ba0b1a0b5b2d00bfe2231c2158efef22.ppt
- Количество слайдов: 41

6 Biology 12 Chapter 6 Energy, Enzymes, and Metabolism

6 Overview/Objectives 6. 1 Cells and the Flow of Energy o Energy flow in ecosystems 6. 2 Metabolic Reactions and Energy Transformations o Coupling the breakdown of ATP with other reactions o Molecules that supply the energy to drive ATP synthesis o Function of ATP in cells 6. 3 Metabolic Pathways and Enzymes o o o Metabolic pathways Function of enzymes Environmental factors that affect enzyme activity How cells regulate enzyme activity and metabolic pathways Enzyme cofactors

6 6. 1 Cells and the Flow of Energy = the capacity to do work or to change things Why do we need energy? Growth Development Movement Metabolism Reproduction To live!
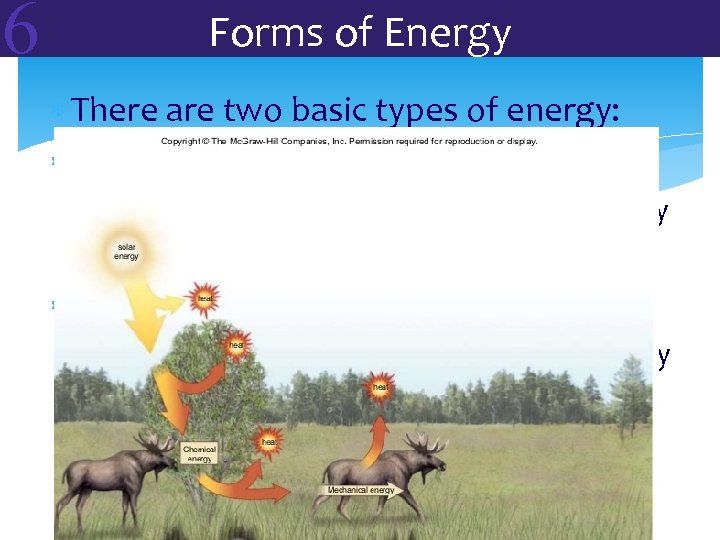
6 Forms of Energy There are two basic types of energy: Kinetic energy = energy of motion Mechanical energy= type of kinetic energy such as running or walking Potential energy = stored energy Chemical energy= type of potential energy Such as food

6 Two Laws of Thermodynamics The First Law of Thermodynamics: Energy cannot be created or destroyed, but it can be changed from one form to another Total initial energy = Total final energy

6 Two Laws of Thermodynamics The Second Law of Thermodynamics: Energy cannot be changed from one form to another without loss of useable energy (often through heat) Every energy transformation makes the world less organized and more disordered = entropy
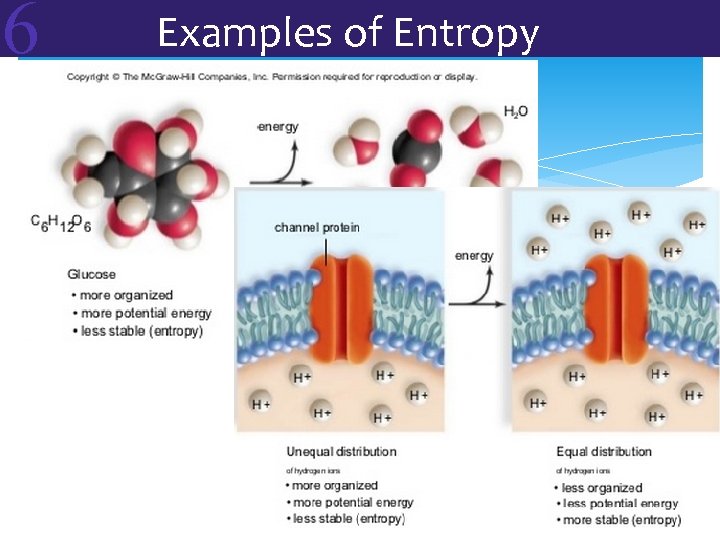
6 Examples of Entropy

