eee50c30a482b5cf25dcb477bc5fc09e.ppt
- Количество слайдов: 147

1 VACCINE PREVENTABLE DISEASES AND VACCINATION Najwa Khuri-Bulos 2012

Jenner
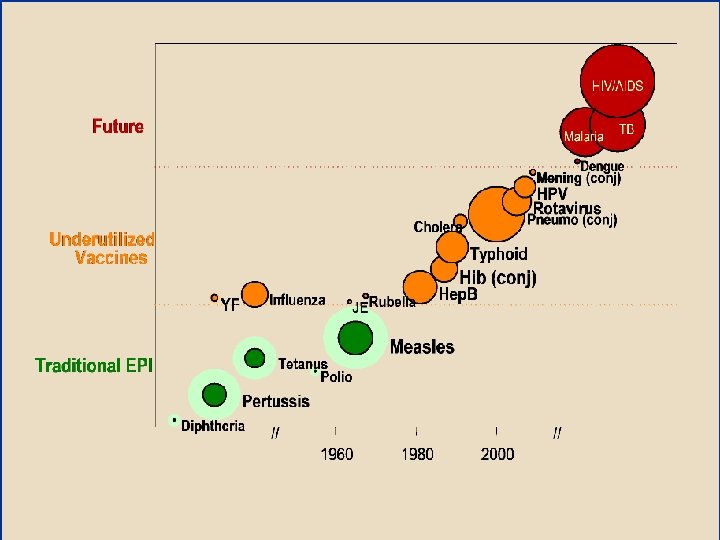
Strong Tools Available/Expected
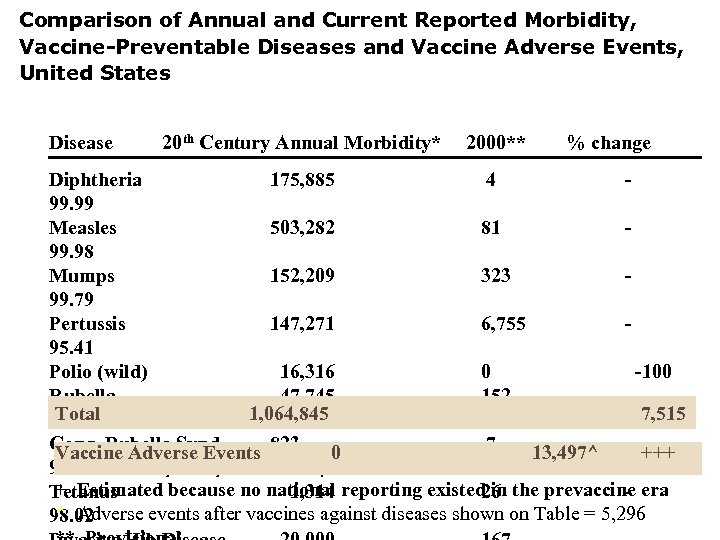
Comparison of Annual and Current Reported Morbidity, Vaccine-Preventable Diseases and Vaccine Adverse Events, United States Disease 20 th Century Annual Morbidity* 2000** % change Diphtheria 175, 885 4 99. 99 Measles 503, 282 81 99. 98 Mumps 152, 209 323 99. 79 Pertussis 147, 271 6, 755 95. 41 Polio (wild) 16, 316 0 -100 Rubella 47, 745 152 Total 1, 064, 845 7, 515 99. 68 Cong. Rubella Synd. 7 Vaccine Adverse Events 823 0 13, 497^ - +++ * Maximum cases reported in pre-vaccine era and year 99. 15 + Estimated because no national reporting existed in the prevaccine era Tetanus 1, 314 26 ^ Adverse events after vaccines against diseases shown on Table = 5, 296 98. 02
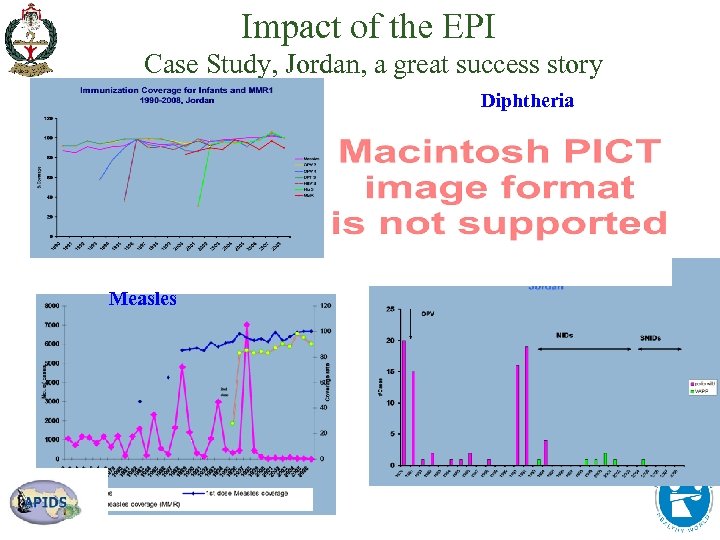
Impact of the EPI Case Study, Jordan, a great success story Diphtheria Measles poliomyelitis

outline 6 Importance of vaccines and vaccinations Basics of vaccination. General principles Routine vaccines in Jordan Recently introduced vaccines world wide Important vaccine information sources

Basics of vaccination 7 Active versus passive immunization Live versus killed vaccines Component vaccines, protein versus polysacharide General rules about minimum age at vaccination General rules about concommitent use of vaccines General rules about vaccine interruption General rules about different vaccine schedules

Principles of Vaccination Active Immunity = Antigen long duration, maybe lifelong Passive Immunity = Antibody limited public health impact, immune compromised hosts, limited duration

Types of vaccine antigens 9 Live attenuated organisms Viral Bacterial Recently parasitic (Malaria) Inactivated Whole organisms Viral bacterial Fractional Protein Polysaccharides Conjugate polysacharide vaccines

Inactivated vaccines general principles 10 Inactivated whole organism and fractional vaccines Safe in immune compromised hosts and pregnancy Protein, better antigens, TH 2 response, effective before age two years Polysacharide vaccines, Not effective before 2 years of age, TH 1 response In general inactivated by freezing Usually do not interfere with each other Need for booster doses to maintain immunity

Polysaccharide versus conjugate vaccines Polysaccharide Vaccines: Pneumococcal Conjugate Vaccines 1: T-cell independent T-cell dependent Not effective in children under 2 years and in the immunocompromised Induce an effective immune response in infants Stimulate an initial response involving both Ig. M and Ig. G while subsequent doses stimulate a primarily Ig. G response Induce immunological memory and can be boosted Result in a primarily Ig. M response Do not induce immunological memory and hence cannot be boosted Do not reduce mucosal carriage of pneumococcus 1 1 Eskola J. PIDJ 2000. 19 (4): 388 -93.

Live vaccines general considerations 12 Live vaccines can be frozen induce longer lasting immunity SHOULD NOT be given to immune compromised and to pregnant women maternal antibody may neutralize the vaccine and make it less effective in early life

Common Live vaccines 13 BCG OPV MMR Rotavirus Chickenpox Intranasal influenza vaccine Live typhoid vaccines

Killed or fractional vaccines 14 IPV Hepatitis a vaccine DTP DTa. P IPV HB HIB Pneumoccal vaccine Meningococcal vaccines Injectable influenza HPV

15 Vaccines in use for children in Jordan and worldwide Vaccines in use in Jordan Diptheria Tetanus Pertussis Polio both IPV and OPV Measles, Mumps Rubella Hemophilus influenza b Hepatitis b BCG Vaccines not yet introduced in Jordan Chickenpox Pneumococcus ( recent donation) Chickenpox/VZV vaccine Rotavirus Hepatitis a Influenza vaccine Acellular pertussis vaccine for adolescents and adults Meningococcal vaccine HPV vaccine

History of Expanded program on Immunization (EPI) – Jordan 16 1979 National Program on Immunization was established Diphtheria, Pertussis, Tetanus, Polio, BCG 1982 First dose Measles 1982 Tetanus toxoid for women in child bearing age 1993 4 th dose of polio 1995 Hepatitis “B” and second dose Measles 2000 MMR replaced 2 nd dose of Measles 2001 HIB introduced 2003 IPV first dose instead of OPV 2006 IPV second dose Plus OPV
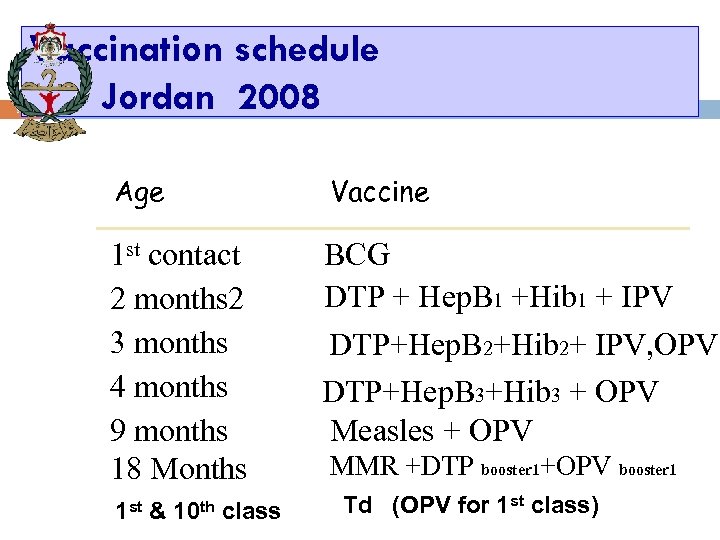
17 Vaccination schedule Jordan 2008 Age 1 st contact 2 months 2 3 months 4 months 9 months 18 Months 1 st & 10 th class Vaccine BCG DTP + Hep. B 1 +Hib 1 + IPV DTP+Hep. B 2+Hib 2+ IPV, OPV DTP+Hep. B 3+Hib 3 + OPV Measles + OPV MMR +DTP booster 1+OPV booster 1 Td (OPV for 1 st class)

18 Vaccination to school age children 1 st elementary class Td +OPV booster 2 Validation of measles and MMR 10 th class Td Second dose of MMR

Vaccine to be discussed 19 Diphtheria Tetanus Pertussis Polio Measles Mumps Rubella HIB HB Hep A Chickenpox Pneumococcal Rotavirus HPV Influenza

Diphtheria 20 Greek diphtheria (leather hide) Gram positive rod, a human pathogen that is transmitted by droplets, both asymptomatic and symptomatic individuals may transmit infection There are four biotypes mitis, intermedius, belfanti, and gravis). All biotypes of C diphtheriae may be either toxigenic or nontoxigenic. Diphtheria is caused by toxigenic strains of Corynebacterium diphtheriae.

Epidemiology of diphtheria 21 Only human pathogen Can be carried in the nasopharynx Bacteriophage induces toxin production Transmitted by droplet Vaccinated individuals can carry the organism but do not get sick

Diphtheria pathogenesis 22 Toxigenic strains express an exotoxin that consists of an enzymatically active A domain and a binding B domain, which promotes the entry of A into the cell. The toxin gene, tox, is carried by a family of related corynebacteria bacteriophages. The toxin inhibits protein synthesis in all cells, including myocardial, renal, and peripheral nerve cells, resulting in myocarditis, acute tubular necrosis, and delayed peripheral nerve conduction. Nontoxigenic strains of C diphtheriae can cause sore throat and other invasive infections.

