fb82a64704128e5e74540e5311602feb.ppt
- Количество слайдов: 31
 A Randomized Comparison of Vascular Complications after TAVR Comparing the SAPIEN vs. The Lower-Profile SAPIEN XT System in Inoperable Aortic Stenosis Patients A PARTNER 2 Substudy Analysis Augusto D. Pichard, MD on behalf of The PARTNER Trial Investigators and The PARTNER Publications Office ACC 2014 | Washington, D. C. | March 29, 2014
A Randomized Comparison of Vascular Complications after TAVR Comparing the SAPIEN vs. The Lower-Profile SAPIEN XT System in Inoperable Aortic Stenosis Patients A PARTNER 2 Substudy Analysis Augusto D. Pichard, MD on behalf of The PARTNER Trial Investigators and The PARTNER Publications Office ACC 2014 | Washington, D. C. | March 29, 2014
 Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship Proctor for Edwards Lifesciences
Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship Proctor for Edwards Lifesciences
 Background • In inoperable patients with severe aortic stenosis, major vascular complications (VC) after TAVR are common and associated with unfavorable outcomes. • The use of large diameter catheters with the first generation devices and the high-risk characteristics of the treated population have been implicated as potential causes. • The frequency and impact of VC with the newer second generation, low-profile SAPIEN XT valve and delivery system is unknown.
Background • In inoperable patients with severe aortic stenosis, major vascular complications (VC) after TAVR are common and associated with unfavorable outcomes. • The use of large diameter catheters with the first generation devices and the high-risk characteristics of the treated population have been implicated as potential causes. • The frequency and impact of VC with the newer second generation, low-profile SAPIEN XT valve and delivery system is unknown.
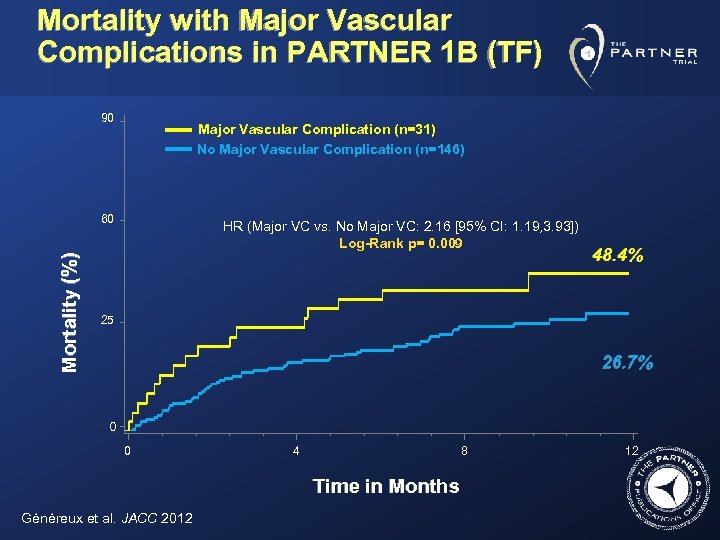 Mortality with Major Vascular Complications in PARTNER 1 B (TF) 90 Major Vascular Complication (n=31) No Major Vascular Complication (n=146) Mortality (%) 60 HR (Major VC vs. No Major VC: 2. 16 [95% CI: 1. 19, 3. 93]) Log-Rank p= 0. 009 48. 4% 25 26. 7% 0 0 4 8 Time in Months Généreux et al. JACC 2012 12
Mortality with Major Vascular Complications in PARTNER 1 B (TF) 90 Major Vascular Complication (n=31) No Major Vascular Complication (n=146) Mortality (%) 60 HR (Major VC vs. No Major VC: 2. 16 [95% CI: 1. 19, 3. 93]) Log-Rank p= 0. 009 48. 4% 25 26. 7% 0 0 4 8 Time in Months Généreux et al. JACC 2012 12
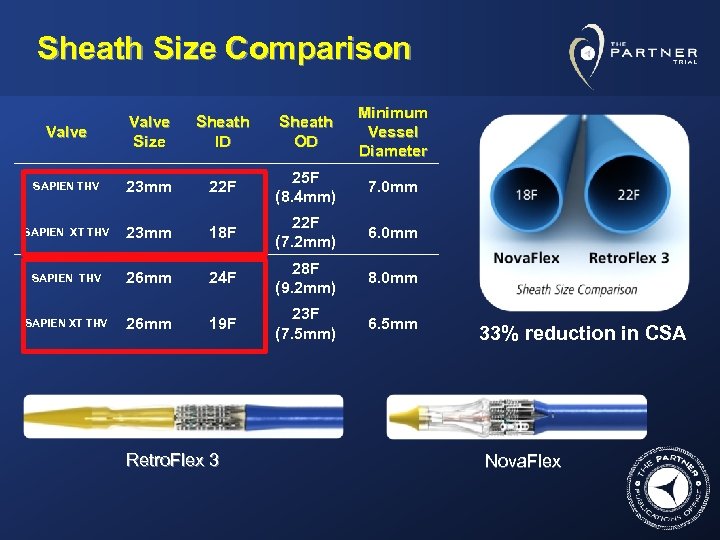 Sheath Size Comparison Valve Size Sheath ID Sheath OD Minimum Vessel Diameter SAPIEN THV 23 mm 22 F 25 F (8. 4 mm) 7. 0 mm SAPIEN XT THV 23 mm 18 F 22 F (7. 2 mm) 6. 0 mm SAPIEN THV 26 mm 24 F 28 F (9. 2 mm) 8. 0 mm SAPIEN XT THV 26 mm 19 F 23 F (7. 5 mm) 6. 5 mm Retro. Flex 3 33% reduction in CSA Nova. Flex
