13a6371345c74917e9de88b47fef8533.ppt
- Количество слайдов: 56
 A Randomiz. Ed Trial of ENt. ERal Glutamine to Minim. IZE Thermal Injury: A multicenter Pragmatic RCT (definitive study) Study Sponsor Dr. Daren Heyland Clinical Evaluation Research Unit Project Leader: Maureen Dansereau
A Randomiz. Ed Trial of ENt. ERal Glutamine to Minim. IZE Thermal Injury: A multicenter Pragmatic RCT (definitive study) Study Sponsor Dr. Daren Heyland Clinical Evaluation Research Unit Project Leader: Maureen Dansereau
 www. criticalcarenutrition. com
www. criticalcarenutrition. com
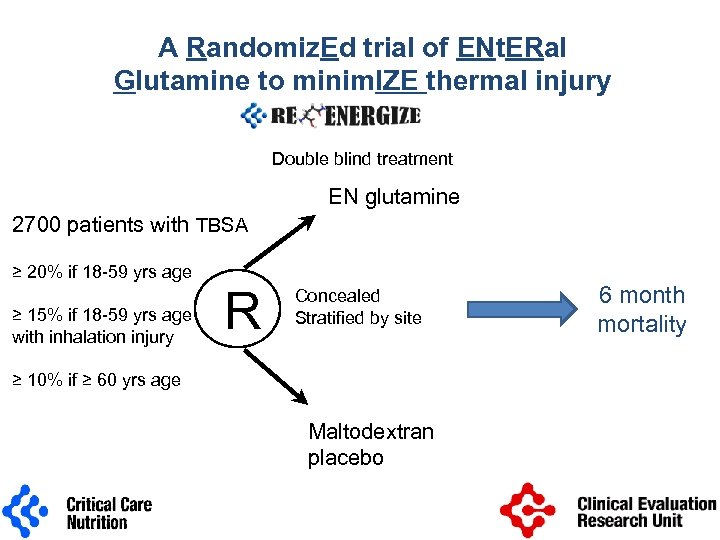 A Randomiz. Ed trial of ENt. ERal Glutamine to minim. IZE thermal injury Double blind treatment EN glutamine 2700 patients with TBSA ≥ 20% if 18 -59 yrs age ≥ 15% if 18 -59 yrs age with inhalation injury R Concealed Stratified by site ≥ 10% if ≥ 60 yrs age Maltodextran placebo 6 month mortality
A Randomiz. Ed trial of ENt. ERal Glutamine to minim. IZE thermal injury Double blind treatment EN glutamine 2700 patients with TBSA ≥ 20% if 18 -59 yrs age ≥ 15% if 18 -59 yrs age with inhalation injury R Concealed Stratified by site ≥ 10% if ≥ 60 yrs age Maltodextran placebo 6 month mortality
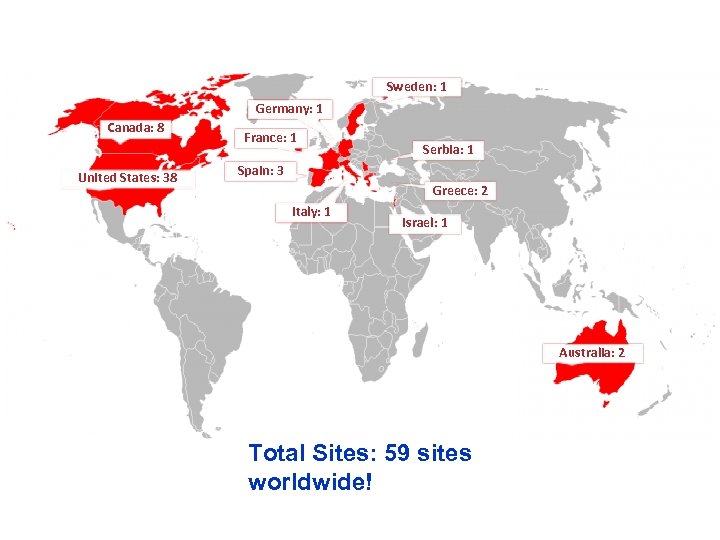 Sweden: 1 Germany: 1 Canada: 8 United States: 38 France: 1 Serbia: 1 Spain: 3 Greece: 2 Italy: 1 Israel: 1 Australia: 2 Total Sites: 59 sites worldwide!
Sweden: 1 Germany: 1 Canada: 8 United States: 38 France: 1 Serbia: 1 Spain: 3 Greece: 2 Italy: 1 Israel: 1 Australia: 2 Total Sites: 59 sites worldwide!
 Why are we doing this trial?
Why are we doing this trial?
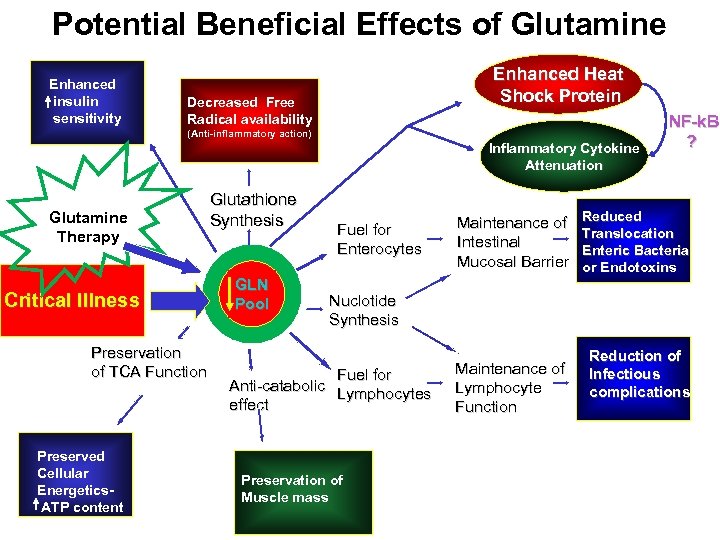 Potential Beneficial Effects of Glutamine Enhanced insulin sensitivity Enhanced Heat Shock Protein Decreased Free Radical availability NF-k. B (Anti-inflammatory action) Inflammatory Cytokine Attenuation Glutamine Therapy Critical Illness Preservation of TCA Function Preserved Cellular Energetics ATP content Glutathione Synthesis GLN Pool pool Fuel for Enterocytes Maintenance of Intestinal Mucosal Barrier ? Reduced Translocation Enteric Bacteria or Endotoxins Nuclotide Synthesis Fuel for Anti-catabolic Lymphocytes effect Preservation of Muscle mass Maintenance of Lymphocyte Function Reduction of Infectious complications
Potential Beneficial Effects of Glutamine Enhanced insulin sensitivity Enhanced Heat Shock Protein Decreased Free Radical availability NF-k. B (Anti-inflammatory action) Inflammatory Cytokine Attenuation Glutamine Therapy Critical Illness Preservation of TCA Function Preserved Cellular Energetics ATP content Glutathione Synthesis GLN Pool pool Fuel for Enterocytes Maintenance of Intestinal Mucosal Barrier ? Reduced Translocation Enteric Bacteria or Endotoxins Nuclotide Synthesis Fuel for Anti-catabolic Lymphocytes effect Preservation of Muscle mass Maintenance of Lymphocyte Function Reduction of Infectious complications
 Glutamine: Important Questions • Is glutamine ‘conditionally essential’? • Is glutamine safe? – Mechanism of harm? – Negative interactions between glutamine and antioxidants • What is the evidence for benefit in Burn patients?
Glutamine: Important Questions • Is glutamine ‘conditionally essential’? • Is glutamine safe? – Mechanism of harm? – Negative interactions between glutamine and antioxidants • What is the evidence for benefit in Burn patients?
 Glutamine: A conditionally essential amino acid? Glutamine levels drop: - following extreme physical exercice - after major surgery - during critical illness Low glutamine levels are associated with: - immune dysfunction - higer mortality in critically ill patients Novak F, Heyland DK, A Avenell et al. , Crit Care Med 2002 O udemans-van Straaten HM, Bosman RJ, Treskes Met al. , Intensive Car Med 2001
Glutamine: A conditionally essential amino acid? Glutamine levels drop: - following extreme physical exercice - after major surgery - during critical illness Low glutamine levels are associated with: - immune dysfunction - higer mortality in critically ill patients Novak F, Heyland DK, A Avenell et al. , Crit Care Med 2002 O udemans-van Straaten HM, Bosman RJ, Treskes Met al. , Intensive Car Med 2001
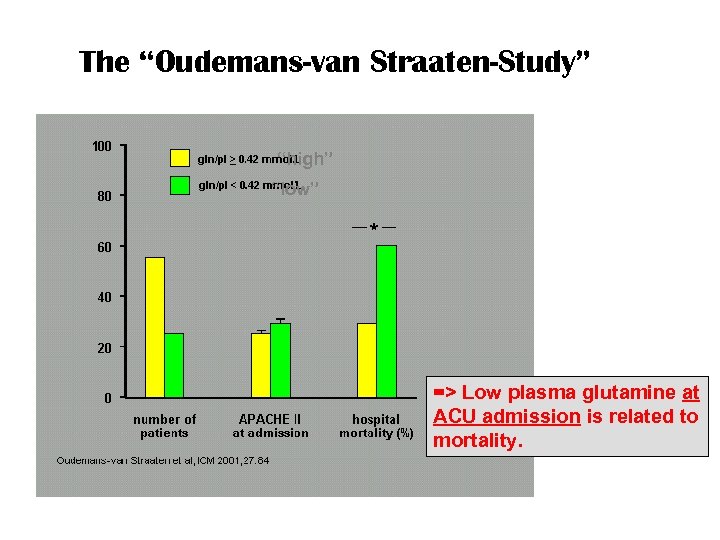 The “Oudemans-van Straaten-Study” “high” “low” => Low plasma glutamine at ACU admission is related to mortality.