Diptheria clinical considerations 23 Usually sick looking and tonsils have a membrane which is grayish and difficult to remove Membrane may involve more than the tonsils Toxin production most important and initially may have difficulty in breathing Arrhythmia with cardiac toxicity occurs in the second week Most common cause of death is cardiac toxicity Neurologic complications occur after three to four weeks Antitoxin should be given before four days

Diphtheria vaccine, toxin only 24 Formalin-inactivated diphtheria toxin Protein antigen Must administer by deep IM Do not freeze Efficacy Approximately 95% Duration Approximately 10 years Amount of antigen higher in children Should be administered with tetanus toxoid as DTP. DTa. P, DT, Td, or Tdap

DTP, DTa. P, DT, and Td 25 Diphtheria DTP, DTa. P, DT 7 -8 Lf units Td, Tdap (adult) 2 -2. 5 Lf units Tetanus 5 -12. 5 Lf units DTP, DTa. P and pediatric DT used through age 6 years. Adult Td for persons 7 years and older. Tdap for persons 10 -18 years (Boostrix) or 11 -64 years (Adacel)

Diphtheria vaccine, schedule, same as Tetanus and pertussis 26 Three doses before the age of one year starting at the age of 2 months Each to be given one to two months apart Repeat booster dose at 18 months Repeat booster dose at 4 -6 years Every ten years thereafter Give only small d after the age of 6 years of life

Diphtheria and Tetanus Toxoids Adverse Reactions and contraindications 27 Reactions Local reactions (erythema, induration) Exaggerated local reactions (Arthus-type) Fever and systemic symptoms not common Contraindications Severe allergic reaction to vaccine component or following a prior dose Moderate or severe acute illness

Tetanus 28 First described by Hippocrates Cl tetani Anaerobic gram-positive, spore-forming bacteria, Spores found in the environment, soil, animal feces; may persist for months to years Multiple toxins produced with growth of bacteria Tetanospasmin estimated human lethal dose = 2. 5 ng/kg It is not contagious to others Clostridium tetani is present in the environment

Pathogenesis of tetanus 29 Anaerobic conditions allow germination of spores but there is little inflammation at the site of infection The vegetative form of C tetani produces a potent plasmid-encoded exotoxin (tetanospasmin) This binds to gangliosides at the myoneural junction of skeletal muscle and on neuronal membranes in the spinal cord, blocking inhibitory impulses to motor neurons. This Leads to unopposed muscle contraction and spasm. The movements mimic seizures

Tetanus Clinical Features 30 Incubation period; 8 days (range, 3 -21 days) The shorter the incubation period the more severe is the disease Three clinical forms: local (uncommon), cephalic (rare), generalized (most common) Generalized tetanus: descending symptoms of trismus (lockjaw), difficulty swallowing, muscle rigidity, spasms Spasms continue for 3 -4 weeks; complete recovery may take months In the newborn only generalized tetanus occurs Maternal vaccination during pregnancy provides maternal antibodies that cross the placenta and help provide antitoxin antibodies to the new born infant

Tetanus Complications 31 The disease lasts for weeks Must be treated with antitoxin and antibiotics but contractions continue Laryngospasm Fractures Hypertension Nosocomial infections Pulmonary embolism, Aspiration pneumonia In the newborn often times the infants become malnourished and wasted

Tetanus vaccine (Tetanus Toxoid) 32 Formalin-inactivated tetanus toxin , Protein antigen Schedule Initially Three or four doses + booster Booster every 10 years Efficacy Duration Approximately 10 years Vaccine content same for children and adults Preferable administered with diphtheria toxoid as DTP, DTa. P, DT, Td, or Tdap Approximately 100%

post exposure wound prophylaxis Tetanus Wound Management 33 Clean, minor wounds All other wounds Vaccination History Td TIG Unknown or <3 doses Yes No Yes 3+ doses No* No No** No * Yes, if >10 years since last dose ** Yes, if >5 years since last dose Dose of TIG is 250 units regardless of age and weight

Pertussis 34 Highly contagious respiratory infection caused by Bordetella pertussis a fastidious gram negative bacterium, only humans Outbreaks first described in 16 th century Bordetella pertussis isolated in 1906 Estimated 285, 000 deaths worldwide in 2001 Recently increased in some parts Transmission Respiratory droplets Communicability Maximum in catarrhal stage Secondary attack rate up to 80%

Pertussis Pathogenesis 35 Attachment to cilia in respiratory tract leading to Local tissue damage in respiratory tract, Systemic disease may be toxin mediated, no bacteremia Antigenic and biologically active components: pertussis toxin (PT) filamentous hemagglutinin (FHA) agglutinogens adenylate cyclase pertactin tracheal cytotoxin NO Bacteremia

Pertussis Clinical Features 36 Incubation period 7 -10 days (range 4 -21 days) Catarrhal stage Paroxysmal cough stage Convalescence 1 -2 weeks 1 -6 weeks Weeks to months Fever usually minimal throughout course of illness unless complicated by superinfection In adults and older children Infection may be asymptomatic, or may present as classic pertussis, these serve as sources of infection to children
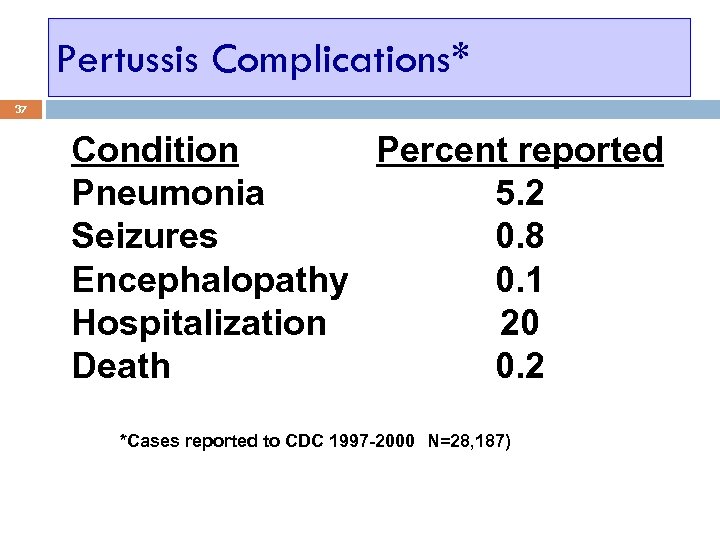
Pertussis Complications* 37 Condition Percent reported Pneumonia 5. 2 Seizures 0. 8 Encephalopathy 0. 1 Hospitalization 20 Death 0. 2 *Cases reported to CDC 1997 -2000 (N=28, 187)
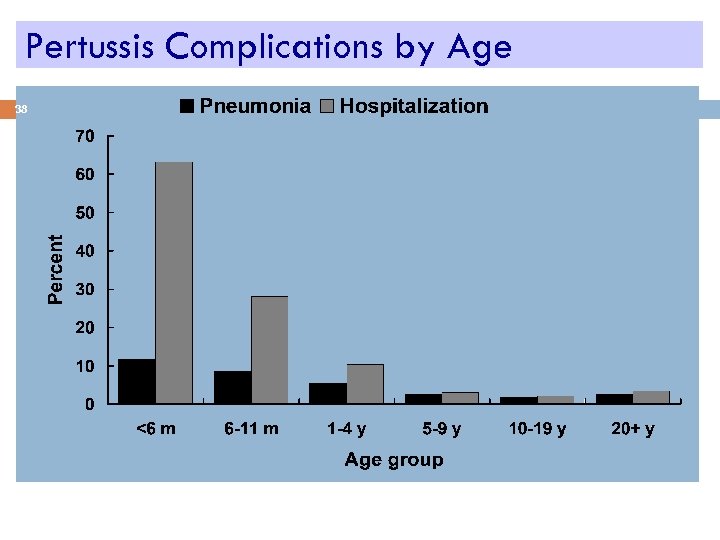
Pertussis Complications by Age 38 *Cases reported to CDC 1997 -2000 (N=28, 187)

Pertussis vaccines 39 Both are inactivated vaccines Whole cell (WC) or acellular pertussis (a. P) WC contraindicated after 6 years of age Immunity decreases with time and hence re vaccination in older individuals is needed but with acellular smaller doses only ONLY acellular P are allowed for use in adolescents and older individuals

Whole-Cell Pertussis Vaccine 40 Developed in mid-1930 s and combined as DTP in mid -1940 s 70%-90% efficacy after 3 doses Protection for 5 -10 years Fever Local reactions Seizures HHE Encephalopathy 40% 35% 1/1750 1/110, 000

Acellular Pertussis Vaccines 41 Local reactions (pain, redness, or swelling at the site of injection) Local reactions more common following 4 th and 5 th doses Reports of swelling of entire limb Extensive swelling after 4 th dose NOT a contraindication to 5 th dose Low-grade fever
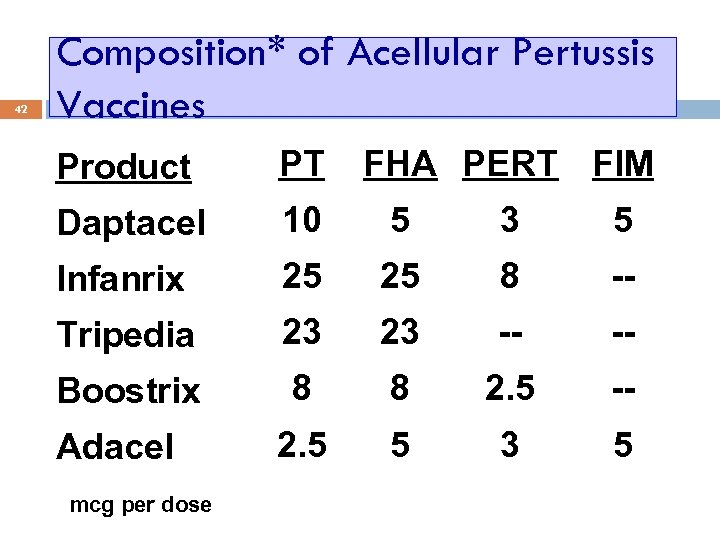
42 Composition* of Acellular Pertussis Vaccines Product PT Daptacel 10 5 3 5 Infanrix 25 25 8 -- Tripedia 23 23 -- -- Boostrix 8 8 2. 5 -- 2. 5 5 3 5 Adacel *mcg per dose FHA PERT FIM

DTP/DTa. P Contraindications 43 Severe allergic reaction to vaccine component or following a prior dose Encephalopathy not due to another identifiable cause occurring within 7 days after vaccination Progressive CNS disease

DTP/DTa. P Precautions* 44 Moderate or severe acute illness Temperature >105°F (40. 5°C) or higher within 48 hours with no other identifiable cause Collapse or shock-like state (hypotonic hyporesponsive episode) within 48 hours Persistent, inconsolable crying lasting >3 hours, occurring within 48 hours Convulsions with or without fever occurring within 3 days *may consider use in outbreaks

