b12a787ce48a1656a4c85358414f6f9c.ppt
- Количество слайдов: 24

2 nd International Conference on Endocrinology October 20 -22, 2014, Chicago, USA DEVELOPING BRAIN AS AN ENDOCRINE ORGAN : A PARADOXIСAL REALITY M. V. Ugrumov Institute of Developmental Biology RAS, Moscow

2 nd International Conference on Endocrinology October 20 -22, 2014, Chicago, USA Developing brain as an endocrine organ: a paradoxical reality Endocrine functions of the brain in adulthood Current concept of neuroendocrine regulations in ontogenesis and contradictions. Revised concept of neuroendocrine regulations in ontogenesis. Summary and prospect
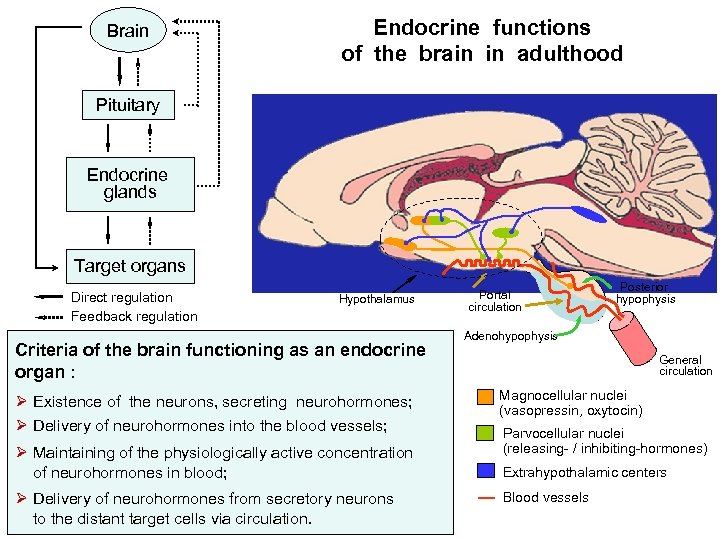
Brain Endocrine functions of the brain in adulthood Pituitary Endocrine glands Target organs Direct regulation Feedback regulation Hypothalamus Criteria of the brain functioning as an endocrine organ : Ø Existence of the neurons, secreting neurohormones; Ø Delivery of neurohormones into the blood vessels; Ø Maintaining of the physiologically active concentration of neurohormones in blood; Ø Delivery of neurohormones from secretory neurons to the distant target cells via circulation. Portal circulation Posterior hypophysis Adenohypophysis General circulation Magnocellular nuclei (vasopressin, oxytocin) Parvocellular nuclei (releasing- / inhibiting-hormones) Extrahypothalamic centers Blood vessels

2 nd International Conference on Endocrinology October 20 -22, 2014, Chicago, USA Developing brain as an endocrine organ: a paradoxical reality Endocrine functions of the brain in adulthood Current concept of neuroendocrine regulations in ontogenesis and contradictions Revised concept of neuroendocrine regulations in ontogenesis Summary and prospect
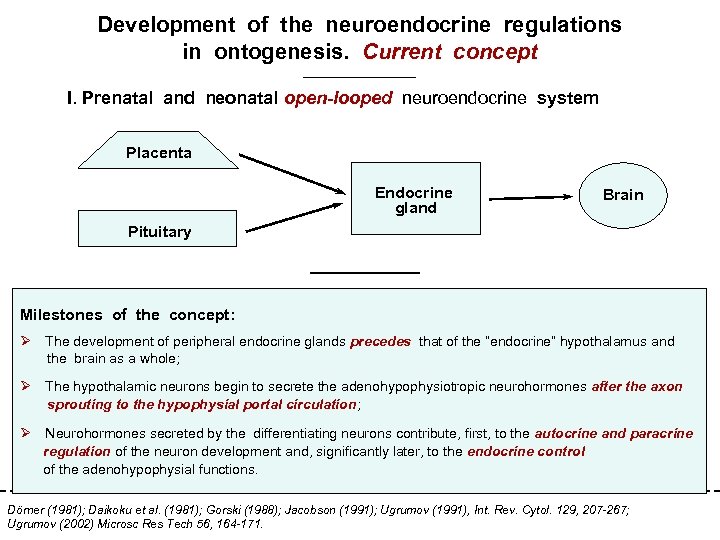