The “Oudemans-van Straaten-Study” “high” “low” => Low plasma glutamine at ACU admission is related to mortality.
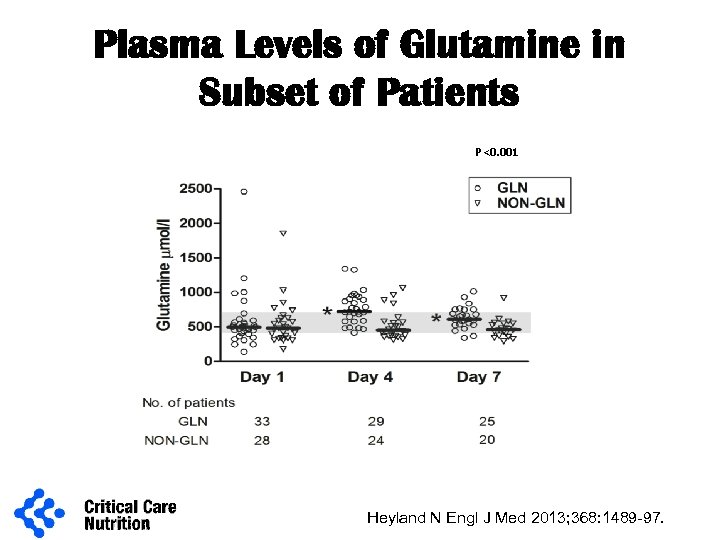 Plasma Levels of Glutamine in Subset of Patients P <0. 001 Heyland N Engl J Med 2013; 368: 1489 -97.
Plasma Levels of Glutamine in Subset of Patients P <0. 001 Heyland N Engl J Med 2013; 368: 1489 -97.
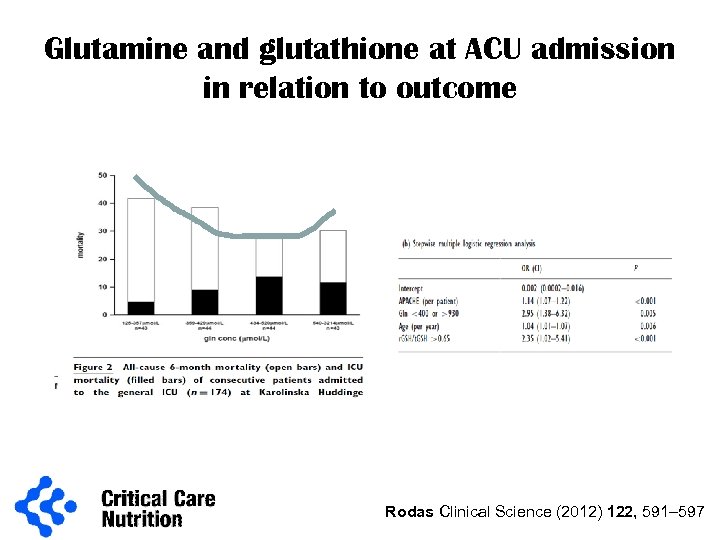 Glutamine and glutathione at ACU admission in relation to outcome Rodas Clinical Science (2012) 122, 591– 597
Glutamine and glutathione at ACU admission in relation to outcome Rodas Clinical Science (2012) 122, 591– 597
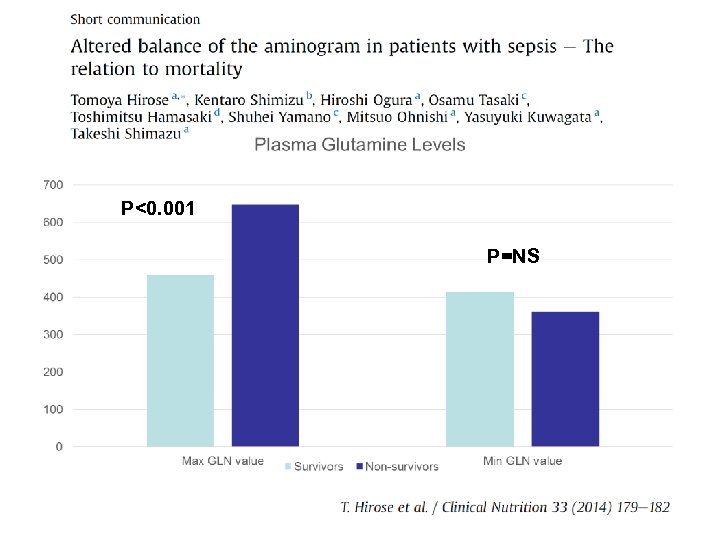 P<0. 001 P=NS
P<0. 001 P=NS
 Future Trials Require Bedside Testing?
Future Trials Require Bedside Testing?
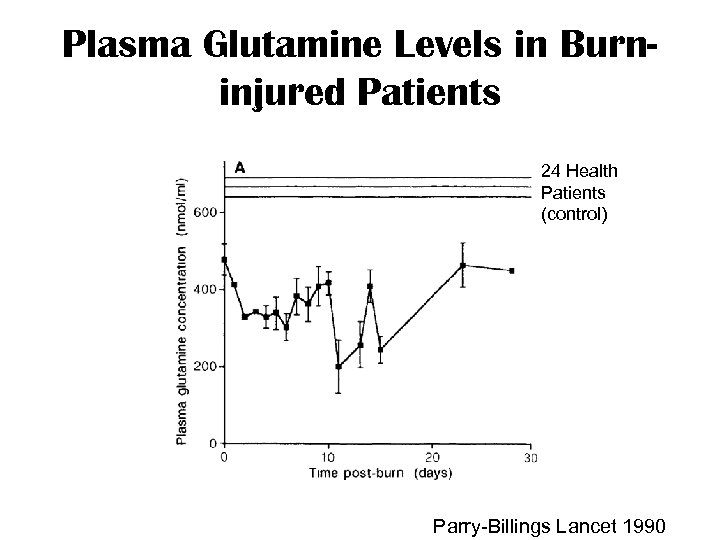 Plasma Glutamine Levels in Burninjured Patients 24 Health Patients (control) Parry-Billings Lancet 1990
Plasma Glutamine Levels in Burninjured Patients 24 Health Patients (control) Parry-Billings Lancet 1990
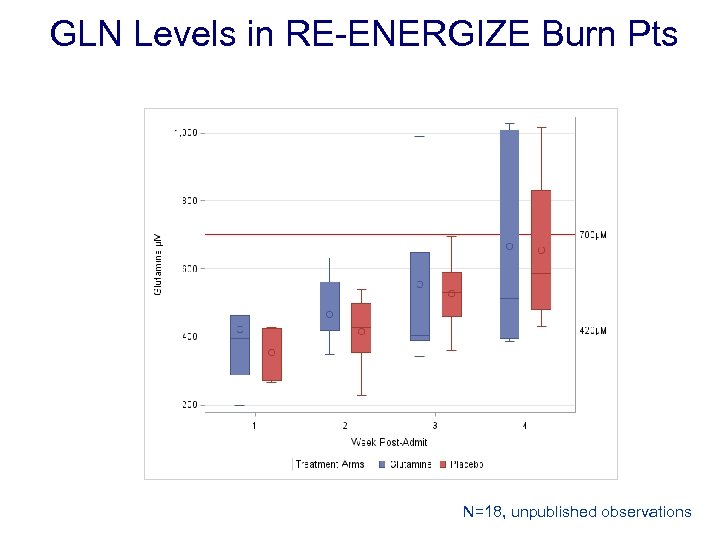 GLN Levels in RE-ENERGIZE Burn Pts N=18, unpublished observations
GLN Levels in RE-ENERGIZE Burn Pts N=18, unpublished observations
 RCTs of IV Glutamine in ACU Patients (n=29) • A trend towards a reduction in mortality – RR, 0. 87, 95 % CI 0. 75, 1. 01, p = 0. 07, 27 trials, • A significant reduction in hospital mortality – RR 0. 70, 95% CI 0. 53, 0. 92, p= 0. 01, 15 trials, • A trend towards a reduction in infectious complications – RR, 0. 89, 95 % CI 0. 77, 1. 03, p = 0. 12, 13 trials, • A significant reduction in hospital length of stay – weighted mean difference in days -2. 56, 95% CI -4. 71, -0. 42, p=0. 02, 11 trials www. criticalcarenutiriont. comCPGs
RCTs of IV Glutamine in ACU Patients (n=29) • A trend towards a reduction in mortality – RR, 0. 87, 95 % CI 0. 75, 1. 01, p = 0. 07, 27 trials, • A significant reduction in hospital mortality – RR 0. 70, 95% CI 0. 53, 0. 92, p= 0. 01, 15 trials, • A trend towards a reduction in infectious complications – RR, 0. 89, 95 % CI 0. 77, 1. 03, p = 0. 12, 13 trials, • A significant reduction in hospital length of stay – weighted mean difference in days -2. 56, 95% CI -4. 71, -0. 42, p=0. 02, 11 trials www. criticalcarenutiriont. comCPGs
 RCTs of EN Glutamine in ACU Patients (n=10) • No significant difference on mortality – RR, 0. 94, 95 % CI 0. 65, 1. 36, p = 0. 74 • No impact on in infectious complications – RR, 0. 93, 95 % CI 0. 79, 1. 10, p = 0. 39, 4 trials • A significant reduction in hospital length of stay – weighted mean difference in days -4. 73, 95% CI -8. 56, 0. 90, p=0. 02, 7 trials. www. criticalcarenutiriont. comCPGs
RCTs of EN Glutamine in ACU Patients (n=10) • No significant difference on mortality – RR, 0. 94, 95 % CI 0. 65, 1. 36, p = 0. 74 • No impact on in infectious complications – RR, 0. 93, 95 % CI 0. 79, 1. 10, p = 0. 39, 4 trials • A significant reduction in hospital length of stay – weighted mean difference in days -4. 73, 95% CI -8. 56, 0. 90, p=0. 02, 7 trials. www. criticalcarenutiriont. comCPGs
 A RANDOMIZED TRIAL OF HIGH-DOSE GLUTAMINE AND ANTIOXIDANTS IN CRITICALLY ILL PATIENTS WITH MULTIORGAN FAILURE The REDOXS study On behalf of the REDOXS Study Investigators N Engl J Med 2013; 368: 1489 -97.
