2c2db8902206e9af4b915d2d0f006e77.ppt
- Количество слайдов: 43

A Prospective, Randomized Comparison of Bivalirudin vs. Heparin Plus Glycoprotein IIb/IIIa Inhibitors During Primary Angioplasty in Acute Myocardial Infarction – 30 Day Results – Gregg W. Stone MD For the HORIZONS AMI Investigators

Disclosures ¼ Gregg W. Stone MD – Research support from The Medicines Company and Boston Scientific – Honoraria from Eli Lilly Co.

Background ¼ ¼ ¼ In addition to suppressing periprocedural ischemia, prevention of hemorrhagic complications has emerged as a priority in patients undergoing PCI In patients with stable angina and NSTEMI, the direct thrombin inhibitor bivalirudin has been shown to result in similar rates of composite ischemia as heparin plus GP IIb/IIIa inhibitors, while significantly reducing major bleeding Whether bivalirudin has comparable safety and efficacy in patients with STEMI undergoing primary PCI is unknown
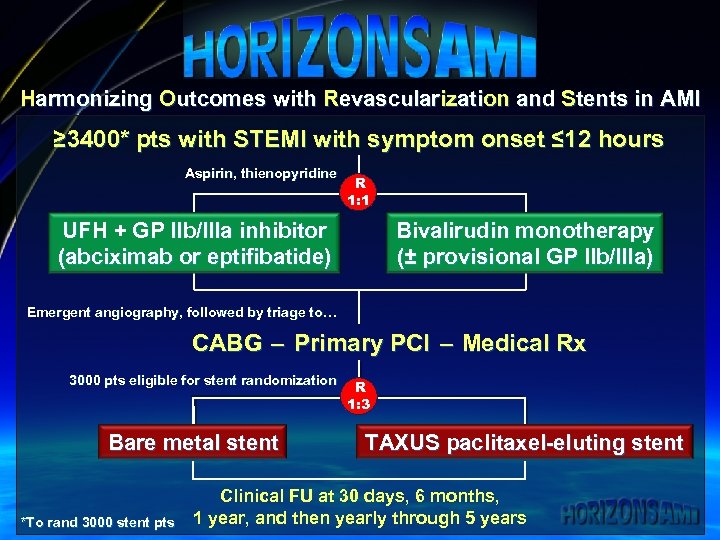
Harmonizing Outcomes with Revascularization and Stents in AMI ≥ 3400* pts with STEMI with symptom onset ≤ 12 hours Aspirin, thienopyridine R 1: 1 UFH + GP IIb/IIIa inhibitor (abciximab or eptifibatide) Bivalirudin monotherapy (± provisional GP IIb/IIIa) Emergent angiography, followed by triage to… CABG – Primary PCI – Medical Rx 3000 pts eligible for stent randomization Bare metal stent *To rand 3000 stent pts R 1: 3 TAXUS paclitaxel-eluting stent Clinical FU at 30 days, 6 months, 1 year, and then yearly through 5 years

Harmonizing Outcomes with Revascularization and Stents in AMI ≥ 3400* pts with STEMI with symptom onset ≤ 12 hours Aspirin, thienopyridine UFH + GP IIb/IIIa inhibitor (abciximab or eptifibatide) R 1: 1 Bivalirudin monotherapy (± provisional GP IIb/IIIa) Pharmacology Arm Primary Endpoints* 30 Day Intention to Treat Population * All stent randomization results are still blinded

30 Day Study Objectives ¼ In patients with STEMI undergoing a primary PCI strategy, compared to UFH plus the routine use of GP IIb/IIIa inhibitors, bivalirudin monotherapy will result in: – Similar or reduced rates of net adverse clinical events (the composite of major adverse cardiovascular events and major bleeding) at 30 days – Similar or reduced rates of major bleeding at 30 days

Inclusion Criteria ¼ STEMI >20 mins and <12 hours in duration – ST-segment elevation of 1 mm in 2 contiguous leads; or – Presumably new left bundle branch block; or – True posterior MI with ST depression of 1 mm in 2 contiguous anterior leads – Patients with cardiogenic shock, left main disease, etc. , were not excluded ¼ Age ≥ 18 years ¼ Written, informed consent

Principal Exclusion Criteria ¼ ¼ ¼ Contraindication to any of the study medications Prior administration of thrombolytic therapy, bivalirudin, GP IIb/IIIa inhibitors, LMWH or fondaparinux for the present admission (prior UFH allowed) Current use of coumadin History of bleeding diathesis or known coagulopathy (including HIT), or will refuse blood transfusions History of intracerebral mass, aneurysm, AVM, or hemorrhagic stroke; stroke or TIA within 6 months or any permanent neurologic deficit; GI or GU bleed within 2 months, or major surgery within 6 weeks; recent or known platelet count <100, 000 cells/mm 3 or hgb <10 g/d. L Planned elective surgical procedure that would necessitate interruption of thienopyridines during the first 6 months post enrollment

Study Medications (i) ¼ Unfractionated heparin – 60 U/kg IV*; subsequent boluses titrated by nomogram to ACT 200 -250 secs; terminated at procedure end unless prolonged antithrombin needed ¼ Bivalirudin – Bolus 0. 75 mg/kg IV**, infusion 1. 75 mg/kg/h, not titrated to ACT; terminated at procedure end unless prolonged antithrombin needed (0. 25 mg/kg/hr infusion) ¼ Glycoprotein IIb/IIIa inhibitors – Routine use in UFH arm; recommended only for giant thrombus or refractory no reflow in bivalirudin arm – Abciximab or double bolus eptifibatide as per investigator discretion – dosing per FDA label, renal adjusted; continued for 12 (abcx) or 12 -18 (eptif) * If pre randomization UFH administered, ACT is checked first ** If pre randomization UFH administered, started 30’ after last bolus