Development of the neuroendocrine regulations in ontogenesis. Current concept I. Prenatal and neonatal open-looped neuroendocrine system Placenta Endocrine gland Brain Pituitary II. Postnatal close-looped neuroendocrine system Milestones of the concept: Direct regulation Ø The development of peripheral endocrine glands precedes that of the “endocrine” hypothalamus and Brain Feedback regulation the brain as a whole; Ø The hypothalamic neurons begin to secrete the adenohypophysiotropic neurohormones after the axon sprouting to the hypophysial portal circulation; Ø Neurohormones secreted by the differentiating neurons contribute, first, to the autocrine and paracrine Endocrine glands regulation of the neuron development and, significantly later, Pituitary to the endocrine control of the adenohypophysial functions. Dörner (1981); Daikoku et al. (1981); Gorski (1988); Jacobson (1991); Ugrumov (1991), Int. Rev. Cytol. 129, 207 -267; Ugrumov (2002) Microsc Res Tech 56, 164 -171.
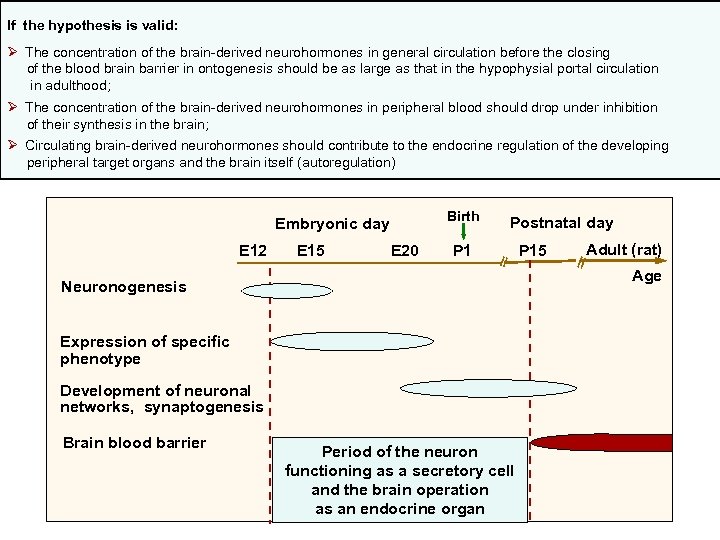
2010. 35, 837 -850. Neurochem. Res. 2010. 35, 837 -850. If the hypothesis is valid: Ø The concentration of the brain-derived neurohormones in general circulation before the closing of the blood brain barrier in ontogenesis should be as large as that in the hypophysial portal circulation in adulthood; Ø The concentration of the brain-derived neurohormones in peripheral blood should drop under inhibition of their synthesis in the brain; Ø Circulating brain-derived neurohormones should contribute to the endocrine regulation of the developing peripheral target organs and the brain itself (autoregulation) Birth E 15 E 20 P 1 Adult (rat) Age Neuronogenesis Expression of specific phenotype Development of neuronal networks, synaptogenesis Brain blood barrier P 15 ‗ E 12 Postnatal day ‗ Embryonic day Period of the neuron functioning as a secretory cell and the brain operation as an endocrine organ