A RANDOMIZED TRIAL OF HIGH-DOSE GLUTAMINE AND ANTIOXIDANTS IN CRITICALLY ILL PATIENTS WITH MULTIORGAN FAILURE The REDOXS study On behalf of the REDOXS Study Investigators N Engl J Med 2013; 368: 1489 -97.
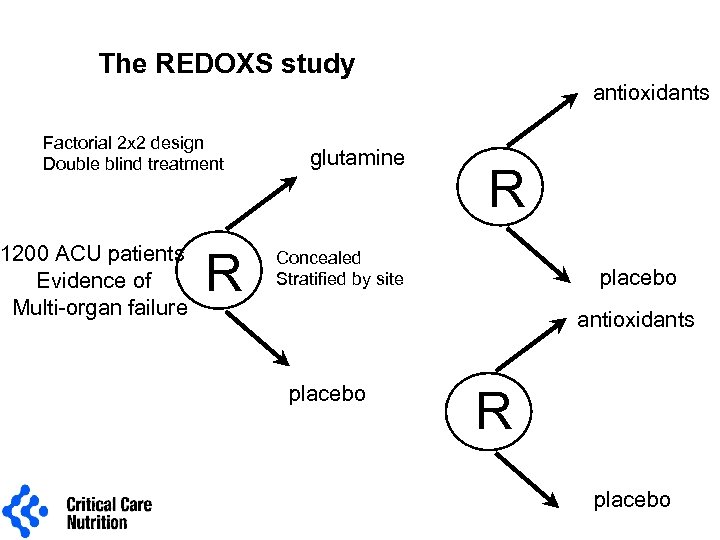 The REDOXS study antioxidants Factorial 2 x 2 design Double blind treatment 1200 ACU patients Evidence of Multi-organ failure R glutamine R Concealed Stratified by site placebo antioxidants placebo R placebo
The REDOXS study antioxidants Factorial 2 x 2 design Double blind treatment 1200 ACU patients Evidence of Multi-organ failure R glutamine R Concealed Stratified by site placebo antioxidants placebo R placebo
 The Research Protocol Inclusion Criteria • Adults (>18) • With 2 or more organ failures related to their acute illness : – Requiring mechanically ventilation (P/F<300) – Clinical evidence of hypoperfusion defined by need for vasopressor agents for more than 2 hour – Renal dysfunction : Cr>171 (>1. 93 mg/d. L) or <500 ml/24 hrs – platelet < 50
The Research Protocol Inclusion Criteria • Adults (>18) • With 2 or more organ failures related to their acute illness : – Requiring mechanically ventilation (P/F<300) – Clinical evidence of hypoperfusion defined by need for vasopressor agents for more than 2 hour – Renal dysfunction : Cr>171 (>1. 93 mg/d. L) or <500 ml/24 hrs – platelet < 50
 Optimizing the Dose of Glutamine Dipeptides and Antioxidants In Critically Ill Patients: A Phase I dose finding study Parenterally Enterally Glutamine/d ay 0. 35 gms/kg 30 gms Antioxidants per day 500 mcg Selenium Vit C 1500 mg Vit E 500 mg B carotene 10 mg Zinc 20 mg Se 300 ug • High dose appears safe • High dose associated with – – no worsening of SOFA Scores greater resolution of oxidative stress greater preservation of glutathione Improved mitochondrial function Heyland JPEN Mar 2007
Optimizing the Dose of Glutamine Dipeptides and Antioxidants In Critically Ill Patients: A Phase I dose finding study Parenterally Enterally Glutamine/d ay 0. 35 gms/kg 30 gms Antioxidants per day 500 mcg Selenium Vit C 1500 mg Vit E 500 mg B carotene 10 mg Zinc 20 mg Se 300 ug • High dose appears safe • High dose associated with – – no worsening of SOFA Scores greater resolution of oxidative stress greater preservation of glutathione Improved mitochondrial function Heyland JPEN Mar 2007
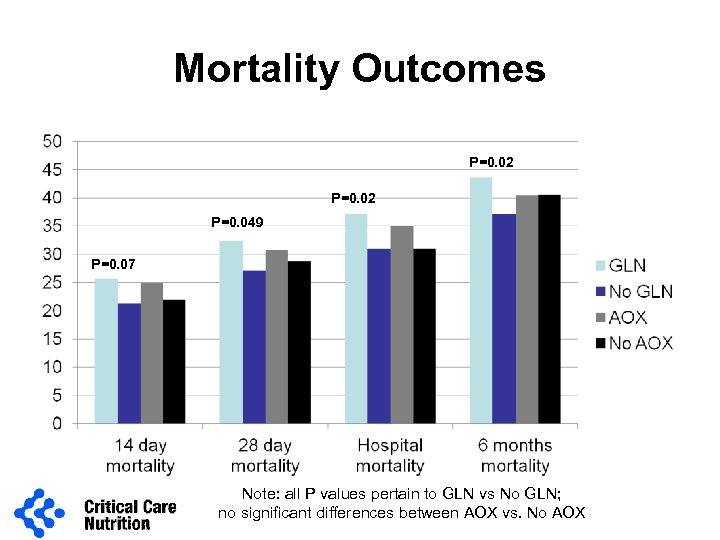 Mortality Outcomes P=0. 02 P=0. 049 P=0. 07 Note: all P values pertain to GLN vs No GLN; no significant differences between AOX vs. No AOX
Mortality Outcomes P=0. 02 P=0. 049 P=0. 07 Note: all P values pertain to GLN vs No GLN; no significant differences between AOX vs. No AOX
 Other Clinical Outcomes • No differences between groups – SOFA – Need for dialysis – Duration of mechanical ventilation – PODS – infections – ACU and Hospital LOS
Other Clinical Outcomes • No differences between groups – SOFA – Need for dialysis – Duration of mechanical ventilation – PODS – infections – ACU and Hospital LOS