Study Medications (ii) ¼ Aspirin – 324 mg chewed non enteric coated or 500 mg IV in the ER, followed by 300 -325 mg/day in-hospital and 75 -81 mg/day as out patient indefinitely ¼ Thienopyridines – Clopidogrel 300 mg or 600 mg loading dose (per investigator discretion) in the ER followed by 75 mg PO QD for at least 6 months (1 year or longer recommended) • Ticlopidine load + daily dose permissible if clopiodgrel is unavailable or patient is allergic ¼ Other – Beta blockers: IV pre procedure followed by PO QD in the absence of contraindications; ACE inhibitors for HTN, CHF or LVEF <40%; Statin if LDL >100 mg/dl

2 Primary Endpoints (at 30 Days) 1) Net Adverse Clinical Events and 2) Major Bleeding (non CABG) • Intracranial bleeding • intraocular bleeding • Retroperitoneal bleeding • Access site bleed requiring intervention/surgery • Hematoma ≥ 5 cm • Hgb ≥ 3 g/d. L with an overt source • Hgb ≥ 4 g/d. L w/o overt source • Reoperation for bleeding • Blood product transfusion

2 Primary Endpoints (at 30 Days) 1) Net Adverse Clinical Events = 2) Major Bleeding (non CABG) or Major adverse cardiovascular events (major secondary endpoint) • All cause death • Reinfarction • Ischemic TVR • Stroke

Statistical Methodology ¼ Randomization stratification i. ii. Administration of non protocol pre-procedural heparin Planned administration of 300 or 600 mg of clopidogrel or 500 mg of ticlopidine prior to cardiac catheterization iii. Whether the patient will receive abciximab or eptifibatide iv. U. S. vs. non-U. S. site ¼ ¼ Primary analysis is by ITT among all randomized pts; secondary analysis is ITT among primary PCI cohort Sequential noninferiority and superiority testing – NI for net adverse clinical events NI for major bleeding Sup for adverse clinical net events – One-sided α=0. 025 for NI; Two-sided α=0. 05 for Sup – Primary analyses conducted using binomial proportions
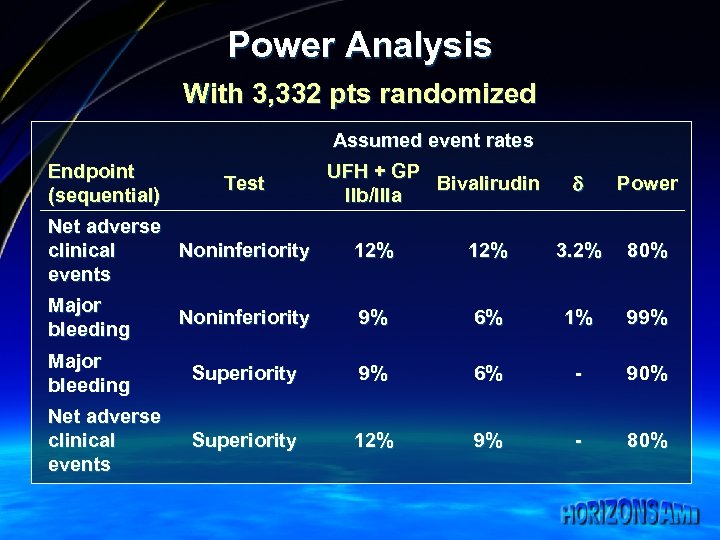
Power Analysis With 3, 332 pts randomized Assumed event rates Endpoint (sequential) Test UFH + GP Bivalirudin IIb/IIIa Power Net adverse clinical Noninferiority events 12% 3. 2% 80% Major bleeding Noninferiority 9% 6% 1% 99% Major bleeding Superiority 9% 6% - 90% Net adverse clinical events Superiority 12% 9% - 80%

Study Organization ¼ Sponsor: The Cardiovascular Research Foundation ¼ Grant Support: Boston Scientific Corporation The Medicines Company ¼ Principal Investigator: Gregg W. Stone MD ¼ Steering Committee: Gregg W. Stone (Chair), Bruce R. Brodie, David A. Cox, Cindy L. Grines, Barry D. Rutherford European Steering Committee: H Bonnier, A Colombo, Eulogio Garcia, E Grube, G Guagliumi, A Kastrati, P Serruys, H Suryapranata Additional Country Leaders: Y Almagor, A Banning, J Belardi, D Dudek, L Grinfeld, K Huber, D Nilsen, G Olivecrona, L Rasmussen Pharmacology Committee: Deepak Bhatt, George Dangas, Fred Feit, Magnus Ohman ¼ ¼ ¼ (unrestricted)