Poliomyelitis 45 First described by Michael Underwood in 1789 Enterovirus, Strict human pathogen Three serotypes: 1, 2, 3 Minimal heterotypic immunity between serotypes Rapidly inactivated by heat, formaldehyde, chlorine, ultraviolet light Entry into mouth/ Replication in pharynx, GI tract, local lymphatics Hematologic spread to lymphatics and central nervous system/Viral spread along nerve fibers Destruction of anterior horn cell (motor neurons) with resultant paralysis
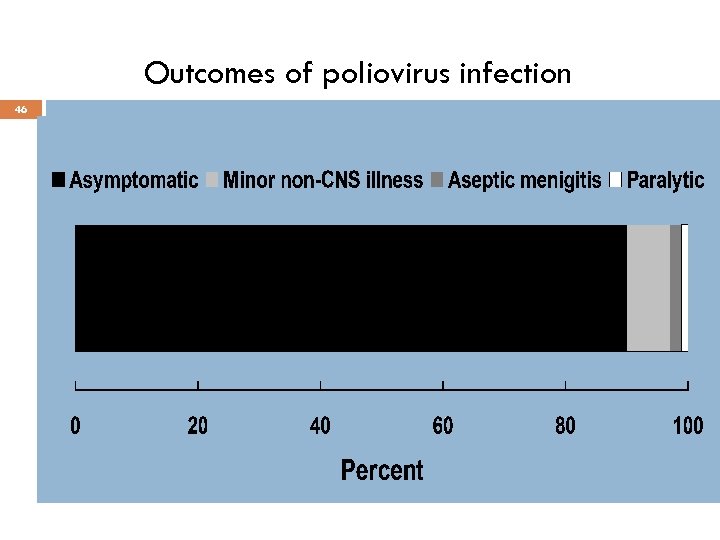
Outcomes of poliovirus infection 46
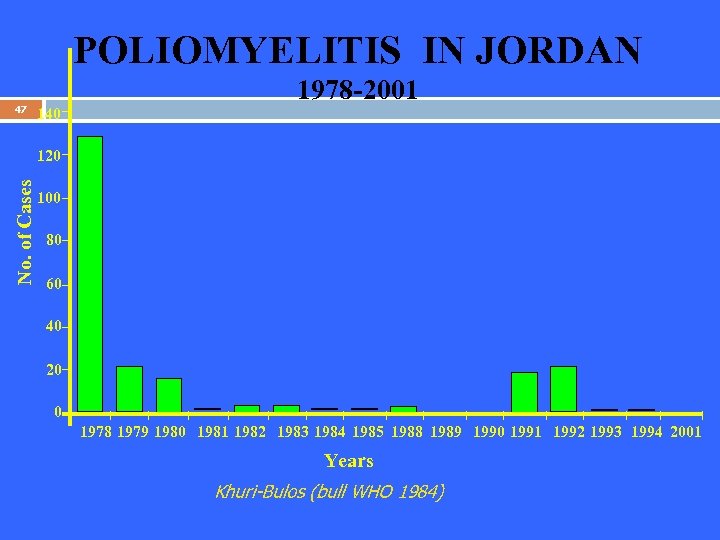
POLIOMYELITIS IN JORDAN 47 140 1978 -2001 No. of Cases 120 100 80 60 40 20 0 1978 1979 1980 1981 1982 1983 1984 1985 1988 1989 1990 1991 1992 1993 1994 2001 Years Khuri-Bulos (bull WHO 1984)
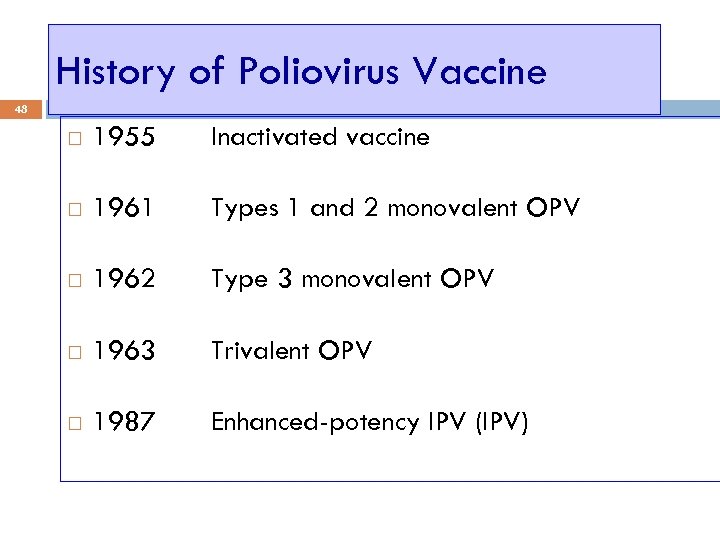
History of Poliovirus Vaccine 48 1955 Inactivated vaccine 1961 Types 1 and 2 monovalent OPV 1962 Type 3 monovalent OPV 1963 Trivalent OPV 1987 Enhanced-potency IPV (IPV)

Oral Polio Vaccine (Sabin) 49 Types 1, 2, 3 live vaccine Shed in stool for up to 6 weeks Shed for prolonged periods in immunecompromised individuals Highly effective in producing immunity to poliovirus 50% immune after 1 dose >95% immune after 3 doses Immunity probably lifelong Immunity less in developing countries? ? Other enteroviruses interfere with immunity Induces both local and systemic immunity
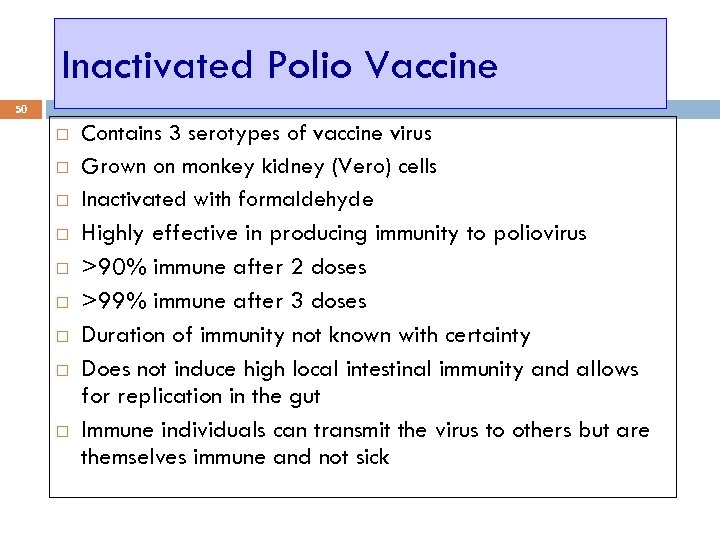
Inactivated Polio Vaccine 50 Contains 3 serotypes of vaccine virus Grown on monkey kidney (Vero) cells Inactivated with formaldehyde Highly effective in producing immunity to poliovirus >90% immune after 2 doses >99% immune after 3 doses Duration of immunity not known with certainty Does not induce high local intestinal immunity and allows for replication in the gut Immune individuals can transmit the virus to others but are themselves immune and not sick

Polio Vaccine Adverse Reactions 51 Rare local reactions (IPV) No serious reactions to IPV have been documented Vaccine associated Paralytic poliomyelitis VAPP ONLY with OPV More likely with the first Rate is 2. 5 million dose Increased risk in persons dose(80%) >18 years Increased risk in persons with immunodeficiency Most cases in healthy children and their household contacts

Polio Vaccine Contraindications and Precautions 52 Immune suppression Pregnancy prednisone in excess of 2 mg/kg/day Contacts of immunecompromised patients Severe allergic reaction to a vaccine component or following a prior dose of vaccine Moderate or severe acute illness

Measles 53 Highly contagious viral illness, infects humans only Almost always clinically apparent illness First described in 7 th century Paramyxovirus (RNA), one antigenic type Respiratory transmission of virus Persists in the air for one or more hours Near universal infection of childhood in prevaccination era Death risk increased if acquired <2 yrs and in malnourished children Communicability 4 days before to 4 days after rash onset

Measles Pathogenesis 54 Respiratory transmission of virus Replication in nasopharynx and regional lymph nodes Primary viremia 2 -3 days after exposure Secondary viremia 5 -7 days after exposure with spread to tissues
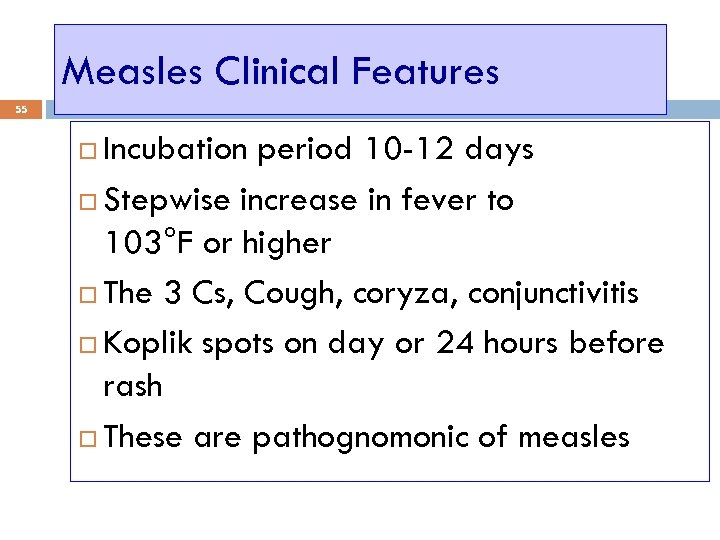
Measles Clinical Features 55 Incubation period 10 -12 days Stepwise increase in fever to 103°F or higher The 3 Cs, Cough, coryza, conjunctivitis Koplik spots on day or 24 hours before rash These are pathognomonic of measles

Measles Clinical Features 56 2 -4 days after prodrome, 14 days after exposure Maculopapular rash, becomes confluent Begins on face and head Persists 5 -6 days Fades in order of appearance
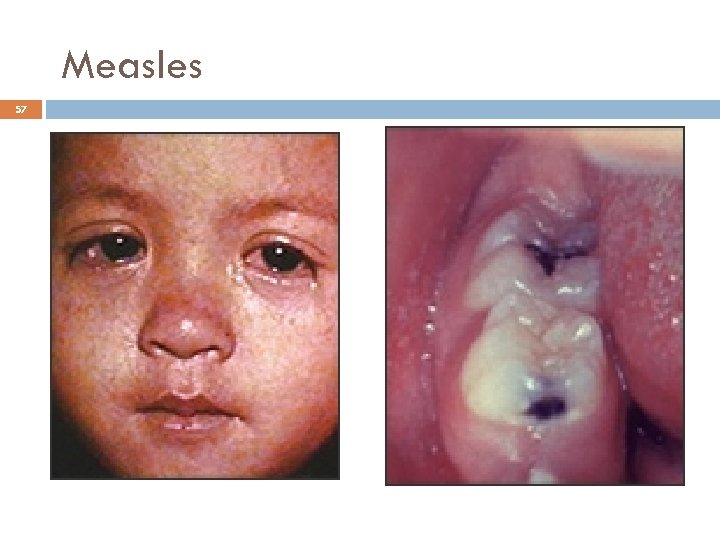
Measles 57
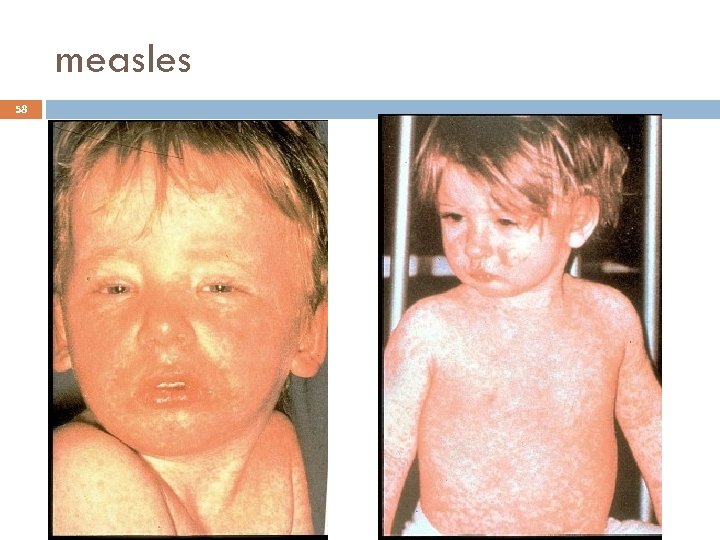
measles 58
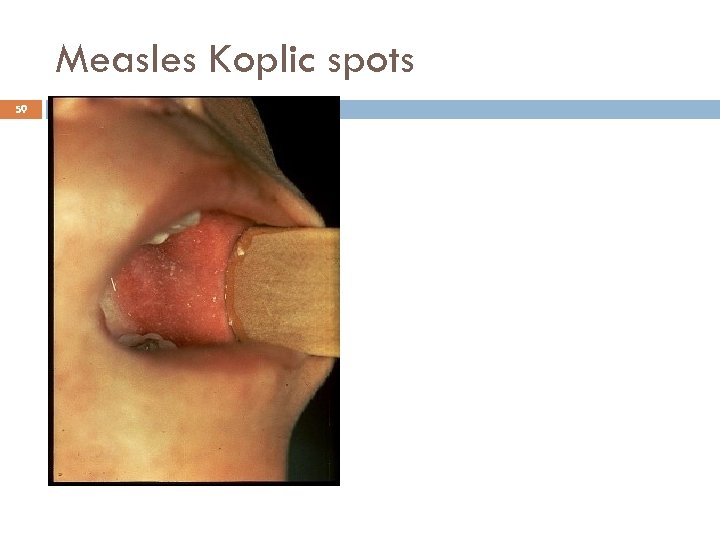
Measles Koplic spots 59