Sheath Size Comparison Valve Size Sheath ID Sheath OD Minimum Vessel Diameter SAPIEN THV 23 mm 22 F 25 F (8. 4 mm) 7. 0 mm SAPIEN XT THV 23 mm 18 F 22 F (7. 2 mm) 6. 0 mm SAPIEN THV 26 mm 24 F 28 F (9. 2 mm) 8. 0 mm SAPIEN XT THV 26 mm 19 F 23 F (7. 5 mm) 6. 5 mm Retro. Flex 3 33% reduction in CSA Nova. Flex
 Study Aims • To examine the effect of device type on vascular complications after TAVR • To characterize the incidence and nature of vascular complications after TAVR • To evaluate the impact of major vascular complications on 1 year all-cause mortality • To determine the predictors of major vascular complications after TAVR
Study Aims • To examine the effect of device type on vascular complications after TAVR • To characterize the incidence and nature of vascular complications after TAVR • To evaluate the impact of major vascular complications on 1 year all-cause mortality • To determine the predictors of major vascular complications after TAVR
 Study Design • As-treated analysis of inoperable patients undergoing TF-TAVR in the PARTNER 2 Cohort B trial with either SAPIEN or SAPIEN XT • Inclusion/exclusion criteria, data collection and monitoring were similar to PARTNER I Cohort B Trial • Key exclusion criteria: – Iliac-femoral anatomy precluding safe sheath insertion (vessel size <7 mm for 23 mm valve or < 8 mm for 26 mm valve. 8 and 9 mm for heavily calcified vessels). – Aortic annulus diameter (echo measurement) < 18 mm or > 25 mm – Significant aorto-iliac disease, including marked tortuosity
Study Design • As-treated analysis of inoperable patients undergoing TF-TAVR in the PARTNER 2 Cohort B trial with either SAPIEN or SAPIEN XT • Inclusion/exclusion criteria, data collection and monitoring were similar to PARTNER I Cohort B Trial • Key exclusion criteria: – Iliac-femoral anatomy precluding safe sheath insertion (vessel size <7 mm for 23 mm valve or < 8 mm for 26 mm valve. 8 and 9 mm for heavily calcified vessels). – Aortic annulus diameter (echo measurement) < 18 mm or > 25 mm – Significant aorto-iliac disease, including marked tortuosity
 Study Design • Events were adjudicated by an independent CEC – Major and minor vascular complications as defined by Valve Academic Research Consortium-2 criteria • Event rates are presented as Kaplan-Meier estimates • Multivariable analyses were performed to identify predictors of major vascular complications using Cox proportional hazards models