2 nd International Conference on Endocrinology October 20 -22, 2014, Chicago, USA Developing brain as an endocrine organ: a paradoxical reality Endocrine functions of the brain in adulthood Current concept of neuroendocrine regulations in ontogenesis and contradictions Revised concept of neuroendocrine regulations in ontogenesis Summary and prospect
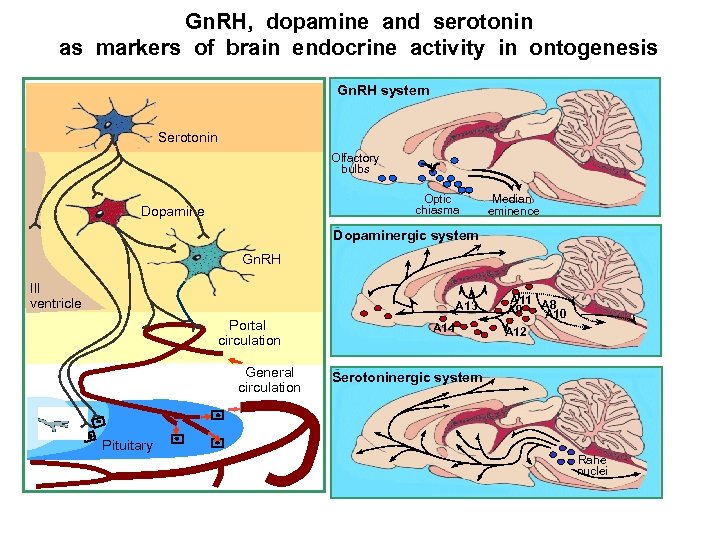
Gn. RH, dopamine and serotonin as markers of brain endocrine activity in ontogenesis Gn. RH system Serotonin Olfactory bulbs Optic chiasma Dopamine Median eminence Dopaminergic system Dopaminergic Gn. RH 3 ventricle III V ventricle system А 11 A 8 A 9 A 10 А 13 Portal circulation General circulation Olfactory A 14 Optic bulbs chiasma Median A 12 eminence 2 Serotoninergic system Pituitary Rahe nuclei

Whether the concentration of neurohormones in peripheral blood in ontogenesis before the blood brain barrier closing is sufficient to provide an endocrine control of the target organs ?
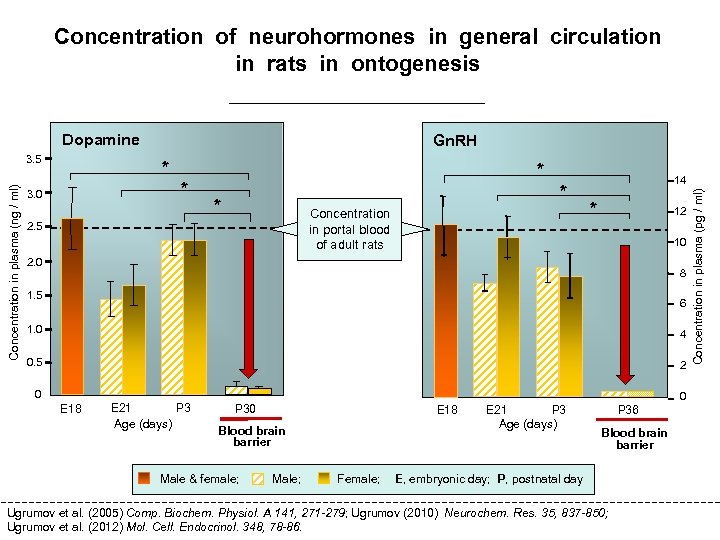
Concentration of neurohormones in general circulation in rats in ontogenesis Dopamine * * 3. 0 * * * Concentration in portal blood of adult rats 2. 5 14 * 12 10 2. 0 8 1. 5 6 1. 0 4 0. 5 2 0 0 E 18 E 21 P 3 Age (days) P 30 E 18 Blood brain barrier Male & female; Male; Female; P 3 E 21 Age (days) P 36 Blood brain barrier E, embryonic day; P, postnatal day Ugrumov et al. (2005) Comp. Biochem. Physiol. A 141, 271 -279; Ugrumov (2010) Neurochem. Res. 35, 837 -850; Ugrumov et al. (2012) Mol. Cell. Endocrinol. 348, 78 -86. Concentration in plasma (pg / ml) Concentration in plasma (ng / ml) 3. 5 Gn. RH

Whether the developing brain is a principal source of circulating neurohormones ?