Measles Complications 60 Condition Diarrhea Otitis media Pneumonia Encephalitis Hospitalization Death Percent reported 8 7 6 0. 1 18 0. 2 Based on 1985 -1992 surveillance data Death rate higher in malnourished and very young children
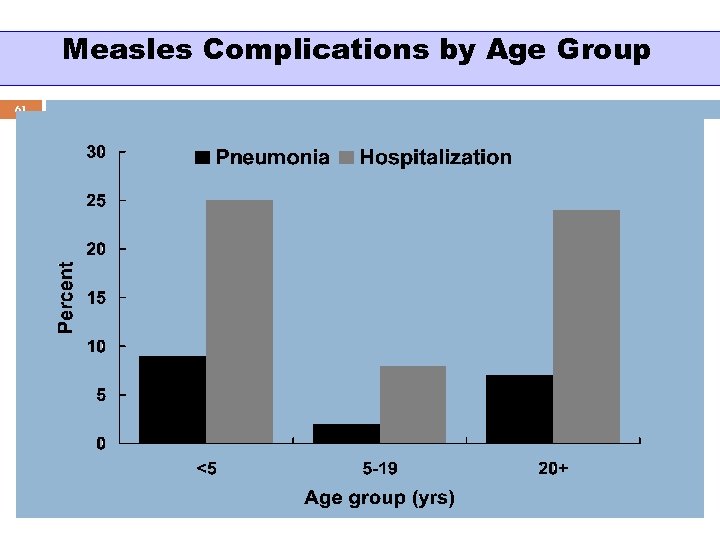
Measles Complications by Age Group 61

Measles Vaccines 62 1963 1965 1967 1968 1971 1989 2005 Live attenuated and killed vaccines Live further attenuated vaccine Killed vaccine withdrawn Live further attenuated vaccine (Edmonston-Enders strain) Licensure of combined measlesmumps-rubella vaccine Two dose schedule Licensure of MMRV

Measles Vaccine 63 Composition Live virus Efficacy 95% (range, 90%-98%) Duration of Immunity Lifelong Schedule 2 doses Should be administered with mumps and rubella as MMR to All infants >12 months of age Susceptible adolescents and adults without documented evidence of immunity should also receive the vaccine
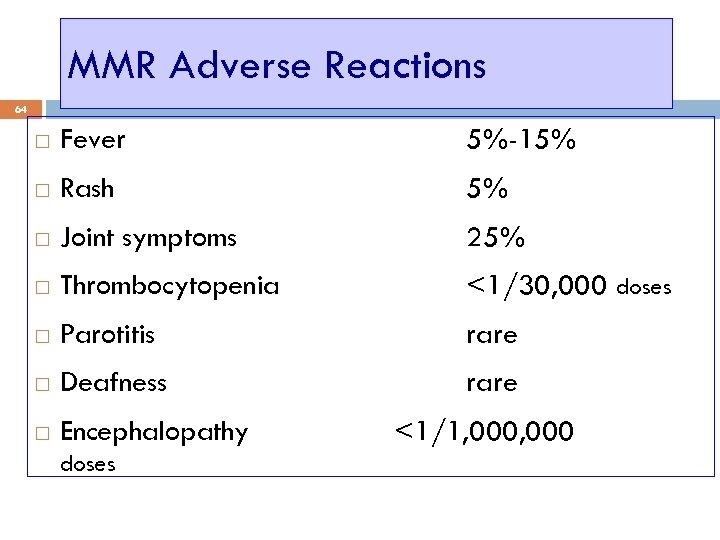
MMR Adverse Reactions 64 Fever 5%-15% Rash 5% Joint symptoms 25% Thrombocytopenia <1/30, 000 doses Parotitis rare Deafness rare Encephalopathy doses <1/1, 000

MMR Vaccine Contraindications and Precautions 65 Severe allergic reaction Pregnancy Immunosuppression Moderate or severe acute illness Studies have demonstrated safety of MMR in egg allergic children even though it is grown in chick embryo Vaccinate without testing, but with observation

Mumps 66 Acute viral illness, caused by mumps virus, a parmyxovirus, an RNA virus one serotype Parotitis and orchitis described by Hippocrates in 5 th century BCE Viral etiology described by Johnson and Goodpasture in 1934 Frequent cause of outbreaks among military personnel in prevaccine era

Mumps Epidemiology 67 Reservoir Transmission Temporal pattern Communicability Human Asymptomatic infections may transmit Respiratory drop nuclei Peak in late winter and spring Three days before to four days after Respiratory transmission of virus Replication in nasopharynx and regional lymph nodes Viremia 12 -25 days after exposure with spread to tissues Multiple tissues infected during viremia

Mumps Clinical Features 68 Incubation period 14 -18 days Nonspecific prodrome of myalgia, malaise, headache, low-grade fever Parotitis in 30%-40% Up to 20% of infections asymptomatic

Mumps Complications 69 CNS involvement Aseptic meningitis 15% of clinical cases Orchitis 20%-50% in post- pubertal males Pancreatitis 2%-5% Deafness 1/20, 000 Death Average 1 per year (1980 – 1999)
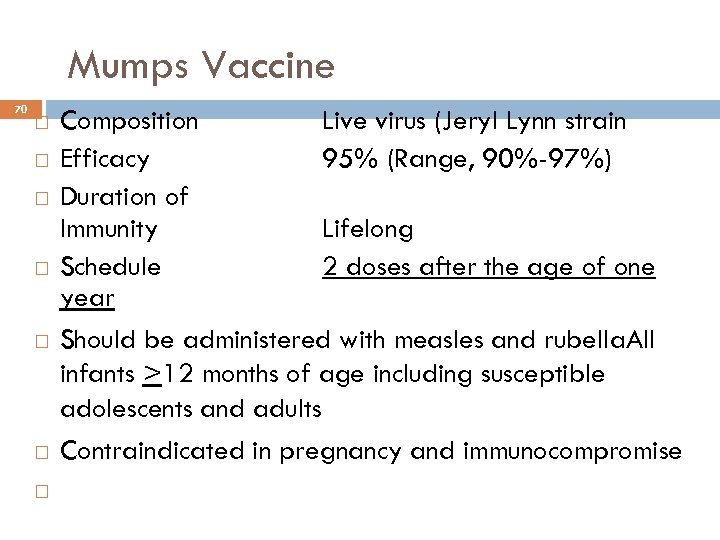
Mumps Vaccine 70 Composition Live virus (Jeryl Lynn strain Efficacy 95% (Range, 90%-97%) Duration of Immunity Lifelong Schedule 2 doses after the age of one year Should be administered with measles and rubella. All infants >12 months of age including susceptible adolescents and adults Contraindicated in pregnancy and immunocompromise

71 Rubella

Rubella 72 From Latin meaning "little red Togavirus RNA virus/one antigenic type Rapidly inactivated by chemical agents, ultraviolet light, low p. H, and heat. Respiratory transmission of virus Human infection only /transmitted by droplet, respiratory Replication in nasopharynx and regional lymph nodes Viremia 5 -7 days after exposure with spread to tissues Placenta and fetus infected during viremia Only primary infection leads to viremia and congenital infection Infection may be symptomatic which makes it mandatory that all mothers who are exposed should be tested. Best is prevention

Rubella Clinical Features/complications 73 Incubation period 14 days (range 12 -23 days) Prodrome of low-grade fever Maculopapular rash 14 -17 days after exposure Lymphadenopathy in second week Complications include arthralgia and arthritis in older females Thrombocytopenia Major complication is that of infection of the fetus This occurs only with primary infection This occurs in early pregnancy less than 16 weeks
rubella 74
75
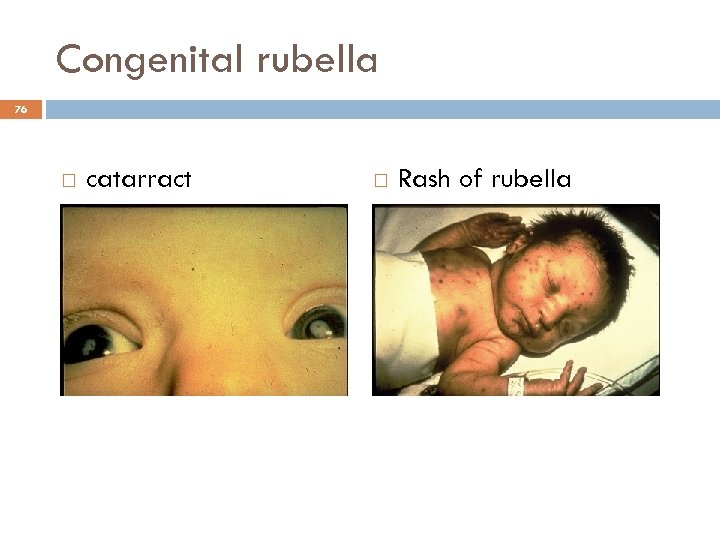
Congenital rubella 76 catarract Rash of rubella

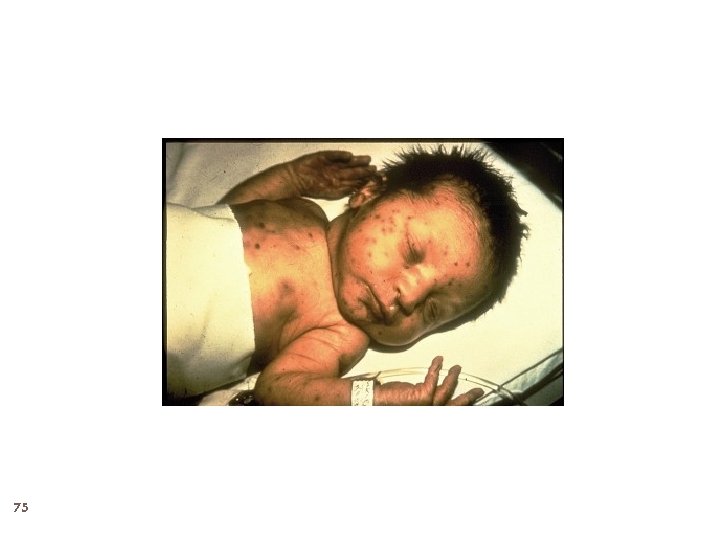
Congenital Rubella Syndrome 77 Occurs only with primary maternal infection and mainly in the first trimester 85% of fetuses Infection may affect all organs. May lead to fetal death or premature delivery Deafness Cataracts Heart defects Microcephaly Mental retardation Bone alterations Liver and spleen damage Severity of damage to fetus depends on gestational age

Rubella Vaccine 78 Composition Live virus (RA 27/3 strain) Efficacy 95% (Range, 90%-97%) Duration of. Immunity Lifelong Schedule >1 Dose Acute arthralgia in about 25% of susceptable adult women, Acute arthritis-like signs and symptoms occurs in about 10% Rare reports of chronic or persistent symptoms Population-based studies have not confirmed association

79 MMR Vaccine Contraindications and Precautions Severe allergic reaction to vaccine component or following a prior dose Pregnancy Immunosuppression Moderate or severe acute illness Recent blood product