Study Design • Events were adjudicated by an independent CEC – Major and minor vascular complications as defined by Valve Academic Research Consortium-2 criteria • Event rates are presented as Kaplan-Meier estimates • Multivariable analyses were performed to identify predictors of major vascular complications using Cox proportional hazards models
 VARC-2 Definitions Major Vascular Complications Minor Vascular Complications 1. Any aortic dissection, rupture, left ventricular perforation, or new apical aneurysm/pseudoaneurysm 1. 2. Access site or access-site related vascular injury (dissection, perforation, stenosis, hematoma, etc. ) leading to death, lifethreatening or major bleeding, visceral ischemia, or neurological impairment Access site or access-site related vascular injury (dissection, perforation, stenosis, hematoma, etc. ) NOT leading to death, lifethreatening or major bleeding, visceral ischemia, or neurological impairment 2. Distal embolization treated with embolectomy and/or thrombectomy and NOT resulting in amputation or end organ damage 3. Distal embolization requiring vascular surgery, amputation, or irreversible end organ damage 3. 4. Unplanned endovascular or surgical intervention associated with death, major bleeding, visceral ischemia, or neurological impairment Unplanned endovascular stenting or unplanned surgical intervention not meeting the criteria for a major vascular complication 4. Vascular repair or the need for vascular repair (via surgery, ultrasound-guided compression, transcatheter embolization, or stent-graft 5. Any new ipsilateral lower extremity ischemia 6. Surgery for access site-related nerve injury 7. Permanent access site-related nerve injury Source: Kappetein, et al. JACC 2012
VARC-2 Definitions Major Vascular Complications Minor Vascular Complications 1. Any aortic dissection, rupture, left ventricular perforation, or new apical aneurysm/pseudoaneurysm 1. 2. Access site or access-site related vascular injury (dissection, perforation, stenosis, hematoma, etc. ) leading to death, lifethreatening or major bleeding, visceral ischemia, or neurological impairment Access site or access-site related vascular injury (dissection, perforation, stenosis, hematoma, etc. ) NOT leading to death, lifethreatening or major bleeding, visceral ischemia, or neurological impairment 2. Distal embolization treated with embolectomy and/or thrombectomy and NOT resulting in amputation or end organ damage 3. Distal embolization requiring vascular surgery, amputation, or irreversible end organ damage 3. 4. Unplanned endovascular or surgical intervention associated with death, major bleeding, visceral ischemia, or neurological impairment Unplanned endovascular stenting or unplanned surgical intervention not meeting the criteria for a major vascular complication 4. Vascular repair or the need for vascular repair (via surgery, ultrasound-guided compression, transcatheter embolization, or stent-graft 5. Any new ipsilateral lower extremity ischemia 6. Surgery for access site-related nerve injury 7. Permanent access site-related nerve injury Source: Kappetein, et al. JACC 2012