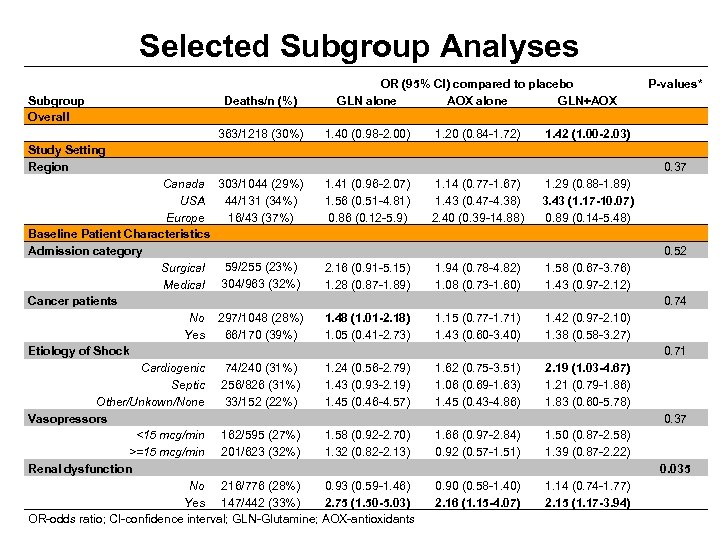 Selected Subgroup Analyses Subgroup Overall Study Setting Region OR (95% CI) compared to placebo GLN alone AOX alone GLN+AOX 1. 40 (0. 98 -2. 00) 1. 20 (0. 84 -1. 72) 1. 42 (1. 00 -2. 03) Canada 1. 41 (0. 96 -2. 07) 1. 14 (0. 77 -1. 67) 1. 29 (0. 88 -1. 89) USA 1. 56 (0. 51 -4. 81) 1. 43 (0. 47 -4. 38) 3. 43 (1. 17 -10. 07) Europe 0. 86 (0. 12 -5. 9) 2. 40 (0. 39 -14. 88) 0. 89 (0. 14 -5. 48) Baseline Patient Characteristics Admission category 59/255 (23%) Surgical 2. 16 (0. 91 -5. 15) 1. 94 (0. 78 -4. 82) 1. 58 (0. 67 -3. 76) Medical 304/963 (32%) 1. 28 (0. 87 -1. 89) 1. 08 (0. 73 -1. 60) 1. 43 (0. 97 -2. 12) Cancer patients No 297/1048 (28%) 1. 48 (1. 01 -2. 18) 1. 15 (0. 77 -1. 71) 1. 42 (0. 97 -2. 10) 66/170 (39%) Yes 1. 05 (0. 41 -2. 73) 1. 43 (0. 60 -3. 40) 1. 38 (0. 58 -3. 27) Etiology of Shock 74/240 (31%) Cardiogenic 1. 24 (0. 56 -2. 79) 1. 62 (0. 75 -3. 51) 2. 19 (1. 03 -4. 67) Septic 256/826 (31%) 1. 43 (0. 93 -2. 19) 1. 06 (0. 69 -1. 63) 1. 21 (0. 79 -1. 86) 33/152 (22%) Other/Unkown/None 1. 45 (0. 46 -4. 57) 1. 45 (0. 43 -4. 86) 1. 83 (0. 60 -5. 78) Vasopressors <15 mcg/min 162/595 (27%) 1. 58 (0. 92 -2. 70) 1. 66 (0. 97 -2. 84) 1. 50 (0. 87 -2. 58) >=15 mcg/min 201/623 (32%) 1. 32 (0. 82 -2. 13) 0. 92 (0. 57 -1. 51) 1. 39 (0. 87 -2. 22) Renal dysfunction No 216/776 (28%) 0. 93 (0. 59 -1. 46) 0. 90 (0. 58 -1. 40) 1. 14 (0. 74 -1. 77) Yes 147/442 (33%) 2. 75 (1. 50 -5. 03) 2. 16 (1. 15 -4. 07) 2. 15 (1. 17 -3. 94) OR-odds ratio; CI-confidence interval; GLN-Glutamine; AOX-antioxidants Deaths/n (%) 363/1218 (30%) 303/1044 (29%) 44/131 (34%) 16/43 (37%) P-values* 0. 37 0. 52 0. 74 0. 71 0. 37 0. 035
Selected Subgroup Analyses Subgroup Overall Study Setting Region OR (95% CI) compared to placebo GLN alone AOX alone GLN+AOX 1. 40 (0. 98 -2. 00) 1. 20 (0. 84 -1. 72) 1. 42 (1. 00 -2. 03) Canada 1. 41 (0. 96 -2. 07) 1. 14 (0. 77 -1. 67) 1. 29 (0. 88 -1. 89) USA 1. 56 (0. 51 -4. 81) 1. 43 (0. 47 -4. 38) 3. 43 (1. 17 -10. 07) Europe 0. 86 (0. 12 -5. 9) 2. 40 (0. 39 -14. 88) 0. 89 (0. 14 -5. 48) Baseline Patient Characteristics Admission category 59/255 (23%) Surgical 2. 16 (0. 91 -5. 15) 1. 94 (0. 78 -4. 82) 1. 58 (0. 67 -3. 76) Medical 304/963 (32%) 1. 28 (0. 87 -1. 89) 1. 08 (0. 73 -1. 60) 1. 43 (0. 97 -2. 12) Cancer patients No 297/1048 (28%) 1. 48 (1. 01 -2. 18) 1. 15 (0. 77 -1. 71) 1. 42 (0. 97 -2. 10) 66/170 (39%) Yes 1. 05 (0. 41 -2. 73) 1. 43 (0. 60 -3. 40) 1. 38 (0. 58 -3. 27) Etiology of Shock 74/240 (31%) Cardiogenic 1. 24 (0. 56 -2. 79) 1. 62 (0. 75 -3. 51) 2. 19 (1. 03 -4. 67) Septic 256/826 (31%) 1. 43 (0. 93 -2. 19) 1. 06 (0. 69 -1. 63) 1. 21 (0. 79 -1. 86) 33/152 (22%) Other/Unkown/None 1. 45 (0. 46 -4. 57) 1. 45 (0. 43 -4. 86) 1. 83 (0. 60 -5. 78) Vasopressors <15 mcg/min 162/595 (27%) 1. 58 (0. 92 -2. 70) 1. 66 (0. 97 -2. 84) 1. 50 (0. 87 -2. 58) >=15 mcg/min 201/623 (32%) 1. 32 (0. 82 -2. 13) 0. 92 (0. 57 -1. 51) 1. 39 (0. 87 -2. 22) Renal dysfunction No 216/776 (28%) 0. 93 (0. 59 -1. 46) 0. 90 (0. 58 -1. 40) 1. 14 (0. 74 -1. 77) Yes 147/442 (33%) 2. 75 (1. 50 -5. 03) 2. 16 (1. 15 -4. 07) 2. 15 (1. 17 -3. 94) OR-odds ratio; CI-confidence interval; GLN-Glutamine; AOX-antioxidants Deaths/n (%) 363/1218 (30%) 303/1044 (29%) 44/131 (34%) 16/43 (37%) P-values* 0. 37 0. 52 0. 74 0. 71 0. 37 0. 035
 Conclusions • Glutamine and antioxidants at doses studied in this study do not improve clinical outcomes in critically ill patients with multi-organ failure • Glutamine may be harmful • For both glutamine and antioxidants, the greatest signal of harm was in patients with multi-organ failure that included renal dysfunction upon study enrollment.
Conclusions • Glutamine and antioxidants at doses studied in this study do not improve clinical outcomes in critically ill patients with multi-organ failure • Glutamine may be harmful • For both glutamine and antioxidants, the greatest signal of harm was in patients with multi-organ failure that included renal dysfunction upon study enrollment.