Haemophilus influenzae 80 Aerobic gram-negativecoccobacillus Transmitted from the NP of humans only Polysaccharide capsule determines serotype and virulence, antibody to this is protective Six different serotypes (a-f) of polysaccharide capsule, 95% of invasive disease caused by type b Organism colonizes nasopharynx only about 4% Invasion of the blood stream leads to invasive disease, Antecedent upper respiratory tract infection may be a contributing factor
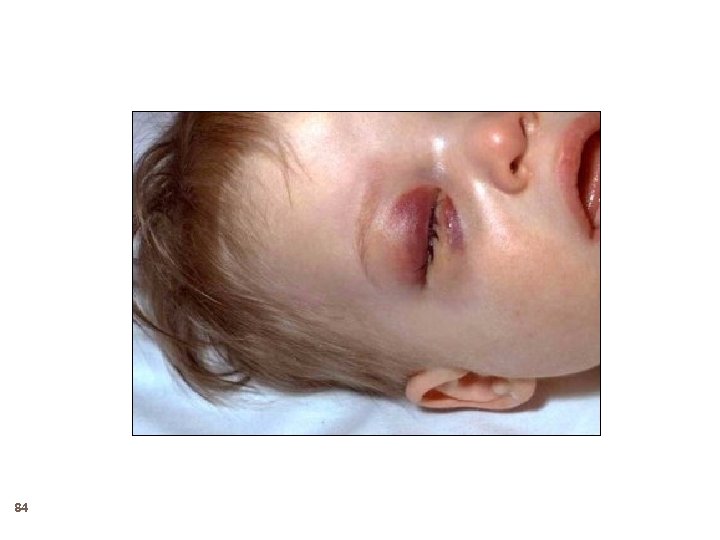
HIB 81
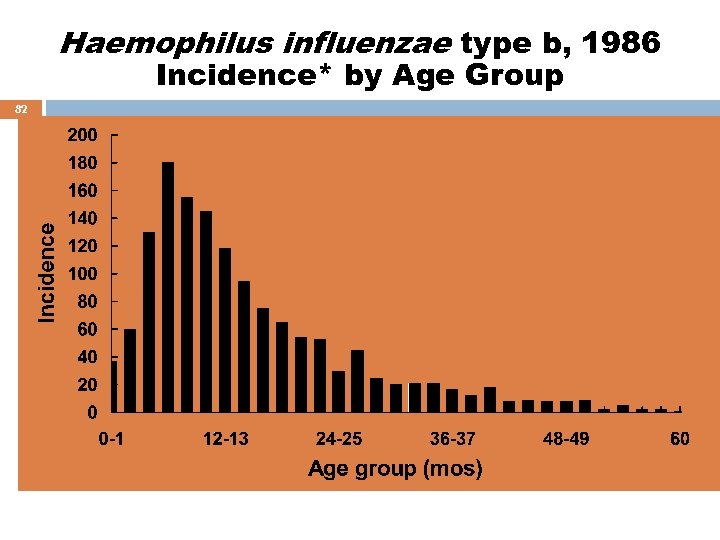
Haemophilus influenzae type b, 1986 Incidence* by Age Group 82 *Rate per 100, 000 population, prevaccine era
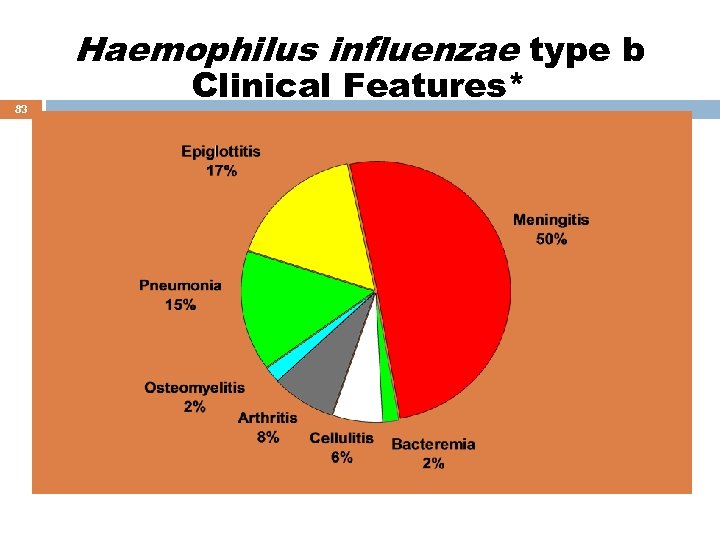
Haemophilus influenzae type b 83 Clinical Features* *prevaccination era
84
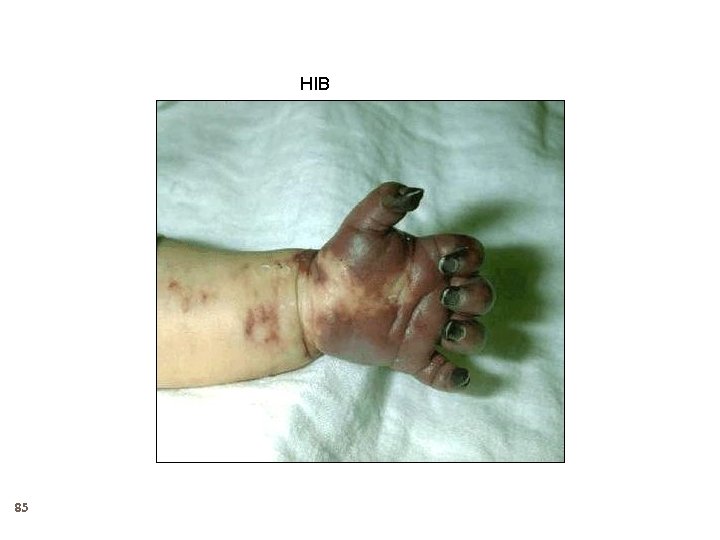
HIB 85
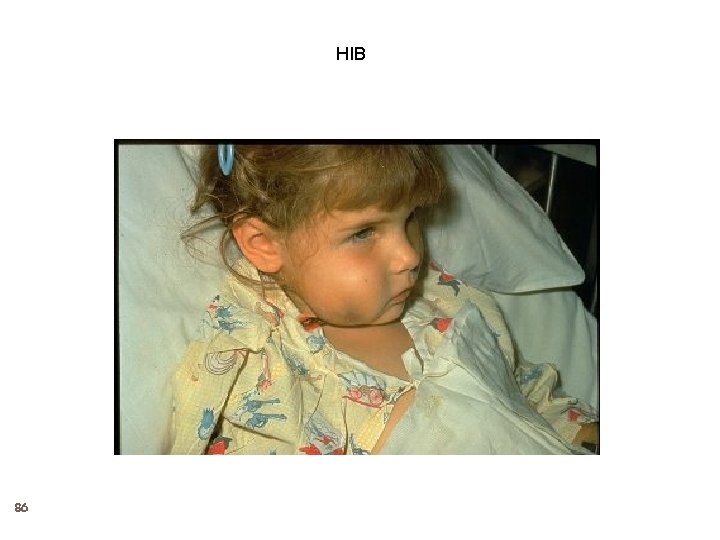
HIB 86

Haemophilus influenzae type b Meningitis 87 Accounted for approximately 50%-65% of cases in the prevaccine era Hearing impairment or neurologic sequelae in 15%30% Case-fatality rate 2%-5% despite of effective antimicrobial therapy Vaccine use almost eradicated the infection Vaccine use also protected the uninfected by decreasing the nasopharyngeal carraige

Haemophilus influenzae type b Conjugate Vaccines 88 Active ingredient made from polysacharide capsule Polysacharide Not effective before age of two years. Must conjugate with a protein to make effective Indicated after age of 6 weeks, NEVER before Give three doses one to two months apart Not indicated after age of 5 years unless immunecompromised Three types of vaccines available all are effective

Haemophilus influenzae type b Vaccine Routine Schedule Vaccine 4 mo Hb. OC x x PRP-T x x PRP-OMP 89 2 mo 6 mo 12 -18 mo x x x

Haemophilus influenzae type b Vaccine Adverse Reactions 90 Swelling, redness, or pain in 5%-30% of recipients Systemic reactions infrequent Serious adverse reactions rare Do not give if Severe allergic reaction to vaccine component or following a prior dose Moderate or severe acute illness Age <6 weeks
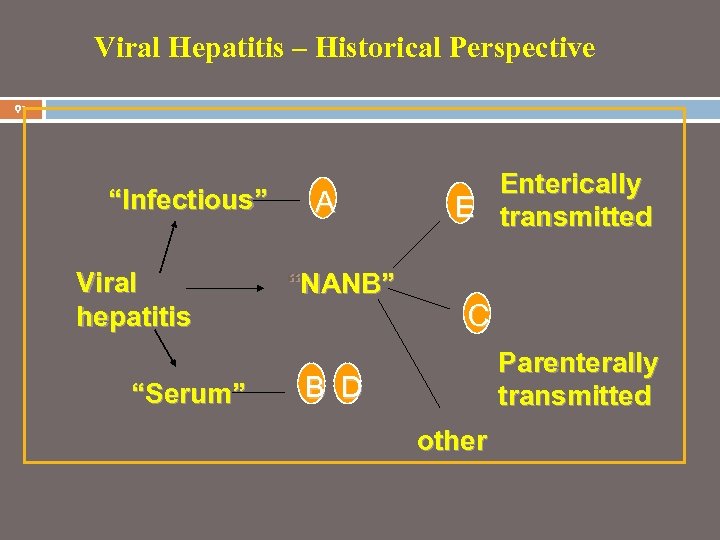
Viral Hepatitis – Historical Perspective 91 “Infectious” Viral hepatitis “Serum” A “NANB” Enterically E transmitted C Parenterally transmitted B D other

Hepatitis vaccines 92 Hepatitis a Hepatitis b No vaccines for the other viruses C, D, E
Hepatitis Cannot distinguish On clinical grounds alone 93
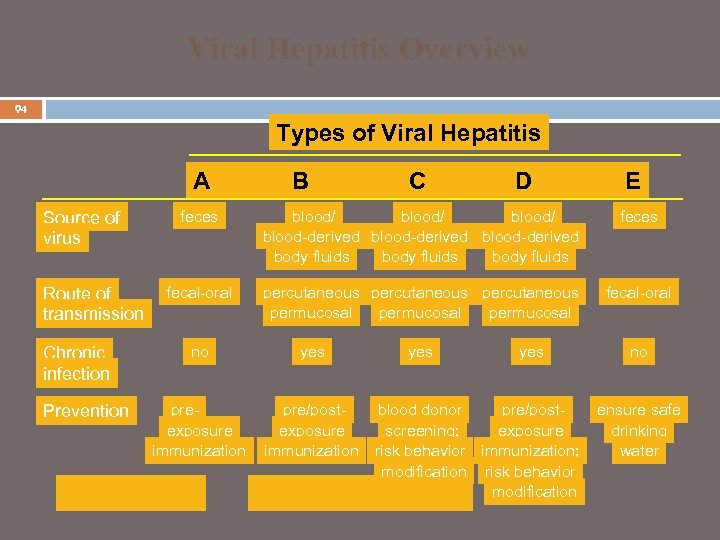
Viral Hepatitis Overview 94 Types of Viral Hepatitis A Source of virus Route of transmission Chronic infection Prevention B C D E feces blood/ blood-derived body fluids feces fecal-oral percutaneous permucosal fecal-oral no yes preexposure immunization pre/postexposure immunization yes blood donor pre/postscreening; exposure risk behavior immunization; modification risk behavior modification no ensure safe drinking water
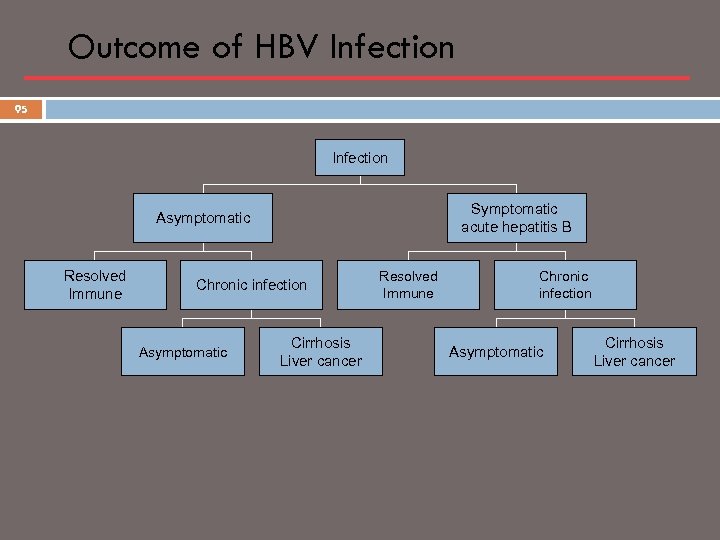
Outcome of HBV Infection 95 Infection Symptomatic acute hepatitis B Asymptomatic Resolved Immune Chronic infection Asymptomatic Cirrhosis Liver cancer
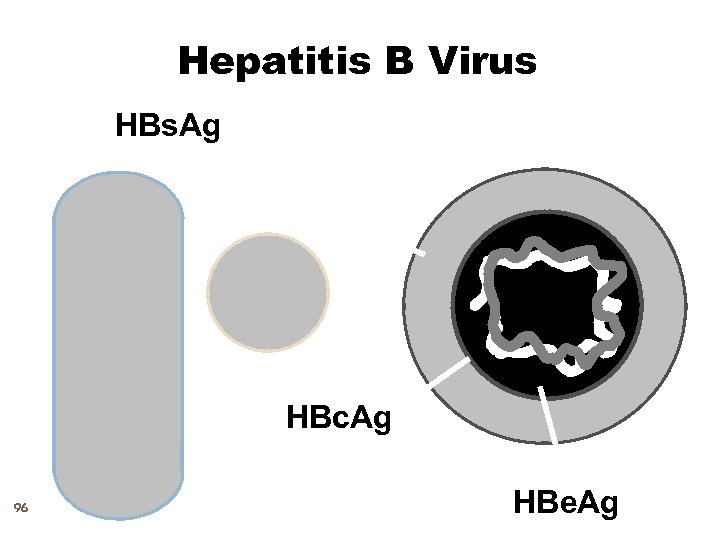
Hepatitis B Virus HBs. Ag HBc. Ag 96 HBe. Ag