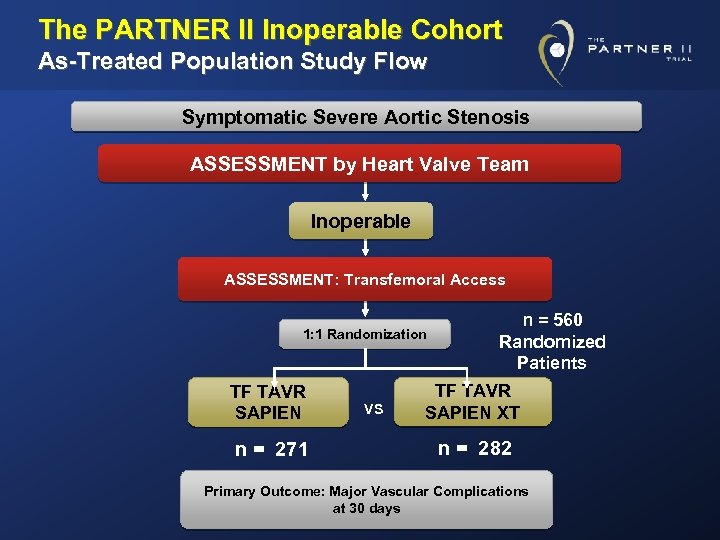 The PARTNER II Inoperable Cohort As-Treated Population Study Flow Symptomatic Severe Aortic Stenosis ASSESSMENT by Heart Valve Team Inoperable ASSESSMENT: Transfemoral Access 1: 1 Randomization TF TAVR SAPIEN n = 271 VS n = 560 Randomized Patients TF TAVR SAPIEN XT n = 282 Primary Outcome: Major Vascular Complications at 30 days
The PARTNER II Inoperable Cohort As-Treated Population Study Flow Symptomatic Severe Aortic Stenosis ASSESSMENT by Heart Valve Team Inoperable ASSESSMENT: Transfemoral Access 1: 1 Randomization TF TAVR SAPIEN n = 271 VS n = 560 Randomized Patients TF TAVR SAPIEN XT n = 282 Primary Outcome: Major Vascular Complications at 30 days
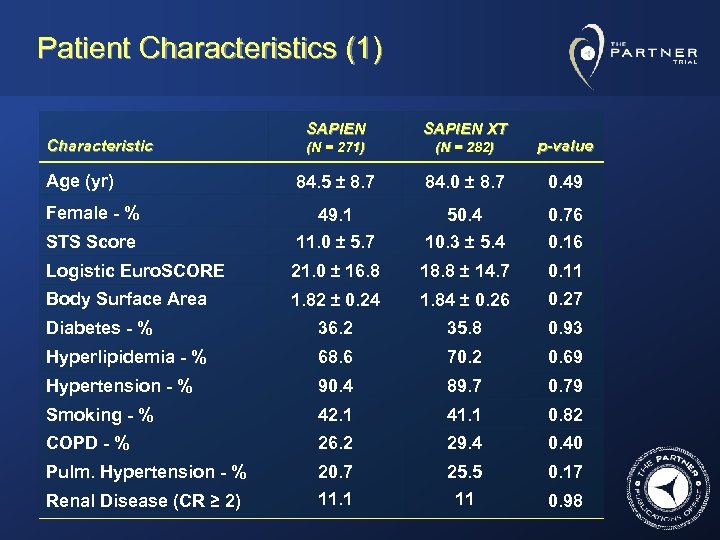 Patient Characteristics (1) SAPIEN XT (N = 271) (N = 282) p-value 84. 5 ± 8. 7 84. 0 ± 8. 7 0. 49 Female - % 49. 1 50. 4 STS Score 11. 0 ± 5. 7 10. 3 ± 5. 4 0. 76 0. 16 Logistic Euro. SCORE 21. 0 ± 16. 8 18. 8 ± 14. 7 0. 11 Body Surface Area 1. 82 ± 0. 24 1. 84 ± 0. 26 0. 27 Diabetes - % 36. 2 35. 8 0. 93 Hyperlipidemia - % 68. 6 70. 2 0. 69 Hypertension - % 90. 4 89. 7 0. 79 Smoking - % 42. 1 41. 1 0. 82 COPD - % 26. 2 29. 4 0. 40 Pulm. Hypertension - % 20. 7 11. 1 25. 5 11 0. 17 Characteristic Age (yr) Renal Disease (CR ≥ 2) 0. 98
Patient Characteristics (1) SAPIEN XT (N = 271) (N = 282) p-value 84. 5 ± 8. 7 84. 0 ± 8. 7 0. 49 Female - % 49. 1 50. 4 STS Score 11. 0 ± 5. 7 10. 3 ± 5. 4 0. 76 0. 16 Logistic Euro. SCORE 21. 0 ± 16. 8 18. 8 ± 14. 7 0. 11 Body Surface Area 1. 82 ± 0. 24 1. 84 ± 0. 26 0. 27 Diabetes - % 36. 2 35. 8 0. 93 Hyperlipidemia - % 68. 6 70. 2 0. 69 Hypertension - % 90. 4 89. 7 0. 79 Smoking - % 42. 1 41. 1 0. 82 COPD - % 26. 2 29. 4 0. 40 Pulm. Hypertension - % 20. 7 11. 1 25. 5 11 0. 17 Characteristic Age (yr) Renal Disease (CR ≥ 2) 0. 98
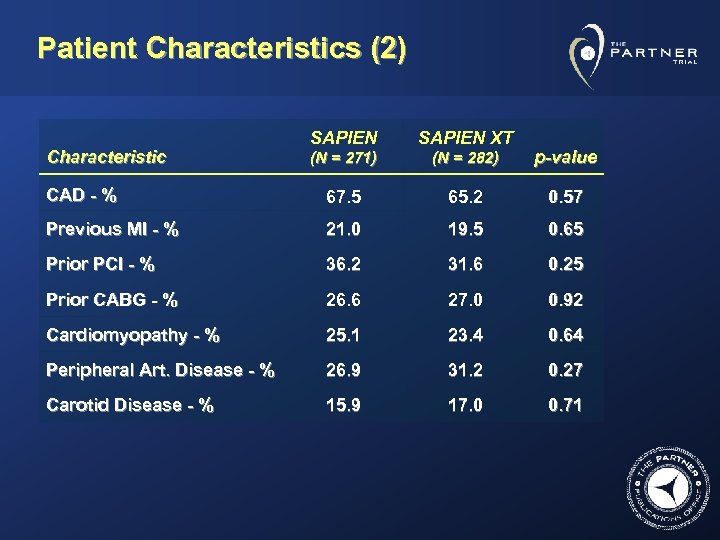 Patient Characteristics (2) SAPIEN XT (N = 271) (N = 282) p-value CAD - % 67. 5 65. 2 0. 57 Previous MI - % 21. 0 19. 5 0. 65 Prior PCI - % 36. 2 31. 6 0. 25 Prior CABG - % 26. 6 27. 0 0. 92 Cardiomyopathy - % 25. 1 23. 4 0. 64 Peripheral Art. Disease - % 26. 9 31. 2 0. 27 Carotid Disease - % 15. 9 17. 0 0. 71 Characteristic
Patient Characteristics (2) SAPIEN XT (N = 271) (N = 282) p-value CAD - % 67. 5 65. 2 0. 57 Previous MI - % 21. 0 19. 5 0. 65 Prior PCI - % 36. 2 31. 6 0. 25 Prior CABG - % 26. 6 27. 0 0. 92 Cardiomyopathy - % 25. 1 23. 4 0. 64 Peripheral Art. Disease - % 26. 9 31. 2 0. 27 Carotid Disease - % 15. 9 17. 0 0. 71 Characteristic
