c9e685d9da6f7e873fa07af7fd1a84e7.ppt
- Количество слайдов: 24

ACE Alera

Objectives 1. Explain the procedure for preparing reagents, calibrators, controls and specimen samples. 2. Identify key components of the ACE analyzer and ISE modules and explain their purpose. 3. Identify each ACE consumable and its use. 4. Describe the role of the function keys when operating the ACE system.

Objectives 7. Perform the four activities required for the ACE system daily start-up. 8. Create QC, calibration and patient requisitions. 9. Place individual sample cups and tubes and create loadlists for running batches of samples. 10. Examine patient reports to determine if the results are possible and if necessary corrective actions are required. 11. Recognize “flags” noted on a QC report and know basic troubleshooting actions. 12. Complete daily shutdown activities.

Objectives 13. List the different malfunctions that one might experience during operation. 14. List the possible causes for malfunctions and the recommended corrective action. 15. Identify the five-step process to help identify and resolve quality control problems.

Reagent Preparation • Ready to use, liquid form • Package insert details information regarding: – Preparation and storage • Room Temp (albumin, direct bilirubin, total bilirubin, calium, creatinine, magnesium, total protein, ISE calibrators A and B, ISE Cleaning and Conditioning, ISE wash, System Diluent, reference Electrode Solution) • Refrigerated( ALT, AST, CO 2, Alkaline Phosphatase, Amylase, Cholesterol, Creatinine Kinase, GGT, Glucose and many more)

Reagent Preparation • On a bottle is opened – Date and time – Initials – Check for bubbles. If bubbles present, use a transfer pipette to remove • Place an evap-cap on the top of reagent container • Bottle is then placed anywhere in the refrigerated compartment • Once the instrument door is closed, the ACE will read the dot codes on the bottle of the reagent to determine expiration dates and stability

Important reminder • Fresh reagents require calibration – GEMCAL • QC verification – Run 2 levels of control each day to verify acceptable system performance

Specimen Preparation • All serum specimens should be allowed to fully clot for 30 minutes before centrifugation • Glucose tests should be completed within 2 hours of sample collection unless the red blood cells were separated from the serum or plasma • Specific requirements are listed in package insert
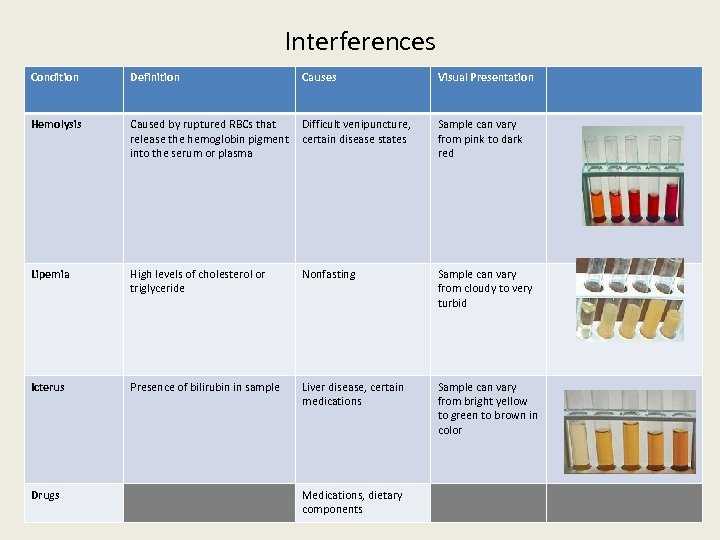
Interferences Condition Definition Causes Visual Presentation Hemolysis Caused by ruptured RBCs that release the hemoglobin pigment into the serum or plasma Difficult venipuncture, certain disease states Sample can vary from pink to dark red Lipemia High levels of cholesterol or triglyceride Nonfasting Sample can vary from cloudy to very turbid Icterus Presence of bilirubin in sample Liver disease, certain medications Sample can vary from bright yellow to green to brown in color Drugs Medications, dietary components