Study Organization ¼ Data Management: CRF, Roxana Mehran (Director), Irene Bihl (Ops), Helen Parise (Stats) ¼ Clinical Events Committee: CRF, S. Chiu Wong (Chair) ¼ Site and Data Monitoring: J. Tyson and Associates (USA), D-Target (Europe), Tango (South America) ¼ Angiographic Core Lab: CRF, Alexandra Lansky (Director), Ecaterina Cristea (Ops) ¼ ECG Core Lab: CRF, James Reiffel (Director) ¼ IVUS Core lab: CRF, Gary Mintz (Director) ¼ DSMB: Bernhard Gersh (Chair), David Faxon, Spencer King, Stuart Pocock, David Williams
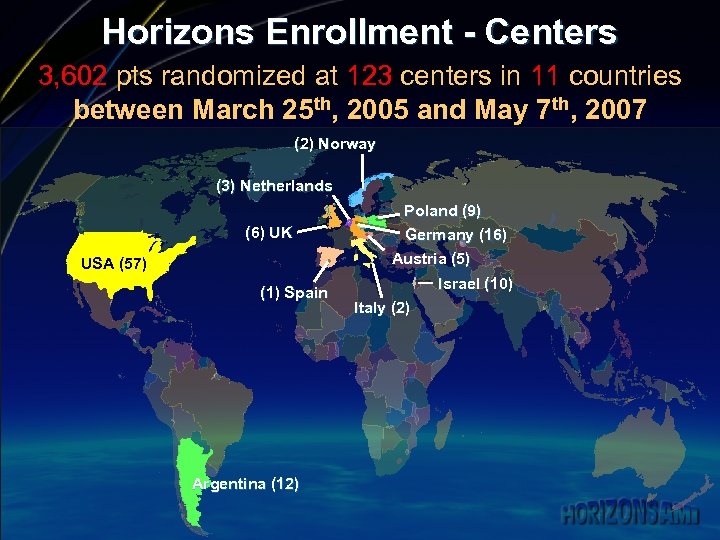
Horizons Enrollment - Centers 3, 602 pts randomized at 123 centers in 11 countries between March 25 th, 2005 and May 7 th, 2007 (2) Norway (3) Netherlands (6) UK USA (57) (1) Spain Argentina (12) Poland (9) Germany (16) Austria (5) Israel (10) Italy (2)
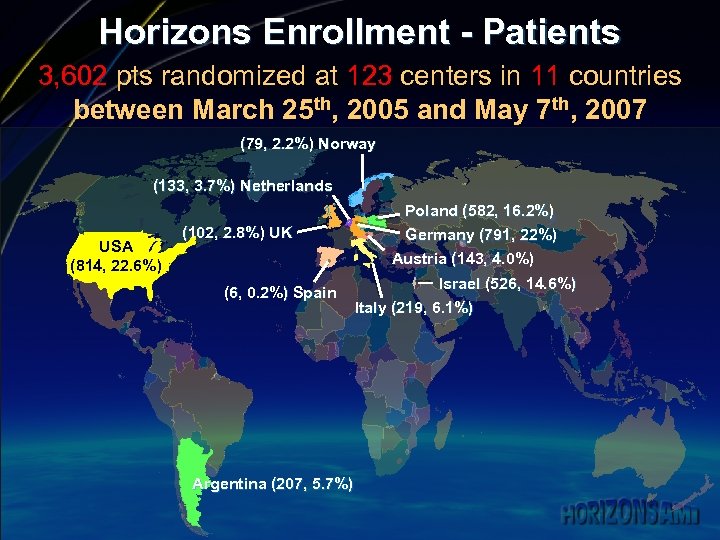
Horizons Enrollment - Patients 3, 602 pts randomized at 123 centers in 11 countries between March 25 th, 2005 and May 7 th, 2007 (79, 2. 2%) Norway (133, 3. 7%) Netherlands USA (814, 22. 6%) (102, 2. 8%) UK (6, 0. 2%) Spain Argentina (207, 5. 7%) Poland (582, 16. 2%) Germany (791, 22%) Austria (143, 4. 0%) Israel (526, 14. 6%) Italy (219, 6. 1%)

Top 20 Enrolling Sites 1. B. Witzenbichler, Germany 11. H. Suryapranata, Netherlands 2. G. Guagliumi, Italy 12. K. Huber, Austria 3. J. Peruga, Poland 13. J. Wöehrle, Germany 4. B. Brodie, USA 14. C. Metzger, USA 5. R. Kornowski, Israel 15. M. Desaga, Germany 6. F. Hartmann, Germany 16. K. Zmudka, Poland 7. M. Moeckel, Germany 17. J. Kochman, Poland 8. A. Ochala, Poland 18. D. Nilsen, Norway 9. W. Ruzyllo, Poland 19. D. Dudek, Poland 10. V. Guetta, Israel 20. A. Finkelstein, Israel