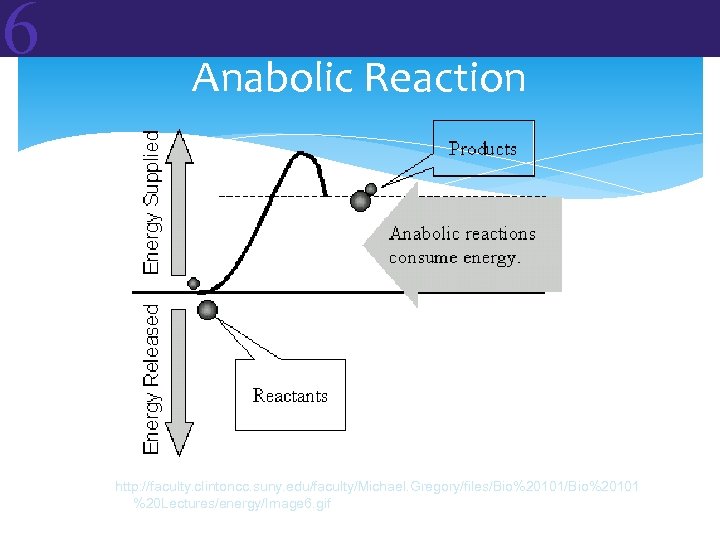
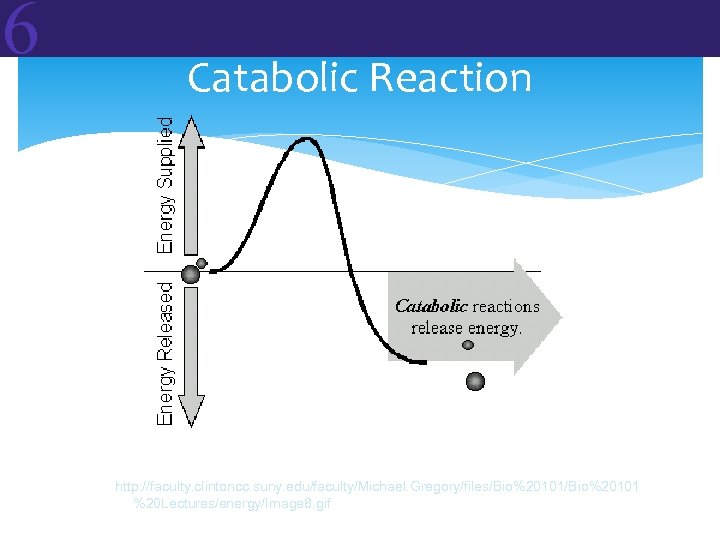
6 6. 2 Metabolic Reactions and Energy Transformations In living organisms, the topic of energy generally focuses on METABOLISM. Metabolism: The sum total of the chemical reactions that occur in an organism In all cells of all organisms, two types of metabolic reactions occur: • Anabolic Reactions = building up • Catabolic Reactions = breaking down Reactants Products

6 Anabolic vs Catabolic Anabolic reactions link simple molecules together to make complex ones. These are energy-storing reactions. Catabolic reactions break down complex molecules into simpler ones. These are energy-releasing reactions.
6 Anabolic Reaction http: //faculty. clintoncc. suny. edu/faculty/Michael. Gregory/files/Bio%20101 %20 Lectures/energy/Image 6. gif
6 Catabolic Reaction http: //faculty. clintoncc. suny. edu/faculty/Michael. Gregory/files/Bio%20101 %20 Lectures/energy/Image 8. gif

6 Free Energy= the amount of energy available, energy that is still “free” to do work, after a reaction has occurred Given the symbol G G = change in free energy after a reaction has occured Free Energy ( G) = useable energy G = Gproducts - Greactants

