3f7a7764603dfb7486078b15c75647e8.ppt
- Количество слайдов: 45
 A Non-Opioid, Non-NSAID Analgesic for Perioperative Pain Management The speaker has been asked to present on the following topic by Mallinckrodt Pharmaceuticals. However, the thoughts and opinions expressed by the Speaker do not necessarily reflect the position or positions of Mallinckrodt.
A Non-Opioid, Non-NSAID Analgesic for Perioperative Pain Management The speaker has been asked to present on the following topic by Mallinckrodt Pharmaceuticals. However, the thoughts and opinions expressed by the Speaker do not necessarily reflect the position or positions of Mallinckrodt.
 ACUTE PERIOPERATIVE PAIN ■ Perioperative pain § Approximately 46 million inpatient procedures and 35 million outpatient surgeries were performed in the US in 20061, 2 § Despite new treatment standards, guidelines, and educational efforts, acute postoperative pain continues to be undertreated, with up to 75% of patients in the US still failing to receive adequate postoperative pain relief 3, 4 § With the advent of Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) surveys, patients are now able to make decisions on hospitals based on quality of care, including quality of pain management 1. De. Frances CJ, Lucas CA, Buie VC, Golosinkskiy A. Natl Health Stat Report. 2008 Jul 30; (5): 1 -20. 2. Cullen KA, Hall MJ, Golosinkskiy. Natl Health Stat Report. 2009 Jan 28; (11): 1 -25. 3. Wu CL, Raja SN. Lancet. 2011; 377: 22152225. 4. Phillips DM. JAMA. 2000; 284(4): 428 -429. 2
ACUTE PERIOPERATIVE PAIN ■ Perioperative pain § Approximately 46 million inpatient procedures and 35 million outpatient surgeries were performed in the US in 20061, 2 § Despite new treatment standards, guidelines, and educational efforts, acute postoperative pain continues to be undertreated, with up to 75% of patients in the US still failing to receive adequate postoperative pain relief 3, 4 § With the advent of Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) surveys, patients are now able to make decisions on hospitals based on quality of care, including quality of pain management 1. De. Frances CJ, Lucas CA, Buie VC, Golosinkskiy A. Natl Health Stat Report. 2008 Jul 30; (5): 1 -20. 2. Cullen KA, Hall MJ, Golosinkskiy. Natl Health Stat Report. 2009 Jan 28; (11): 1 -25. 3. Wu CL, Raja SN. Lancet. 2011; 377: 22152225. 4. Phillips DM. JAMA. 2000; 284(4): 428 -429. 2
 THE HISTORICAL ACUTE PAIN PARADIGM +++ Severe Pain Opioids Moderate Pain ++ Opioids + Mild Pain Opioids Aubrun et al. , 2003 Aubrun F, Langeron O, Quesnel C, Coriat P, Riou B. Anesthesiology. 2003; 98(6): 1415 -1421. 3
THE HISTORICAL ACUTE PAIN PARADIGM +++ Severe Pain Opioids Moderate Pain ++ Opioids + Mild Pain Opioids Aubrun et al. , 2003 Aubrun F, Langeron O, Quesnel C, Coriat P, Riou B. Anesthesiology. 2003; 98(6): 1415 -1421. 3
 THE JOINT COMMISSION SENTINEL EVENT ALERT ■ Joint Commission Sentinel Event Alert Entitled “Safe use of opioids in hospitals” § Focuses on the need for assessing and managing pain to help avoid accidental opioid overdose among hospital inpatients § Provides a number of actions that can be taken to avoid the unintended consequences ■ Recommendations include advising clinicians who prescribe pain medications to use both non-pharmacologic and pharmacologic alternatives § Non-pharmacologic therapies: physical therapy, acupuncture, manipulation or massage, ice, etc. § Pharmacologic treatment: non-opioid analgesics, such as acetaminophen, NSAIDs, antidepressants, anticonvulsants, and muscle relaxants, can be used before prescribing an opioid § When used in combination with opioids, these non-opioid pharmacologic treatments may reduce the dose of opioids required to effectively manage pain The Joint Commission Sentinel Event Alert. Safe use of opioids in hospitals. Issue 49; August 8, 2012. http: //www. jointcommission. org. 4
THE JOINT COMMISSION SENTINEL EVENT ALERT ■ Joint Commission Sentinel Event Alert Entitled “Safe use of opioids in hospitals” § Focuses on the need for assessing and managing pain to help avoid accidental opioid overdose among hospital inpatients § Provides a number of actions that can be taken to avoid the unintended consequences ■ Recommendations include advising clinicians who prescribe pain medications to use both non-pharmacologic and pharmacologic alternatives § Non-pharmacologic therapies: physical therapy, acupuncture, manipulation or massage, ice, etc. § Pharmacologic treatment: non-opioid analgesics, such as acetaminophen, NSAIDs, antidepressants, anticonvulsants, and muscle relaxants, can be used before prescribing an opioid § When used in combination with opioids, these non-opioid pharmacologic treatments may reduce the dose of opioids required to effectively manage pain The Joint Commission Sentinel Event Alert. Safe use of opioids in hospitals. Issue 49; August 8, 2012. http: //www. jointcommission. org. 4
 MULTIMODAL TECHNIQUES FOR PERIOPERATIVE PAIN MANAGEMENT ■ Multimodal analgesia combines two or more analgesic agents or techniques that act by different mechanisms to provide analgesia 1 ■ ASA, WHO, ASPMN, and The Joint Commission recommend use of a multimodal approach 1 -4 ■ Opioid dose-sparing effects can be achieved via the use of non-opioid agents and regional blocks 1 ■ ASA Task Force recommendations: § Unless contraindicated, all patients should receive an around-the-clock regimen of a non-opioid agent § Non-steroidal anti-inflammatory drugs (NSAIDs) § Cyclooxygenase-2 specific drugs (COXIBs) § Acetaminophen § Consider supplemental regional anesthesia techniques 1. ASA Task Force. Anesthesiology. 2012; 116(2): 248 -273. 2. World Health Organization. Pain relief ladder. http: //www. who. int/cancer/palliative/painladder/en/. Accessed September 10, 2014. 3. Jarzyna D, et al. Pain Manage Nurs 2011; 12: 118 -145. 4. The Joint Commission Sentinel Event Alert. Safe use of opioids in hospitals. Issue 49; August 8, 2012. www. jointcommission. org/assets/1/18/SEA_49_opioids_8_2_12_finalpdf. 5
MULTIMODAL TECHNIQUES FOR PERIOPERATIVE PAIN MANAGEMENT ■ Multimodal analgesia combines two or more analgesic agents or techniques that act by different mechanisms to provide analgesia 1 ■ ASA, WHO, ASPMN, and The Joint Commission recommend use of a multimodal approach 1 -4 ■ Opioid dose-sparing effects can be achieved via the use of non-opioid agents and regional blocks 1 ■ ASA Task Force recommendations: § Unless contraindicated, all patients should receive an around-the-clock regimen of a non-opioid agent § Non-steroidal anti-inflammatory drugs (NSAIDs) § Cyclooxygenase-2 specific drugs (COXIBs) § Acetaminophen § Consider supplemental regional anesthesia techniques 1. ASA Task Force. Anesthesiology. 2012; 116(2): 248 -273. 2. World Health Organization. Pain relief ladder. http: //www. who. int/cancer/palliative/painladder/en/. Accessed September 10, 2014. 3. Jarzyna D, et al. Pain Manage Nurs 2011; 12: 118 -145. 4. The Joint Commission Sentinel Event Alert. Safe use of opioids in hospitals. Issue 49; August 8, 2012. www. jointcommission. org/assets/1/18/SEA_49_opioids_8_2_12_finalpdf. 5
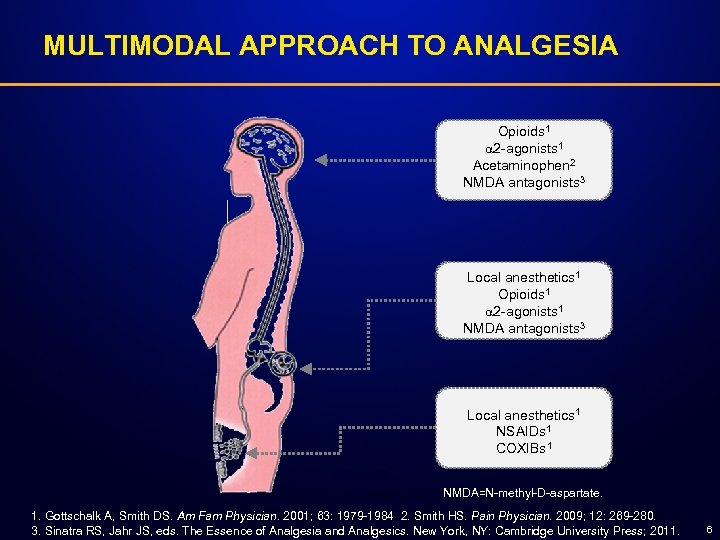 MULTIMODAL APPROACH TO ANALGESIA Opioids 1 2 -agonists 1 Acetaminophen 2 NMDA antagonists 3 Local anesthetics 1 Opioids 1 2 -agonists 1 NMDA antagonists 3 Trauma Local anesthetics 1 NSAIDs 1 COXIBs 1 NMDA=N-methyl-D-aspartate. 1. Gottschalk A, Smith DS. Am Fam Physician. 2001; 63: 1979 -1984 2. Smith HS. Pain Physician. 2009; 12: 269 -280. 3. Sinatra RS, Jahr JS, eds. The Essence of Analgesia and Analgesics. New York, NY: Cambridge University Press; 2011. 6
MULTIMODAL APPROACH TO ANALGESIA Opioids 1 2 -agonists 1 Acetaminophen 2 NMDA antagonists 3 Local anesthetics 1 Opioids 1 2 -agonists 1 NMDA antagonists 3 Trauma Local anesthetics 1 NSAIDs 1 COXIBs 1 NMDA=N-methyl-D-aspartate. 1. Gottschalk A, Smith DS. Am Fam Physician. 2001; 63: 1979 -1984 2. Smith HS. Pain Physician. 2009; 12: 269 -280. 3. Sinatra RS, Jahr JS, eds. The Essence of Analgesia and Analgesics. New York, NY: Cambridge University Press; 2011. 6
 MULTIMODAL APPROACH TO ACUTE PAIN MANAGEMENT Severe Pain STEP 3 STEP 2 and Higher doses of opioids STEP 2 Moderate Pain STEP 1 and Low doses of opioids STEP 1 Mild Pain Acetaminophen, NSAIDs, or COXIBs and Local/regional anesthesia 1. Crews JC. JAMA. 2002; 288: 629 -632. 2. World Health Organization. Pain relief ladder. http: //www. who. int/cancer/palliative/painladder/en/. Accessed September 10, 2014. 3. Ventafridda V, Tamburini M, Caraceni A, De Conno F, Naldi F. Cancer. 1987; 59: 850 -856. 4. ASA Task Force. Anesthesiology. 2004; 100: 1573 -1581. 7
MULTIMODAL APPROACH TO ACUTE PAIN MANAGEMENT Severe Pain STEP 3 STEP 2 and Higher doses of opioids STEP 2 Moderate Pain STEP 1 and Low doses of opioids STEP 1 Mild Pain Acetaminophen, NSAIDs, or COXIBs and Local/regional anesthesia 1. Crews JC. JAMA. 2002; 288: 629 -632. 2. World Health Organization. Pain relief ladder. http: //www. who. int/cancer/palliative/painladder/en/. Accessed September 10, 2014. 3. Ventafridda V, Tamburini M, Caraceni A, De Conno F, Naldi F. Cancer. 1987; 59: 850 -856. 4. ASA Task Force. Anesthesiology. 2004; 100: 1573 -1581. 7
 FOCUS OF HCAHPS ON PAIN AND PATIENT SATISFACTION ■ Pain § Establishing and maintaining an institutional pain performance improvement plan is a Joint Commission requirement 1 ■ Patient satisfaction § Local, regional, or national patient satisfaction data are now being reported via Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS, also known as CAHPS® hospital survey)2 § As part of the Affordable Care Act 2010, the Centers for Medicare and Medicaid (CMS) have established hospital reimbursement based on HCAHPS scores 3 § Effective beginning October 1, 2012 1. Wells N, Pasero C, Mc. Caffery M. In: Hughes RG, ed. Patient Safety and Quality: an Evidence-Based Handbook for Nurses. AHRQ Publication No. 80 -0043. 2. US Department of Health and Human Services, Centers for Medicare and Medicaid. HCAHPS: Patients' Perspectives of Care Survey. http: //www. cms. gov/Hospital. Quality. Inits/30_Hospital. HCAHPS. asp. Accessed September 10, 2014. 3. American Hospital Association (AHA) Hospital-based purchasing program: the final rule. May 24, 2011. http: //www. americangovernance. com/americangovernance/webinar/policy/pdf/final_rule_vbp_regulatory_advisory. pdf. 8 Accessed September 10, 2014.