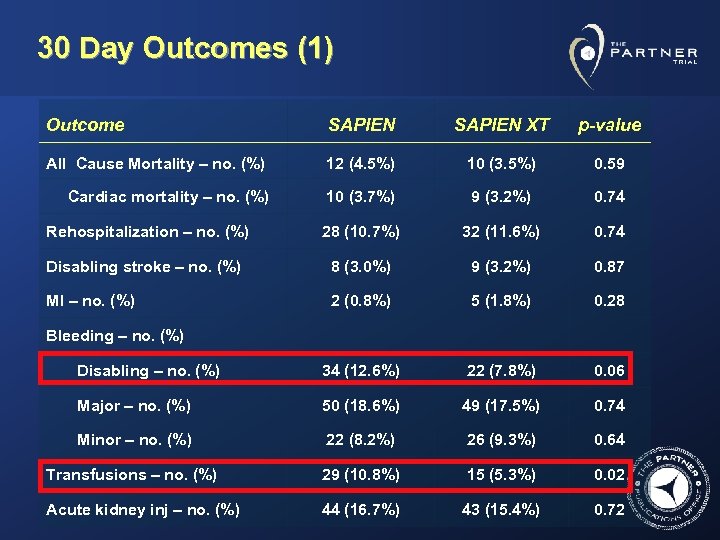 30 Day Outcomes (1) Outcome SAPIEN XT p-value All Cause Mortality – no. (%) 12 (4. 5%) 10 (3. 5%) 0. 59 Cardiac mortality – no. (%) 10 (3. 7%) 9 (3. 2%) 0. 74 Rehospitalization – no. (%) 28 (10. 7%) 32 (11. 6%) 0. 74 Disabling stroke – no. (%) 8 (3. 0%) 9 (3. 2%) 0. 87 MI – no. (%) 2 (0. 8%) 5 (1. 8%) 0. 28 Disabling – no. (%) 34 (12. 6%) 22 (7. 8%) 0. 06 Major – no. (%) 50 (18. 6%) 49 (17. 5%) 0. 74 Minor – no. (%) 22 (8. 2%) 26 (9. 3%) 0. 64 Transfusions – no. (%) 29 (10. 8%) 15 (5. 3%) 0. 02 Acute kidney inj – no. (%) 44 (16. 7%) 43 (15. 4%) 0. 72 Bleeding – no. (%)
30 Day Outcomes (1) Outcome SAPIEN XT p-value All Cause Mortality – no. (%) 12 (4. 5%) 10 (3. 5%) 0. 59 Cardiac mortality – no. (%) 10 (3. 7%) 9 (3. 2%) 0. 74 Rehospitalization – no. (%) 28 (10. 7%) 32 (11. 6%) 0. 74 Disabling stroke – no. (%) 8 (3. 0%) 9 (3. 2%) 0. 87 MI – no. (%) 2 (0. 8%) 5 (1. 8%) 0. 28 Disabling – no. (%) 34 (12. 6%) 22 (7. 8%) 0. 06 Major – no. (%) 50 (18. 6%) 49 (17. 5%) 0. 74 Minor – no. (%) 22 (8. 2%) 26 (9. 3%) 0. 64 Transfusions – no. (%) 29 (10. 8%) 15 (5. 3%) 0. 02 Acute kidney inj – no. (%) 44 (16. 7%) 43 (15. 4%) 0. 72 Bleeding – no. (%)
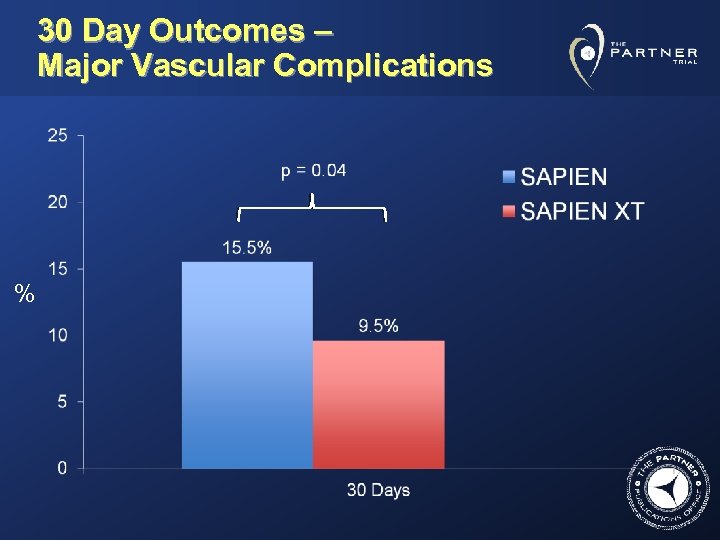 30 Day Outcomes – Major Vascular Complications %
30 Day Outcomes – Major Vascular Complications %
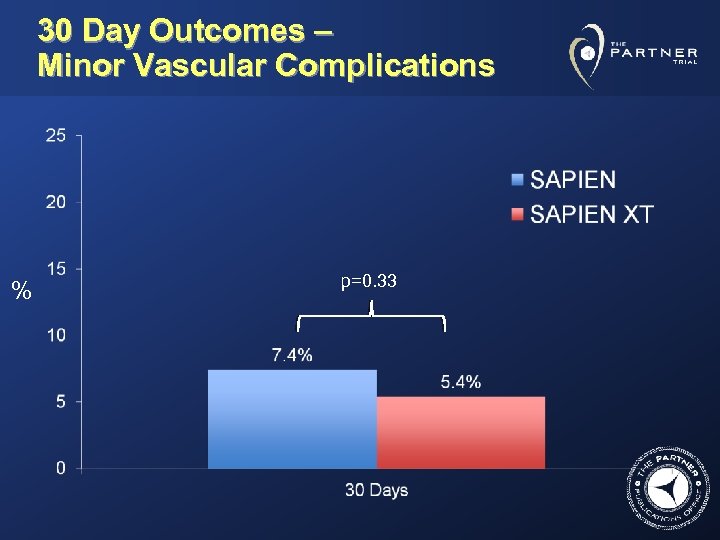 30 Day Outcomes – Minor Vascular Complications % p=0. 33
30 Day Outcomes – Minor Vascular Complications % p=0. 33
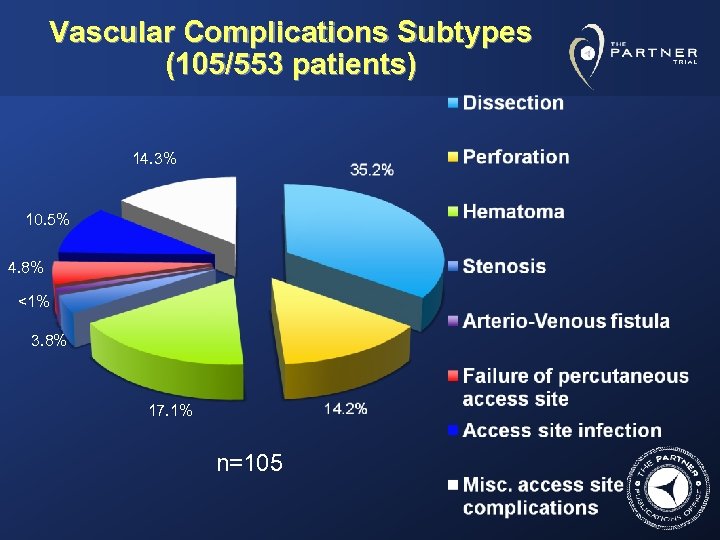 Vascular Complications Subtypes (105/553 patients) 14. 3% 10. 5% 4. 8% <1% 3. 8% 17. 1% n=105
Vascular Complications Subtypes (105/553 patients) 14. 3% 10. 5% 4. 8% <1% 3. 8% 17. 1% n=105
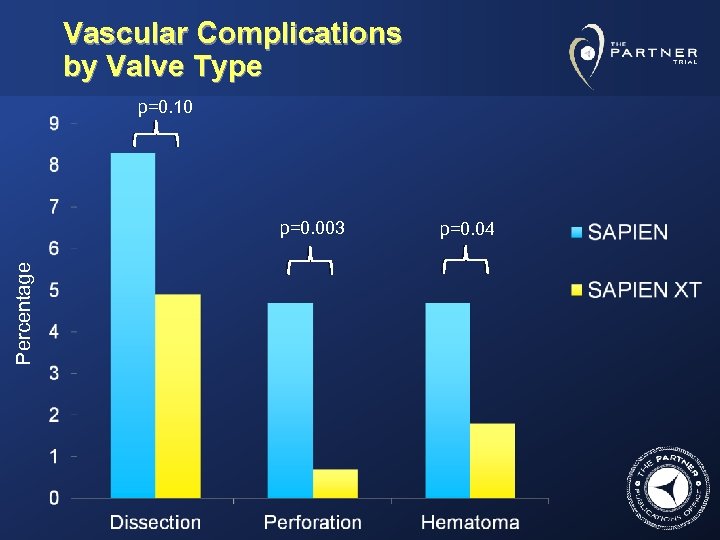 Vascular Complications by Valve Type p=0. 10 Percentage p=0. 003 p=0. 04
Vascular Complications by Valve Type p=0. 10 Percentage p=0. 003 p=0. 04
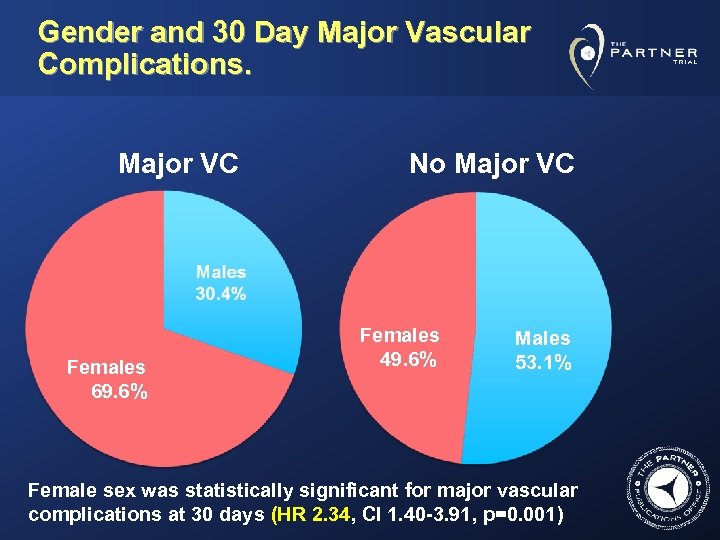 Gender and 30 Day Major Vascular Complications. Major VC Females 69. 6% No Major VC Females 49. 6% Males 53. 1% Female sex was statistically significant for major vascular complications at 30 days (HR 2. 34, CI 1. 40 -3. 91, p=0. 001)
Gender and 30 Day Major Vascular Complications. Major VC Females 69. 6% No Major VC Females 49. 6% Males 53. 1% Female sex was statistically significant for major vascular complications at 30 days (HR 2. 34, CI 1. 40 -3. 91, p=0. 001)
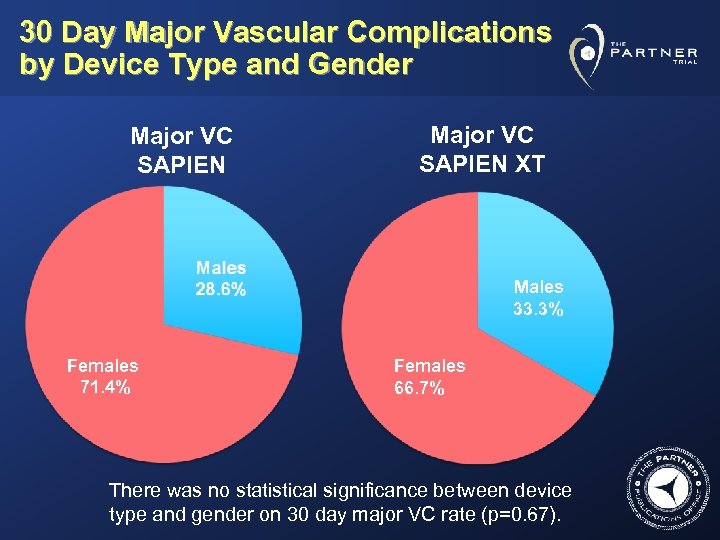 30 Day Major Vascular Complications by Device Type and Gender Major VC SAPIEN XT Males 33. 3% Females 71. 4% Females 66. 7% There was no statistical significance between device type and gender on 30 day major VC rate (p=0. 67).