6 Two Types of Reactions ENDERGONIC REACTIONS (endothermic) • DG is positive (+) free energy is required • Products have more free energy than reactants • This is the case for anabolic reactions EXERGONIC REACTIONS (exothermic) DG is negative (–) free energy is released Products have less free energy than reactants This is the case for catabolic reactions
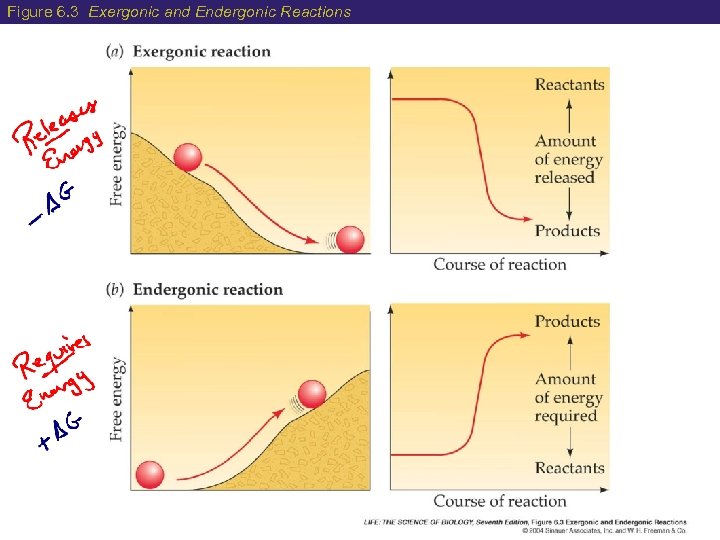
Figure 6. 3 Exergonic and Endergonic Reactions
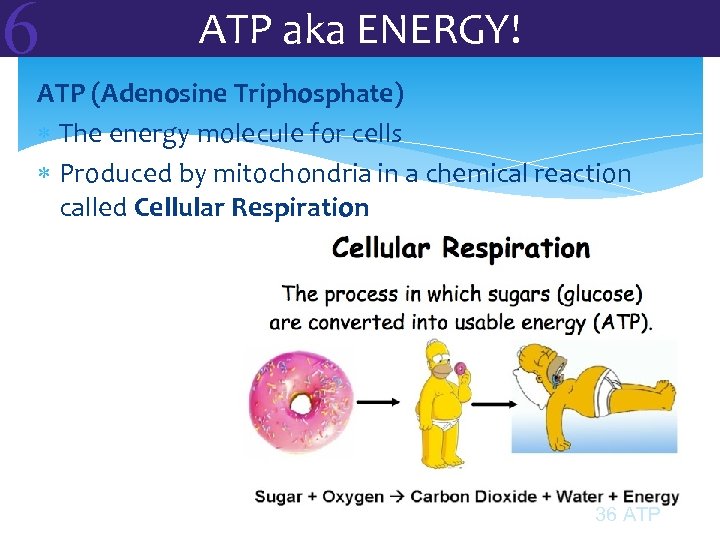
6 ATP aka ENERGY! ATP (Adenosine Triphosphate) The energy molecule for cells Produced by mitochondria in a chemical reaction called Cellular Respiration 36 ATP
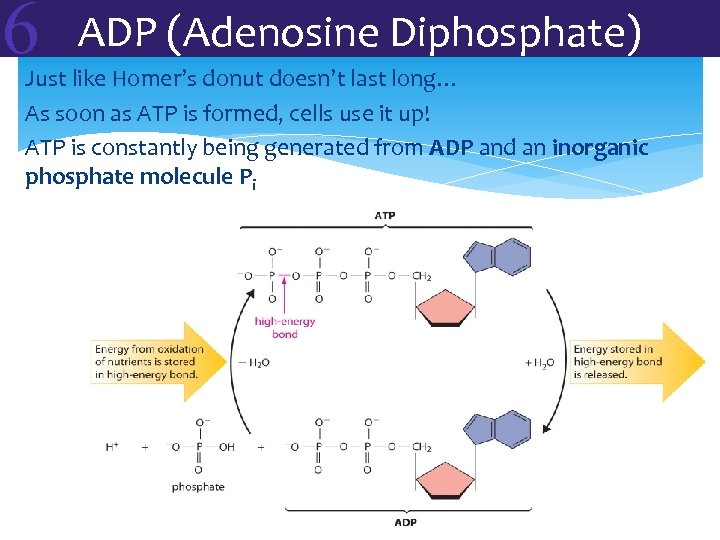
6 ADP (Adenosine Diphosphate) Just like Homer’s donut doesn’t last long… As soon as ATP is formed, cells use it up! ATP is constantly being generated from ADP and an inorganic phosphate molecule Pi