 Metaplus Study • 14 ACUs in the Netherlands, Germany, France, and Belgium. • 301 adult patients who were expected to be ventilated and to require EN for more than 72 hours • Randomized to intervention feed – High-protein EN enriched with 21 grams of glutamine, extra antioxidants including an additional 275 mcg of selenium and an additional 7. 5 grams of fish oils, n = 152) – or control (standard high-protein EN, n = 149). • Intention-to-treat analysis for total population as well as predefined medical, surgical, and trauma subpopulations. Van Zanten JAMA 2014; 312: 514
Metaplus Study • 14 ACUs in the Netherlands, Germany, France, and Belgium. • 301 adult patients who were expected to be ventilated and to require EN for more than 72 hours • Randomized to intervention feed – High-protein EN enriched with 21 grams of glutamine, extra antioxidants including an additional 275 mcg of selenium and an additional 7. 5 grams of fish oils, n = 152) – or control (standard high-protein EN, n = 149). • Intention-to-treat analysis for total population as well as predefined medical, surgical, and trauma subpopulations. Van Zanten JAMA 2014; 312: 514
 Metaplus Study • No differences in infection (primary end point) – 53% in the enriched group vs 52% in the control group (P =. 96). • No differences in any secondary end points including mortality, Sequential Organ Failure Assessment (SOFA) scores, mechanical ventilation duration, ACU and hospital lengths of stay, and subtypes of infections according to CDC definitions. • hazard ratio of 1. 57 (95%CI, 1. 03 -2. 39; P =. 04) for 6 -month mortality adjusted for age and Acute Physiology and Chronic Health Evaluation II score • A higher 6 -month mortality rate in the medical subgroup: 54% (95%CI, 40%-67%) in the enriched group vs 35% (95%CI, 22%49%) in the control group (P =. 04). • No signal of harm in renal failure Van Zanten JAMA 2014; 312: 514
Metaplus Study • No differences in infection (primary end point) – 53% in the enriched group vs 52% in the control group (P =. 96). • No differences in any secondary end points including mortality, Sequential Organ Failure Assessment (SOFA) scores, mechanical ventilation duration, ACU and hospital lengths of stay, and subtypes of infections according to CDC definitions. • hazard ratio of 1. 57 (95%CI, 1. 03 -2. 39; P =. 04) for 6 -month mortality adjusted for age and Acute Physiology and Chronic Health Evaluation II score • A higher 6 -month mortality rate in the medical subgroup: 54% (95%CI, 40%-67%) in the enriched group vs 35% (95%CI, 22%49%) in the control group (P =. 04). • No signal of harm in renal failure Van Zanten JAMA 2014; 312: 514
 2015 Canadian Nutrition CPGs: IV Glutamine Recommendation: • When parenteral nutrition is prescribed to critically ill patients, we recommend parenteral supplementation with glutamine NOT be used*. • There are insufficient data to generate recommendations for intravenous glutamine in critically ill patients receiving enteral nutrition, but given the safety concerns we also recommend intravenous glutamine NOT be used in enterally fed critically ill patients. *downgraded from ‘should be considered’
2015 Canadian Nutrition CPGs: IV Glutamine Recommendation: • When parenteral nutrition is prescribed to critically ill patients, we recommend parenteral supplementation with glutamine NOT be used*. • There are insufficient data to generate recommendations for intravenous glutamine in critically ill patients receiving enteral nutrition, but given the safety concerns we also recommend intravenous glutamine NOT be used in enterally fed critically ill patients. *downgraded from ‘should be considered’
 2015 Canadian Nutrition CPGs: Combined IV+ EN Glutamine Recommendation: • Based on one level 1 study (REDOXS) and 1 level 2 study, we strongly recommend that high dose combined parenteral and enteral glutamine supplementation NOT be used in critically ill patients
2015 Canadian Nutrition CPGs: Combined IV+ EN Glutamine Recommendation: • Based on one level 1 study (REDOXS) and 1 level 2 study, we strongly recommend that high dose combined parenteral and enteral glutamine supplementation NOT be used in critically ill patients
 Key Question • If we conclude that glutamine has potential for harm, what impact does this have on our clinical recommendations for burn patients?
Key Question • If we conclude that glutamine has potential for harm, what impact does this have on our clinical recommendations for burn patients?
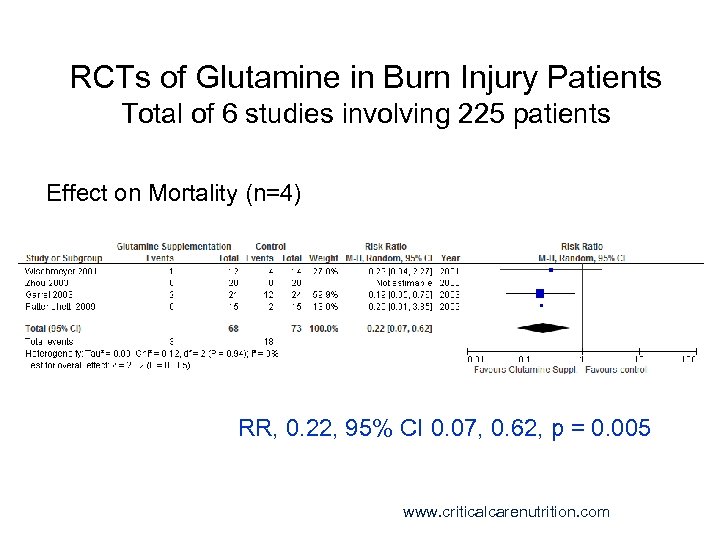 RCTs of Glutamine in Burn Injury Patients Total of 6 studies involving 225 patients Effect on Mortality (n=4) RR, 0. 22, 95% CI 0. 07, 0. 62, p = 0. 005 www. criticalcarenutrition. com
RCTs of Glutamine in Burn Injury Patients Total of 6 studies involving 225 patients Effect on Mortality (n=4) RR, 0. 22, 95% CI 0. 07, 0. 62, p = 0. 005 www. criticalcarenutrition. com
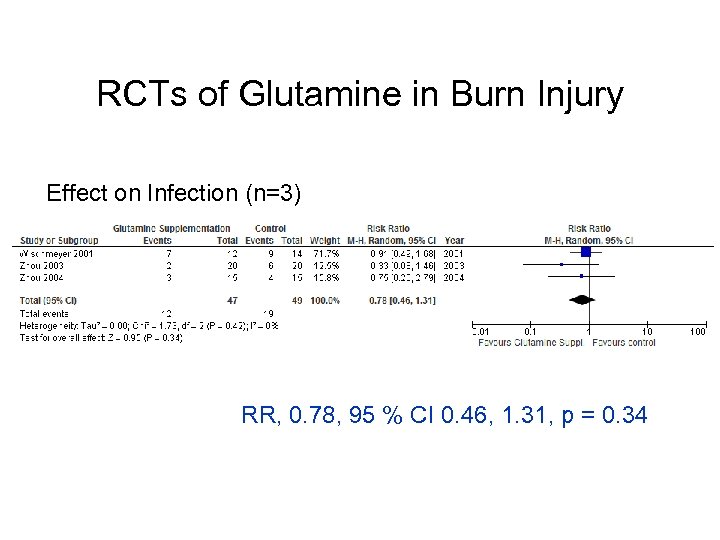 RCTs of Glutamine in Burn Injury Effect on Infection (n=3) RR, 0. 78, 95 % CI 0. 46, 1. 31, p = 0. 34
