs related to response and toxicity; and potential new therapeutic targets (eg, GRB-7) ¡ Multiple Myeloma: established superiority of low dose dexamethasone plus lenalidomide in induction therapy eastern cooperative oncology group 10
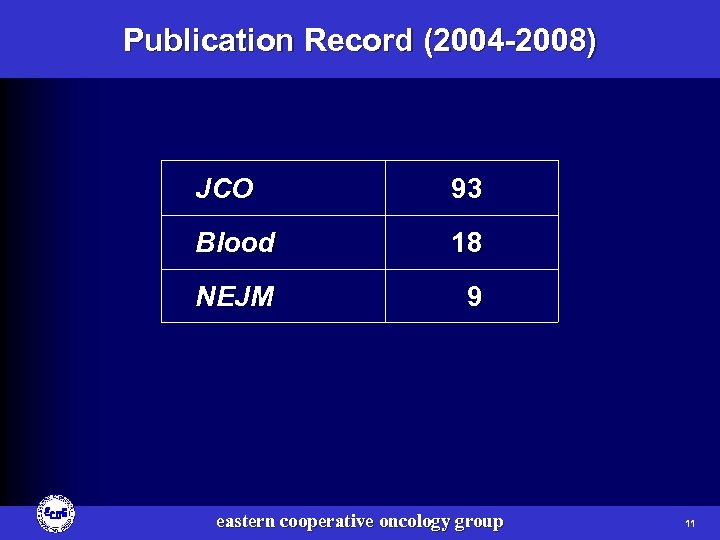
Publication Record (2004 -2008) JCO 93 Blood 18 NEJM 9 eastern cooperative oncology group 11

Peer-Review Funding: 2004 -2008 ¡ Laboratory Science and Pathology Committee (LSPC) w Reviews, comments on and approves all ECOG related grant studies prior to submission ¡ 22 R 01 and R 21 proposals were reviewed and approved by LSPC ¡ 50% of RO 1 and R 21 submissions were funded by NIH eastern cooperative oncology group 12
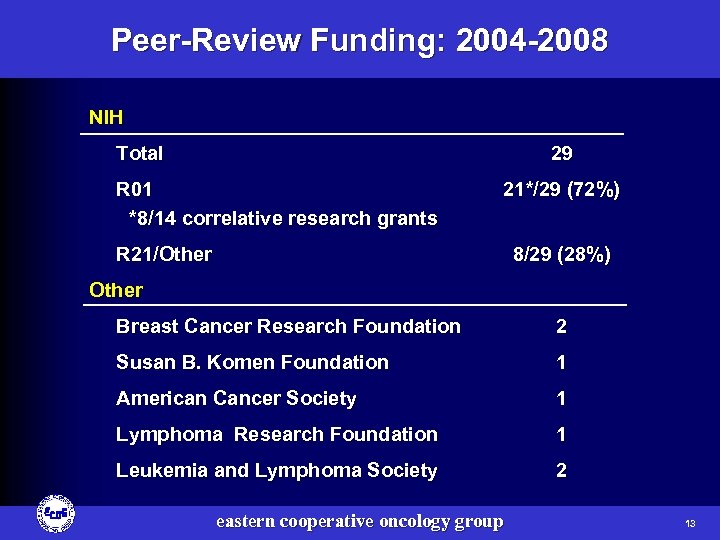
Peer-Review Funding: 2004 -2008 NIH Total 29 R 01 *8/14 correlative research grants 21*/29 (72%) R 21/Other 8/29 (28%) Other Breast Cancer Research Foundation 2 Susan B. Komen Foundation 1 American Cancer Society 1 Lymphoma Research Foundation 1 Leukemia and Lymphoma Society 2 eastern cooperative oncology group 13

Major Accomplishments ¡ Key components of our Translational Science effort ¡ Laboratory Science and Pathology Committee w PCO-RL and other core laboratories ¡ Developmental Therapeutics Committee w Pharmacogenetics subcommittee studies ¡ Leukemia Laboratory Committee and Leukemia Translational Research Laboratory ¡ Translational Science Team ¡ IT infrastructure 15 eastern cooperative oncology group 14