6 Advantages of Using ATP Advantages of ATP as a carrier of energy: 1) Provides a common currency of energy (It’s universal!) 2) When ATP ADP + P, there is enough energy released to be utilized by living cells (It works!) 3) When ATP is broken down, it is coupled with endergonic reactions to minimize energy loss (It’s efficient!)
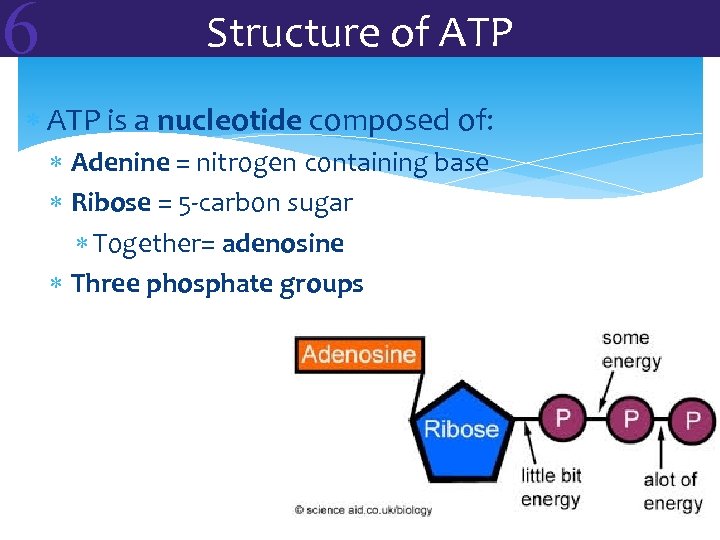
6 Structure of ATP is a nucleotide composed of: Adenine = nitrogen containing base Ribose = 5 -carbon sugar Together= adenosine Three phosphate groups
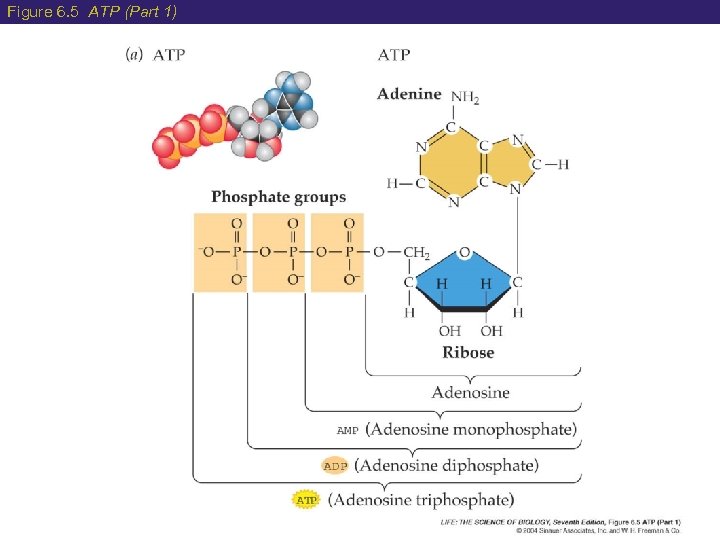
Figure 6. 5 ATP (Part 1)
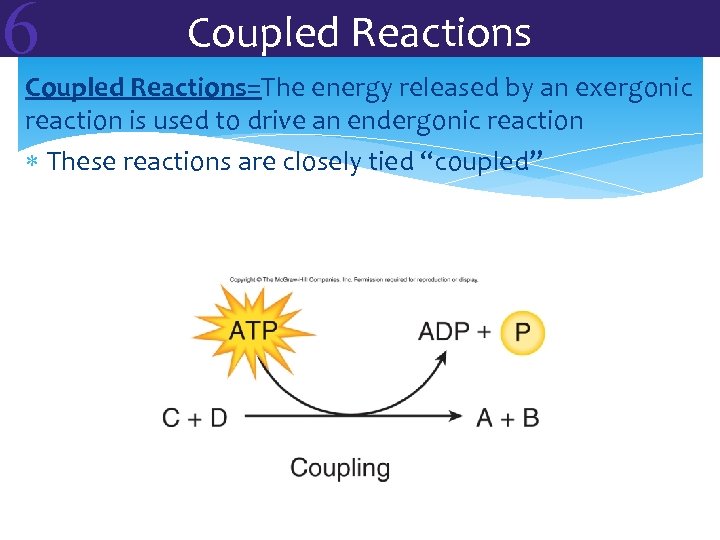
6 Coupled Reactions=The energy released by an exergonic reaction is used to drive an endergonic reaction These reactions are closely tied “coupled”
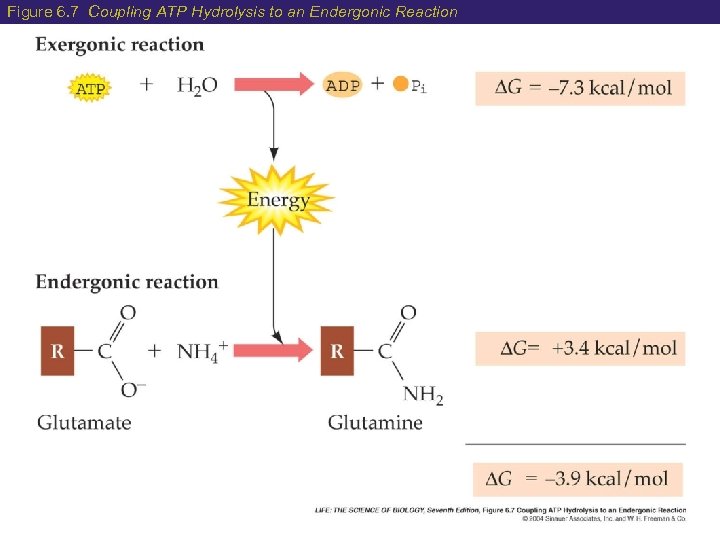
Figure 6. 7 Coupling ATP Hydrolysis to an Endergonic Reaction