FOCUS OF HCAHPS ON PAIN AND PATIENT SATISFACTION ■ Pain § Establishing and maintaining an institutional pain performance improvement plan is a Joint Commission requirement 1 ■ Patient satisfaction § Local, regional, or national patient satisfaction data are now being reported via Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS, also known as CAHPS® hospital survey)2 § As part of the Affordable Care Act 2010, the Centers for Medicare and Medicaid (CMS) have established hospital reimbursement based on HCAHPS scores 3 § Effective beginning October 1, 2012 1. Wells N, Pasero C, Mc. Caffery M. In: Hughes RG, ed. Patient Safety and Quality: an Evidence-Based Handbook for Nurses. AHRQ Publication No. 80 -0043. 2. US Department of Health and Human Services, Centers for Medicare and Medicaid. HCAHPS: Patients' Perspectives of Care Survey. http: //www. cms. gov/Hospital. Quality. Inits/30_Hospital. HCAHPS. asp. Accessed September 10, 2014. 3. American Hospital Association (AHA) Hospital-based purchasing program: the final rule. May 24, 2011. http: //www. americangovernance. com/americangovernance/webinar/policy/pdf/final_rule_vbp_regulatory_advisory. pdf. 8 Accessed September 10, 2014.
 OFIRMEV® (acetaminophen) Injection Overview
OFIRMEV® (acetaminophen) Injection Overview
 OFIRMEV® (ACETAMINOPHEN) INJECTION INDICATIONS AND USAGE ■ OFIRMEV (acetaminophen) injection is indicated for the: § management of mild to moderate pain § management of moderate to severe pain with adjunctive opioid analgesics § reduction of fever IMPORTANT RISK INFORMATION WARNING: RISK OF MEDICATION ERRORS AND HEPATOTOXICITY Take care when prescribing, preparing, and administering OFIRMEV injection to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that: • the dose in milligrams (mg) and milliliters (m. L) is not confused; • the dosing is based on weight for patients under 50 kg; • infusion pumps are properly programmed; and • the total daily dose of acetaminophen from all sources does not exceed maximum daily limits. OFIRMEV contains acetaminophen. Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed the recommended maximum daily limits, and often involve more than one acetaminophen-containing product. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 10
OFIRMEV® (ACETAMINOPHEN) INJECTION INDICATIONS AND USAGE ■ OFIRMEV (acetaminophen) injection is indicated for the: § management of mild to moderate pain § management of moderate to severe pain with adjunctive opioid analgesics § reduction of fever IMPORTANT RISK INFORMATION WARNING: RISK OF MEDICATION ERRORS AND HEPATOTOXICITY Take care when prescribing, preparing, and administering OFIRMEV injection to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that: • the dose in milligrams (mg) and milliliters (m. L) is not confused; • the dosing is based on weight for patients under 50 kg; • infusion pumps are properly programmed; and • the total daily dose of acetaminophen from all sources does not exceed maximum daily limits. OFIRMEV contains acetaminophen. Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed the recommended maximum daily limits, and often involve more than one acetaminophen-containing product. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 10
 IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONTRAINDICATIONS ■ Acetaminophen is contraindicated in patients with: § known hypersensitivity to acetaminophen or to any of the excipients in the intravenous (IV) formulation. § severe hepatic impairment or severe active liver disease. WARNINGS AND PRECAUTIONS ■ Administration of acetaminophen in doses higher than recommended may result in hepatic injury, including the risk of liver failure and death. Do not exceed the maximum recommended daily dose of acetaminophen. The maximum recommended daily dose of acetaminophen includes all routes of acetaminophen administration and all acetaminophen-containing products administered, including combination products. Dosing errors could result in accidental overdose and death. ■ Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, alcoholism, chronic malnutrition, severe hypovolemia (e. g. , due to dehydration or blood loss), or severe renal impairment (creatinine clearance ≤ 30 m. L/min). OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 11
IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONTRAINDICATIONS ■ Acetaminophen is contraindicated in patients with: § known hypersensitivity to acetaminophen or to any of the excipients in the intravenous (IV) formulation. § severe hepatic impairment or severe active liver disease. WARNINGS AND PRECAUTIONS ■ Administration of acetaminophen in doses higher than recommended may result in hepatic injury, including the risk of liver failure and death. Do not exceed the maximum recommended daily dose of acetaminophen. The maximum recommended daily dose of acetaminophen includes all routes of acetaminophen administration and all acetaminophen-containing products administered, including combination products. Dosing errors could result in accidental overdose and death. ■ Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, alcoholism, chronic malnutrition, severe hypovolemia (e. g. , due to dehydration or blood loss), or severe renal impairment (creatinine clearance ≤ 30 m. L/min). OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 11
 IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONT. WARNINGS AND PRECAUTIONS, cont. ■ Rarely, acetaminophen may cause serious skin reactions such as acute generalized exanthematous pustulosis (AGEP), Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. ■ Hypersensitivity and anaphylaxis associated with the use of acetaminophen have been reported. Clinical signs included swelling of the face, mouth, and throat, respiratory distress, urticaria, rash, and pruritis. ■ The antipyretic effects of OFIRMEV may mask fever. ADVERSE REACTIONS ■ Serious adverse reactions may include hepatic injury, serious skin reactions, hypersensitivity, and anaphylaxis. ■ Common adverse reactions in adults include nausea, vomiting, headache, and insomnia. Common adverse reactions in pediatric patients include nausea, vomiting, constipation, pruritus, agitation, and atelectasis. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 12
IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONT. WARNINGS AND PRECAUTIONS, cont. ■ Rarely, acetaminophen may cause serious skin reactions such as acute generalized exanthematous pustulosis (AGEP), Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. ■ Hypersensitivity and anaphylaxis associated with the use of acetaminophen have been reported. Clinical signs included swelling of the face, mouth, and throat, respiratory distress, urticaria, rash, and pruritis. ■ The antipyretic effects of OFIRMEV may mask fever. ADVERSE REACTIONS ■ Serious adverse reactions may include hepatic injury, serious skin reactions, hypersensitivity, and anaphylaxis. ■ Common adverse reactions in adults include nausea, vomiting, headache, and insomnia. Common adverse reactions in pediatric patients include nausea, vomiting, constipation, pruritus, agitation, and atelectasis. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 12
 IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONT. USE IN SPECIFIC POPULATIONS ■ Pregnancy: Pregnancy Category C. OFIRMEV should be given to a pregnant woman only if clearly needed. ■ Breast Feeding: While studies with OFIRMEV have not been conducted, acetaminophen is secreted in human milk in small quantities after oral administration. ■ Pediatrics: The effectiveness of OFIRMEV for the treatment of acute pain and fever has not been studied in pediatric patients <2 years of age. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 13
IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONT. USE IN SPECIFIC POPULATIONS ■ Pregnancy: Pregnancy Category C. OFIRMEV should be given to a pregnant woman only if clearly needed. ■ Breast Feeding: While studies with OFIRMEV have not been conducted, acetaminophen is secreted in human milk in small quantities after oral administration. ■ Pediatrics: The effectiveness of OFIRMEV for the treatment of acute pain and fever has not been studied in pediatric patients <2 years of age. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 13
 Pharmacokinetics and Metabolism of OFIRMEV® ■ Rapid time to reach Cmax § Peak levels achieved at 15 minutes of administration in both pain and fever studies 1 § Measurable CSF levels at 15 minutes 2 ■ Peak effect: within an hour of administration 1 § OFIRMEV® should only be administered as a 15 minute infusion ■ Dosing interval: 4 to 6 hours 1 ■ IV acetaminophen bypasses first-pass liver exposure and metabolism 1 The relationship between OFIRMEV pharmacokinetics and clinical efficacy has not been established. 1. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 2. Singla NK, Parulan C, Samson R, et al. Pain Pract. 2012; 12(7): 523 -532. 14