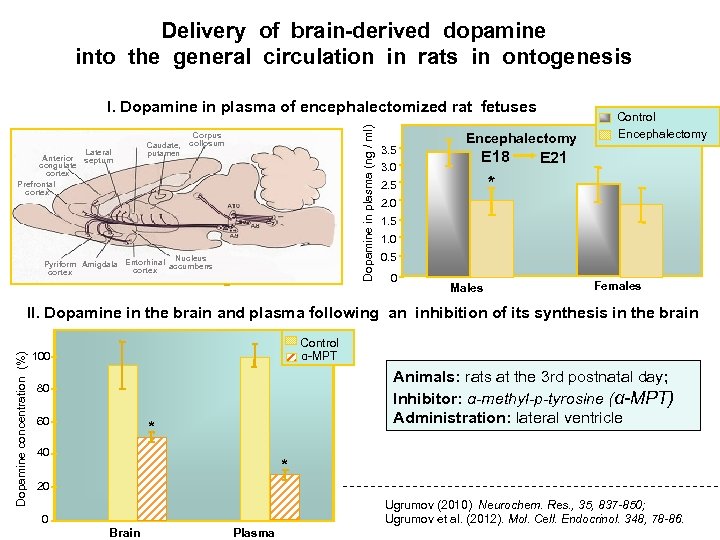
Delivery of brain-derived dopamine into the general circulation in rats in ontogenesis Anterior congulate cortex Olfactory Prefrontal bulb cortex Lateral septum Caudate, putamen Dopamine in plasma (ng / ml) I. Dopamine in plasma of encephalectomized rat fetuses Corpus collosum Pituitary Nucleus Retina Pyriform Amigdala Entorhinal accumbens A 17 Median cortex eminence 3. 5 3. 0 Encephalectomy E 18 Control Encephalectomy E 21 * 2. 5 2. 0 1. 5 1. 0 0. 5 0 Males Females Dopamine concentration (%) II. Dopamine in the brain and plasma following an inhibition of its synthesis in the brain Control α-МPТ 100 Animals: rats at the 3 rd postnatal day; Inhibitor: α-methyl-p-tyrosine (α-МPТ) Administration: lateral ventricle 80 60 * 40 * 20 Ugrumov (2010) Neurochem. Res. , 35, 837 -850; Ugrumov et al. (2012). Mol. Cell. Endocrinol. 348, 78 -86. 0 Brain Plasma
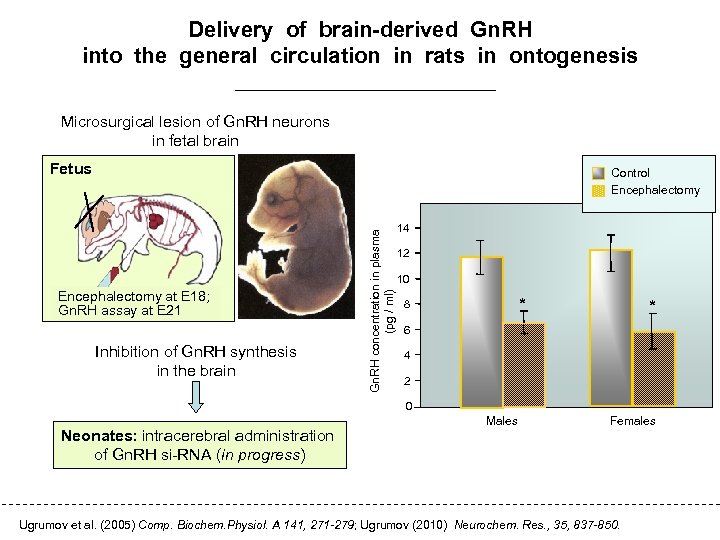
Delivery of brain-derived Gn. RH into the general circulation in rats in ontogenesis Microsurgical lesion of Gn. RH neurons in fetal brain Fetus Encephalectomy at E 18; Gn. RH assay at E 21 Inhibition of Gn. RH synthesis in the brain Gn. RH concentration in plasma (pg / ml) Control Encephalectomy 14 12 10 * 8 * 6 4 2 0 Neonates: intracerebral administration of Gn. RH si-RNA (in progress) Males Females Ugrumov et al. (2005) Comp. Biochem. Physiol. A 141, 271 -279; Ugrumov (2010) Neurochem. Res. , 35, 837 -850.