Harmonizing Outcomes with Revascularization and Stents in AMI 3602 pts with STEMI R 1: 1 Randomized UFH + GP IIb/IIIa N=1802 9 15 Bivalirudin Monotherapy N=1800 • • • Withdrew • • • Lost to FU • • • 10 13 30 day FU* N=1778 (98. 7%) N=1777 (98. 7%) ITT population N=1802 N=1800 * Range ± 7 days
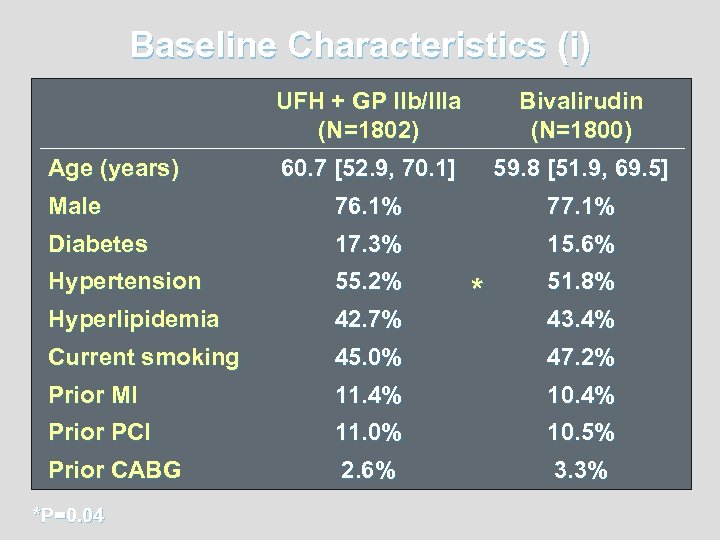
Baseline Characteristics (i) UFH + GP IIb/IIIa (N=1802) Bivalirudin (N=1800) 60. 7 [52. 9, 70. 1] 59. 8 [51. 9, 69. 5] Male 76. 1% 77. 1% Diabetes 17. 3% 15. 6% Hypertension 55. 2% Hyperlipidemia 42. 7% Current smoking 45. 0% 47. 2% Prior MI 11. 4% 10. 4% Prior PCI 11. 0% 10. 5% Prior CABG 2. 6% 3. 3% Age (years) *P=0. 04 * 51. 8% 43. 4%
![Baseline Characteristics (ii) UFH + GP IIb/IIIa (N=1802) Bivalirudin (N=1800) 80 [71, 90] 2. Baseline Characteristics (ii) UFH + GP IIb/IIIa (N=1802) Bivalirudin (N=1800) 80 [71, 90] 2.](https://present5.com/presentation/2c2db8902206e9af4b915d2d0f006e77/image-22.jpg)
Baseline Characteristics (ii) UFH + GP IIb/IIIa (N=1802) Bivalirudin (N=1800) 80 [71, 90] 2. 1 [1. 3, 3. 9] 2. 2 [1. 3, 4. 0] Killip class 2 -4 8. 5% Anterior MI 43. 9% 41. 2% 50 [41, 59] 50 [45, 60] Femoral a. access 93. 6% 93. 9% Venous access 8. 4% 9. 3% Closure device 27. 7% 28. 3% Weight (kg) Chest pain – ER, hrs LVEF