Pharmacokinetics and Metabolism of OFIRMEV® ■ Rapid time to reach Cmax § Peak levels achieved at 15 minutes of administration in both pain and fever studies 1 § Measurable CSF levels at 15 minutes 2 ■ Peak effect: within an hour of administration 1 § OFIRMEV® should only be administered as a 15 minute infusion ■ Dosing interval: 4 to 6 hours 1 ■ IV acetaminophen bypasses first-pass liver exposure and metabolism 1 The relationship between OFIRMEV pharmacokinetics and clinical efficacy has not been established. 1. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 2. Singla NK, Parulan C, Samson R, et al. Pain Pract. 2012; 12(7): 523 -532. 14
 ABSORPTION OF ORAL ANALGESICS MAY BE COMPROMISED IN THE PERIOPERATIVE SETTING ■ Absorption of oral acetaminophen is diminished due to compromised gastric function 1, 2 ■ Significant delays in gastric emptying occur with the administration of IV opioid analgesics 1 ■ Do not exceed the maximum daily limits of acetaminophen by all routes 1. Berger MM, Berger-Gryllaki M, Wiesel PH, et al. Crit Care Med 2000; 28(7): 2217 -2223. 2. Petring OU, Dawson PJ, Blake DW, Jones DJ, Biorksten AR, Libreri FC. Br J Anaesth 1995; 74: 257 -260. 15
ABSORPTION OF ORAL ANALGESICS MAY BE COMPROMISED IN THE PERIOPERATIVE SETTING ■ Absorption of oral acetaminophen is diminished due to compromised gastric function 1, 2 ■ Significant delays in gastric emptying occur with the administration of IV opioid analgesics 1 ■ Do not exceed the maximum daily limits of acetaminophen by all routes 1. Berger MM, Berger-Gryllaki M, Wiesel PH, et al. Crit Care Med 2000; 28(7): 2217 -2223. 2. Petring OU, Dawson PJ, Blake DW, Jones DJ, Biorksten AR, Libreri FC. Br J Anaesth 1995; 74: 257 -260. 15
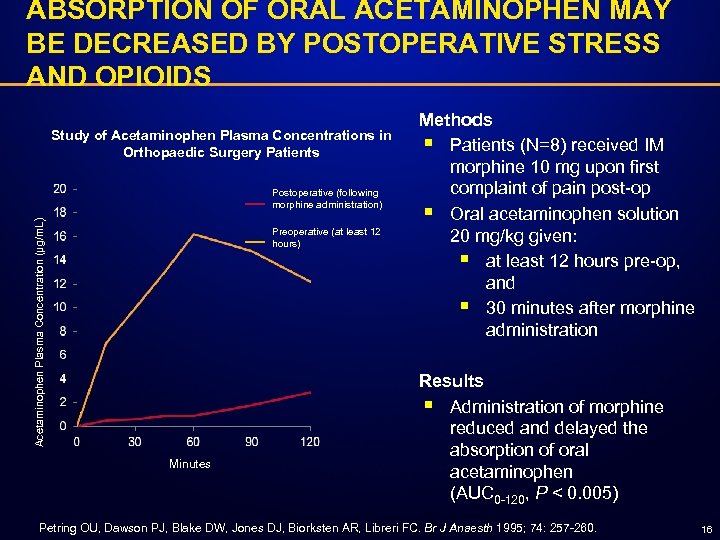 ABSORPTION OF ORAL ACETAMINOPHEN MAY BE DECREASED BY POSTOPERATIVE STRESS AND OPIOIDS Study of Acetaminophen Plasma Concentrations in Orthopaedic Surgery Patients Acetaminophen Plasma Concentration (µg/m. L) Postoperative (following morphine administration) Preoperative (at least 12 hours) Minutes Methods § Patients (N=8) received IM morphine 10 mg upon first complaint of pain post-op § Oral acetaminophen solution 20 mg/kg given: § at least 12 hours pre-op, and § 30 minutes after morphine administration Results § Administration of morphine reduced and delayed the absorption of oral acetaminophen (AUC 0 -120, P < 0. 005) Petring OU, Dawson PJ, Blake DW, Jones DJ, Biorksten AR, Libreri FC. Br J Anaesth 1995; 74: 257 -260. 16
ABSORPTION OF ORAL ACETAMINOPHEN MAY BE DECREASED BY POSTOPERATIVE STRESS AND OPIOIDS Study of Acetaminophen Plasma Concentrations in Orthopaedic Surgery Patients Acetaminophen Plasma Concentration (µg/m. L) Postoperative (following morphine administration) Preoperative (at least 12 hours) Minutes Methods § Patients (N=8) received IM morphine 10 mg upon first complaint of pain post-op § Oral acetaminophen solution 20 mg/kg given: § at least 12 hours pre-op, and § 30 minutes after morphine administration Results § Administration of morphine reduced and delayed the absorption of oral acetaminophen (AUC 0 -120, P < 0. 005) Petring OU, Dawson PJ, Blake DW, Jones DJ, Biorksten AR, Libreri FC. Br J Anaesth 1995; 74: 257 -260. 16
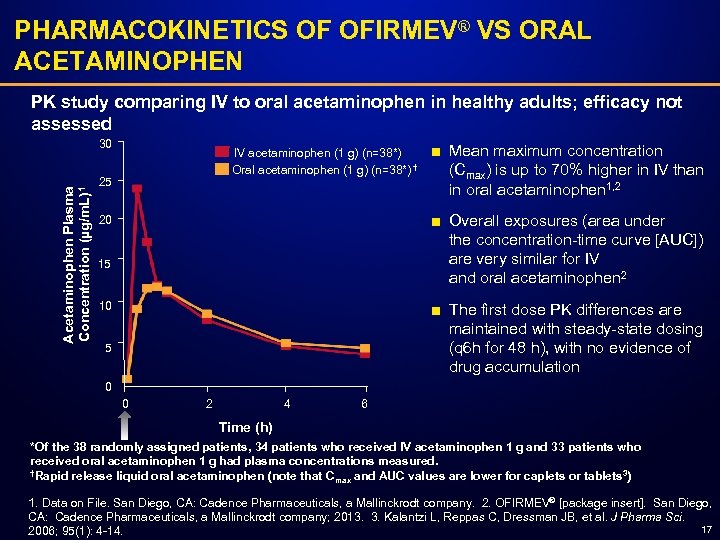 PHARMACOKINETICS OF OFIRMEV® VS ORAL ACETAMINOPHEN PK study comparing IV to oral acetaminophen in healthy adults; efficacy not assessed Acetaminophen Plasma Concentration (µg/m. L)1 30 IV acetaminophen (1 g) (n=38*) Oral acetaminophen (1 g) (n=38*) † 25 ■ Mean maximum concentration (Cmax) is up to 70% higher in IV than in oral acetaminophen 1, 2 ■ Overall exposures (area under the concentration-time curve [AUC]) are very similar for IV and oral acetaminophen 2 20 15 10 ■ The first dose PK differences are maintained with steady-state dosing (q 6 h for 48 h), with no evidence of drug accumulation 5 0 0 2 4 6 Time (h) *Of the 38 randomly assigned patients, 34 patients who received IV acetaminophen 1 g and 33 patients who received oral acetaminophen 1 g had plasma concentrations measured. †Rapid release liquid oral acetaminophen (note that C 3 max and AUC values are lower for caplets or tablets ) 1. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 2. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 3. Kalantzi L, Reppas C, Dressman JB, et al. J Pharma Sci. 17 2006; 95(1): 4 -14.
PHARMACOKINETICS OF OFIRMEV® VS ORAL ACETAMINOPHEN PK study comparing IV to oral acetaminophen in healthy adults; efficacy not assessed Acetaminophen Plasma Concentration (µg/m. L)1 30 IV acetaminophen (1 g) (n=38*) Oral acetaminophen (1 g) (n=38*) † 25 ■ Mean maximum concentration (Cmax) is up to 70% higher in IV than in oral acetaminophen 1, 2 ■ Overall exposures (area under the concentration-time curve [AUC]) are very similar for IV and oral acetaminophen 2 20 15 10 ■ The first dose PK differences are maintained with steady-state dosing (q 6 h for 48 h), with no evidence of drug accumulation 5 0 0 2 4 6 Time (h) *Of the 38 randomly assigned patients, 34 patients who received IV acetaminophen 1 g and 33 patients who received oral acetaminophen 1 g had plasma concentrations measured. †Rapid release liquid oral acetaminophen (note that C 3 max and AUC values are lower for caplets or tablets ) 1. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 2. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 3. Kalantzi L, Reppas C, Dressman JB, et al. J Pharma Sci. 17 2006; 95(1): 4 -14.
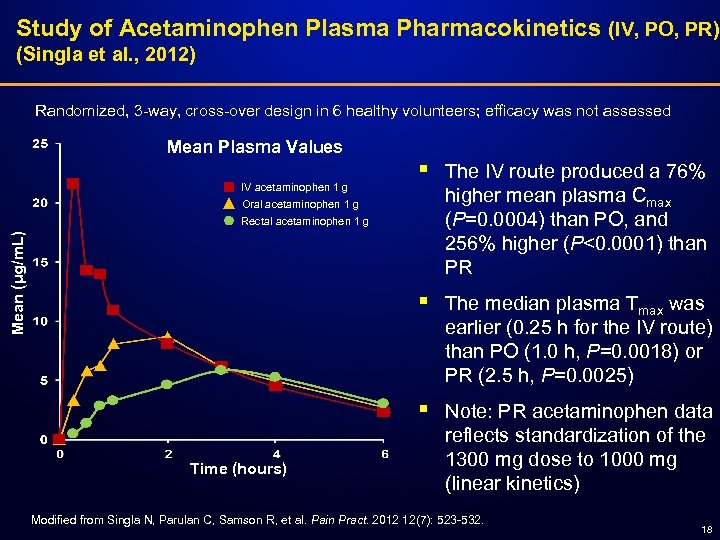 Study of Acetaminophen Plasma Pharmacokinetics (IV, PO, PR) (Singla et al. , 2012) Randomized, 3 -way, cross-over design in 6 healthy volunteers; efficacy was not assessed Mean Plasma Values § The IV route produced a 76% higher mean plasma Cmax (P=0. 0004) than PO, and 256% higher (P<0. 0001) than PR § The median plasma Tmax was earlier (0. 25 h for the IV route) than PO (1. 0 h, P=0. 0018) or PR (2. 5 h, P=0. 0025) § IV acetaminophen 1 g Note: PR acetaminophen data reflects standardization of the 1300 mg dose to 1000 mg (linear kinetics) Oral acetaminophen 1 g Mean (μg/m. L) Rectal acetaminophen 1 g Time (hours) Modified from Singla N, Parulan C, Samson R, et al. Pain Pract. 2012 12(7): 523 -532. 18
Study of Acetaminophen Plasma Pharmacokinetics (IV, PO, PR) (Singla et al. , 2012) Randomized, 3 -way, cross-over design in 6 healthy volunteers; efficacy was not assessed Mean Plasma Values § The IV route produced a 76% higher mean plasma Cmax (P=0. 0004) than PO, and 256% higher (P<0. 0001) than PR § The median plasma Tmax was earlier (0. 25 h for the IV route) than PO (1. 0 h, P=0. 0018) or PR (2. 5 h, P=0. 0025) § IV acetaminophen 1 g Note: PR acetaminophen data reflects standardization of the 1300 mg dose to 1000 mg (linear kinetics) Oral acetaminophen 1 g Mean (μg/m. L) Rectal acetaminophen 1 g Time (hours) Modified from Singla N, Parulan C, Samson R, et al. Pain Pract. 2012 12(7): 523 -532. 18
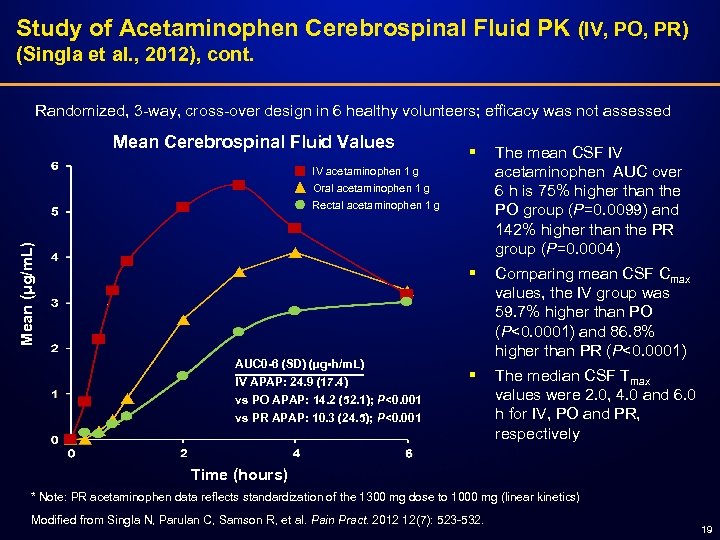 Study of Acetaminophen Cerebrospinal Fluid PK (IV, PO, PR) (Singla et al. , 2012), cont. Randomized, 3 -way, cross-over design in 6 healthy volunteers; efficacy was not assessed Mean Cerebrospinal Fluid Values § The mean CSF IV acetaminophen AUC over 6 h is 75% higher than the PO group (P=0. 0099) and 142% higher than the PR group (P=0. 0004) § Comparing mean CSF Cmax values, the IV group was 59. 7% higher than PO (P<0. 0001) and 86. 8% higher than PR (P<0. 0001) § The median CSF Tmax values were 2. 0, 4. 0 and 6. 0 h for IV, PO and PR, respectively IV acetaminophen 1 g Oral acetaminophen 1 g Mean (μg/m. L) Rectal acetaminophen 1 g AUC 0 -6 (SD) (µg • h/m. L) IV APAP: 24. 9 (17. 4) vs PO APAP: 14. 2 (52. 1); P<0. 001 vs PR APAP: 10. 3 (24. 5); P<0. 001 Time (hours) * Note: PR acetaminophen data reflects standardization of the 1300 mg dose to 1000 mg (linear kinetics) Modified from Singla N, Parulan C, Samson R, et al. Pain Pract. 2012 12(7): 523 -532. 19
Study of Acetaminophen Cerebrospinal Fluid PK (IV, PO, PR) (Singla et al. , 2012), cont. Randomized, 3 -way, cross-over design in 6 healthy volunteers; efficacy was not assessed Mean Cerebrospinal Fluid Values § The mean CSF IV acetaminophen AUC over 6 h is 75% higher than the PO group (P=0. 0099) and 142% higher than the PR group (P=0. 0004) § Comparing mean CSF Cmax values, the IV group was 59. 7% higher than PO (P<0. 0001) and 86. 8% higher than PR (P<0. 0001) § The median CSF Tmax values were 2. 0, 4. 0 and 6. 0 h for IV, PO and PR, respectively IV acetaminophen 1 g Oral acetaminophen 1 g Mean (μg/m. L) Rectal acetaminophen 1 g AUC 0 -6 (SD) (µg • h/m. L) IV APAP: 24. 9 (17. 4) vs PO APAP: 14. 2 (52. 1); P<0. 001 vs PR APAP: 10. 3 (24. 5); P<0. 001 Time (hours) * Note: PR acetaminophen data reflects standardization of the 1300 mg dose to 1000 mg (linear kinetics) Modified from Singla N, Parulan C, Samson R, et al. Pain Pract. 2012 12(7): 523 -532. 19