30 Day Major Vascular Complications by Device Type and Gender Major VC SAPIEN XT Males 33. 3% Females 71. 4% Females 66. 7% There was no statistical significance between device type and gender on 30 day major VC rate (p=0. 67).
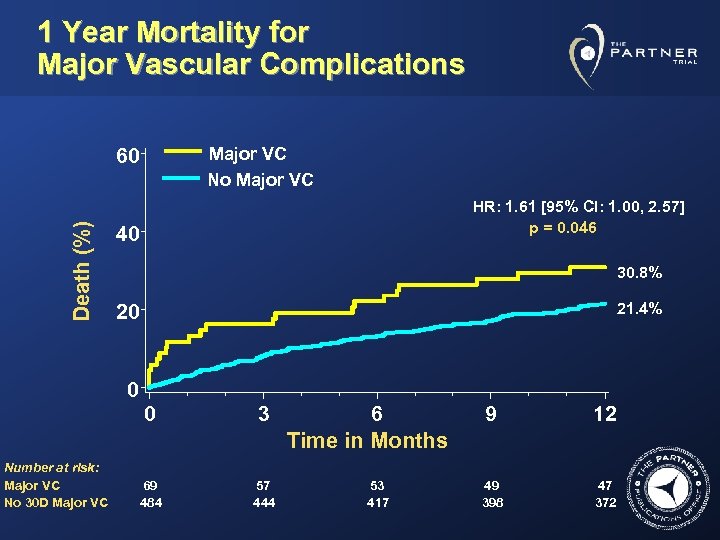 1 Year Mortality for Major Vascular Complications Major VC No Major VC Death (%) 60 HR: 1. 61 [95% CI: 1. 00, 2. 57] p = 0. 046 40 30. 8% 21. 4% 20 0 0 Number at risk: Major VC No 30 D Major VC 3 69 484 57 444 6 Time in Months 53 417 9 12 49 398 47 372
1 Year Mortality for Major Vascular Complications Major VC No Major VC Death (%) 60 HR: 1. 61 [95% CI: 1. 00, 2. 57] p = 0. 046 40 30. 8% 21. 4% 20 0 0 Number at risk: Major VC No 30 D Major VC 3 69 484 57 444 6 Time in Months 53 417 9 12 49 398 47 372
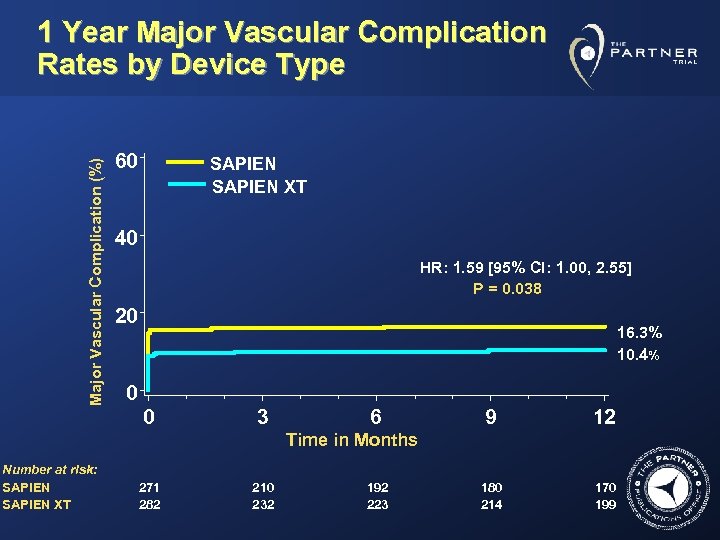 Major Vascular Complication (%) 1 Year Major Vascular Complication Rates by Device Type 60 SAPIEN XT 40 HR: 1. 59 [95% CI: 1. 00, 2. 55] P = 0. 038 20 16. 3% 10. 4% 0 0 3 6 9 12 180 214 170 199 Time in Months Number at risk: SAPIEN XT 271 282 210 232 192 223
Major Vascular Complication (%) 1 Year Major Vascular Complication Rates by Device Type 60 SAPIEN XT 40 HR: 1. 59 [95% CI: 1. 00, 2. 55] P = 0. 038 20 16. 3% 10. 4% 0 0 3 6 9 12 180 214 170 199 Time in Months Number at risk: SAPIEN XT 271 282 210 232 192 223
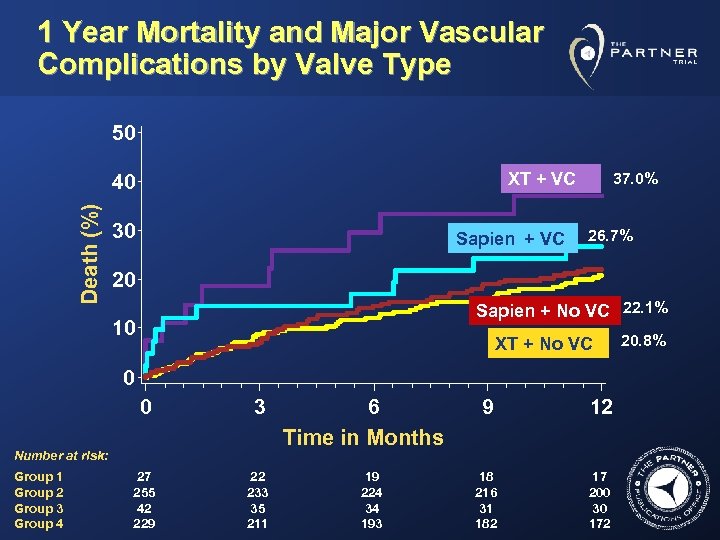 1 Year Mortality and Major Vascular Complications by Valve Type 50 XT + VC Death (%) 40 30 Sapien + VC 37. 0% 26. 7% 20 Sapien + No VC 22. 1% 10 XT + No VC 0 0 3 9 12 18 216 31 182 17 200 30 172 Time in Months Number at risk: Group 1 Group 2 Group 3 Group 4 6 27 255 42 229 22 233 35 211 19 224 34 193 20. 8%
1 Year Mortality and Major Vascular Complications by Valve Type 50 XT + VC Death (%) 40 30 Sapien + VC 37. 0% 26. 7% 20 Sapien + No VC 22. 1% 10 XT + No VC 0 0 3 9 12 18 216 31 182 17 200 30 172 Time in Months Number at risk: Group 1 Group 2 Group 3 Group 4 6 27 255 42 229 22 233 35 211 19 224 34 193 20. 8%
 Predictors of 30 Day Major Vascular Complications • Multivariable analysis* demonstrated only two statistical significant predictors: – Female sex (HR 2. 34, CI 1. 40 -3. 91, p=0. 0001) – • SAPIEN THV (HR 1. 64, CI 1. 01 -2. 67, p=0. 043) Interaction testing between gender and device type was not statistically significant for 30 day major vascular complications. *Baseline Covariates: Age, gender, BMI, STS score, diabetes, smoker, peripheral arterial disease, renal insufficiency, and device type