Study Drugs UFH + GP IIb/IIIa Bivalirudin (N=1802) (N=1800) UFH pre randomization 65. 6% Antithrombin in CCL - UFH 98. 9% 4. 1% - Bivalirudin 0. 4% 96. 9% - Peak ACT 264 [228, 320] 357 [300, 402] GP IIb/IIIa in CCL 94. 5%* 7. 2%* - Bail-out per protocol** 4. 4% - Abciximab 49. 9% 4. 0% - Eptifibatide 44. 4% 3. 1% - Tirofiban 0. 2% 0. 1% *97. 7% and 7. 5% during PCI. ** For giant thrombus or refractory no reflow after PCI. CCL = cardiac catheterization laboratory
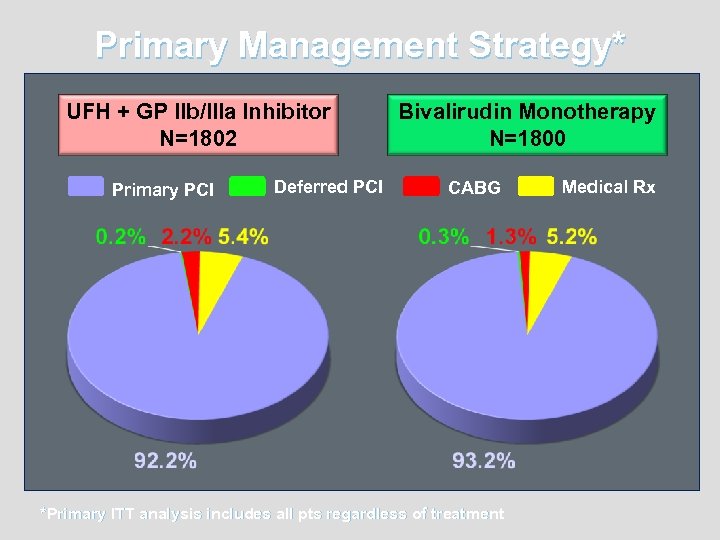
Primary Management Strategy* UFH + GP IIb/IIIa Inhibitor N=1802 Primary PCI Deferred PCI Bivalirudin Monotherapy N=1800 CABG *Primary ITT analysis includes all pts regardless of treatment Medical Rx
![Primary Outcome Measures (ITT) Diff = -2. 9% [-4. 9, -0. 8] RR = Primary Outcome Measures (ITT) Diff = -2. 9% [-4. 9, -0. 8] RR =](https://present5.com/presentation/2c2db8902206e9af4b915d2d0f006e77/image-25.jpg)
Primary Outcome Measures (ITT) Diff = -2. 9% [-4. 9, -0. 8] RR = 0. 76 [0. 63, 0. 92] Diff = -3. 3% [-5. 0, -1. 6] RR = 0. 60 [0. 46, 0. 77] Diff = 0. 0% [-1. 6, 1. 5] RR = 0. 99 [0. 76, 1. 30] PNI ≤ 0. 0001 Psup = 0. 006 PNI ≤ 0. 0001 Psup = 1. 00 1 endpoint *Not related to CABG **MACE = All cause death, reinfarction, ischemic TVR or stroke
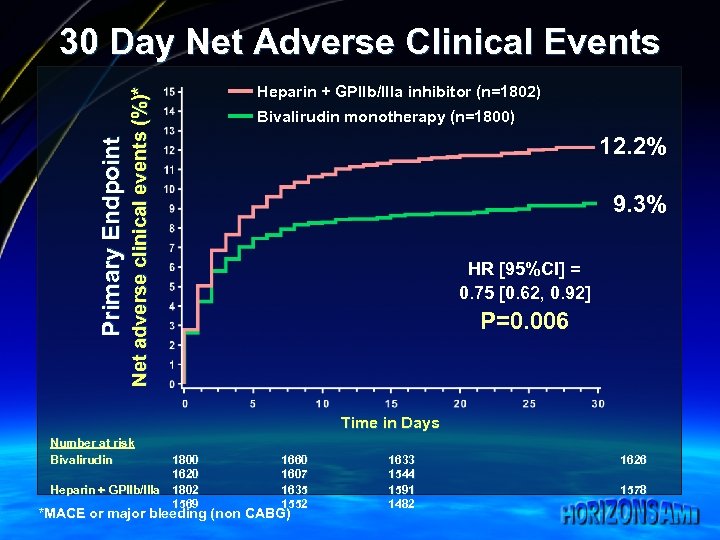
Net adverse clinical events (%)* Primary Endpoint 30 Day Net Adverse Clinical Events Heparin + GPIIb/IIIa inhibitor (n=1802) Bivalirudin monotherapy (n=1800) 12. 2% 9. 3% HR [95%CI] = 0. 75 [0. 62, 0. 92] P=0. 006 Time in Days Number at risk Bivalirudin 1800 1620 Heparin + GPIIb/IIIa 1802 1569 1660 1607 1635 1552 *MACE or major bleeding (non CABG) 1633 1544 1591 1482 1626 1578
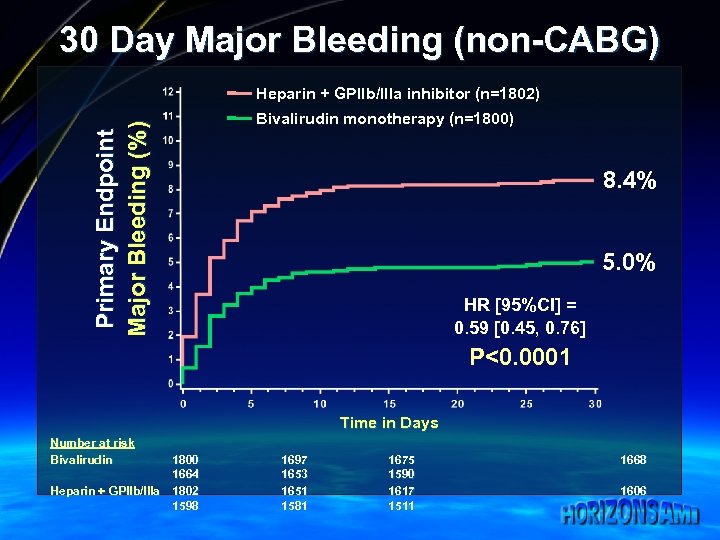
30 Day Major Bleeding (non-CABG) Primary Endpoint Major Bleeding (%) Heparin + GPIIb/IIIa inhibitor (n=1802) Bivalirudin monotherapy (n=1800) 8. 4% 5. 0% HR [95%CI] = 0. 59 [0. 45, 0. 76] P<0. 0001 Time in Days Number at risk Bivalirudin 1800 1664 Heparin + GPIIb/IIIa 1802 1598 1697 1653 1651 1581 1675 1590 1617 1511 1668 1606
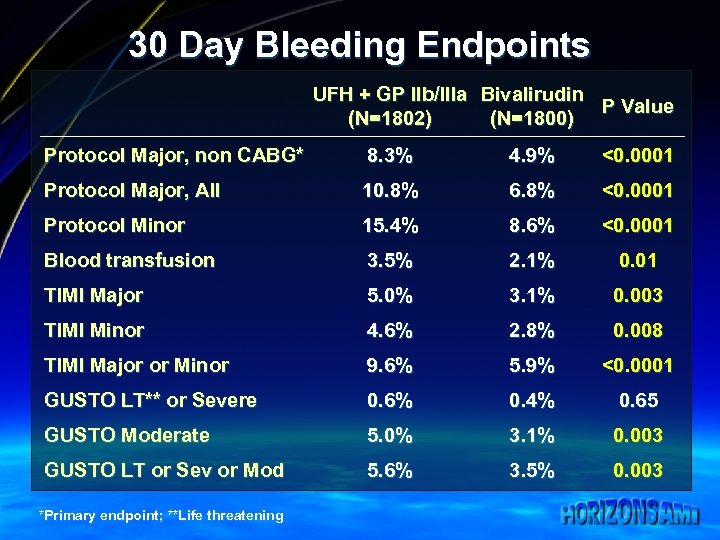