 Clinical Studies of OFIRMEV® (acetaminophen) Injection
Clinical Studies of OFIRMEV® (acetaminophen) Injection
 STUDY IN MAJOR ORTHOPAEDIC SURGERY (SINATRA ET AL. , 2005) ■ Randomized, double-blind, placebo-controlled multi-dose study in total hip or knee arthroplasty ■ 7 US centers, N=101 patients (plus 50 patients treated with propacetamol) ■ Started on postoperative day 1 to allow for anesthesia washout (combinations of general, spinal or epidural anesthesia allowed) and to ensure a stable baseline ■ Moderate to severe pain; patients randomly assigned to 1 of 3 treatment groups § 1 g OFIRMEV® § 2 g propacetamol IV (Note: Propacetamol is not FDA approved in the United States. ) § Placebo ■ Rescue medication: patient-controlled analgesia (PCA) morphine plus as needed (PRN) bolus doses available to all patients ■ Endpoints: pain intensity, pain relief, patient satisfaction and morphine use were measured at selected intervals IMPORTANT RISK INFORMATION Serious adverse reactions may include hepatic injury, serious skin reactions, hypersensitivity, and anaphylaxis. Common adverse reactions in adult patients include nausea, vomiting, headache, and insomnia; and in pediatric patients include nausea, vomiting, constipation, pruritus, agitation, and atelectasis. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102: 822 -831. 21
STUDY IN MAJOR ORTHOPAEDIC SURGERY (SINATRA ET AL. , 2005) ■ Randomized, double-blind, placebo-controlled multi-dose study in total hip or knee arthroplasty ■ 7 US centers, N=101 patients (plus 50 patients treated with propacetamol) ■ Started on postoperative day 1 to allow for anesthesia washout (combinations of general, spinal or epidural anesthesia allowed) and to ensure a stable baseline ■ Moderate to severe pain; patients randomly assigned to 1 of 3 treatment groups § 1 g OFIRMEV® § 2 g propacetamol IV (Note: Propacetamol is not FDA approved in the United States. ) § Placebo ■ Rescue medication: patient-controlled analgesia (PCA) morphine plus as needed (PRN) bolus doses available to all patients ■ Endpoints: pain intensity, pain relief, patient satisfaction and morphine use were measured at selected intervals IMPORTANT RISK INFORMATION Serious adverse reactions may include hepatic injury, serious skin reactions, hypersensitivity, and anaphylaxis. Common adverse reactions in adult patients include nausea, vomiting, headache, and insomnia; and in pediatric patients include nausea, vomiting, constipation, pruritus, agitation, and atelectasis. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102: 822 -831. 21
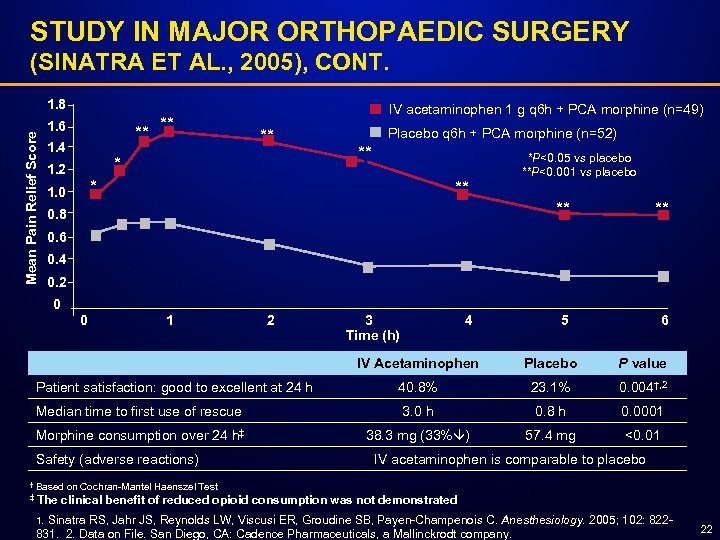 STUDY IN MAJOR ORTHOPAEDIC SURGERY (SINATRA ET AL. , 2005), CONT. Mean Pain Relief Score 1. 8 1. 6 ** 1. 4 ** IV acetaminophen 1 g q 6 h + PCA morphine (n=49) ** * 1. 2 Placebo q 6 h + PCA morphine (n=52) ** *P<0. 05 vs placebo * 1. 0 ** **P<0. 001 vs placebo ** 0. 8 ** 5 6 0. 4 0. 2 0 0 1 2 3 Time (h) 4 IV Acetaminophen Placebo P value 40. 8% 23. 1% 0. 004†, 2 Median time to first use of rescue 3. 0 h 0. 8 h 0. 0001 Morphine consumption over 24 h‡ 38. 3 mg (33% ) 57. 4 mg <0. 01 Patient satisfaction: good to excellent at 24 h Safety (adverse reactions) IV acetaminophen is comparable to placebo † Based on Cochran-Mantel Haenszel Test ‡ The clinical benefit of reduced opioid consumption was not demonstrated 1. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102: 822 - 831. 2. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 22
STUDY IN MAJOR ORTHOPAEDIC SURGERY (SINATRA ET AL. , 2005), CONT. Mean Pain Relief Score 1. 8 1. 6 ** 1. 4 ** IV acetaminophen 1 g q 6 h + PCA morphine (n=49) ** * 1. 2 Placebo q 6 h + PCA morphine (n=52) ** *P<0. 05 vs placebo * 1. 0 ** **P<0. 001 vs placebo ** 0. 8 ** 5 6 0. 4 0. 2 0 0 1 2 3 Time (h) 4 IV Acetaminophen Placebo P value 40. 8% 23. 1% 0. 004†, 2 Median time to first use of rescue 3. 0 h 0. 8 h 0. 0001 Morphine consumption over 24 h‡ 38. 3 mg (33% ) 57. 4 mg <0. 01 Patient satisfaction: good to excellent at 24 h Safety (adverse reactions) IV acetaminophen is comparable to placebo † Based on Cochran-Mantel Haenszel Test ‡ The clinical benefit of reduced opioid consumption was not demonstrated 1. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102: 822 - 831. 2. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 22
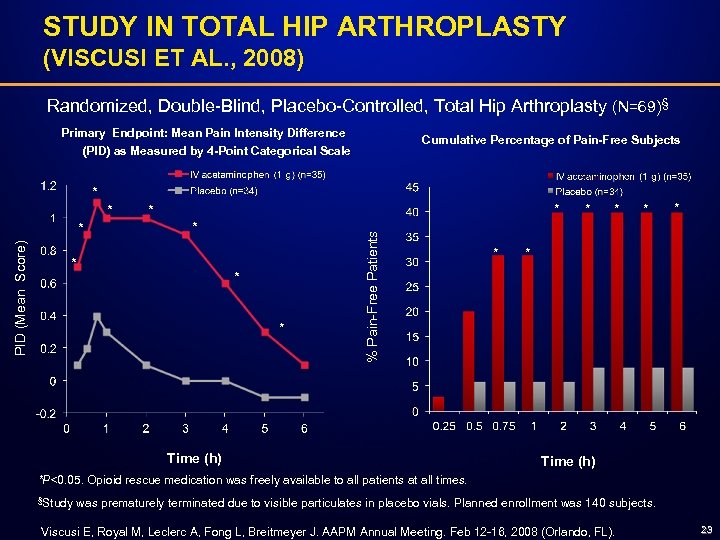 STUDY IN TOTAL HIP ARTHROPLASTY (VISCUSI ET AL. , 2008) Randomized, Double-Blind, Placebo-Controlled, Total Hip Arthroplasty (N=69)§ Primary Endpoint: Mean Pain Intensity Difference (PID) as Measured by 4 -Point Categorical Scale Cumulative Percentage of Pain-Free Subjects * PID (Mean Score) * * * * % Pain-Free Patients * Time (h) * * * Time (h) *P<0. 05. Opioid rescue medication was freely available to all patients at all times. §Study was prematurely terminated due to visible particulates in placebo vials. Planned enrollment was 140 subjects. Viscusi E, Royal M, Leclerc A, Fong L, Breitmeyer J. AAPM Annual Meeting. Feb 12 -16, 2008 (Orlando, FL). 23
STUDY IN TOTAL HIP ARTHROPLASTY (VISCUSI ET AL. , 2008) Randomized, Double-Blind, Placebo-Controlled, Total Hip Arthroplasty (N=69)§ Primary Endpoint: Mean Pain Intensity Difference (PID) as Measured by 4 -Point Categorical Scale Cumulative Percentage of Pain-Free Subjects * PID (Mean Score) * * * * % Pain-Free Patients * Time (h) * * * Time (h) *P<0. 05. Opioid rescue medication was freely available to all patients at all times. §Study was prematurely terminated due to visible particulates in placebo vials. Planned enrollment was 140 subjects. Viscusi E, Royal M, Leclerc A, Fong L, Breitmeyer J. AAPM Annual Meeting. Feb 12 -16, 2008 (Orlando, FL). 23
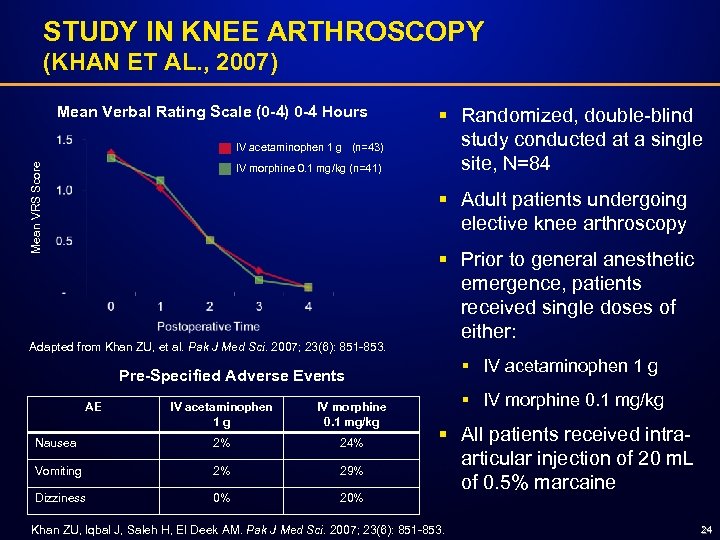 STUDY IN KNEE ARTHROSCOPY (KHAN ET AL. , 2007) Mean Verbal Rating Scale (0 -4) 0 -4 Hours Mean VRS Score IV acetaminophen 1 g (n=43) IV morphine 0. 1 mg/kg (n=41) § Randomized, double-blind study conducted at a single site, N=84 § Adult patients undergoing elective knee arthroscopy Adapted from Khan ZU, et al. Pak J Med Sci. 2007; 23(6): 851 -853. § Prior to general anesthetic emergence, patients received single doses of either: § IV acetaminophen 1 g Pre-Specified Adverse Events AE IV acetaminophen 1 g IV morphine 0. 1 mg/kg Nausea 2% 24% Vomiting 2% 29% Dizziness 0% 20% § IV morphine 0. 1 mg/kg § All patients received intraarticular injection of 20 m. L of 0. 5% marcaine Khan ZU, Iqbal J, Saleh H, El Deek AM. Pak J Med Sci. 2007; 23(6): 851 -853. 24
STUDY IN KNEE ARTHROSCOPY (KHAN ET AL. , 2007) Mean Verbal Rating Scale (0 -4) 0 -4 Hours Mean VRS Score IV acetaminophen 1 g (n=43) IV morphine 0. 1 mg/kg (n=41) § Randomized, double-blind study conducted at a single site, N=84 § Adult patients undergoing elective knee arthroscopy Adapted from Khan ZU, et al. Pak J Med Sci. 2007; 23(6): 851 -853. § Prior to general anesthetic emergence, patients received single doses of either: § IV acetaminophen 1 g Pre-Specified Adverse Events AE IV acetaminophen 1 g IV morphine 0. 1 mg/kg Nausea 2% 24% Vomiting 2% 29% Dizziness 0% 20% § IV morphine 0. 1 mg/kg § All patients received intraarticular injection of 20 m. L of 0. 5% marcaine Khan ZU, Iqbal J, Saleh H, El Deek AM. Pak J Med Sci. 2007; 23(6): 851 -853. 24
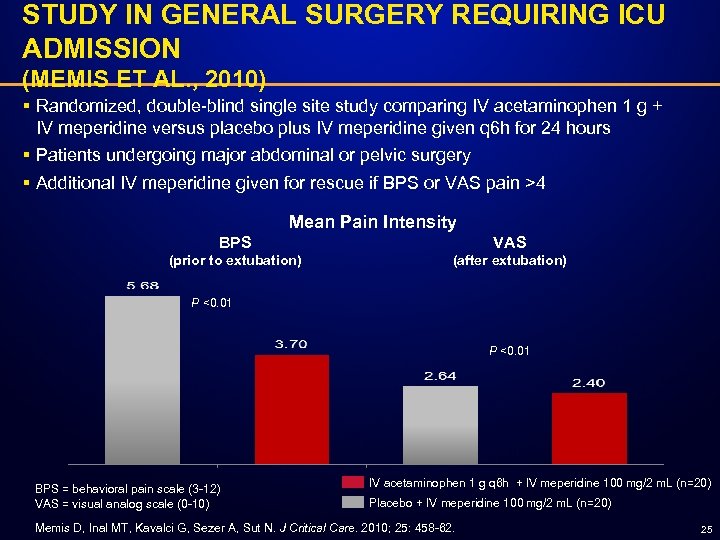 STUDY IN GENERAL SURGERY REQUIRING ICU ADMISSION (MEMIS ET AL. , 2010) § Randomized, double-blind single site study comparing IV acetaminophen 1 g + IV meperidine versus placebo plus IV meperidine given q 6 h for 24 hours § Patients undergoing major abdominal or pelvic surgery § Additional IV meperidine given for rescue if BPS or VAS pain >4 Mean Pain Intensity BPS VAS (prior to extubation) (after extubation) P <0. 01 BPS = behavioral pain scale (3 -12) VAS = visual analog scale (0 -10) IV acetaminophen 1 g q 6 h + IV meperidine 100 mg/2 m. L (n=20) Placebo + IV meperidine 100 mg/2 m. L (n=20) Memis D, Inal MT, Kavalci G, Sezer A, Sut N. J Critical Care. 2010; 25: 458 -62. 25
STUDY IN GENERAL SURGERY REQUIRING ICU ADMISSION (MEMIS ET AL. , 2010) § Randomized, double-blind single site study comparing IV acetaminophen 1 g + IV meperidine versus placebo plus IV meperidine given q 6 h for 24 hours § Patients undergoing major abdominal or pelvic surgery § Additional IV meperidine given for rescue if BPS or VAS pain >4 Mean Pain Intensity BPS VAS (prior to extubation) (after extubation) P <0. 01 BPS = behavioral pain scale (3 -12) VAS = visual analog scale (0 -10) IV acetaminophen 1 g q 6 h + IV meperidine 100 mg/2 m. L (n=20) Placebo + IV meperidine 100 mg/2 m. L (n=20) Memis D, Inal MT, Kavalci G, Sezer A, Sut N. J Critical Care. 2010; 25: 458 -62. 25