Hepatitis B Virus 97 Hepadnaviridae family (DNA) Numerous antigenic components Humans are only known host May retain infectivity for at least 1 month at room temperature Stable in the enviroment and can infect if microabrasions in the skin Contagious also by intimate contact and exposure to blood and all body secretions that may be contaminted with blood

Hepatitis B Clinical Features 98 Incubation period 60 -150 days (average 90 days) Nonspecific prodrome of malaise, fever, headache, myalgia Illness not specific for hepatitis B At least 50% of infections asymptomatic

99 Global Patterns of Chronic HBV Infection High (>8%): 45% of global population lifetime risk of infection >60% early childhood infections common Intermediate (2%-7%): 43% of global population lifetime risk of infection 20%-60% infections occur in all age groups Low (<2%): 12% of global population lifetime risk of infection <20% most infections occur in adult risk groups

Hepatitis B Complications 100 Fulminant hepatitis Hospitalization Cirrhosis Hepatocellular carcinoma Death
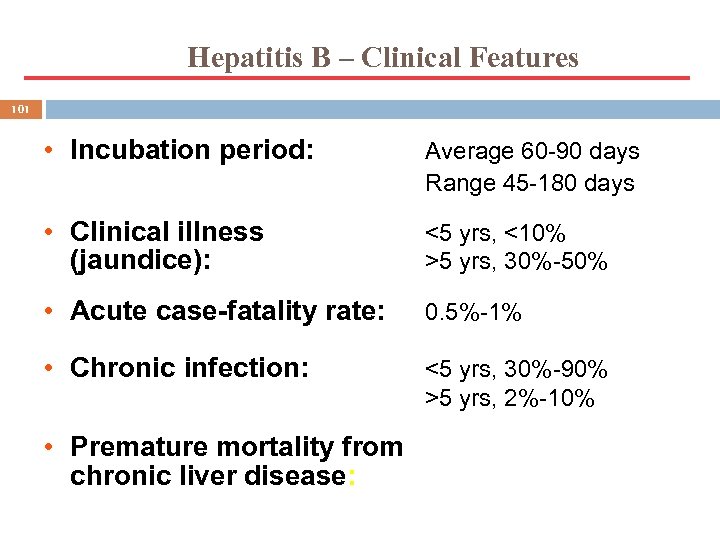
Hepatitis B – Clinical Features 101 • Incubation period: Average 60 -90 days Range 45 -180 days • Clinical illness (jaundice): <5 yrs, <10% >5 yrs, 30%-50% • Acute case-fatality rate: 0. 5%-1% • Chronic infection: <5 yrs, 30%-90% >5 yrs, 2%-10% • Premature mortality from chronic liver disease: 15%-25%

Hepatitis B Epidemiology 102 Reservoir Human Transmission Bloodborne Subclinical cases can also transmit infection Communicability 1 -2 months before and after onset of symptoms Chronic carriers major source of infection Assume in health care setting that ALL patients may be contagious

Hepatitis B Perinatal Transmission* 103 If mother positive for HBs. Ag and HBe. Ag 70%-90% of infants infected 90% of infected infants become chronically infected If positive for HBs. Ag only 5%-20% of infants infected 90% of infected infants become chronically infected *in the absence of postexposure prophylaxis
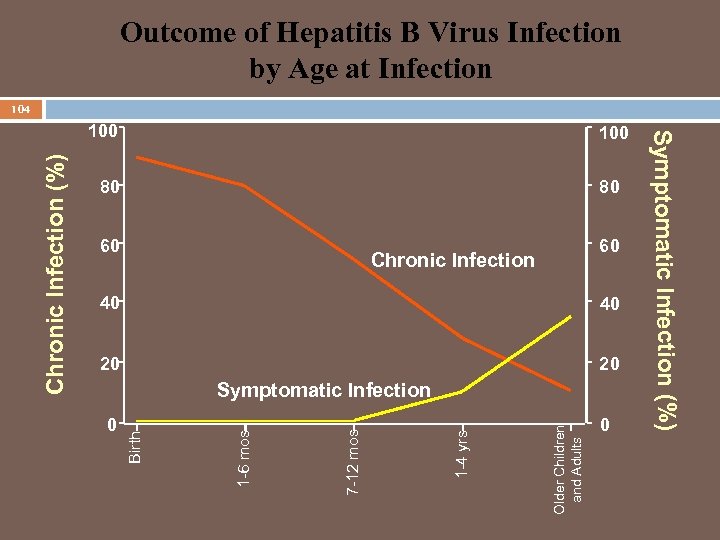
Outcome of Hepatitis B Virus Infection by Age at Infection 104 80 80 60 60 Chronic Infection 40 40 20 20 Older Children and Adults 1 -4 yrs 7 -12 mos 0 1 -6 mos Symptomatic Infection Birth Chronic Infection (%) 100 0 Symptomatic Infection (%) 100
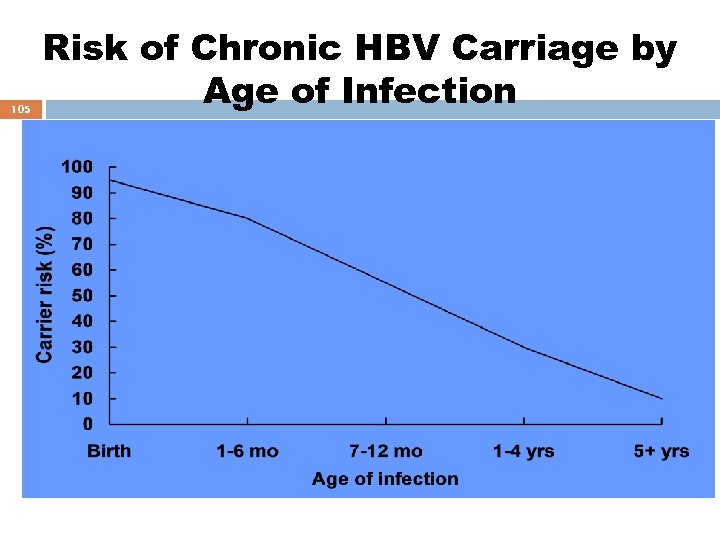
105 Risk of Chronic HBV Carriage by Age of Infection

Hepatitis B Vaccine composed only of Surface antigen 106 1965 Discovery of Australian antigen 1973 Successful HBV infection of chimpanzees 1981 Licensure of plasma-derived vaccine 1986 Licensure of recombinant vaccine 1991 Universal infant vaccination 1996 Universal adolescent vaccination

107 Hepatitis B Vaccine, surface antigen only Composition Recombinant HBs. Ag Efficacy 95% (Range, 80%-100%) Duration of Immunity >15 years Schedule 3 Doses Booster doses not routinely recommended

Hepatitis B Vaccine Formulations 108 Recombivax HB (Merck) - 5 mcg/0. 5 m. L (pediatric) - 10 mcg/1 m. L (adult) - 40 mcg/1 m. L (dialysis) Engerix-B (GSK) - 10 mcg/0. 5 m. L (pediatric) - 20 mcg/1 m. L (adult)
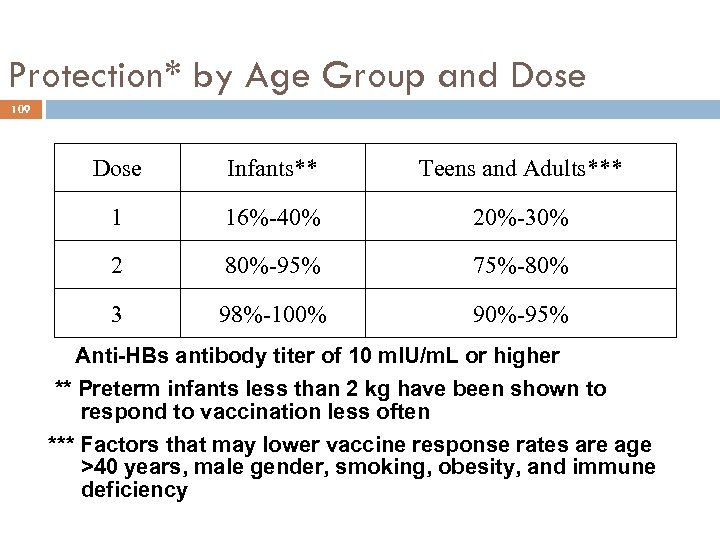
Protection* by Age Group and Dose 109 Dose Infants** Teens and Adults*** 1 16%-40% 20%-30% 2 80%-95% 75%-80% 3 98%-100% 90%-95% * Anti-HBs antibody titer of 10 m. IU/m. L or higher ** Preterm infants less than 2 kg have been shown to respond to vaccination less often *** Factors that may lower vaccine response rates are age >40 years, male gender, smoking, obesity, and immune deficiency

Recommended Dose of Hepatitis B Vaccine 110 Recombivax HB Engerix-B Dose (mcg) 0. 5 m. L (5) Dose (mcg) 0. 5 m. L (10) Adolescents 11 -19 years 0. 5 m. L (5) 0. 5 m. L (10) Adults >20 years 1. 0 m. L (10) 1. 0 m. L (20) Infants and children <11 years of age

111 Hepatitis B Vaccine Long-term Efficacy Immunologic memory established following vaccination Exposure to HBV results in anamnestic anti-HBs response Chronic infection rarely documented among vaccine responders

Hepatitis B Vaccine 112 Routine booster doses are NOT routinely recommended for any group

113 Hepatitis B Vaccine Routine Infant Schedule Dose+ Primary 1 Primary 2 Primary 3 Usual Age Minimum Interval Birth --1 - 2 months 4 weeks 6 -18 months* 8 weeks** * infants who mothers are HBs. Ag+ or whose HBs. Ag status is unknown should receive third dose at 6 months of age ** at least 16 weeks after the first dose +an additional dose at 4 months is acceptable if the clinician prefers to use a combination vaccine that contains hepatitis B vaccine