RCTs of Glutamine in Burn Injury Effect on Infection (n=3) RR, 0. 78, 95 % CI 0. 46, 1. 31, p = 0. 34
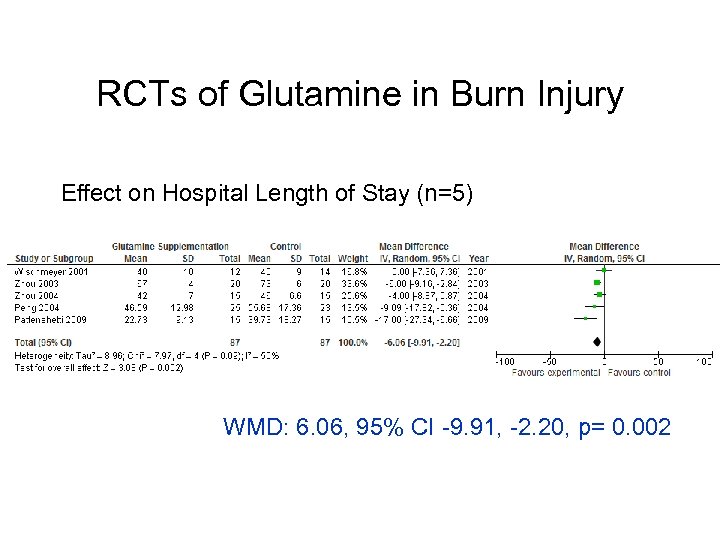 RCTs of Glutamine in Burn Injury Effect on Hospital Length of Stay (n=5) WMD: 6. 06, 95% CI -9. 91, -2. 20, p= 0. 002
RCTs of Glutamine in Burn Injury Effect on Hospital Length of Stay (n=5) WMD: 6. 06, 95% CI -9. 91, -2. 20, p= 0. 002
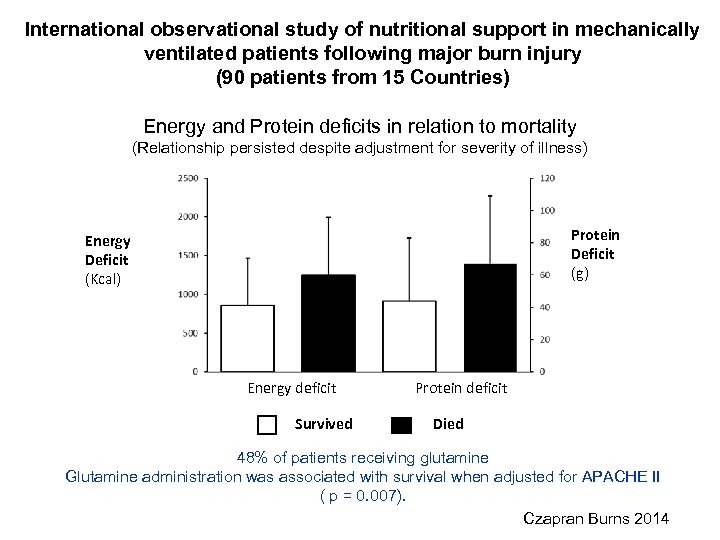 International observational study of nutritional support in mechanically ventilated patients following major burn injury (90 patients from 15 Countries) Energy and Protein deficits in relation to mortality (Relationship persisted despite adjustment for severity of illness) Protein Deficit (g) Energy Deficit (Kcal) Energy deficit Survived Protein deficit Died 48% of patients receiving glutamine Glutamine administration was associated with survival when adjusted for APACHE II ( p = 0. 007). Czapran Burns 2014
International observational study of nutritional support in mechanically ventilated patients following major burn injury (90 patients from 15 Countries) Energy and Protein deficits in relation to mortality (Relationship persisted despite adjustment for severity of illness) Protein Deficit (g) Energy Deficit (Kcal) Energy deficit Survived Protein deficit Died 48% of patients receiving glutamine Glutamine administration was associated with survival when adjusted for APACHE II ( p = 0. 007). Czapran Burns 2014
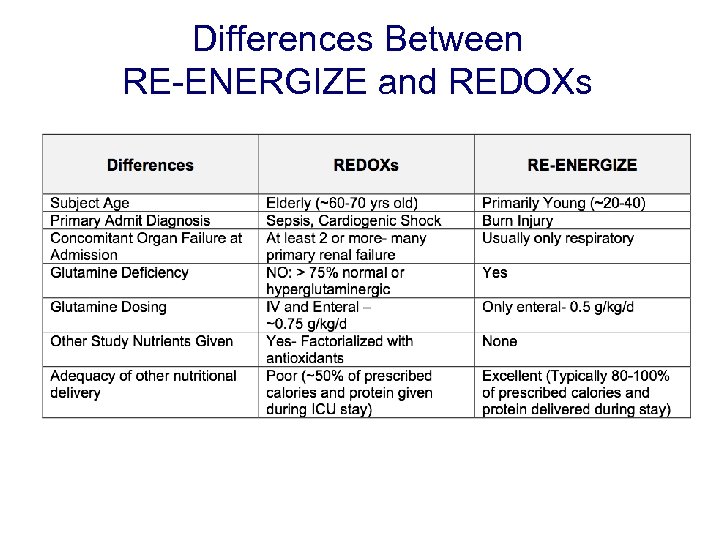 Differences Between RE-ENERGIZE and REDOXs
Differences Between RE-ENERGIZE and REDOXs
 Compelling Need to Answer the Study Question Primary research question: In patients with severe, life-threatening burn injury, what is the effect of enteral glutamine on 6 month mortality? Secondary Research question: In patients with severe, life-threatening burn injury, what is the effect of enteral glutamine on -time to discharge alive from hospital, -hospital-acquired blood infections, -hospital mortality, -duration of stay in ACU and hospital, -health-related quality of life -and health care resources?
Compelling Need to Answer the Study Question Primary research question: In patients with severe, life-threatening burn injury, what is the effect of enteral glutamine on 6 month mortality? Secondary Research question: In patients with severe, life-threatening burn injury, what is the effect of enteral glutamine on -time to discharge alive from hospital, -hospital-acquired blood infections, -hospital mortality, -duration of stay in ACU and hospital, -health-related quality of life -and health care resources?
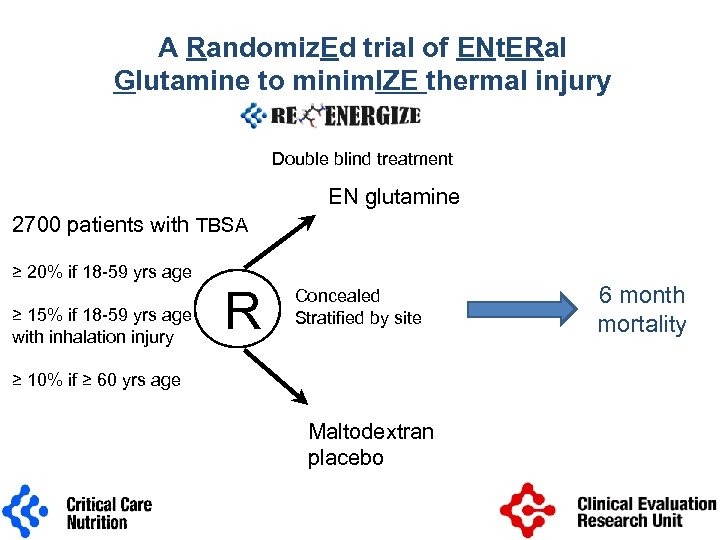 A Randomiz. Ed trial of ENt. ERal Glutamine to minim. IZE thermal injury Double blind treatment EN glutamine 2700 patients with TBSA ≥ 20% if 18 -59 yrs age ≥ 15% if 18 -59 yrs age with inhalation injury R Concealed Stratified by site ≥ 10% if ≥ 60 yrs age Maltodextran placebo 6 month mortality
A Randomiz. Ed trial of ENt. ERal Glutamine to minim. IZE thermal injury Double blind treatment EN glutamine 2700 patients with TBSA ≥ 20% if 18 -59 yrs age ≥ 15% if 18 -59 yrs age with inhalation injury R Concealed Stratified by site ≥ 10% if ≥ 60 yrs age Maltodextran placebo 6 month mortality
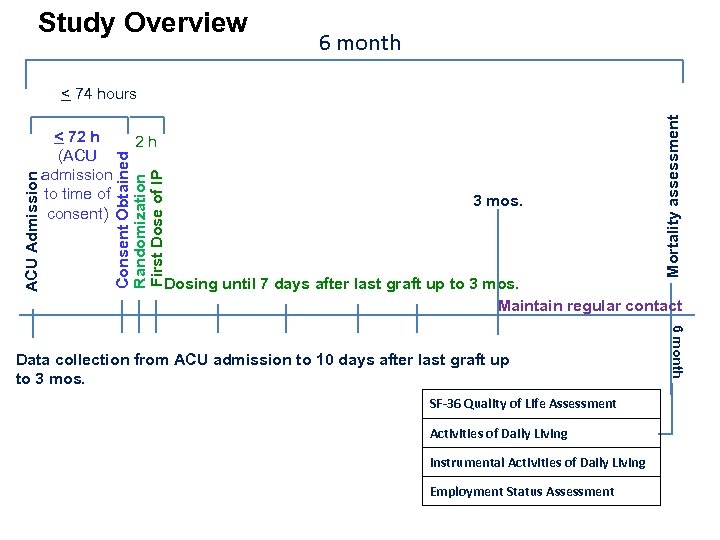 Study Overview 6 month 2 h 3 mos. Mortality assessment < 72 h (ACU admission to time of consent) Consent Obtained Randomization First Dose of IP ACU Admission < 74 hours Dosing until 7 days after last graft up to 3 mos. Maintain regular contact SF-36 Quality of Life Assessment Activities of Daily Living Instrumental Activities of Daily Living Employment Status Assessment 6 month Data collection from ACU admission to 10 days after last graft up to 3 mos.
Study Overview 6 month 2 h 3 mos. Mortality assessment < 72 h (ACU admission to time of consent) Consent Obtained Randomization First Dose of IP ACU Admission < 74 hours Dosing until 7 days after last graft up to 3 mos. Maintain regular contact SF-36 Quality of Life Assessment Activities of Daily Living Instrumental Activities of Daily Living Employment Status Assessment 6 month Data collection from ACU admission to 10 days after last graft up to 3 mos.
 Definitive Study: Inclusion Criteria • Deep 2 nd and/or 3 rd degree burns requiring skin grafting*. • For patients age 18 – 59 years we require a burn of ≥ 20% TBSA (Total Body Surface Area) or a minimum of 15 % TBSA when concomitant inhalation injury is present. • For patients aged 60 years or older we require a burn of ≥ 10% TBSA. Smoke inhalation injury is defined as: restricted to injury below the glottis caused by products of combustion. Diagnosis of inhalation injury requires both of the following: 1) history of exposure to products of combustion 2) bronchoscopy revealing one of the following below the glottis evidence of carbonaceous material signs of edema or ulceration *Note: there is no minimum area that requires grafting. As long as the total burn size meets the inclusion criteria and a graft is required, the patient is eligible.