6 Functions of ATP There at least 3 functions ATP: 1) Chemical Work: ATP supplies the energy to synthesize macromolecules 2) Transport Work: ATP supplies the energy for active transport across the cell membrane 3) Mechanical Work: ATP supplies the energy to contract muscles, beat cilia and flagella, etc.

6 6. 3 Metabolic Pathways and Enzymes Metabolic Pathway = series of linked reactions E 1 E 2 E 3 A B C D E = Enzyme = a protein molecule that functions as an organic catalyst. Speeds up chemical reactions Catalyst = any substance that speeds up a chemical reaction without itself being used up

6 Enzymes: Biological Catalysts How do enzymes speed up chemical reactions? Lowers the activation energy (EA) needed for a reaction to occur EA = Energy required for the reaction to go from reactants products Much less energy is required when an enzyme is present The result of the reaction does not change, but it does occur much faster!
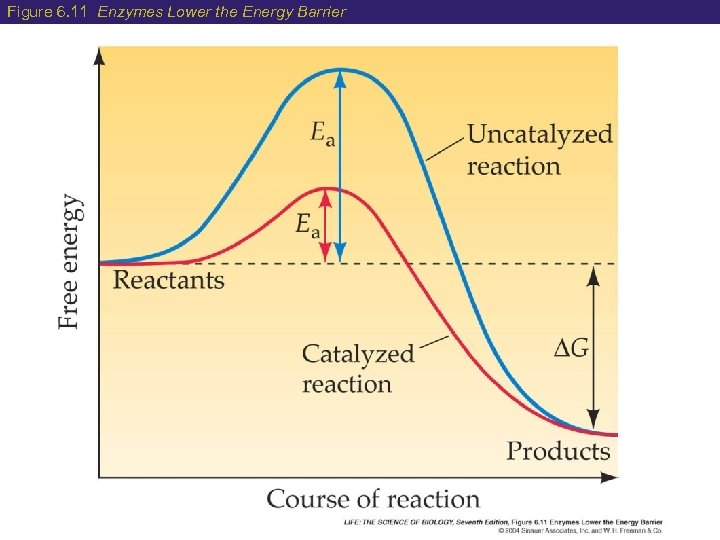
Figure 6. 11 Enzymes Lower the Energy Barrier
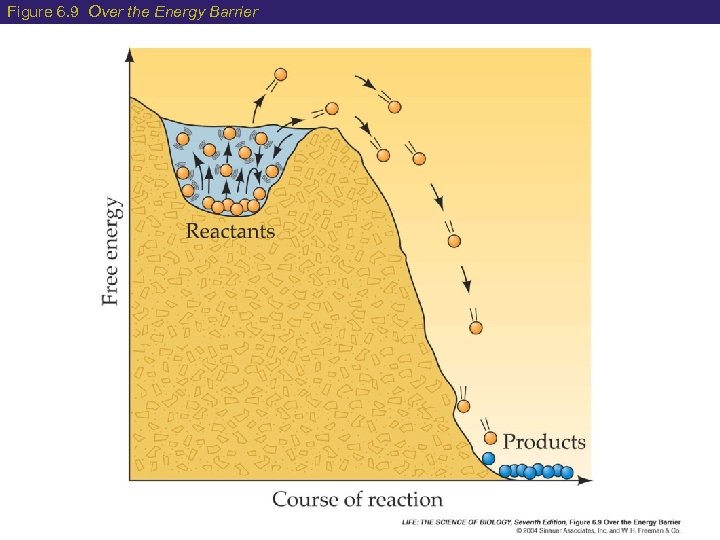
Figure 6. 9 Over the Energy Barrier
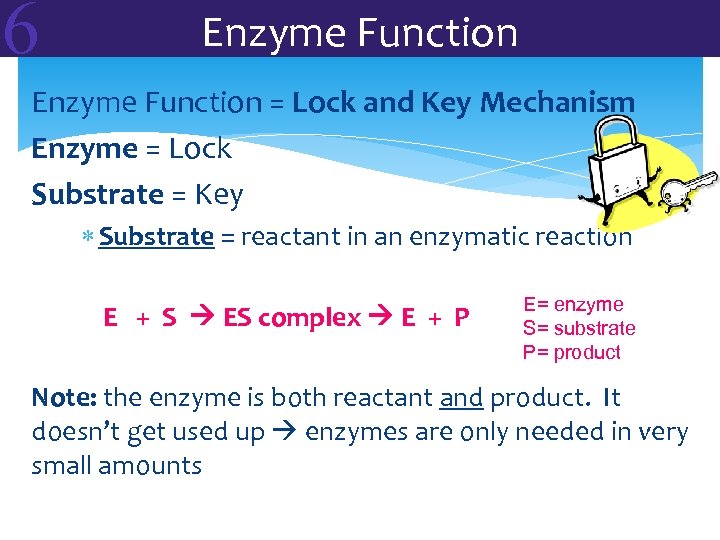
6 Enzyme Function = Lock and Key Mechanism Enzyme = Lock Substrate = Key Substrate = reactant in an enzymatic reaction E + S ES complex E + P E= enzyme S= substrate P= product Note: the enzyme is both reactant and product. It doesn’t get used up enzymes are only needed in very small amounts
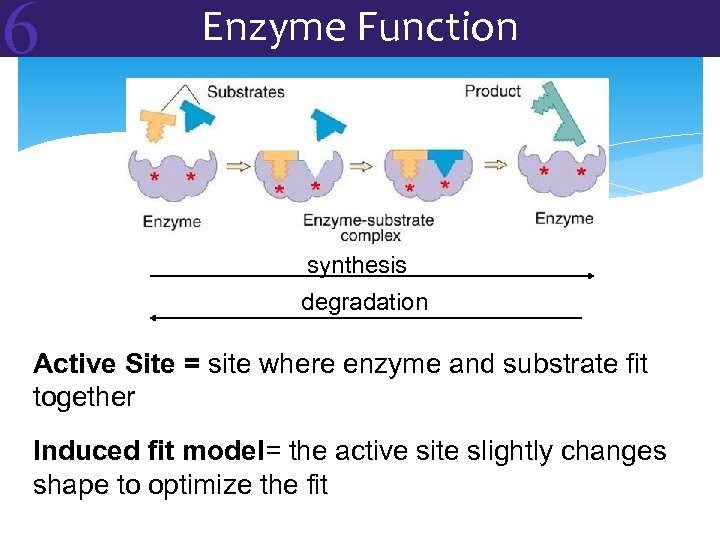
6 Enzyme Function synthesis degradation Active Site = site where enzyme and substrate fit together Induced fit model= the active site slightly changes shape to optimize the fit
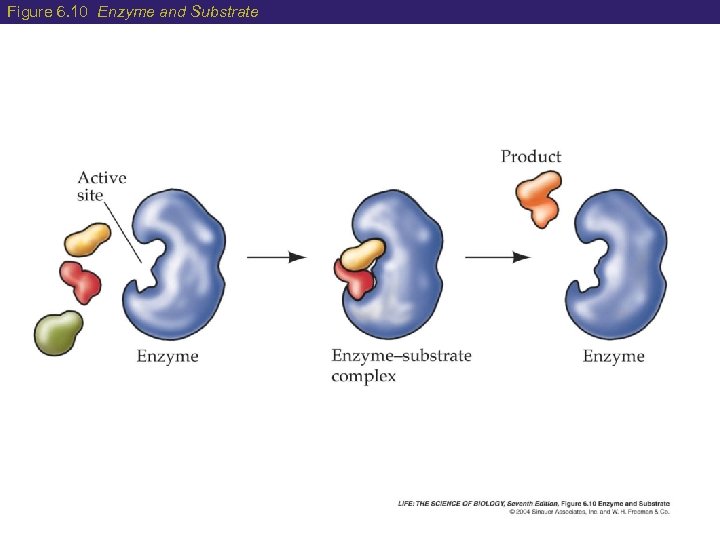
Figure 6. 10 Enzyme and Substrate
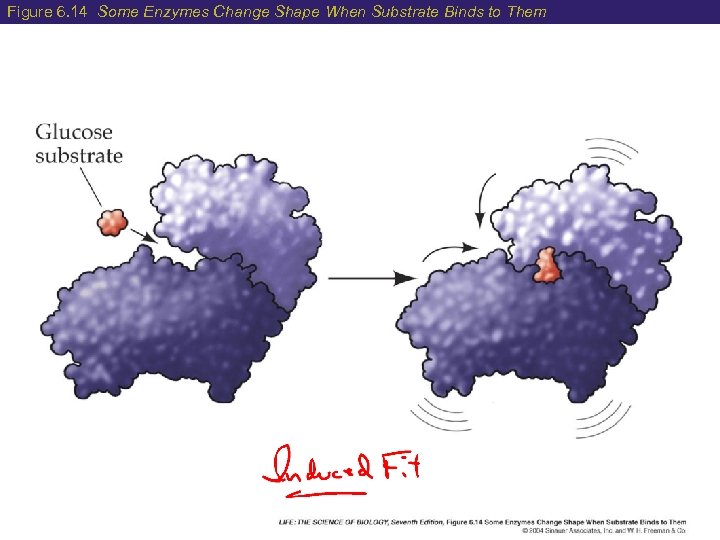
Figure 6. 14 Some Enzymes Change Shape When Substrate Binds to Them