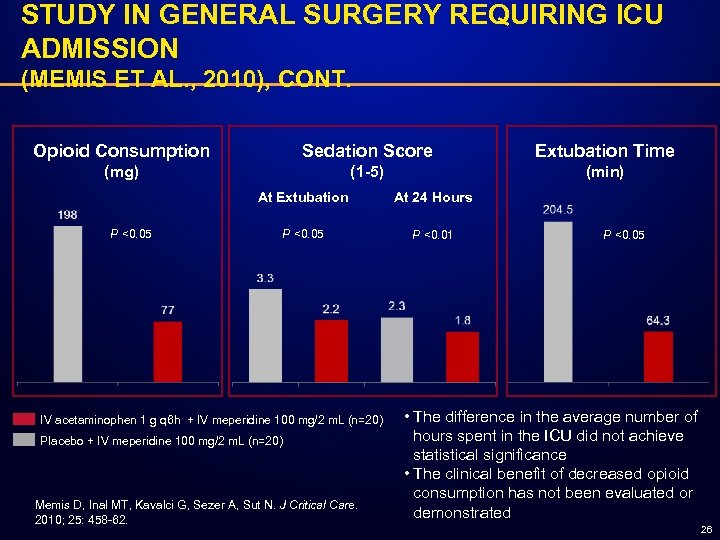 STUDY IN GENERAL SURGERY REQUIRING ICU ADMISSION (MEMIS ET AL. , 2010), CONT. Opioid Consumption Sedation Score Extubation Time (1 -5) (min) (mg) At Extubation P <0. 05 At 24 Hours P <0. 05 P <0. 01 IV acetaminophen 1 g q 6 h + IV meperidine 100 mg/2 m. L (n=20) Placebo + IV meperidine 100 mg/2 m. L (n=20) Memis D, Inal MT, Kavalci G, Sezer A, Sut N. J Critical Care. 2010; 25: 458 -62. P <0. 05 • The difference in the average number of hours spent in the ICU did not achieve statistical significance • The clinical benefit of decreased opioid consumption has not been evaluated or demonstrated 26
STUDY IN GENERAL SURGERY REQUIRING ICU ADMISSION (MEMIS ET AL. , 2010), CONT. Opioid Consumption Sedation Score Extubation Time (1 -5) (min) (mg) At Extubation P <0. 05 At 24 Hours P <0. 05 P <0. 01 IV acetaminophen 1 g q 6 h + IV meperidine 100 mg/2 m. L (n=20) Placebo + IV meperidine 100 mg/2 m. L (n=20) Memis D, Inal MT, Kavalci G, Sezer A, Sut N. J Critical Care. 2010; 25: 458 -62. P <0. 05 • The difference in the average number of hours spent in the ICU did not achieve statistical significance • The clinical benefit of decreased opioid consumption has not been evaluated or demonstrated 26
 STUDY IN RENAL COLIC (BEKTAS ET AL. , 2009) ■ A randomized, double-blind, placebo-controlled, single dose clinical study of adult patients admitted to the ER with suspected renal colic ■ Patients were randomized to receive: § IV acetaminophen 1 g (n=46) § IV morphine 0. 1 mg/kg – weight self-reported (n=49) § Placebo (n=51) ■ Analgesia assessed over a 30 -minute period ■ Fentanyl available as rescue medication following the 30 -minute period ■ Endpoints § 100 mm Visual Analog Scale (VAS) § 4 -point Verbal Rating Scale (VRS) § Amount of rescue consumed Bektas F, Eken C, Karadeniz O, Goksu E, Cubuk M, Cete Y. Ann Emerg Med. 2009; 54(4): 568 -574. 27
STUDY IN RENAL COLIC (BEKTAS ET AL. , 2009) ■ A randomized, double-blind, placebo-controlled, single dose clinical study of adult patients admitted to the ER with suspected renal colic ■ Patients were randomized to receive: § IV acetaminophen 1 g (n=46) § IV morphine 0. 1 mg/kg – weight self-reported (n=49) § Placebo (n=51) ■ Analgesia assessed over a 30 -minute period ■ Fentanyl available as rescue medication following the 30 -minute period ■ Endpoints § 100 mm Visual Analog Scale (VAS) § 4 -point Verbal Rating Scale (VRS) § Amount of rescue consumed Bektas F, Eken C, Karadeniz O, Goksu E, Cubuk M, Cete Y. Ann Emerg Med. 2009; 54(4): 568 -574. 27
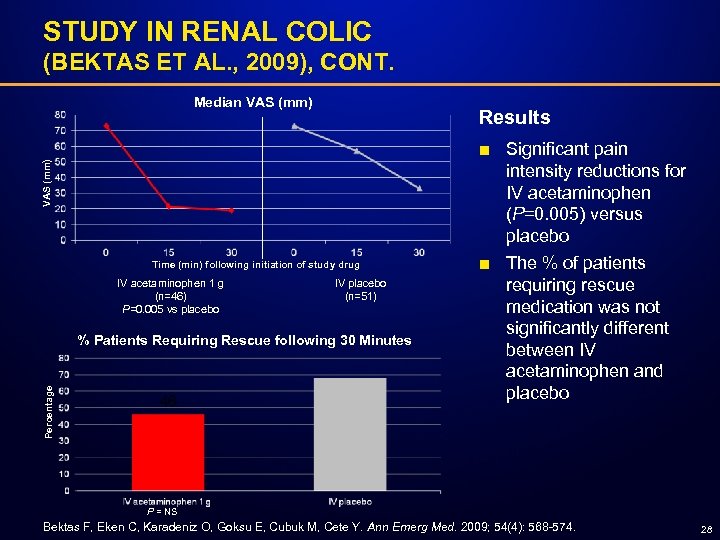 STUDY IN RENAL COLIC (BEKTAS ET AL. , 2009), CONT. Median VAS (mm) Results Time (min) following initiation of study drug IV acetaminophen 1 g (n=46) P=0. 005 vs placebo IV placebo (n=51) Percentage % Patients Requiring Rescue following 30 Minutes ■ Significant pain intensity reductions for IV acetaminophen (P=0. 005) versus placebo ■ The % of patients requiring rescue medication was not significantly different between IV acetaminophen and placebo P = NS Bektas F, Eken C, Karadeniz O, Goksu E, Cubuk M, Cete Y. Ann Emerg Med. 2009; 54(4): 568 -574. 28
STUDY IN RENAL COLIC (BEKTAS ET AL. , 2009), CONT. Median VAS (mm) Results Time (min) following initiation of study drug IV acetaminophen 1 g (n=46) P=0. 005 vs placebo IV placebo (n=51) Percentage % Patients Requiring Rescue following 30 Minutes ■ Significant pain intensity reductions for IV acetaminophen (P=0. 005) versus placebo ■ The % of patients requiring rescue medication was not significantly different between IV acetaminophen and placebo P = NS Bektas F, Eken C, Karadeniz O, Goksu E, Cubuk M, Cete Y. Ann Emerg Med. 2009; 54(4): 568 -574. 28
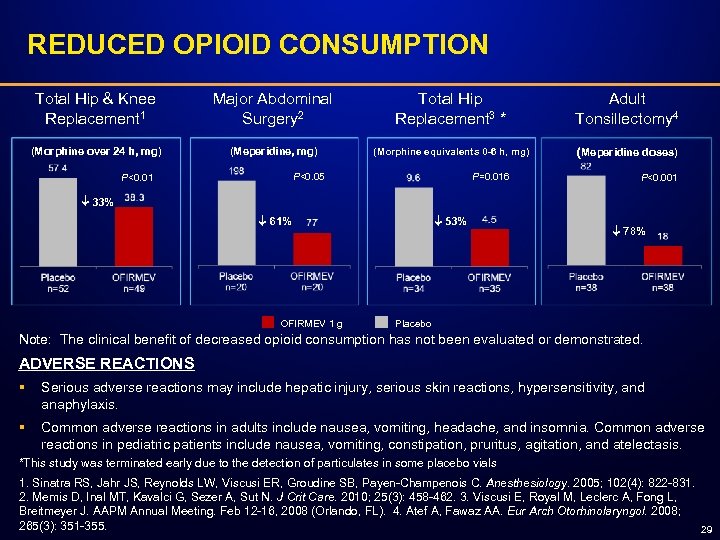 REDUCED OPIOID CONSUMPTION Total Hip & Knee Replacement 1 Major Abdominal Surgery 2 Total Hip Replacement 3 * Adult Tonsillectomy 4 (Morphine over 24 h, mg) (Meperidine, mg) (Morphine equivalents 0 -6 h, mg) (Meperidine doses) P<0. 05 P<0. 01 P=0. 016 P<0. 001 33% 61% OFIRMEV 1 g 53% 78% Placebo Note: The clinical benefit of decreased opioid consumption has not been evaluated or demonstrated. ADVERSE REACTIONS § Serious adverse reactions may include hepatic injury, serious skin reactions, hypersensitivity, and anaphylaxis. § Common adverse reactions in adults include nausea, vomiting, headache, and insomnia. Common adverse reactions in pediatric patients include nausea, vomiting, constipation, pruritus, agitation, and atelectasis. *This study was terminated early due to the detection of particulates in some placebo vials 1. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102(4): 822 -831. 2. Memis D, Inal MT, Kavalci G, Sezer A, Sut N. J Crit Care. 2010; 25(3): 458 -462. 3. Viscusi E, Royal M, Leclerc A, Fong L, Breitmeyer J. AAPM Annual Meeting. Feb 12 -16, 2008 (Orlando, FL). 4. Atef A, Fawaz AA. Eur Arch Otorhinolaryngol. 2008; 265(3): 351 -355. 29
REDUCED OPIOID CONSUMPTION Total Hip & Knee Replacement 1 Major Abdominal Surgery 2 Total Hip Replacement 3 * Adult Tonsillectomy 4 (Morphine over 24 h, mg) (Meperidine, mg) (Morphine equivalents 0 -6 h, mg) (Meperidine doses) P<0. 05 P<0. 01 P=0. 016 P<0. 001 33% 61% OFIRMEV 1 g 53% 78% Placebo Note: The clinical benefit of decreased opioid consumption has not been evaluated or demonstrated. ADVERSE REACTIONS § Serious adverse reactions may include hepatic injury, serious skin reactions, hypersensitivity, and anaphylaxis. § Common adverse reactions in adults include nausea, vomiting, headache, and insomnia. Common adverse reactions in pediatric patients include nausea, vomiting, constipation, pruritus, agitation, and atelectasis. *This study was terminated early due to the detection of particulates in some placebo vials 1. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102(4): 822 -831. 2. Memis D, Inal MT, Kavalci G, Sezer A, Sut N. J Crit Care. 2010; 25(3): 458 -462. 3. Viscusi E, Royal M, Leclerc A, Fong L, Breitmeyer J. AAPM Annual Meeting. Feb 12 -16, 2008 (Orlando, FL). 4. Atef A, Fawaz AA. Eur Arch Otorhinolaryngol. 2008; 265(3): 351 -355. 29
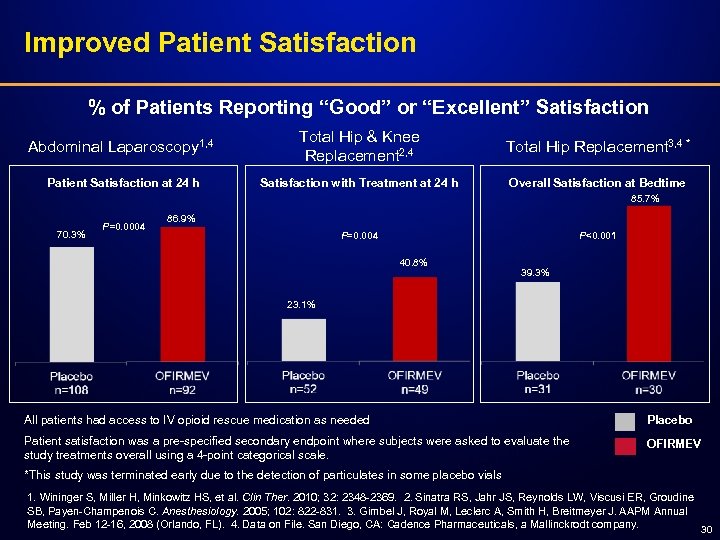 Improved Patient Satisfaction % of Patients Reporting “Good” or “Excellent” Satisfaction Abdominal Laparoscopy 1, 4 Total Hip & Knee Replacement 2, 4 Total Hip Replacement 3, 4 * Patient Satisfaction at 24 h Satisfaction with Treatment at 24 h Overall Satisfaction at Bedtime 85. 7% 70. 3% P=0. 0004 86. 9% P=0. 004 P<0. 001 40. 8% 39. 3% 23. 1% All patients had access to IV opioid rescue medication as needed Placebo Patient satisfaction was a pre-specified secondary endpoint where subjects were asked to evaluate the study treatments overall using a 4 -point categorical scale. OFIRMEV *This study was terminated early due to the detection of particulates in some placebo vials 1. Wininger S, Miller H, Minkowitz HS, et al. Clin Ther. 2010; 32: 2348 -2369. 2. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102: 822 -831. 3. Gimbel J, Royal M, Leclerc A, Smith H, Breitmeyer J. AAPM Annual Meeting. Feb 12 -16, 2008 (Orlando, FL). 4. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 30
Improved Patient Satisfaction % of Patients Reporting “Good” or “Excellent” Satisfaction Abdominal Laparoscopy 1, 4 Total Hip & Knee Replacement 2, 4 Total Hip Replacement 3, 4 * Patient Satisfaction at 24 h Satisfaction with Treatment at 24 h Overall Satisfaction at Bedtime 85. 7% 70. 3% P=0. 0004 86. 9% P=0. 004 P<0. 001 40. 8% 39. 3% 23. 1% All patients had access to IV opioid rescue medication as needed Placebo Patient satisfaction was a pre-specified secondary endpoint where subjects were asked to evaluate the study treatments overall using a 4 -point categorical scale. OFIRMEV *This study was terminated early due to the detection of particulates in some placebo vials 1. Wininger S, Miller H, Minkowitz HS, et al. Clin Ther. 2010; 32: 2348 -2369. 2. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102: 822 -831. 3. Gimbel J, Royal M, Leclerc A, Smith H, Breitmeyer J. AAPM Annual Meeting. Feb 12 -16, 2008 (Orlando, FL). 4. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 30
 Safety and Tolerability of OFIRMEV® (acetaminophen) Injection
Safety and Tolerability of OFIRMEV® (acetaminophen) Injection
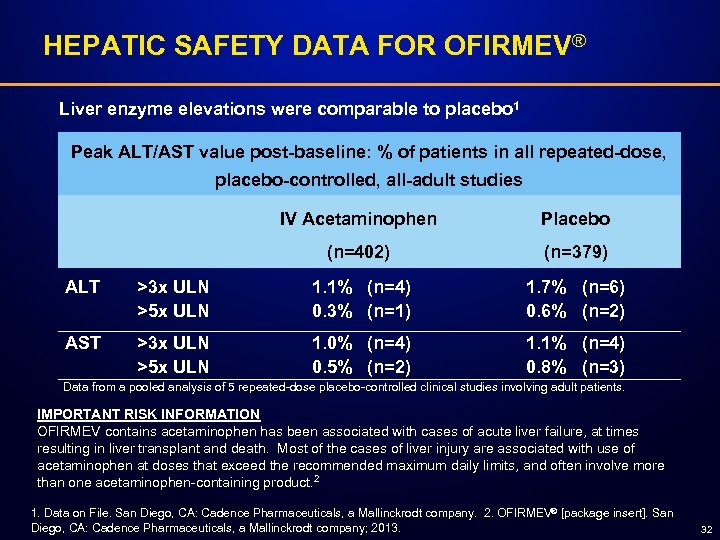 HEPATIC SAFETY DATA FOR OFIRMEV® Liver enzyme elevations were comparable to placebo 1 Peak ALT/AST value post-baseline: % of patients in all repeated-dose, placebo-controlled, all-adult studies IV Acetaminophen (n=402) Placebo (n=379) ALT >3 x ULN >5 x ULN 1. 1% (n=4) 0. 3% (n=1) 1. 7% (n=6) 0. 6% (n=2) AST >3 x ULN >5 x ULN 1. 0% (n=4) 0. 5% (n=2) 1. 1% (n=4) 0. 8% (n=3) Data from a pooled analysis of 5 repeated-dose placebo-controlled clinical studies involving adult patients. IMPORTANT RISK INFORMATION OFIRMEV contains acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with use of acetaminophen at doses that exceed the recommended maximum daily limits, and often involve more than one acetaminophen-containing product. 2 1. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 2. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 32