Preterm Infants 114 Birth dose and HBIG if mother HBs. Ag positive Preterm infants <2, 000 grams have a decreased response to vaccine administered before 1 month of age Delay first dose until chronologic age 1 month if mother HBs. Ag negative
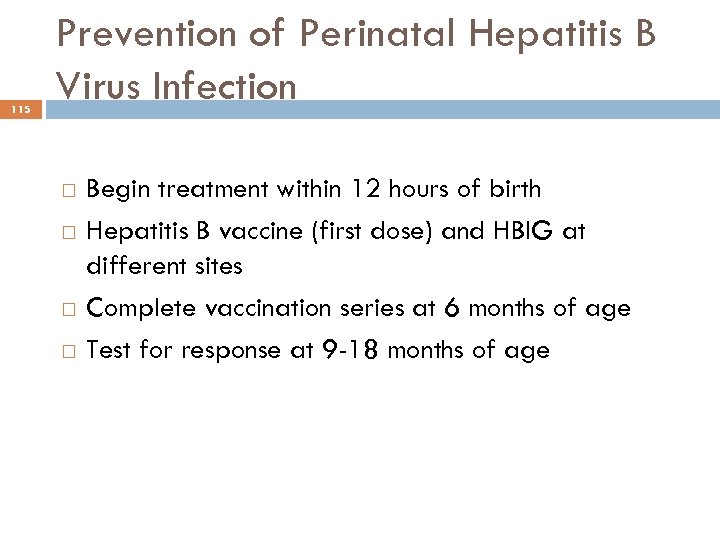
115 Prevention of Perinatal Hepatitis B Virus Infection Begin treatment within 12 hours of birth Hepatitis B vaccine (first dose) and HBIG at different sites Complete vaccination series at 6 months of age Test for response at 9 -18 months of age
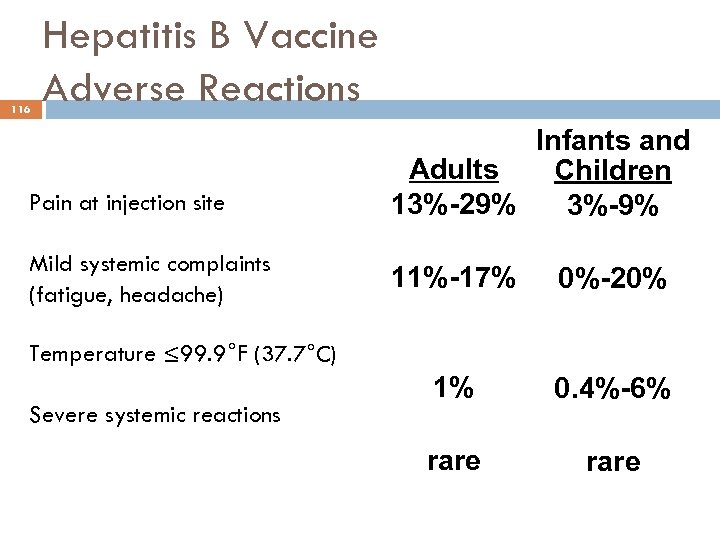
116 Hepatitis B Vaccine Adverse Reactions Pain at injection site Mild systemic complaints (fatigue, headache) Infants and Adults Children 13%-29% 3%-9% 11%-17% 0%-20% 1% 0. 4%-6% rare Temperature ≤ 99. 9°F (37. 7°C) Severe systemic reactions

Hepatitis B Vaccine 117 Contraindications and Precautions Severe allergic reaction to a vaccine component or following a prior dose Moderate or severe acute illness

The pneumococcus and the vaccine The pneumococcus Diseases caused by the pneumococcus and the global disease burden Immune response to pneumococcus Types of pneumococcal vaccines Pneumococcal conjugate vaccine Importance of serotype and moving forward with the decision to include the vaccine in the Jordan EPI

Streptococcus pneumoniae Gram positive diplococci Many serotypes (90) Serotypes depend on polysaccharide capsule Serotypes define invasiveness and protective antibody Acquired by close contact with humans Invasive disease more common in early childhood and in certain high risk individuals Immunity mainly serotype specific 119 Major impact on child health
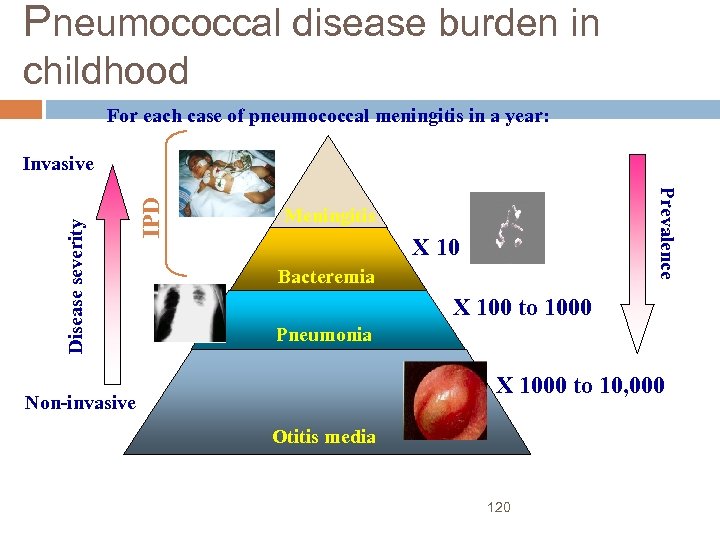
Pneumococcal disease burden in childhood For each case of pneumococcal meningitis in a year: IPD Prevalence Disease severity Invasive Meningitis X 10 Bacteremia X 100 to 1000 Pneumonia X 1000 to 10, 000 Non-invasive Otitis media Adapted from Abramson JS et al. , Pediatrics, 2000, ; 106(2): 362 -6 120
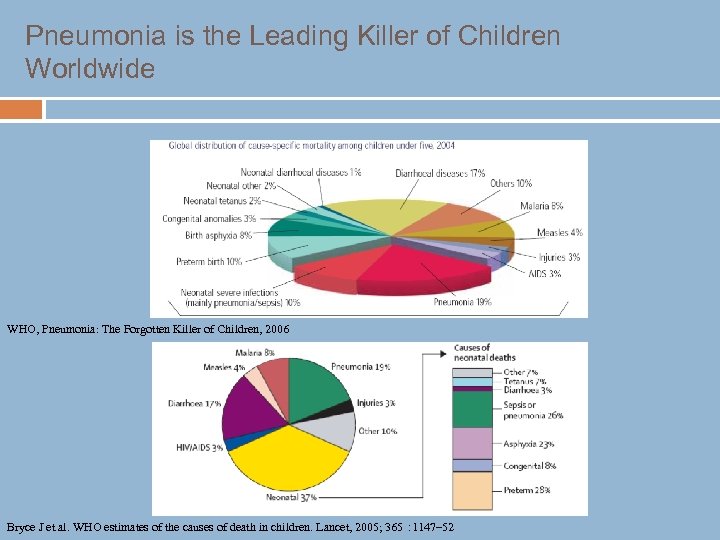
Pneumonia is the Leading Killer of Children Worldwide WHO, Pneumonia: The Forgotten Killer of Children, 2006 Bryce J et al. WHO estimates of the causes of death in children. Lancet, 2005; 365 : 1147– 52
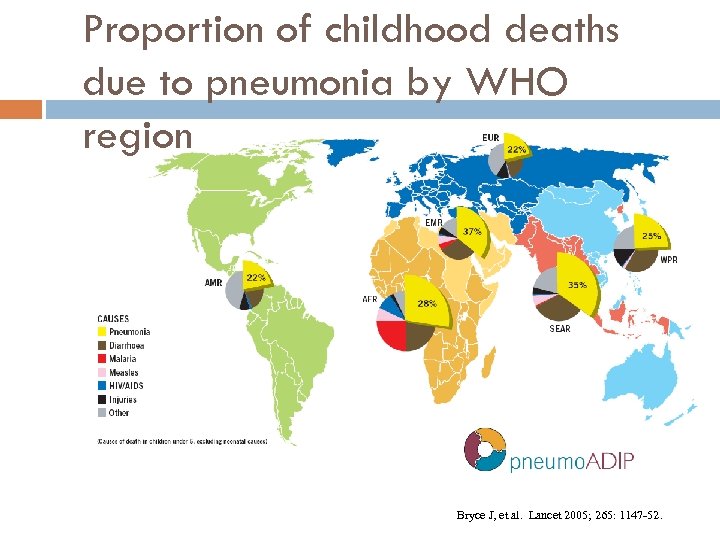
Proportion of childhood deaths due to pneumonia by WHO region Bryce J, et al. Lancet 2005; 265: 1147 -52.

Vaccine-preventable Deaths in Children <5 Years of Age, Globally The WHO estimates that Streptococcus pneumoniae (SP) causes >1. 6 million deaths/yr 800, 000 are in children <5 yrs Pneumonia is the leading cause of pneumococcal deaths worldwide Rotavirus Measles 16% 21% N. meningitidis <1% Of all causes of bacterial meningitis, SP has the highest case fatality rate Rotavirus causes approximately 400, 000 death/yr in those <5 yrs HPV is a vaccine-preventable cause of deaths in adults, estimated to be 260, 000 deaths annually worldwide SP is the most common cause of vaccinepreventable deaths worldwide Bilous J. Lancet. 2006; 367: 1464 -1465. WHO Global Immunization Vision and Strategy, April 2005 www. who. int/vaccines/GIVS/english/Global_imm. _data_EN. pdf. www. who. int/reproductive-health/publications/hpvvaccines_techinfo/index. html. Hib 15% S. pneumoniae 28% Tetanus 8% Pertussis 12%

Global Perspective Estimated number of deaths (WHO 2002) Vaccine-preventable Deaths (WHO) Pneumococcal disease Measles Rotavirus Hib Pertussis Tetanus Other* Meningococcus Streptococcus pneumoniae is the leading cause of vaccine-preventable deaths globally *Polio, diphtheria, yellow fever WHO. 2004 Global Immunization Data. Available at: http: //www. who. int/immunization_monitoring/data/Global. Immunization. Data. pdf. Accessed July 11, 2008.
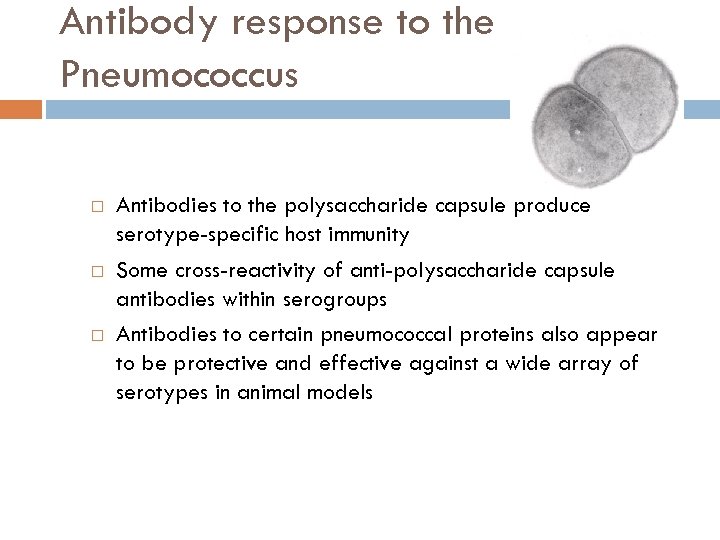
Antibody response to the Pneumococcus Antibodies to the polysaccharide capsule produce serotype-specific host immunity Some cross-reactivity of anti-polysaccharide capsule antibodies within serogroups Antibodies to certain pneumococcal proteins also appear to be protective and effective against a wide array of serotypes in animal models

Three types of inactivated pneumococcal vaccine Polysaccharide vaccines: long chains of sugars taken from the capsule of various pneumococcal serotypes Pneumococcal conjugate vaccines (PCV): the serotype-specific capsular polysaccharide is linked with a carrier protein Protein vaccines: use bacterial proteins to evoke an immune response and may be effective over a wide array of pneumococcal serotypes USAID. Immunization Essentials: A Practical Field Guide. 2003. WHO. State of the Art of New Vaccines: Research and Development. 2006.

Polysaccharide versus conjugate vaccines Polysaccharide Vaccines: Pneumococcal Conjugate Vaccines 1: T-cell independent T-cell dependent Not effective in children under 2 years and in the immunocompromised Induce an effective immune response in infants Stimulate an initial response involving both Ig. M and Ig. G while subsequent doses stimulate a primarily Ig. G response Induce immunological memory and can be boosted Result in a primarily Ig. M response Do not induce immunological memory and hence cannot be boosted Do not reduce mucosal carriage of pneumococcus 1 1 Eskola J. PIDJ 2000. 19 (4): 388 -93.