6 Enzyme Names Every reaction requires a specific enzyme (… ends in “ase”) *The enzyme that is present and active will determine the reaction* Substrate Enzyme Lipid Lipase Urease Lactose Lactase

6 Factors Affecting Enzyme Speed There are 5 factors that affect enzymatic speed: 1) Substrate concentration 2) Temperature and p. H 3) Enzyme activation 4) Enzyme inhibition 5) Enzyme cofactors

6 Factors Affecting Enzyme Speed 1. Substrate Concentration Enzyme activity increases as substrate concentration increases Because more collisions occur between substrate and enzyme Until there are too many “keys” and not enough “locks” enzyme saturation

6 Factors Affecting Enzyme Speed 2) Temperature and p. H As temperature increases, reaction rate will increase Because higher temperature causes more effective collisions between enzyme and substrate Until the temperature becomes too high and the enzyme denatures Enzymes have an optimal p. H range and work best in it. Changes in p. H change the shape of the enzyme… Key won’t fit!
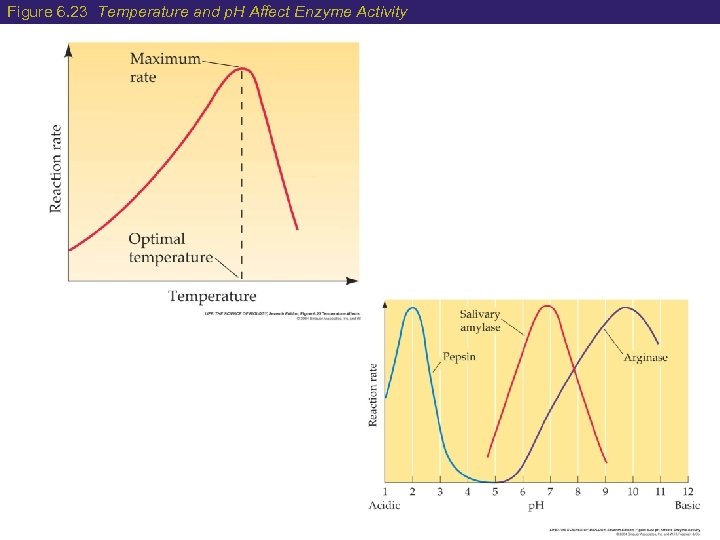
Figure 6. 23 Temperature and p. H Affect Enzyme Activity