30 Day Bleeding Endpoints UFH + GP IIb/IIIa Bivalirudin P Value (N=1802) (N=1800) Protocol Major, non CABG* 8. 3% 4. 9% <0. 0001 Protocol Major, All 10. 8% 6. 8% <0. 0001 Protocol Minor 15. 4% 8. 6% <0. 0001 Blood transfusion 3. 5% 2. 1% 0. 01 TIMI Major 5. 0% 3. 1% 0. 003 TIMI Minor 4. 6% 2. 8% 0. 008 TIMI Major or Minor 9. 6% 5. 9% <0. 0001 GUSTO LT** or Severe 0. 6% 0. 4% 0. 65 GUSTO Moderate 5. 0% 3. 1% 0. 003 GUSTO LT or Sev or Mod 5. 6% 3. 5% 0. 003 *Primary endpoint; **Life threatening
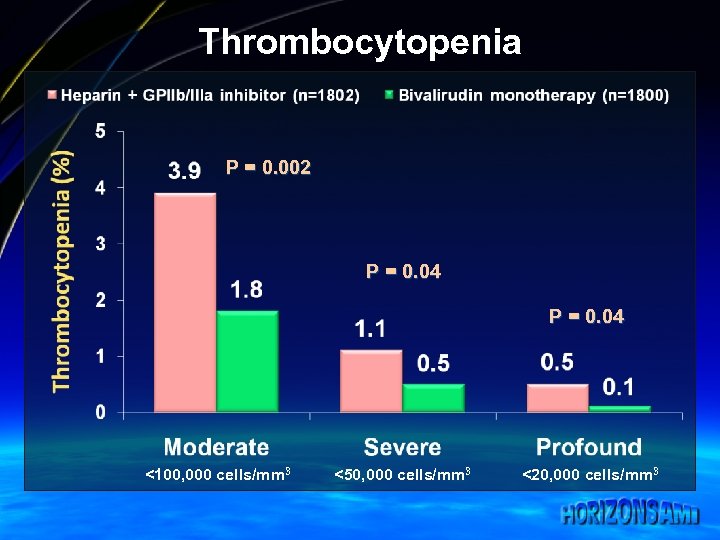
Thrombocytopenia P = 0. 002 P = 0. 04 <100, 000 cells/mm 3 <50, 000 cells/mm 3 <20, 000 cells/mm 3
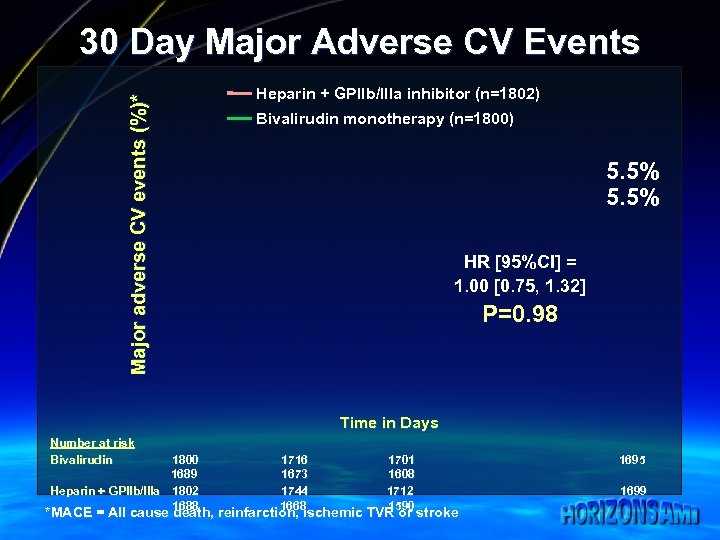
Major adverse CV events (%)* 30 Day Major Adverse CV Events Heparin + GPIIb/IIIa inhibitor (n=1802) Bivalirudin monotherapy (n=1800) 5. 5% HR [95%CI] = 1. 00 [0. 75, 1. 32] P=0. 98 Time in Days Number at risk Bivalirudin 1800 1689 Heparin + GPIIb/IIIa 1802 1688 1716 1673 1744 1668 1701 1608 1712 1590 *MACE = All cause death, reinfarction, ischemic TVR or stroke 1695 1699
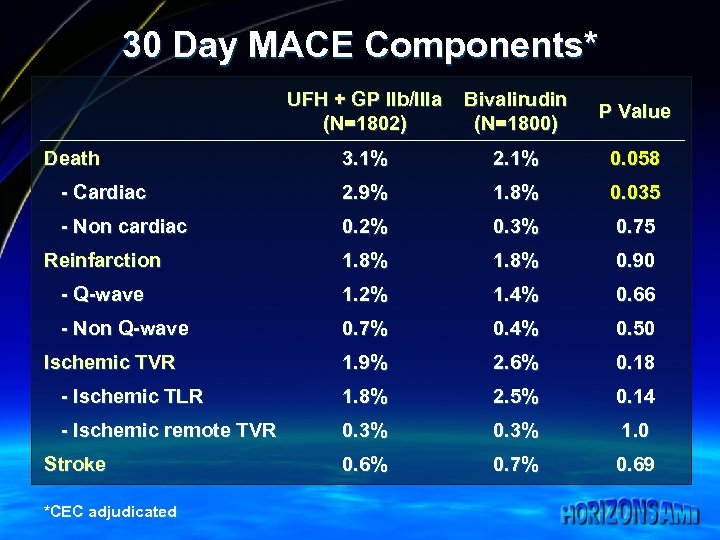
30 Day MACE Components* UFH + GP IIb/IIIa (N=1802) Bivalirudin (N=1800) P Value 3. 1% 2. 1% 0. 058 - Cardiac 2. 9% 1. 8% 0. 035 - Non cardiac 0. 2% 0. 3% 0. 75 Reinfarction 1. 8% 0. 90 - Q-wave 1. 2% 1. 4% 0. 66 - Non Q-wave 0. 7% 0. 4% 0. 50 1. 9% 2. 6% 0. 18 - Ischemic TLR 1. 8% 2. 5% 0. 14 - Ischemic remote TVR 0. 3% 1. 0 0. 6% 0. 7% 0. 69 Death Ischemic TVR Stroke *CEC adjudicated
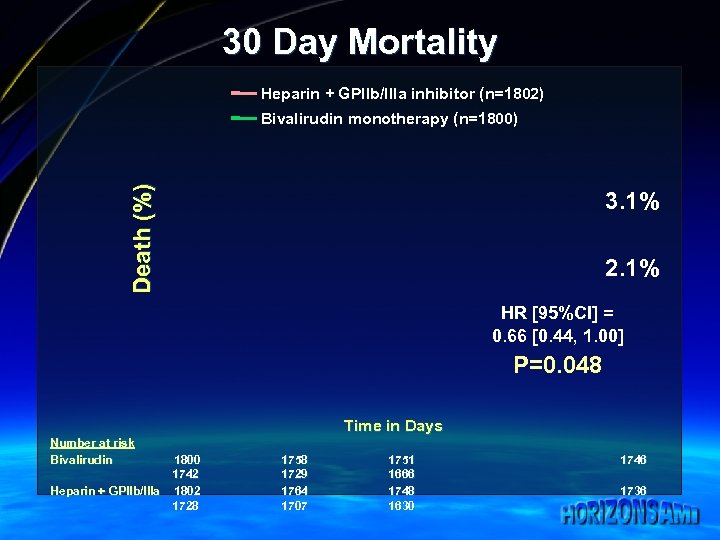
30 Day Mortality Heparin + GPIIb/IIIa inhibitor (n=1802) Death (%) Bivalirudin monotherapy (n=1800) 3. 1% 2. 1% HR [95%CI] = 0. 66 [0. 44, 1. 00] P=0. 048 Time in Days Number at risk Bivalirudin 1800 1742 Heparin + GPIIb/IIIa 1802 1728 1758 1729 1764 1707 1751 1666 1748 1630 1746 1736
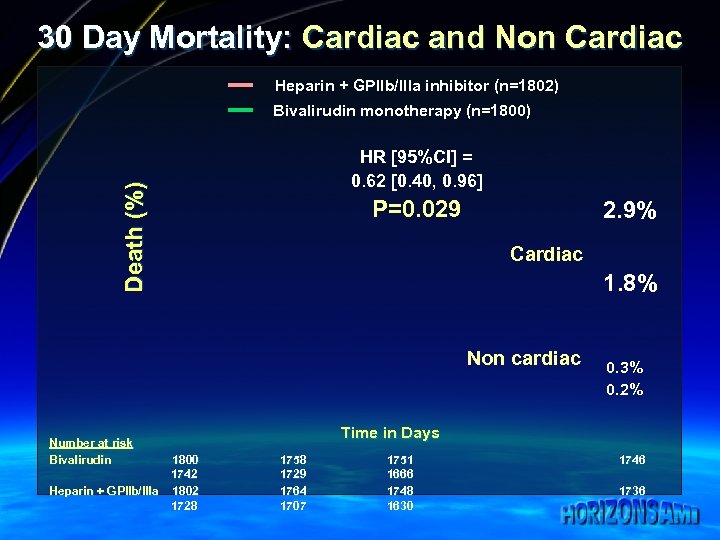
30 Day Mortality: Cardiac and Non Cardiac Heparin + GPIIb/IIIa inhibitor (n=1802) Bivalirudin monotherapy (n=1800) Death (%) HR [95%CI] = 0. 62 [0. 40, 0. 96] P=0. 029 2. 9% Cardiac 1. 8% Non cardiac Number at risk Bivalirudin 1800 1742 Heparin + GPIIb/IIIa 1802 1728 0. 3% 0. 2% Time in Days 1758 1729 1764 1707 1751 1666 1748 1630 1746 1736
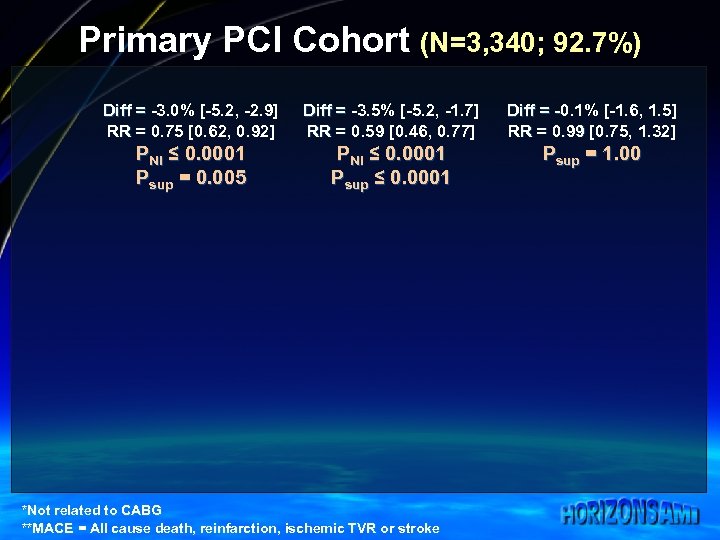
Primary PCI Cohort (N=3, 340; 92. 7%) Diff = -3. 0% [-5. 2, -2. 9] RR = 0. 75 [0. 62, 0. 92] Diff = -3. 5% [-5. 2, -1. 7] RR = 0. 59 [0. 46, 0. 77] Diff = -0. 1% [-1. 6, 1. 5] RR = 0. 99 [0. 75, 1. 32] PNI ≤ 0. 0001 Psup = 0. 005 PNI ≤ 0. 0001 Psup = 1. 00 *Not related to CABG **MACE = All cause death, reinfarction, ischemic TVR or stroke