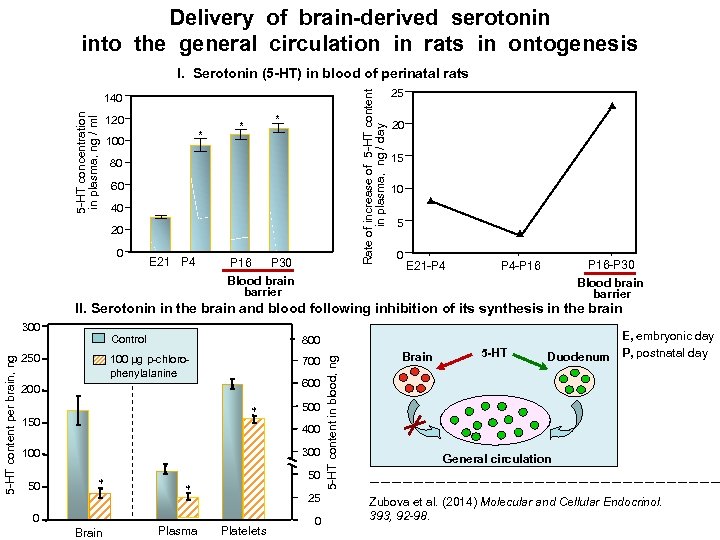
Delivery of brain-derived serotonin into the general circulation in rats in ontogenesis Rate of increase of 5 -HT content in plasma, ng / day I. Serotonin (5 -HT) in blood of perinatal rats 5 -HT concentration in plasma, ng / ml 140 120 * 100 * * 80 60 40 20 0 E 21 P 4 P 16 P 30 Blood brain barrier 25 20 15 10 5 0 E 21 -P 4 P 16 -P 30 Blood brain barrier P 4 -P 16 II. Serotonin in the brain and blood following inhibition of its synthesis in the brain Control 100 µg p-chlorophenylalanine 250 800 700 600 200 * 150 400 300 100 50 500 * 0 Brain 50 * Plasma 25 Platelets 0 5 -HT content in blood, ng 5 -HT content per brain, ng 300 Brain 5 -HT E, embryonic day Duodenum P, postnatal day General circulation Zubova et al. (2014) Molecular and Cellular Endocrinol. 393, 92 -98.

Whether the brain-derived neurohormones provide a direct endocrine action on peripheral targets and the brain itself ?
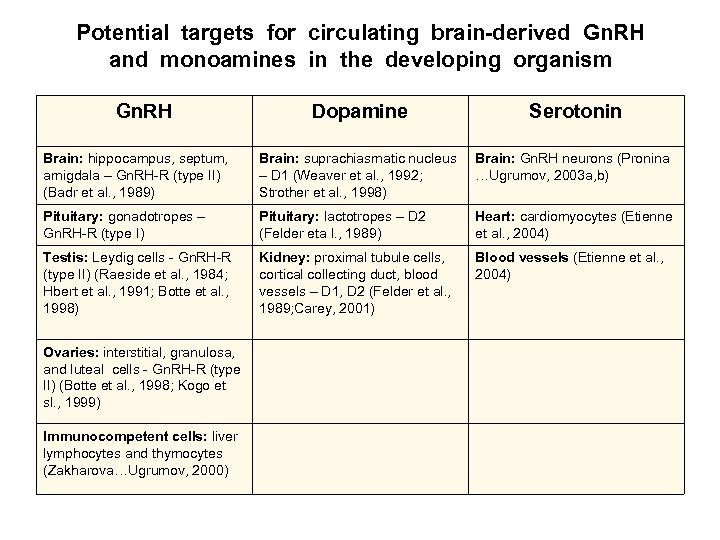
Potential targets for circulating brain-derived Gn. RH and monoamines in the developing organism Gn. RH Dopamine Serotonin Brain: hippocampus, septum, amigdala – Gn. RH-R (type II) (Badr et al. , 1989) Brain: suprachiasmatic nucleus – D 1 (Weaver et al. , 1992; Strother et al. , 1998) Brain: Gn. RH neurons (Pronina …Ugrumov, 2003 a, b) Pituitary: gonadotropes – Gn. RH-R (type I) Pituitary: lactotropes – D 2 (Felder eta l. , 1989) Heart: cardiomyocytes (Etienne et al. , 2004) Testis: Leydig cells - Gn. RH-R (type II) (Raeside et al. , 1984; Hbert et al. , 1991; Botte et al. , 1998) Kidney: proximal tubule cells, cortical collecting duct, blood vessels – D 1, D 2 (Felder et al. , 1989; Carey, 2001) Blood vessels (Etienne et al. , 2004) Ovaries: interstitial, granulosa, and luteal cells - Gn. RH-R (type II) (Botte et al. , 1998; Kogo et sl. , 1999) Immunocompetent cells: liver lymphocytes and thymocytes (Zakharova…Ugrumov, 2000)
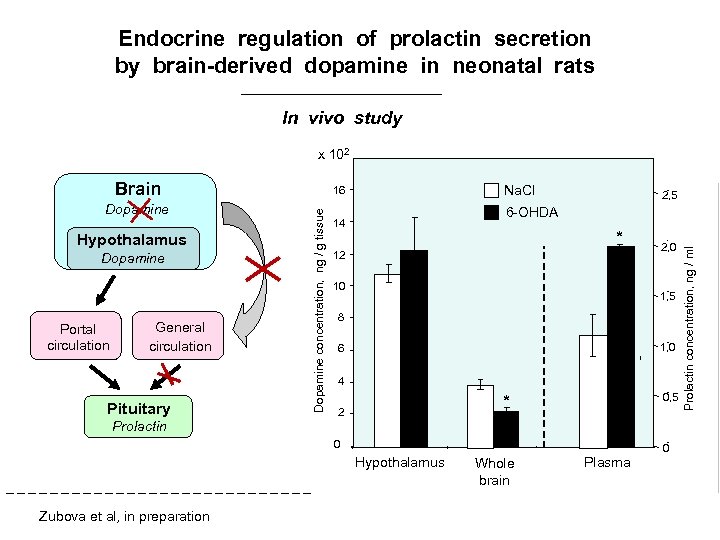
Endocrine regulation of prolactin secretion by brain-derived dopamine in neonatal rats In vivo study x 102 Hypothalamus Dopamine Portal circulation General circulation Pituitary Prolactin Dopamine concentration, ng / g tissue Dopamine Na. Cl 16 2, 5 6 -OHDA 14 * 12 10 1, 5 8 1, 0 6 4 0, 5 * 2 0 0 Hypothalamus Zubova et al, in preparation 2, 0 Whole brain Plasma Prolactin concentration, ng / ml Brain
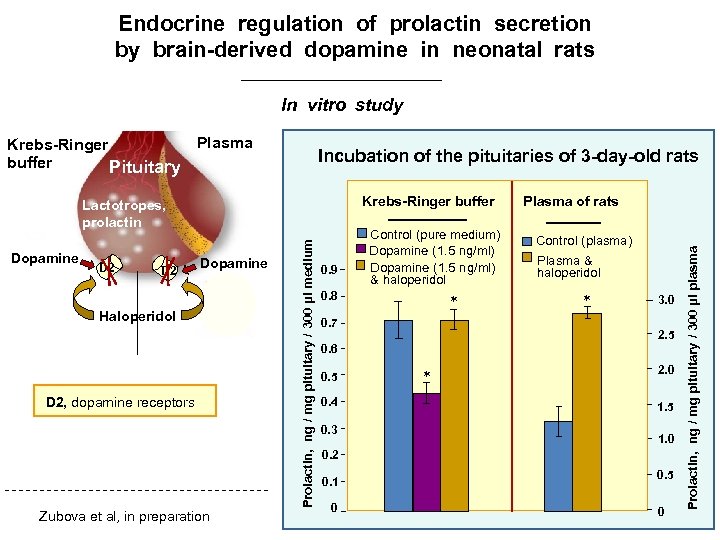
Endocrine regulation of prolactin secretion by brain-derived dopamine in neonatal rats In vitro study Plasma Incubation of the pituitaries of 3 -day-old rats D 2 Krebs-Ringer buffer Dopamine D 2 Dopamine Haloperidol D 2, dopamine receptors Zubova et al, in preparation Prolactin, ng / mg pituitary / 300 μl medium Lactotropes, prolactin 0. 