Predictors of 30 Day Major Vascular Complications • Multivariable analysis* demonstrated only two statistical significant predictors: – Female sex (HR 2. 34, CI 1. 40 -3. 91, p=0. 0001) – • SAPIEN THV (HR 1. 64, CI 1. 01 -2. 67, p=0. 043) Interaction testing between gender and device type was not statistically significant for 30 day major vascular complications. *Baseline Covariates: Age, gender, BMI, STS score, diabetes, smoker, peripheral arterial disease, renal insufficiency, and device type
 Conclusions • In this randomized evaluation of SAPIEN vs. the lower profile SAPIEN XT system: – SAPIEN XT patients had a lower rate of major vascular complications, disabling bleeding and transfusions – The most common vascular complications were dissection, perforation, and hematoma, which were all lower with SAPIEN XT – Major vascular complications resulted in increased 1 year mortality – Female sex was associated with a greater likelihood of developing major vascular complications at 30 days (independent of device type).
Conclusions • In this randomized evaluation of SAPIEN vs. the lower profile SAPIEN XT system: – SAPIEN XT patients had a lower rate of major vascular complications, disabling bleeding and transfusions – The most common vascular complications were dissection, perforation, and hematoma, which were all lower with SAPIEN XT – Major vascular complications resulted in increased 1 year mortality – Female sex was associated with a greater likelihood of developing major vascular complications at 30 days (independent of device type).
 Limitations • CT scans, angiograms, and peripheral ultrasound performed prior to randomization was not available in case report forms. • Information regarding operator experience, and strategies to prevent and/or treat major vascular complications was unavailable at the time of this analysis.
Limitations • CT scans, angiograms, and peripheral ultrasound performed prior to randomization was not available in case report forms. • Information regarding operator experience, and strategies to prevent and/or treat major vascular complications was unavailable at the time of this analysis.
 Implications • The newer low-profile SAPIEN XT and Nova. Flex delivery system demonstrated markedly lower major vascular complications rates. • SAPIEN XT should be the preferred transcatheter valve to minimize morbidity from major vascular complications in inoperable patients undergoing TAVR.