HEPATIC SAFETY DATA FOR OFIRMEV® Liver enzyme elevations were comparable to placebo 1 Peak ALT/AST value post-baseline: % of patients in all repeated-dose, placebo-controlled, all-adult studies IV Acetaminophen (n=402) Placebo (n=379) ALT >3 x ULN >5 x ULN 1. 1% (n=4) 0. 3% (n=1) 1. 7% (n=6) 0. 6% (n=2) AST >3 x ULN >5 x ULN 1. 0% (n=4) 0. 5% (n=2) 1. 1% (n=4) 0. 8% (n=3) Data from a pooled analysis of 5 repeated-dose placebo-controlled clinical studies involving adult patients. IMPORTANT RISK INFORMATION OFIRMEV contains acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with use of acetaminophen at doses that exceed the recommended maximum daily limits, and often involve more than one acetaminophen-containing product. 2 1. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 2. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 32
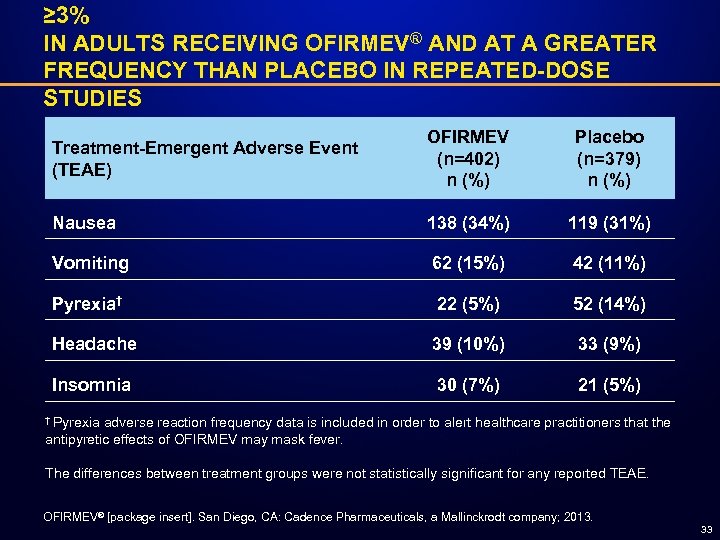 ≥ 3% IN ADULTS RECEIVING OFIRMEV® AND AT A GREATER FREQUENCY THAN PLACEBO IN REPEATED-DOSE STUDIES Treatment-Emergent Adverse Event (TEAE) OFIRMEV (n=402) n (%) Placebo (n=379) n (%) Nausea 138 (34%) 119 (31%) Vomiting 62 (15%) 42 (11%) Pyrexia† 22 (5%) 52 (14%) Headache 39 (10%) 33 (9%) Insomnia 30 (7%) 21 (5%) † Pyrexia adverse reaction frequency data is included in order to alert healthcare practitioners that the antipyretic effects of OFIRMEV may mask fever. The differences between treatment groups were not statistically significant for any reported TEAE. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 33
≥ 3% IN ADULTS RECEIVING OFIRMEV® AND AT A GREATER FREQUENCY THAN PLACEBO IN REPEATED-DOSE STUDIES Treatment-Emergent Adverse Event (TEAE) OFIRMEV (n=402) n (%) Placebo (n=379) n (%) Nausea 138 (34%) 119 (31%) Vomiting 62 (15%) 42 (11%) Pyrexia† 22 (5%) 52 (14%) Headache 39 (10%) 33 (9%) Insomnia 30 (7%) 21 (5%) † Pyrexia adverse reaction frequency data is included in order to alert healthcare practitioners that the antipyretic effects of OFIRMEV may mask fever. The differences between treatment groups were not statistically significant for any reported TEAE. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 33
 Recommended Dosing of OFIRMEV® Dosing of OFIRMEV for Adults, Adolescents, and Children ≥ 2 Years Old Age Group Adults and adolescents (≥ 13 years old) ≥ 50 kg Adults and adolescents (≥ 13 years old) <50 kg Dose Given Every 4 h* Dose Given Every 6 h* Maximum Single Dose* Maximum Total Daily Dose of Acetaminophen (by all routes) 650 mg 1000 mg 4000 mg in 24 h 15 mg/kg (up to 750 mg) 75 mg/kg in 24 h (up to 3750 mg) 12. 5 mg/kg Children ≥ 2 to 12 years old * Each m. L contains 10 mg of OFIRMEV. WARNINGS AND PRECAUTIONS Administration of acetaminophen by any route in doses higher than recommended may result in hepatic injury, including the risk of liver failure and death. Please see Important Risk Information, including complete boxed warning, in Full Prescribing Information. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 34
Recommended Dosing of OFIRMEV® Dosing of OFIRMEV for Adults, Adolescents, and Children ≥ 2 Years Old Age Group Adults and adolescents (≥ 13 years old) ≥ 50 kg Adults and adolescents (≥ 13 years old) <50 kg Dose Given Every 4 h* Dose Given Every 6 h* Maximum Single Dose* Maximum Total Daily Dose of Acetaminophen (by all routes) 650 mg 1000 mg 4000 mg in 24 h 15 mg/kg (up to 750 mg) 75 mg/kg in 24 h (up to 3750 mg) 12. 5 mg/kg Children ≥ 2 to 12 years old * Each m. L contains 10 mg of OFIRMEV. WARNINGS AND PRECAUTIONS Administration of acetaminophen by any route in doses higher than recommended may result in hepatic injury, including the risk of liver failure and death. Please see Important Risk Information, including complete boxed warning, in Full Prescribing Information. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 34
 Administration of OFIRMEV® § OFIRMEV® should be administered only as a 15 -minute IV infusion § Minimum dosing interval is 4 hours, not to exceed 4 g of acetaminophen by all routes in 24 hours § An infusion pump is not required except when delivering weight-based calculated doses less than 600 mg (60 m. L). Use a syringe pump for administering small volume doses, particularly in young children § No dosage adjustment is required when transitioning to oral acetaminophen OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 35
Administration of OFIRMEV® § OFIRMEV® should be administered only as a 15 -minute IV infusion § Minimum dosing interval is 4 hours, not to exceed 4 g of acetaminophen by all routes in 24 hours § An infusion pump is not required except when delivering weight-based calculated doses less than 600 mg (60 m. L). Use a syringe pump for administering small volume doses, particularly in young children § No dosage adjustment is required when transitioning to oral acetaminophen OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 35
 SUMMARY OF OFIRMEV® DATA ■ OFIRMEV® should be considered as a foundational approach to multimodal analgesia ■ OFIRMEV has demonstrated: § Significant reductions in pain intensity 1 -4 § Reduced opioid consumption 1, 3, 4 § Improved patient satisfaction 1, 2 ■ OFIRMEV has an established safety profile and was well tolerated in clinical trials 1 -5 *Note: The clinical benefit of reduced opioid consumption has not been evaluated or demonstrated IMPORTANT RISK INFORMATION Do not exceed the maximum recommended daily limit of acetaminophen by all routes. 5 Acetaminophen should be used with caution in patients with the following conditions: hepatic impairment or active hepatic disease, alcoholism, chronic malnutrition, severe hypovolemia, or severe renal impairment. 5 1. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102: 822 -831. 2. Wininger SJ, Miller H, Minkowitz, et al. Clin Ther. 2010; 32: 2348 -69. 3. Memis D, Inal MT, Kavalci G, Sezer A, Sut N. J Crit Care. 2010; 25: 458 -462. 4. Atef A, Fawaz AAl. Eur Arch Otorhinolaryngol. 2008; 265: 351 -355. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 36
SUMMARY OF OFIRMEV® DATA ■ OFIRMEV® should be considered as a foundational approach to multimodal analgesia ■ OFIRMEV has demonstrated: § Significant reductions in pain intensity 1 -4 § Reduced opioid consumption 1, 3, 4 § Improved patient satisfaction 1, 2 ■ OFIRMEV has an established safety profile and was well tolerated in clinical trials 1 -5 *Note: The clinical benefit of reduced opioid consumption has not been evaluated or demonstrated IMPORTANT RISK INFORMATION Do not exceed the maximum recommended daily limit of acetaminophen by all routes. 5 Acetaminophen should be used with caution in patients with the following conditions: hepatic impairment or active hepatic disease, alcoholism, chronic malnutrition, severe hypovolemia, or severe renal impairment. 5 1. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Anesthesiology. 2005; 102: 822 -831. 2. Wininger SJ, Miller H, Minkowitz, et al. Clin Ther. 2010; 32: 2348 -69. 3. Memis D, Inal MT, Kavalci G, Sezer A, Sut N. J Crit Care. 2010; 25: 458 -462. 4. Atef A, Fawaz AAl. Eur Arch Otorhinolaryngol. 2008; 265: 351 -355. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 36
 IMPORTANT SAFETY INFORMATION FOR OFIRMEV® (ACETAMINOPHEN) INJECTION INDICATIONS AND USAGE ■ OFIRMEV (acetaminophen) injection is indicated for the: § management of mild to moderate pain § management of moderate to severe pain with adjunctive opioid analgesics § reduction of fever IMPORTANT RISK INFORMATION WARNING: RISK OF MEDICATION ERRORS AND HEPATOTOXICITY Take care when prescribing, preparing, and administering OFIRMEV injection to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that: • the dose in milligrams (mg) and milliliters (m. L) is not confused; • the dosing is based on weight for patients under 50 kg; • infusion pumps are properly programmed; and • the total daily dose of acetaminophen from all sources does not exceed maximum daily limits. OFIRMEV contains acetaminophen. Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed the recommended maximum daily limits, and often involve more than one acetaminophen-containing product. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 37
IMPORTANT SAFETY INFORMATION FOR OFIRMEV® (ACETAMINOPHEN) INJECTION INDICATIONS AND USAGE ■ OFIRMEV (acetaminophen) injection is indicated for the: § management of mild to moderate pain § management of moderate to severe pain with adjunctive opioid analgesics § reduction of fever IMPORTANT RISK INFORMATION WARNING: RISK OF MEDICATION ERRORS AND HEPATOTOXICITY Take care when prescribing, preparing, and administering OFIRMEV injection to avoid dosing errors which could result in accidental overdose and death. In particular, be careful to ensure that: • the dose in milligrams (mg) and milliliters (m. L) is not confused; • the dosing is based on weight for patients under 50 kg; • infusion pumps are properly programmed; and • the total daily dose of acetaminophen from all sources does not exceed maximum daily limits. OFIRMEV contains acetaminophen. Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at doses that exceed the recommended maximum daily limits, and often involve more than one acetaminophen-containing product. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 37
 IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONTRAINDICATIONS ■ Acetaminophen is contraindicated in patients with: § known hypersensitivity to acetaminophen or to any of the excipients in the intravenous (IV) formulation. § severe hepatic impairment or severe active liver disease. WARNINGS AND PRECAUTIONS ■ Administration of acetaminophen in doses higher than recommended may result in hepatic injury, including the risk of liver failure and death. Do not exceed the maximum recommended daily dose of acetaminophen. The maximum recommended daily dose of acetaminophen includes all routes of acetaminophen administration and all acetaminophen-containing products administered, including combination products. Dosing errors could result in accidental overdose and death. ■ Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, alcoholism, chronic malnutrition, severe hypovolemia (e. g. , due to dehydration or blood loss), or severe renal impairment (creatinine clearance ≤ 30 m. L/min). OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 38
IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONTRAINDICATIONS ■ Acetaminophen is contraindicated in patients with: § known hypersensitivity to acetaminophen or to any of the excipients in the intravenous (IV) formulation. § severe hepatic impairment or severe active liver disease. WARNINGS AND PRECAUTIONS ■ Administration of acetaminophen in doses higher than recommended may result in hepatic injury, including the risk of liver failure and death. Do not exceed the maximum recommended daily dose of acetaminophen. The maximum recommended daily dose of acetaminophen includes all routes of acetaminophen administration and all acetaminophen-containing products administered, including combination products. Dosing errors could result in accidental overdose and death. ■ Use caution when administering acetaminophen in patients with the following conditions: hepatic impairment or active hepatic disease, alcoholism, chronic malnutrition, severe hypovolemia (e. g. , due to dehydration or blood loss), or severe renal impairment (creatinine clearance ≤ 30 m. L/min). OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 38
 IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONT. WARNINGS AND PRECAUTIONS, cont. ■ Rarely, acetaminophen may cause serious skin reactions such as acute generalized exanthematous pustulosis (AGEP), Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. ■ Hypersensitivity and anaphylaxis associated with the use of acetaminophen have been reported. Clinical signs included swelling of the face, mouth, and throat, respiratory distress, urticaria, rash, and pruritis. ■ The antipyretic effects of OFIRMEV may mask fever. ADVERSE REACTIONS ■ Serious adverse reactions may include hepatic injury, serious skin reactions, hypersensitivity, and anaphylaxis. ■ Common adverse reactions in adults include nausea, vomiting, headache, and insomnia. Common adverse reactions in pediatric patients include nausea, vomiting, constipation, pruritus, agitation, and atelectasis. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 39
IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONT. WARNINGS AND PRECAUTIONS, cont. ■ Rarely, acetaminophen may cause serious skin reactions such as acute generalized exanthematous pustulosis (AGEP), Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. ■ Hypersensitivity and anaphylaxis associated with the use of acetaminophen have been reported. Clinical signs included swelling of the face, mouth, and throat, respiratory distress, urticaria, rash, and pruritis. ■ The antipyretic effects of OFIRMEV may mask fever. ADVERSE REACTIONS ■ Serious adverse reactions may include hepatic injury, serious skin reactions, hypersensitivity, and anaphylaxis. ■ Common adverse reactions in adults include nausea, vomiting, headache, and insomnia. Common adverse reactions in pediatric patients include nausea, vomiting, constipation, pruritus, agitation, and atelectasis. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 39
 IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONT. USE IN SPECIFIC POPULATIONS ■ Pregnancy: Pregnancy Category C. OFIRMEV should be given to a pregnant woman only if clearly needed. ■ Breast Feeding: While studies with OFIRMEV have not been conducted, acetaminophen is secreted in human milk in small quantities after oral administration. ■ Pediatrics: The effectiveness of OFIRMEV for the treatment of acute pain and fever has not been studied in pediatric patients <2 years of age. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 40
IMPORTANT RISK INFORMATION FOR OFIRMEV®, CONT. USE IN SPECIFIC POPULATIONS ■ Pregnancy: Pregnancy Category C. OFIRMEV should be given to a pregnant woman only if clearly needed. ■ Breast Feeding: While studies with OFIRMEV have not been conducted, acetaminophen is secreted in human milk in small quantities after oral administration. ■ Pediatrics: The effectiveness of OFIRMEV for the treatment of acute pain and fever has not been studied in pediatric patients <2 years of age. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. 40
 REFERENCES De. Frances CJ, Lucas CA, Buie VC, Golosinkskiy A. 2006 National Hospital Discharge Survey. Natl Health Stat Report. 2008 Jul 30; (5): 1 -20. Cullen KA, Hall MJ, Golosinkskiy A. Ambulatory surgery in the United States, 2006. Natl Health Stat Report. 2009 Jan 28; (11): 1 -25. Wu CL, Raja SN, Treatment of acute postoperative pain. Lancet. 2011; 377: 2215 -2225. Phillips DM. JCAHO pain management standards unveiled. JAMA. 2000; 284(4): 428 -429. Aubrun F, Langeron O, Quesnel C, Coriat P, Riou B. Relationships between measurement of pain using visual analog score and morphine requirements during postoperative intravenous morphine titration. Anesthesiology. 2003; 98(6): 1415 -1421. The Joint Commission Sentinel Event Alert. Safe use of opioids in hospitals. Issue 49; August 8, 2012. www. jointcommission. org/assets/1/18/SEA_49_opioids_8_2_12_finalpdf. American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2012; 116(2): 248 -273. World Health Organization. Pain relief ladder. http: //www. who. int/cancer/palliative/painladder/en/. Accessed September 10, 2014. Jarzyna D, Jungquist CR, Pasero C, et al. American Society of Pain Management Nursing guidelines on monitoring for opioidinduced sedation and respiratory depression. Pain Manag Nurs. 2011; 12(3): 118 -145. Gottschalk A, Smith DS. New concepts in acute pain therapy: preemptive analgesia. Am Fam Physician. 2001; 63: 1979 -1984. Smith HS. Potential analgesic mechanisms of acetaminophen. Pain Physician. 2009; 12: 269 -280. Sinatra RS, Jahr JS, eds. The Essence of Analgesia and Analgesics. New York, NY: Cambridge University Press; 2011. Crews JC. Multimodal pain management strategies for office-based and ambulatory procedures. JAMA. 2002; 288: 629 -632. 41
REFERENCES De. Frances CJ, Lucas CA, Buie VC, Golosinkskiy A. 2006 National Hospital Discharge Survey. Natl Health Stat Report. 2008 Jul 30; (5): 1 -20. Cullen KA, Hall MJ, Golosinkskiy A. Ambulatory surgery in the United States, 2006. Natl Health Stat Report. 2009 Jan 28; (11): 1 -25. Wu CL, Raja SN, Treatment of acute postoperative pain. Lancet. 2011; 377: 2215 -2225. Phillips DM. JCAHO pain management standards unveiled. JAMA. 2000; 284(4): 428 -429. Aubrun F, Langeron O, Quesnel C, Coriat P, Riou B. Relationships between measurement of pain using visual analog score and morphine requirements during postoperative intravenous morphine titration. Anesthesiology. 2003; 98(6): 1415 -1421. The Joint Commission Sentinel Event Alert. Safe use of opioids in hospitals. Issue 49; August 8, 2012. www. jointcommission. org/assets/1/18/SEA_49_opioids_8_2_12_finalpdf. American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2012; 116(2): 248 -273. World Health Organization. Pain relief ladder. http: //www. who. int/cancer/palliative/painladder/en/. Accessed September 10, 2014. Jarzyna D, Jungquist CR, Pasero C, et al. American Society of Pain Management Nursing guidelines on monitoring for opioidinduced sedation and respiratory depression. Pain Manag Nurs. 2011; 12(3): 118 -145. Gottschalk A, Smith DS. New concepts in acute pain therapy: preemptive analgesia. Am Fam Physician. 2001; 63: 1979 -1984. Smith HS. Potential analgesic mechanisms of acetaminophen. Pain Physician. 2009; 12: 269 -280. Sinatra RS, Jahr JS, eds. The Essence of Analgesia and Analgesics. New York, NY: Cambridge University Press; 2011. Crews JC. Multimodal pain management strategies for office-based and ambulatory procedures. JAMA. 2002; 288: 629 -632. 41