Definitive Study: Inclusion Criteria • Deep 2 nd and/or 3 rd degree burns requiring skin grafting*. • For patients age 18 – 59 years we require a burn of ≥ 20% TBSA (Total Body Surface Area) or a minimum of 15 % TBSA when concomitant inhalation injury is present. • For patients aged 60 years or older we require a burn of ≥ 10% TBSA. Smoke inhalation injury is defined as: restricted to injury below the glottis caused by products of combustion. Diagnosis of inhalation injury requires both of the following: 1) history of exposure to products of combustion 2) bronchoscopy revealing one of the following below the glottis evidence of carbonaceous material signs of edema or ulceration *Note: there is no minimum area that requires grafting. As long as the total burn size meets the inclusion criteria and a graft is required, the patient is eligible.
 Definitive Study: Exclusion Criteria 1. >72 hrs from admission to ACU (Acute Care Unit) to time of consent 2. Patients younger than 18 years of age (age of maturity for an eligible patient to obtain consent is 18 years in Canada and in the US) 3. In patients without known renal disease, renal dysfunction defined as a q serum creatinine >171 μmol/L or >1. 93 mg/d. L or q urine output of less than 500 ml/last 24 hours (or 80 ml/last 4 hours if a 24 hour period of observation is not available). q In patients with acute on chronic renal failure (pre-dialysis), an absolute increase of >80 μmol/L or >0. 9 mg/d. L from baseline or pre-admission creatinine or a urine output of <500 ml/last 24 hours (or 80 ml/last 4 hours) will be required. q Patients with chronic renal failure on dialysis will be excluded. Changed to REDOXS criteria to address reviewer’s concerns
Definitive Study: Exclusion Criteria 1. >72 hrs from admission to ACU (Acute Care Unit) to time of consent 2. Patients younger than 18 years of age (age of maturity for an eligible patient to obtain consent is 18 years in Canada and in the US) 3. In patients without known renal disease, renal dysfunction defined as a q serum creatinine >171 μmol/L or >1. 93 mg/d. L or q urine output of less than 500 ml/last 24 hours (or 80 ml/last 4 hours if a 24 hour period of observation is not available). q In patients with acute on chronic renal failure (pre-dialysis), an absolute increase of >80 μmol/L or >0. 9 mg/d. L from baseline or pre-admission creatinine or a urine output of <500 ml/last 24 hours (or 80 ml/last 4 hours) will be required. q Patients with chronic renal failure on dialysis will be excluded. Changed to REDOXS criteria to address reviewer’s concerns
 Definitive Study: Exclusion Criteria 4. Liver cirrhosis -Child’s class C liver disease 5. Pregnancy (urine/blood tests for pregnancy will be done on all women of childbearing age by each site as part of standard ACU practice) 6. Contra-indication for EN: intestinal occlusion or perforation, intra-abdominal injury 7. Patients with injuries from high voltage electrical contact 8. Patients who are moribund (not expected to survive the next 72 hours) 9. Patients with extreme body sizes: BMI < 18 or > 50 kg/m 2 10. Enrolment in another industry sponsored ACU intervention study (coenrollment in academic studies will be considered on a case by case basis) 11. Received glutamine supplement for > 24 hrs prior to randomization 12. Known allergy to maltodextrin, corn starch, corn products or glutamine
Definitive Study: Exclusion Criteria 4. Liver cirrhosis -Child’s class C liver disease 5. Pregnancy (urine/blood tests for pregnancy will be done on all women of childbearing age by each site as part of standard ACU practice) 6. Contra-indication for EN: intestinal occlusion or perforation, intra-abdominal injury 7. Patients with injuries from high voltage electrical contact 8. Patients who are moribund (not expected to survive the next 72 hours) 9. Patients with extreme body sizes: BMI < 18 or > 50 kg/m 2 10. Enrolment in another industry sponsored ACU intervention study (coenrollment in academic studies will be considered on a case by case basis) 11. Received glutamine supplement for > 24 hrs prior to randomization 12. Known allergy to maltodextrin, corn starch, corn products or glutamine
 Study Intervention OR MALTRIN® Maltodextrin Enteral glutamine (0. 5 gm/kg/day) or maltodextrin will be given through the feeding tube, q 4 h until 7 days post last successful graft
Study Intervention OR MALTRIN® Maltodextrin Enteral glutamine (0. 5 gm/kg/day) or maltodextrin will be given through the feeding tube, q 4 h until 7 days post last successful graft
 Study Intervention • The study intervention is to be mixed in 50 m. L of water per 5 g packet and given as a bolus every 4 hours via the enteral route. • Administer the study intervention immediately after mixing with water to avoid thickening of the mixture. • If the mixture becomes too thick to administer as a bolus, add water. • Have not observed increase in tube blockages • When the patient is tolerating oral feeds, the boluses may be switched to TID or QID via the oral route according to the patient’s preference, as long as the patient receives the daily prescribed number of packets.
Study Intervention • The study intervention is to be mixed in 50 m. L of water per 5 g packet and given as a bolus every 4 hours via the enteral route. • Administer the study intervention immediately after mixing with water to avoid thickening of the mixture. • If the mixture becomes too thick to administer as a bolus, add water. • Have not observed increase in tube blockages • When the patient is tolerating oral feeds, the boluses may be switched to TID or QID via the oral route according to the patient’s preference, as long as the patient receives the daily prescribed number of packets.
 Study Intervention • Avoid mixing the IP in water when given orally. Patients who participated in the pilot study reported it did not taste good when mixed with water. • There should be little to no difference in taste of the glutamine and maltodextrin. • Do NOT mix the study intervention with: – soda or – highly acidic juices, such as: • grapefruit juice • orange juice • lemonade. Why? • The study intervention becomes unstable in an acidic medium.
Study Intervention • Avoid mixing the IP in water when given orally. Patients who participated in the pilot study reported it did not taste good when mixed with water. • There should be little to no difference in taste of the glutamine and maltodextrin. • Do NOT mix the study intervention with: – soda or – highly acidic juices, such as: • grapefruit juice • orange juice • lemonade. Why? • The study intervention becomes unstable in an acidic medium.
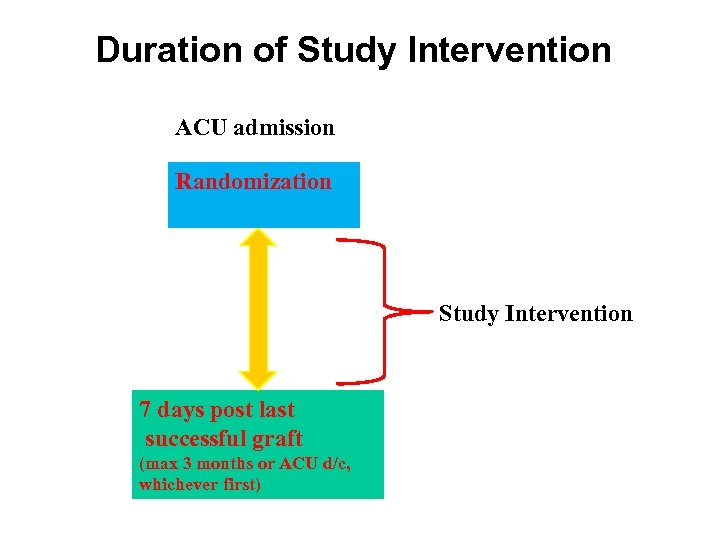 Duration of Study Intervention ACU admission Randomization Study Intervention 7 days post last successful graft (max 3 months or ACU d/c, whichever first)
Duration of Study Intervention ACU admission Randomization Study Intervention 7 days post last successful graft (max 3 months or ACU d/c, whichever first)
 Co-interventions: Standardization of Nutrition • Prescribed calorie ranges • Prescribed protein ranges • Vitamin/mineral prescription • Optimization of the Delivery of Enteral Nutrition • Glycemic control • Arginine enriched formulas (> 6 gms arginine/L) i. e. Pivot, Perative • Glutamine supplements or formulas containing higher than standard amounts of glutamine i. e. Juven
Co-interventions: Standardization of Nutrition • Prescribed calorie ranges • Prescribed protein ranges • Vitamin/mineral prescription • Optimization of the Delivery of Enteral Nutrition • Glycemic control • Arginine enriched formulas (> 6 gms arginine/L) i. e. Pivot, Perative • Glutamine supplements or formulas containing higher than standard amounts of glutamine i. e. Juven

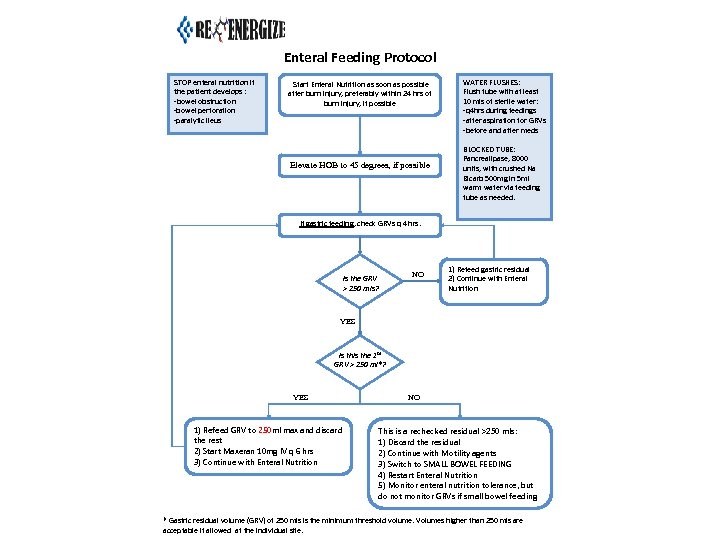 Enteral Feeding Protocol STOP enteral nutrition if the patient develops : -bowel obstruction -bowel perforation -paralytic ileus Start Enteral Nutrition as soon as possible after burn injury, preferably within 24 hrs of burn injury, if possible Elevate HOB to 45 degrees, if possible WATER FLUSHES: Flush tube with at least 10 mls of sterile water: -q 4 hrs during feedings -after aspiration for GRVs -before and after meds BLOCKED TUBE: Pancrealipase, 8000 units, with crushed Na Bicarb 500 mg in 5 ml warm water via feeding tube as needed. If gastric feeding, check GRVs q 4 hrs. Is the GRV > 250 mls? NO 1) Refeed gastric residual 2) Continue with Enteral Nutrition YES Is this the 1 st GRV > 250 ml*? YES 1) Refeed GRV to 250 ml max and discard the rest 2) Start Maxeran 10 mg IV q 6 hrs 3) Continue with Enteral Nutrition NO This is a rechecked residual >250 mls: 1) Discard the residual 2) Continue with Motility agents 3) Switch to SMALL BOWEL FEEDING 4) Restart Enteral Nutrition 5) Monitor enteral nutrition tolerance, but do not monitor GRVs if small bowel feeding * Gastric residual volume (GRV) of 250 mls is the minimum threshold volume. Volumes higher than 250 mls are acceptable if allowed at the individual site.