Implications • The newer low-profile SAPIEN XT and Nova. Flex delivery system demonstrated markedly lower major vascular complications rates. • SAPIEN XT should be the preferred transcatheter valve to minimize morbidity from major vascular complications in inoperable patients undergoing TAVR.
 A Special Thank You to the Writing Group
A Special Thank You to the Writing Group
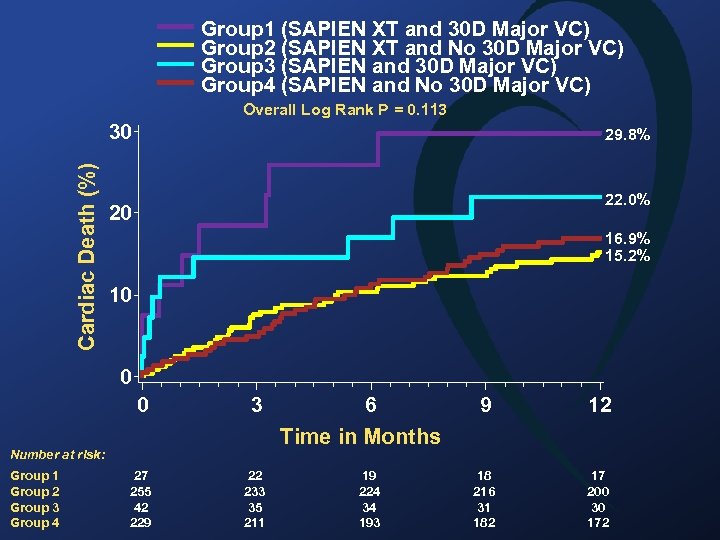 Group 1 (SAPIEN XT and 30 D Major VC) Group 2 (SAPIEN XT and No 30 D Major VC) Group 3 (SAPIEN and 30 D Major VC) Group 4 (SAPIEN and No 30 D Major VC) Overall Log Rank P = 0. 113 Cardiac Death (%) 30 29. 8% 22. 0% 20 16. 9% 15. 2% 10 0 0 3 9 12 18 216 31 182 17 200 30 172 Time in Months Number at risk: Group 1 Group 2 Group 3 Group 4 6 27 255 42 229 22 233 35 211 19 224 34 193
Group 1 (SAPIEN XT and 30 D Major VC) Group 2 (SAPIEN XT and No 30 D Major VC) Group 3 (SAPIEN and 30 D Major VC) Group 4 (SAPIEN and No 30 D Major VC) Overall Log Rank P = 0. 113 Cardiac Death (%) 30 29. 8% 22. 0% 20 16. 9% 15. 2% 10 0 0 3 9 12 18 216 31 182 17 200 30 172 Time in Months Number at risk: Group 1 Group 2 Group 3 Group 4 6 27 255 42 229 22 233 35 211 19 224 34 193
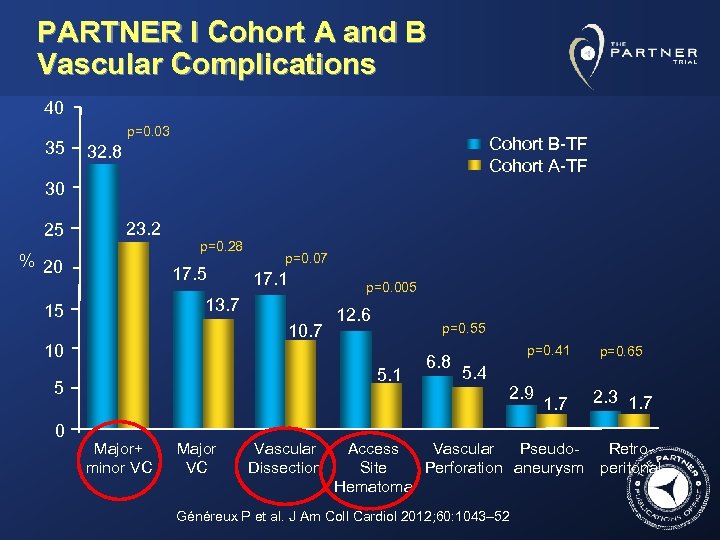 PARTNER I Cohort A and B Vascular Complications 40 35 p=0. 03 Cohort B-TF Cohort A-TF 32. 8 30 25 23. 2 % 20 p=0. 28 17. 5 p=0. 07 17. 1 13. 7 15 10. 7 p=0. 005 12. 6 p=0. 55 10 5. 1 5 0 Major+ minor VC Major VC Vascular Dissection 6. 8 p=0. 41 5. 4 2. 9 1. 7 p=0. 65 2. 3 1. 7 Access Vascular Pseudo. Retro. Site Perforation aneurysm peritonal Hematoma Généreux P et al. J Am Coll Cardiol 2012; 60: 1043– 52
PARTNER I Cohort A and B Vascular Complications 40 35 p=0. 03 Cohort B-TF Cohort A-TF 32. 8 30 25 23. 2 % 20 p=0. 28 17. 5 p=0. 07 17. 1 13. 7 15 10. 7 p=0. 005 12. 6 p=0. 55 10 5. 1 5 0 Major+ minor VC Major VC Vascular Dissection 6. 8 p=0. 41 5. 4 2. 9 1. 7 p=0. 65 2. 3 1. 7 Access Vascular Pseudo. Retro. Site Perforation aneurysm peritonal Hematoma Généreux P et al. J Am Coll Cardiol 2012; 60: 1043– 52
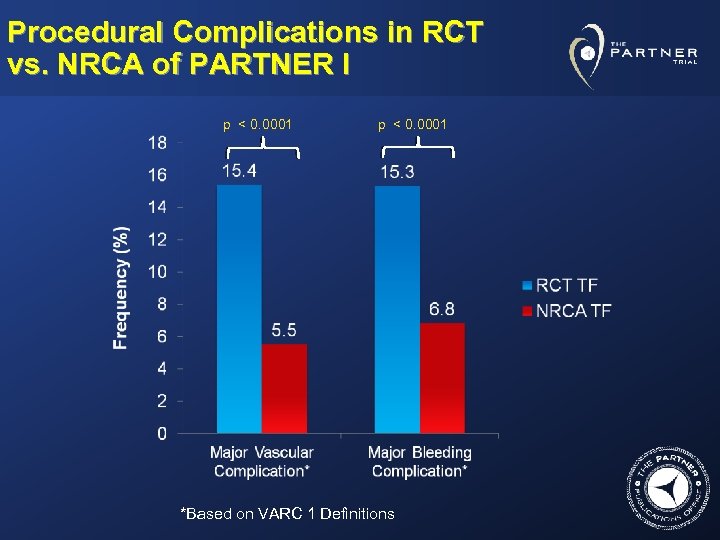 Procedural Complications in RCT vs. NRCA of PARTNER I p < 0. 0001 *Based on VARC 1 Definitions
Procedural Complications in RCT vs. NRCA of PARTNER I p < 0. 0001 *Based on VARC 1 Definitions
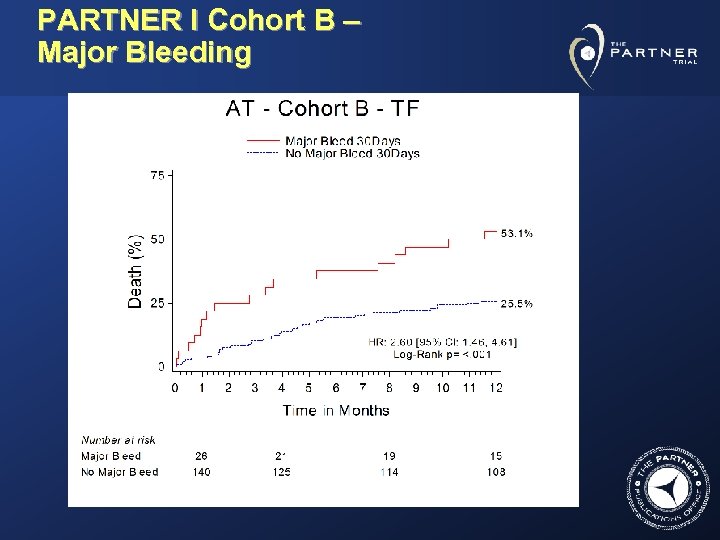 PARTNER I Cohort B – Major Bleeding
PARTNER I Cohort B – Major Bleeding