Major Accomplishments ¡ Established scientific infrastructure to perform large-scale biomarker driven studies ¡ Launched two large marker-driven phase III studies w E 5202 – stage II colon cancer (MSI; 18 q LOH) w TAILORx (PACCT-1) – node negative breast cancer (Oncotype-DX) 16 eastern cooperative oncology group 15
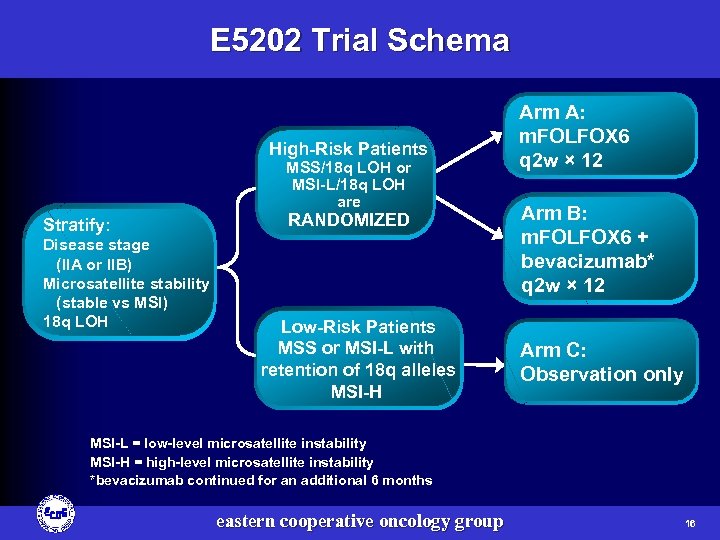
E 5202 Trial Schema High-Risk Patients MSS/18 q LOH or MSI-L/18 q LOH are Stratify: Disease stage (IIA or IIB) Microsatellite stability (stable vs MSI) 18 q LOH RANDOMIZED Low-Risk Patients MSS or MSI-L with retention of 18 q alleles MSI-H Arm A: m. FOLFOX 6 q 2 w × 12 Arm B: m. FOLFOX 6 + bevacizumab* q 2 w × 12 Arm C: Observation only MSI-L = low-level microsatellite instability MSI-H = high-level microsatellite instability *bevacizumab continued for an additional 6 months eastern cooperative oncology group 16
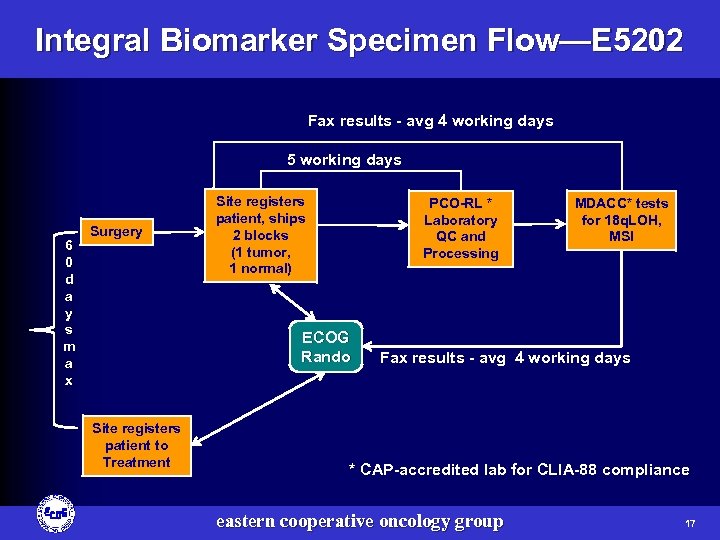
Integral Biomarker Specimen Flow—E 5202 Fax results - avg 4 working days 5 working days 6 0 d a y s m a x Surgery Site registers patient, ships 2 blocks (1 tumor, 1 normal) PCO-RL * Laboratory QC and Processing ECOG Rando Site registers patient to Treatment MDACC* tests for 18 q. LOH, MSI Fax results - avg 4 working days * CAP-accredited lab for CLIA-88 compliance eastern cooperative oncology group 17

General Themes for the Future ¡ Study “targeted” therapies with the ultimate goal of developing new potentially curative strategies ¡ Define new predictive and prognostic factors which lead to hypothesis driven clinical trials ¡ Continue to develop both laboratory-based and IT infrastructure ¡ Build upon new data from existing pivotal studies undergoing completion and analysis eastern cooperative oncology group 19
cc49cc34f7b2844bf59e161a600b87f8.ppt