Enteral Feeding Protocol STOP enteral nutrition if the patient develops : -bowel obstruction -bowel perforation -paralytic ileus Start Enteral Nutrition as soon as possible after burn injury, preferably within 24 hrs of burn injury, if possible Elevate HOB to 45 degrees, if possible WATER FLUSHES: Flush tube with at least 10 mls of sterile water: -q 4 hrs during feedings -after aspiration for GRVs -before and after meds BLOCKED TUBE: Pancrealipase, 8000 units, with crushed Na Bicarb 500 mg in 5 ml warm water via feeding tube as needed. If gastric feeding, check GRVs q 4 hrs. Is the GRV > 250 mls? NO 1) Refeed gastric residual 2) Continue with Enteral Nutrition YES Is this the 1 st GRV > 250 ml*? YES 1) Refeed GRV to 250 ml max and discard the rest 2) Start Maxeran 10 mg IV q 6 hrs 3) Continue with Enteral Nutrition NO This is a rechecked residual >250 mls: 1) Discard the residual 2) Continue with Motility agents 3) Switch to SMALL BOWEL FEEDING 4) Restart Enteral Nutrition 5) Monitor enteral nutrition tolerance, but do not monitor GRVs if small bowel feeding * Gastric residual volume (GRV) of 250 mls is the minimum threshold volume. Volumes higher than 250 mls are acceptable if allowed at the individual site.
 Outcomes Primary Outcome: 6 month mortality Secondary Outcome: Time to discharge alive Teritiary Outcomes: • Health-related quality of life (SF-36 @ 6 months) • Incidence of acquired bacteremias due to Gram negative organisms • Hospital Mortality • Duration of mechanical ventilation • ACU stay and hospital stay
Outcomes Primary Outcome: 6 month mortality Secondary Outcome: Time to discharge alive Teritiary Outcomes: • Health-related quality of life (SF-36 @ 6 months) • Incidence of acquired bacteremias due to Gram negative organisms • Hospital Mortality • Duration of mechanical ventilation • ACU stay and hospital stay
 Results of Pilot Study • 203 patients enrolled in 8 sites • Approx 1 patient/site/month • >90% of prescribed doses of study medication were delivered. • Only 2% of patients have been lost to follow-up at 6 months. • Good compliance with study procedures and filling out the case report form • Plan to roll the data from the pilot trial into this current proposal so we have not analyzed the trial by group.
Results of Pilot Study • 203 patients enrolled in 8 sites • Approx 1 patient/site/month • >90% of prescribed doses of study medication were delivered. • Only 2% of patients have been lost to follow-up at 6 months. • Good compliance with study procedures and filling out the case report form • Plan to roll the data from the pilot trial into this current proposal so we have not analyzed the trial by group.
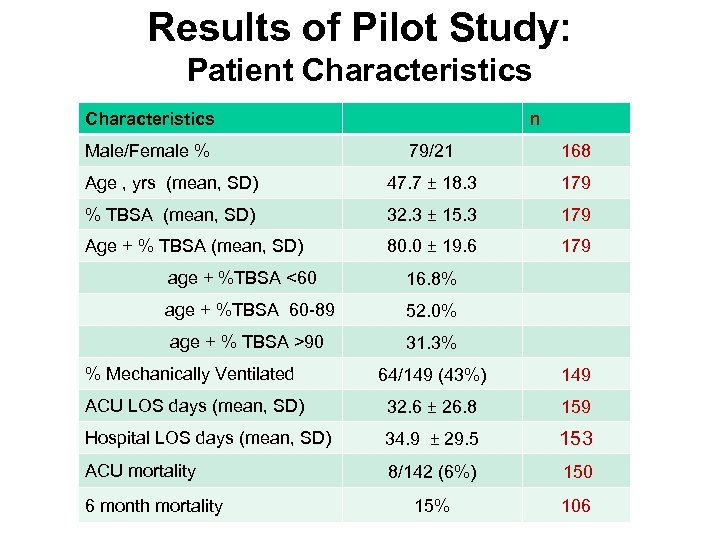 Results of Pilot Study: Patient Characteristics Male/Female % n 79/21 168 Age , yrs (mean, SD) 47. 7 ± 18. 3 179 % TBSA (mean, SD) 32. 3 ± 15. 3 179 Age + % TBSA (mean, SD) 80. 0 ± 19. 6 179 age + %TBSA <60 age + %TBSA 60 -89 age + % TBSA >90 16. 8% 52. 0% 31. 3% % Mechanically Ventilated 64/149 (43%) 149 ACU LOS days (mean, SD) 32. 6 ± 26. 8 159 Hospital LOS days (mean, SD) 34. 9 ± 29. 5 153 ACU mortality 8/142 (6%) 150 15% 106 6 month mortality
Results of Pilot Study: Patient Characteristics Male/Female % n 79/21 168 Age , yrs (mean, SD) 47. 7 ± 18. 3 179 % TBSA (mean, SD) 32. 3 ± 15. 3 179 Age + % TBSA (mean, SD) 80. 0 ± 19. 6 179 age + %TBSA <60 age + %TBSA 60 -89 age + % TBSA >90 16. 8% 52. 0% 31. 3% % Mechanically Ventilated 64/149 (43%) 149 ACU LOS days (mean, SD) 32. 6 ± 26. 8 159 Hospital LOS days (mean, SD) 34. 9 ± 29. 5 153 ACU mortality 8/142 (6%) 150 15% 106 6 month mortality
 Current Status and Timelines • Definitive Trial Funded by CIHR • Site start-up in progress – Pilot sites resume by Dec 1, 2015 – Rest of Canadian sites initiate by end of 2015 – New sites on board by end of second quarter 2016 • Continue to develop network of interested burn units • 4 years of enrolling patients
Current Status and Timelines • Definitive Trial Funded by CIHR • Site start-up in progress – Pilot sites resume by Dec 1, 2015 – Rest of Canadian sites initiate by end of 2015 – New sites on board by end of second quarter 2016 • Continue to develop network of interested burn units • 4 years of enrolling patients
 Questions and Discussion Points? • • Patients? Study Products Feeding protocol 6 month follow up Adequate research infrastructure Competing studies Financials and Timelines
Questions and Discussion Points? • • Patients? Study Products Feeding protocol 6 month follow up Adequate research infrastructure Competing studies Financials and Timelines
 Conclusions • RE-ENERGIZE trial – Will ask a clinically important question – Has the potential to save lives and reduce morbidity whether positive or negative – Largest burn trial ever! • Thank you for your interest and support
Conclusions • RE-ENERGIZE trial – Will ask a clinically important question – Has the potential to save lives and reduce morbidity whether positive or negative – Largest burn trial ever! • Thank you for your interest and support
 Questions?
Questions?