ACE System • Analysis module • Computer workstation – – – Computer Monitor Keyboard Priner Uninteruptible power supply
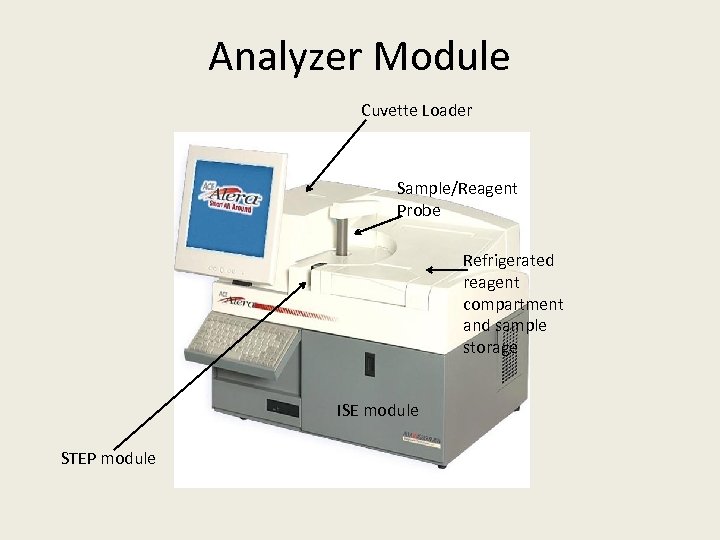
Analyzer Module Cuvette Loader Sample/Reagent Probe Refrigerated reagent compartment and sample storage ISE module STEP module

Consumables: disposable products • • Plastic sample cups Segments Seglets Well Segments Cuvettes Evap-caps Biobags

ISE Components • Valves – Control the movement of fluid and air in and out of the measuring chamber • Sample sensor – Detects proper aspiration and timing of all ISA fluids and air • Measuring Chamber – Contains 3 electrodes that measure sodium, potassium and chloride. The fourth electrode is the reference • Peristaltic Pump and Tubing • The pump transports liquids and air bubbles throughout the ISE module

Daily Operation • • • Daily Start-up Create Requisitions Load and Run Samples Interpret Results Daily Shut-down

Daily Start-Up • Verify Power Ø Look for a green light to indicate power is on • • UPS Monitor Computer Printer Ø Listen for fans • Status Checks Ø Hit Status {F 3} on keyboard, check reagent tray, calibration, sample ring and hopper to check each item Ø Prepare GEMCAL, QC products, reagents

Daily Start-Up • Maintenance – Hit Instrument Access {F 12} to check, inspect and service: • • Clean reagent compartment condensation and camera lens Re-fill container compartment Fill Hopper Empty cuvette waste box – Perform a probe alignment and cleaning – Clean the wash bath and probe pathway • ISE daily maintenance – Main menu, utilities, ISE, press enter three times to select Calibrate, Condition, Clean – Select Accept {F 10} This will take about 13 minutes

Create Requisition • Types – Quality Control • Used to order tests on QC materials • Separate requisition is needed for each control – Calibration • Used to order tests for calibration • One requisition is required for all calibrator materials in a run • When? – Calibration is expired – New lot of reagents introduced – Software/Hardware changes – Patient • Order tests on a patient

Creating a Requisition for a New Patient • Requires – Patient first and last name – ID – Accession number – Selection of tests to run

Loading and Running Samples • Loadlist or worksheet – List of each sample to be run – Shows where to place each sample cup on the segment – Created prior to running samples – Maximum of 15 samples

Loading and Running Samples • Important Points – Requisition must be created PRIOR to loading and running – All samples must be run within 4 hours of loading

Placing Sample Tubes • STEP: Sample Tube Entry Port – Allows sample to be introduced directly to system – Detects level of sample and withdraws sample for analysis

Results Interpretation • Quality Control – Verify that all results are in range, if not trouble shoot. • Patient – Verify that no “flags” are present • Examples: “H” or “L” – Check for critical values • Examples: “HH” or “LL”
![Shut Down • • • Instrument Access [F 12] – Containers • Verify fluid(s) Shut Down • • • Instrument Access [F 12] – Containers • Verify fluid(s)](https://present5.com/presentation/c9e685d9da6f7e873fa07af7fd1a84e7/image-23.jpg)
Shut Down • • • Instrument Access [F 12] – Containers • Verify fluid(s) levels – Segment • Remove segments notated as “inactive” or “done” – Accept [F 10] Clean-Up Back-up – Main menu • Utilities- backup • Insert disk- hit [F 10] then accept • Remove disk

Maintenance • • Daily Weekly Monthly Bi-Annual
c9e685d9da6f7e873fa07af7fd1a84e7.ppt