6 Factors Affecting Enzyme Speed 3) Enzyme Activation Cells regulate metabolism by regulating the presence and activity of enzymes Genes controlling protein synthesis and enzyme production can be turned on or off Phosphorylation can activate enzymes the addition of a phosphate molecule on the enzyme makes it active (requires kinase)

6 Factors Affecting Enzyme Speed 4) Enzyme Inhibition Occurs when the substrate is unable to bind to the active site of the enzyme (3 ways): a) Competitive Inhibition- both the product and substrate can bind to the active site sets up a competition. b) Non-Competitive Inhibition- the product binds to a different site (allosteric site) which causes a change of shape to the active site. c) Poisons permanently block the active site of an enzyme stopping activity
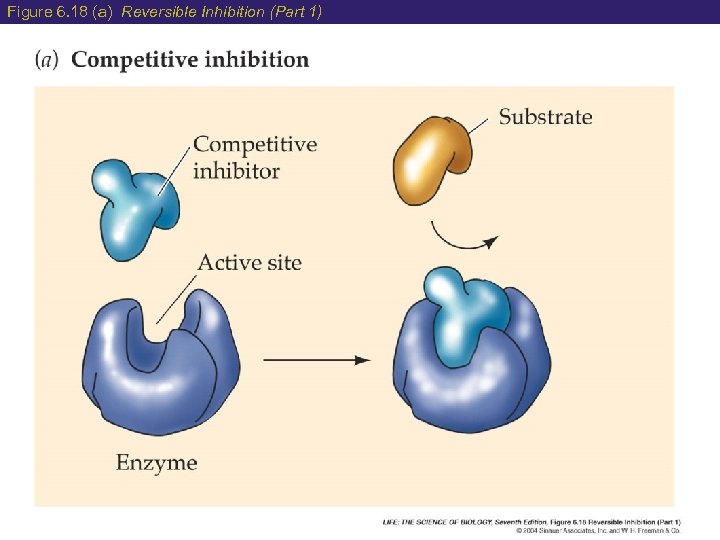
Figure 6. 18 (a) Reversible Inhibition (Part 1)
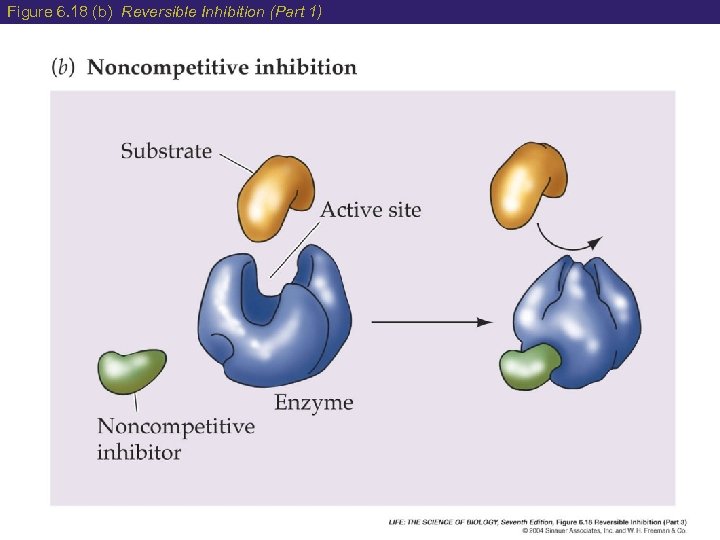
Figure 6. 18 (b) Reversible Inhibition (Part 1)

6 Factors Affecting Enzyme Speed 5) Enzyme Cofactors Many enzymes require a non-protein helper to function properly: a) Cofactors=Inorganic helpers • metals like copper, zinc or iron b) Coenzymes= Organic helpers • Vitamins • Note: deficiency in a vitamin results in a lack of coenzymes lack of enzymatic action *Nutrition is so important!*

6 You Did It! Questions?
ba0b1a0b5b2d00bfe2231c2158efef22.ppt