Net Adverse Clinical Events (%)* 30 Day Net Clinical Events: PCI Cohort Heparin + GPIIb/IIIa inhibitor (n=1662) Bivalirudin monotherapy (n=1678) 12. 3% 9. 2% HR [95%CI] = 0. 74 [0. 60, 0. 92] P=0. 005 Time in Days Number at risk Bivalirudin 1678 1516 Heparin + GPIIb/IIIa 1662 1448 1554 1505 1510 1436 *MACE or major bleeding (non CABG) 1528 1448 1467 1372 1521 1456

Major Bleeding (Non-CABG) (%) 30 Day Major Bleeding: PCI Cohort Heparin + GPIIb/IIIa inhibitor (n=1662) Bivalirudin monotherapy (n=1678) 8. 6% 5. 1% HR [95%CI] = 0. 58 [0. 44, 0. 76] P<0. 0001 Time in Days Number at risk Bivalirudin 1678 1558 Heparin + GPIIb/IIIa 1662 1475 1590 1548 1525 1463 1568 1490 1492 1399 1561 1482
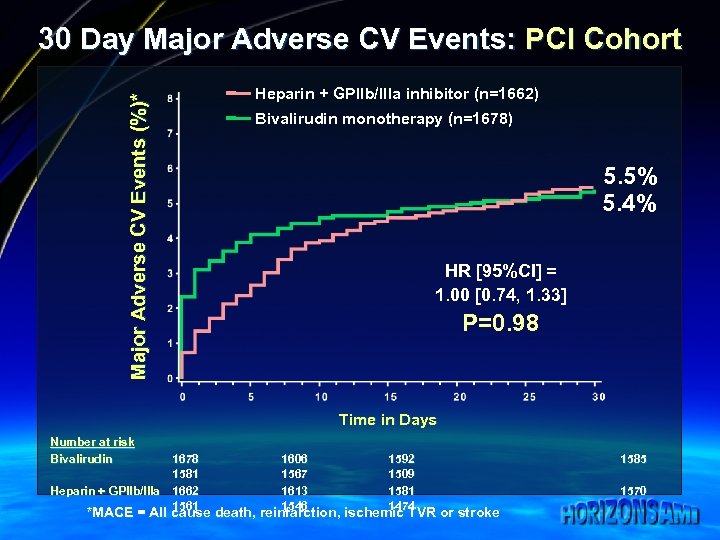
Major Adverse CV Events (%)* 30 Day Major Adverse CV Events: PCI Cohort Heparin + GPIIb/IIIa inhibitor (n=1662) Bivalirudin monotherapy (n=1678) 5. 5% 5. 4% HR [95%CI] = 1. 00 [0. 74, 1. 33] P=0. 98 Time in Days Number at risk Bivalirudin 1678 1581 Heparin + GPIIb/IIIa 1662 1561 1606 1567 1613 1546 1592 1509 1581 1474 *MACE = All cause death, reinfarction, ischemic TVR or stroke 1585 1570