9 Control (pure medium) Dopamine (1. 5 ng/ml) & haloperidol 0. 8 * 0. 7 Plasma of rats Control (plasma) Plasma & haloperidol * 3. 0 2. 5 0. 6 0. 5 0. 4 0. 3 * 2. 0 1. 5 1. 0 0. 2 0. 1 0 0. 5 0 Prolactin, ng / mg pituitary / 300 μl plasma Krebs-Ringer buffer Pituitary
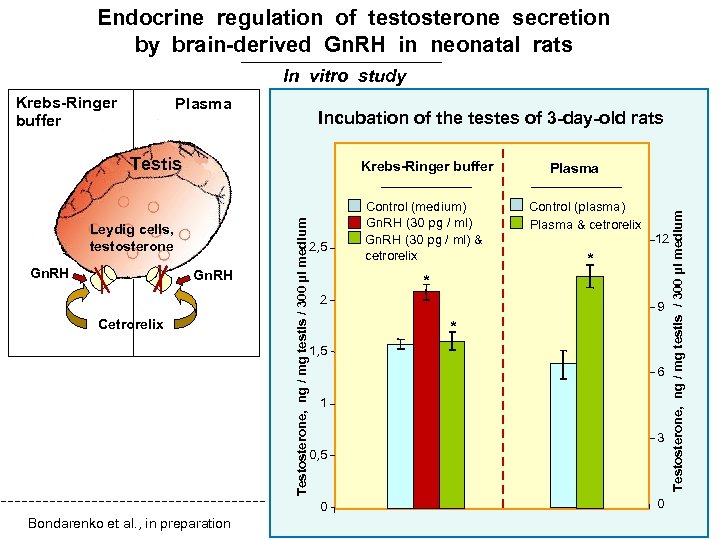
Endocrine regulation of testosterone secretion by brain-derived Gn. RH in neonatal rats In vitro study Plasma Incubation of the testes of 3 -day-old rats Testis Leydig cells, testosterone Gn. RH Cetrorelix Testosterone, ng / mg testis / 300 µl medium Krebs-Ringer buffer 2, 5 Control (plasma) Plasma & cetrorelix 12 * * 2 9 * 1, 5 6 1 3 0, 5 0 Bondarenko et al. , in preparation Control (medium) Gn. RH (30 pg / ml) & cetrorelix Plasma 0 Testosterone, ng / mg testis / 300 µl medium Krebs-Ringer buffer
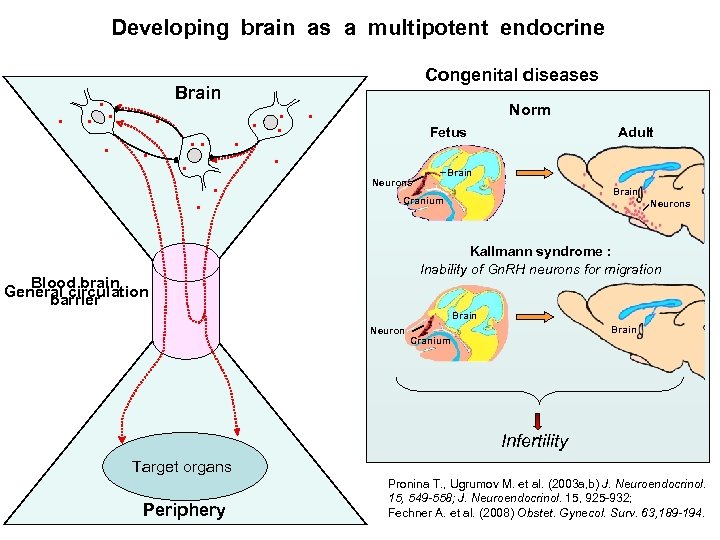
Developing brain as a multipotent endocrine Brain Congenital diseases Norm Fetus Adult Brain Neurons Brain Cranium Neurons Kallmann syndrome : Inability of Gn. RH neurons for migration Blood brain General circulation barrier Brain Neuron Brain Cranium Infertility Target organs Periphery Pronina T. , Ugrumov M. et al. (2003 a, b) J. Neuroendocrinol. 15, 549 -558; J. Neuroendocrinol. 15, 925 -932; Fechner A. et al. (2008) Obstet. Gynecol. Surv. 63, 189 -194.