 REFERENCES, CONT. Ventafridda V, Tamburini M, Caraceni A, De Conno F, Naldi F. A validation study of the WHO method for cancer pain relief. Cancer. 1987; 59: 850 -856. American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2004; 100: 1573 -1581. Wells N, Pasero C, Mc. Caffery M. Improving the quality of care through pain assessment and management. In: Hughes RG, ed. Patient Safety and Quality: an Evidence-based Handbook for Nurses. AHRQ Publication No. 08 -0043. Rockville, MD: Agency for Healthcare Research and Quality; March 2008. US Department of Health and Human Services, Centers for Medicare and Medicaid. HCAHPS: Patients' Perspectives of Care Survey. http: //www. cms. gov/Hospital. Quality. Inits/30_Hospital. HCAHPS. asp. Accessed September 10, 2014. American Hospital Association (AHA) Hospital-based purchasing program: the final rule. May 24, 2011. http: //www. americangovernance. com/americangovernance/webinar/policy/pdf/final_rule_vbp_regulatory_advisory. pdf. Accessed September 10, 2014. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. Singla NK, Parulan C, Samson R, et al. Plasma and cerebrospinal fluid pharmacokinetic parameters after single-dose administration of intravenous, oral, or rectal acetaminophen. Pain Pract. 2012; 12(7): 523 -532. Berger MM, Berger-Gryllaki M, Wiesel PH, et. al. Intestinal absorption in patients after cardiac surgery. Crit Care Med. 2000; 28(7): 2217 -2223. Petring OU, Dawson PJ, Blake DW, Jones DJ, Bjorksten AR, Libreri FC. Normal postoperative gastric emptying after orthopaedic surgery with spinal anaesthesia and i. m. ketorolac as the first postoperative analgesic. Br J Anaesth. 1995; 74: 257 -260. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 42
REFERENCES, CONT. Ventafridda V, Tamburini M, Caraceni A, De Conno F, Naldi F. A validation study of the WHO method for cancer pain relief. Cancer. 1987; 59: 850 -856. American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2004; 100: 1573 -1581. Wells N, Pasero C, Mc. Caffery M. Improving the quality of care through pain assessment and management. In: Hughes RG, ed. Patient Safety and Quality: an Evidence-based Handbook for Nurses. AHRQ Publication No. 08 -0043. Rockville, MD: Agency for Healthcare Research and Quality; March 2008. US Department of Health and Human Services, Centers for Medicare and Medicaid. HCAHPS: Patients' Perspectives of Care Survey. http: //www. cms. gov/Hospital. Quality. Inits/30_Hospital. HCAHPS. asp. Accessed September 10, 2014. American Hospital Association (AHA) Hospital-based purchasing program: the final rule. May 24, 2011. http: //www. americangovernance. com/americangovernance/webinar/policy/pdf/final_rule_vbp_regulatory_advisory. pdf. Accessed September 10, 2014. OFIRMEV® [package insert]. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company; 2013. Singla NK, Parulan C, Samson R, et al. Plasma and cerebrospinal fluid pharmacokinetic parameters after single-dose administration of intravenous, oral, or rectal acetaminophen. Pain Pract. 2012; 12(7): 523 -532. Berger MM, Berger-Gryllaki M, Wiesel PH, et. al. Intestinal absorption in patients after cardiac surgery. Crit Care Med. 2000; 28(7): 2217 -2223. Petring OU, Dawson PJ, Blake DW, Jones DJ, Bjorksten AR, Libreri FC. Normal postoperative gastric emptying after orthopaedic surgery with spinal anaesthesia and i. m. ketorolac as the first postoperative analgesic. Br J Anaesth. 1995; 74: 257 -260. Data on File. San Diego, CA: Cadence Pharmaceuticals, a Mallinckrodt company. 42
 REFERENCES, CONT. Kalantzi L, Reppas C, Dressman JB, et al. Biowaiver monographs for immediate release solid oral dosage forms: Acetaminophen (paracetamol). J Pharma Sci. 2006; 95(1): 4 -14. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Efficacy and safety of single and repeated administration of 1 gram intravenous acetaminophen injection (paracetamol) for pain management after major orthopedic surgery. Anesthesiology. 2005; 102: 822 -831. Viscusi G, Royal M, Leclerc A, Fong L, Breitmeyer J. Pharmacokinetics, efficacy and safety of IV acetaminophen in the treatment of pain following total hip arthroplasty: results of a double-blind, randomized, placebo-controlled, single-dose study. Presented at 24 th Annual Meeting of the American Academy of Pain Medicine. February 12 -16, 2008 (Orlando, FL). Khan ZU, Iqbal J, Saleh H, El Deek AM. Intravenous paracetamol is as effective as morphine in knee arthroscopic day surgery procedures. Pak J Med Sci. 2007; 23(6): 851– 853. Memis D, Inal MT, Kavalci G, Sezer A, Sut N. Intravenous paracetamol reduced the use of opioids, extubation time, and opioidrelated adverse effects after major surgery in intensive care unit. J Crit Care. 2010; 25(3): 458 462. Bektas F, Eken C, Karadeniz O, Goksu E, Cubuk M, Cete Y. Intravenous paracetamol or morphine for the treatment of renal colic: A randomized, placebo-controlled trial. Ann Emer g. Med. 2009; 54: 568 574. Serinken M, Eken C, Turkcuer I, Elicabuk H, Uyanik E, Schultz CH. Intravenous paracetamol versus morphine for renal colic in the emergency department: a randomised double-blind controlled trial. Emerg Med J. 2012; 29: 902 905. Atef A, Fawaz AA. Intravenous paracetamol is highly effective in pain treatment after tonsillectomy in adults. Eur Arch Otorhinolaryngol. 2008; 265: 351 -355. Wininger SJ, Miller H, Minkowitz HS, et al. A randomized, double-blind, placebo-controlled, multi-center, repeat-dose study of two intravenous acetaminophen dosing regimens for the treatment of pain after abdominal laparoscopic surgery. Clin Ther. 2010; 32(14): 2348 2369. 43
REFERENCES, CONT. Kalantzi L, Reppas C, Dressman JB, et al. Biowaiver monographs for immediate release solid oral dosage forms: Acetaminophen (paracetamol). J Pharma Sci. 2006; 95(1): 4 -14. Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, Payen-Champenois C. Efficacy and safety of single and repeated administration of 1 gram intravenous acetaminophen injection (paracetamol) for pain management after major orthopedic surgery. Anesthesiology. 2005; 102: 822 -831. Viscusi G, Royal M, Leclerc A, Fong L, Breitmeyer J. Pharmacokinetics, efficacy and safety of IV acetaminophen in the treatment of pain following total hip arthroplasty: results of a double-blind, randomized, placebo-controlled, single-dose study. Presented at 24 th Annual Meeting of the American Academy of Pain Medicine. February 12 -16, 2008 (Orlando, FL). Khan ZU, Iqbal J, Saleh H, El Deek AM. Intravenous paracetamol is as effective as morphine in knee arthroscopic day surgery procedures. Pak J Med Sci. 2007; 23(6): 851– 853. Memis D, Inal MT, Kavalci G, Sezer A, Sut N. Intravenous paracetamol reduced the use of opioids, extubation time, and opioidrelated adverse effects after major surgery in intensive care unit. J Crit Care. 2010; 25(3): 458 462. Bektas F, Eken C, Karadeniz O, Goksu E, Cubuk M, Cete Y. Intravenous paracetamol or morphine for the treatment of renal colic: A randomized, placebo-controlled trial. Ann Emer g. Med. 2009; 54: 568 574. Serinken M, Eken C, Turkcuer I, Elicabuk H, Uyanik E, Schultz CH. Intravenous paracetamol versus morphine for renal colic in the emergency department: a randomised double-blind controlled trial. Emerg Med J. 2012; 29: 902 905. Atef A, Fawaz AA. Intravenous paracetamol is highly effective in pain treatment after tonsillectomy in adults. Eur Arch Otorhinolaryngol. 2008; 265: 351 -355. Wininger SJ, Miller H, Minkowitz HS, et al. A randomized, double-blind, placebo-controlled, multi-center, repeat-dose study of two intravenous acetaminophen dosing regimens for the treatment of pain after abdominal laparoscopic surgery. Clin Ther. 2010; 32(14): 2348 2369. 43
 REFERENCES, CONT. Gimbel J, Royal M, Leclerc A, Smith H, Breitmeyer J. Efficacy and safety of IV acetaminophen in the treatment of pain following primary total hip arthroplasty: results of a double-blind, randomized, placebo-controlled, multiple-dose, 24 hour study. Poster Presented at: 24 th Annual Meeting of the American Academy of Pain Management (February 12 16, 2008; Orlando, FL). 44
REFERENCES, CONT. Gimbel J, Royal M, Leclerc A, Smith H, Breitmeyer J. Efficacy and safety of IV acetaminophen in the treatment of pain following primary total hip arthroplasty: results of a double-blind, randomized, placebo-controlled, multiple-dose, 24 hour study. Poster Presented at: 24 th Annual Meeting of the American Academy of Pain Management (February 12 16, 2008; Orlando, FL). 44
 Thank You Mallinckrodt, the “M” brand mark, the Mallinckrodt Pharmaceuticals logo and other brands are trademarks of a Mallinckrodt company. © 2014 Mallinckrodt.
Thank You Mallinckrodt, the “M” brand mark, the Mallinckrodt Pharmaceuticals logo and other brands are trademarks of a Mallinckrodt company. © 2014 Mallinckrodt.