30 Day Stent Thrombosis (N=3, 124) UFH + Bivalirudin P GP IIb/IIIa (N=1571) Value (N=1553) ARC definite or probable* 1. 9% 2. 5% 0. 33 - definite 1. 4% 2. 2% 0. 11 - probable 0. 5% 0. 3% 0. 26 - acute (≤ 24 hrs) 0. 3% 1. 3% 0. 0009 - subacute (>24 hrs – 30 d) 1. 7% 1. 2% 0. 30 *Protocol definition of stent thrombosis, CEC adjudicated
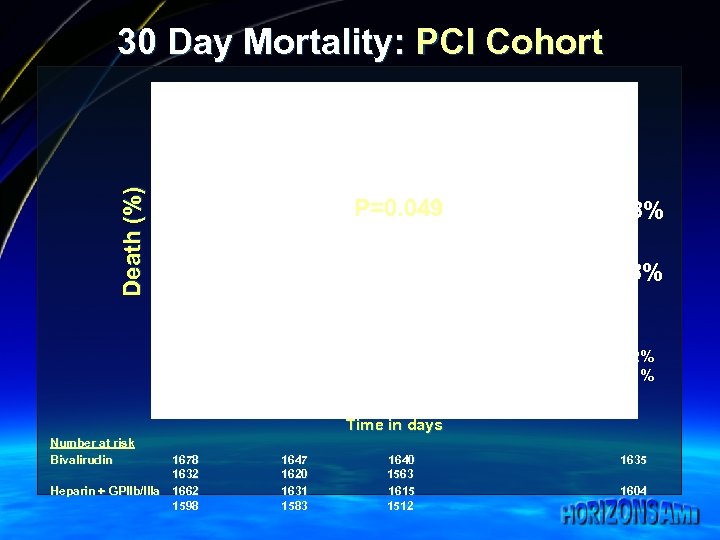
30 Day Mortality: PCI Cohort Heparin + GPIIb/IIIa inhibitor (n=1662) Bivalirudin monotherapy (n=1678) Death (%) HR [95%CI] = 0. 63 [0. 40, 0. 99] P=0. 049 2. 8% Cardiac 1. 8% Non cardiac 0. 2% 0. 1% Time in days Number at risk Bivalirudin 1678 1632 Heparin + GPIIb/IIIa 1662 1598 1647 1620 1631 1583 1640 1563 1615 1512 1635 1604

Limitations ¼ Open label design – Potential bias was mitigated by high protocol procedure compliance and use of blinded clinical event adjudication committees and core laboratories ¼ Underpowered for MACE and low frequency safety endpoints – The virtually identical rates of MACE in the bivalirudin and UFH plus GP IIb/IIIa inhibitor arms makes it unlikely major differences favoring control exist, especially given the lower cardiac mortality with bivalirudin

Conclusions ¼ In this large scale, prospective, randomized trial of pts with STEMI undergoing a primary PCI management strategy, compared to UFH plus the routine use of GP IIb/IIIa inhibitors, bivalirudin monotherapy with GP IIb/IIIa inhibitors reserved for suboptimal PCI outcomes resulted in: – A significant 24% reduction in the 30 day primary endpoint of net adverse clinical events – A significant 40% reduction in the 30 day primary endpoint of major bleeding

Conclusions ¼ ¼ The long-term impact of treatment with bivalirudin monotherapy rather than heparin plus GP IIb/IIIa inhibitors in patients with STEMI undergoing primary PCI will be determined by the ongoing 5 year follow-up of patients randomized in this trial The primary endpoint results of the second randomized arm of the HORIZONS AMI trial (paclitaxel-eluting vs. bare metal stents) will be available for presentation at this time in 2008

2c2db8902206e9af4b915d2d0f006e77.ppt