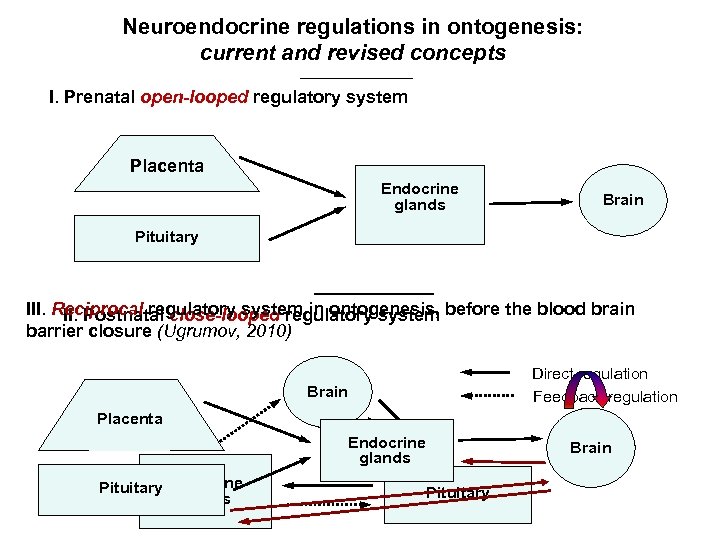
Neuroendocrine regulations in ontogenesis: current and revised concepts I. Prenatal open-looped regulatory system Placenta Endocrine glands Brain Pituitary III. Reciprocal regulatory system in ontogenesis, before the blood brain II. Postnatal close-looped regulatory system barrier closure (Ugrumov, 2010) Direct regulation Feedback regulation Brain Placenta Endocrine glands Pituitary Endocrine glands Brain Pituitary

2 nd International Conference on Endocrinology October 20 -22, 2014, Chicago, USA Developing brain as an endocrine organ: a paradoxical reality Endocrine functions of the brain in adulthood Current concept of neuroendocrine regulations in ontogenesis and contradictions Revised concept of neuroendocrine regulations in ontogenesis Summary and prospect

The concept on the role of the brain in neuroendocrine regulations in ontogenesis: in the past, at present and in the future In the past. The brain does not participate in neuroendocrine regulations up to its complete “maturation”, in rodents at prepuberty. At present. The developing brain provides the paraadenohypophysial endocrine regulations of peripheral target organs and the brain itself in ontogenesis, over a period from the neuron origin till the establishment of synaptic interneuronal relations and the blood brain barrier closing. In the future. It should be ascertain: Ø Whether the brain-derived neurohormones contribute to the paradenohypophysial endocrine regulation either of functioning or morphogenesis of the target organs; Ø What is the functional significance of a temporal combination of paracrine and endocrine regulations of the target cells by the same neurohormones; Ø What is a role of impairing metabolism of the brain-derived neurohormones in the development of congenital diseases

Laboratory of Hormonal Regulations Yu. Saifityarova (Ph. D student) A. Sapronova (Ph. D), E. Volina (Ph. D) Thanks for your attention T. Pronina (Ph. D) L. Dilmuchametova (Ph. D student) V. Khaindrava (Ph. D) E. Kozina (Ph. D student) E. Degtyareva (Ph. D student) Michael V. UGRUMOV e-mail: Chakimova michael. ugrumov@mail. ru; G. (Ph. D student) Tel. +7 499 135 88 42 V. Kirillova (technician)
b12a787ce48a1656a4c85358414f6f9c.ppt