PCV-7 serotypes account for most disease in young US children 7 -valent PCV serotypes Non- vaccine serotypes Distribution of Pneumococcal Serotypes by Frequency: Children <2 years, USA (1999)

PCV-7 serotypes account for most antibiotic resistance in young US children 7 -valent PCV serotypes Distribution of Serotypes by Penicillin Resistance: Children <2 years, USA (1999) Non- vaccine serotypes
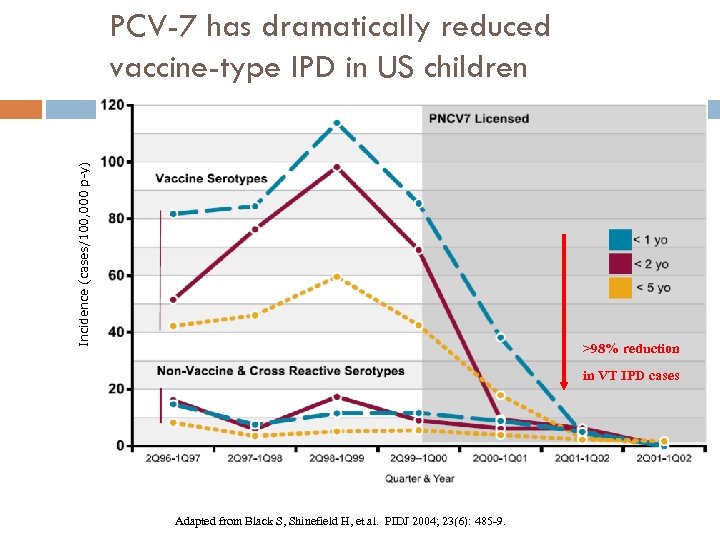
Incidence (cases/100, 000 p-y) PCV-7 has dramatically reduced vaccine-type IPD in US children >98% reduction in VT IPD cases Adapted from Black S, Shinefield H, et al. PIDJ 2004; 23(6): 485 -9.
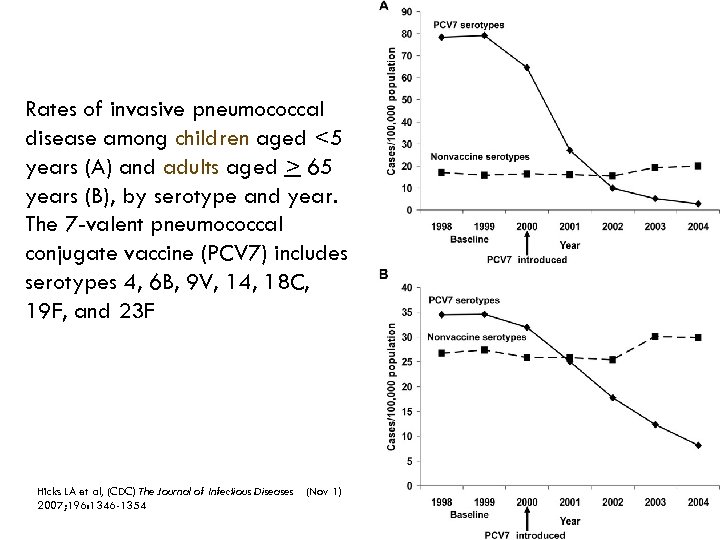
Rates of invasive pneumococcal disease among children aged <5 years (A) and adults aged > 65 years (B), by serotype and year. The 7 -valent pneumococcal conjugate vaccine (PCV 7) includes serotypes 4, 6 B, 9 V, 14, 18 C, 19 F, and 23 F Hicks LA et al, (CDC) The Journal of Infectious Diseases 2007; 196: 1346 -1354 (Nov 1)

PCV has an indirect effect on reducing IPD in the community as a whole The indirect effect of PCV provides protection against pneumococcal disease even among unimmunized individuals Lower NP colonization of vaccine serotypes in immunized children decreases transmission to others in the community This type of immunity is also known as herd immunity O’Brien KL, Dagan R. Vaccine 2003; 21: 1815 -25. Hammitt LL, Bruden DL, et al. JID 2006; 193: 1487 -94.

PCV Impact: Key Points Less IPD in all age groups in US due to herd immunity Less antibiotic-resistant pneumococcal disease in US Reduces nasopharyngeal (NP) colonization with vaccine serotypes In US, disease prevented by PCV use outweighs small increase in non-vaccine type IPD

What about pneumococcal vaccine for our region? ? 134

Pneumococcal Serotypes • There are over 90 strains (or serotypes) of the pneumococcal bacterium. • The distribution of disease –causing pneumococcal serotypes affecting children varies geographically. • Seven pneumococcal serotypes are common to all regions and comprise 58 -66% of invasive childhood pneumococcal disease in every region of the world 8* Information courtesy of the International Vaccine Access Center (IVAC) • Seven common serotypes are: 1, 5, 6 A, 6 B, 14, 19 F, 23 F Source: 8. Johnson HL, Deloria-Knoll M, Levine OS, et al: Pneumococcal global serotype project. Submitted 12/09.
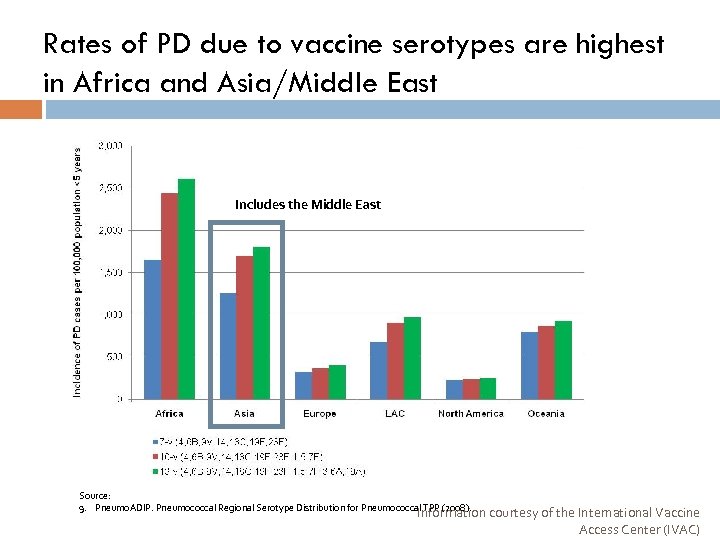
Rates of PD due to vaccine serotypes are highest in Africa and Asia/Middle East Includes the Middle East Source: 9. Pneumo. ADIP. Pneumococcal Regional Serotype Distribution for Pneumococcal TPP (2008). Information courtesy of the International Vaccine Access Center (IVAC)
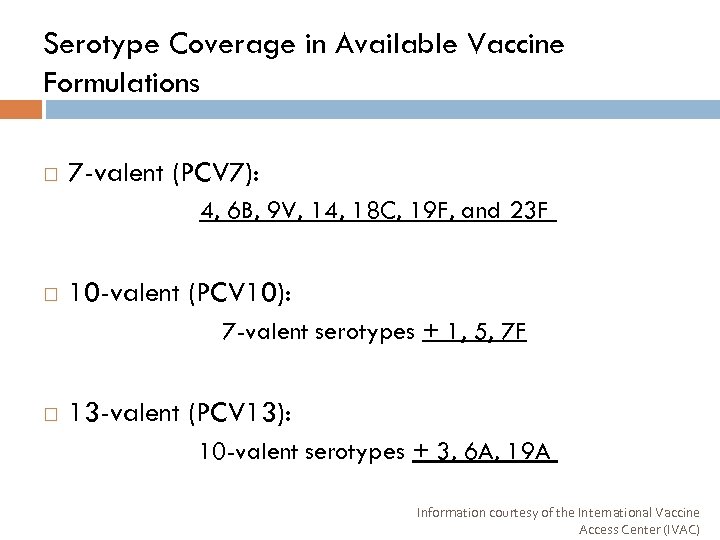
Serotype Coverage in Available Vaccine Formulations 7 -valent (PCV 7): 4, 6 B, 9 V, 14, 18 C, 19 F, and 23 F 10 -valent (PCV 10): 7 -valent serotypes + 1, 5, 7 F 13 -valent (PCV 13): 10 -valent serotypes + 3, 6 A, 19 A Information courtesy of the International Vaccine Access Center (IVAC)
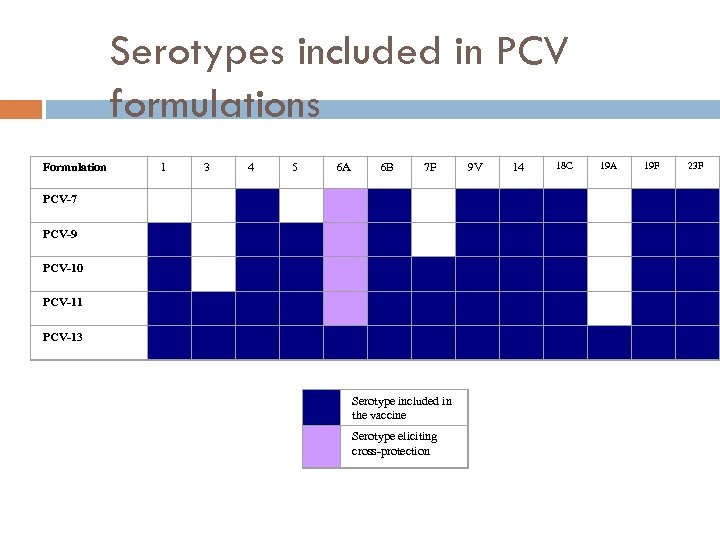
Serotypes included in PCV formulations Formulation 1 3 4 5 6 A 6 B 7 F 9 V 14 18 C 19 A 19 F 23 F PCV-7 PCV-9 PCV-10 PCV-11 PCV-13 Serotype included in the vaccine Serotype eliciting cross-protection

Rationale for inclusion of additional serotypes in PCV 13 1 • Cause of epidemic disease • Important cause of pneumococcal pneumonia; predominant serotype in empyema in children • Important cause of pneumococcal disease in many regions 3 • Important cause of pneumococcal disease including pneumonia and AOM • Among the leading serotypes in CAP and IPD in adults • Increasingly reported in IPD in Europe 5 • Cause epidemic disease • Among the leading serotypes in Africa and South America 6 A 7 F 19 A 143 • Important cause of pneumococcal disease, particularly AOM • Commonly found in carriage and frequently antibiotic resistant • Decrease in 6 A pediatric IPD following use of PCV 7, remaining disease may be mostly 6 C • Cause epidemic disease • Important cause of pneumococcal disease globally, increasing in many European countries • Significantly increased in IPD globally • Commonly found in AOM • Commonly found in carriage and frequently antibiotic resistant • 19 F antibodies from PCV 7 vaccination do not cross-protect for 19 A infection Dagan et al. J Infect Dis 2008; 197(8): 1094 -102. Dagan et al. Clin Infect Dis 2000; 30(2): 319 -21. Brueggemann et al. Journal of Clinical Microbiology 2003; 41(11): 4966 -70, Gratten Med J Aust 1993; 158(5): 340 -2, Nunes et al. Clin Microbiol Infect 2008; 14(1): 82 -4, Romney et al. Clin Infect Dis 2008; 47(6): 768 -74, Ruckinger et al. Pediatr Infect Dis J 2009; 28(2): 118 -22, Whitney et al. Lancet 2006; 368(9546): 1495 -502, Dagan & Klugman Lancet Infect Dis, 8(12), 785 -795 (2008), Byington et al. Pediatr Infect Dis J, 25(3), 250 -254 (2006), Fletcher et al. Pediatr Infect Dis J, 25(6), 559 -560 (2006), Cohen et al. Vaccine, (2009) in press, Hausdorff WP, Vaccine, 25(13), 2406 -2412 (2007), Imohl et al. Clin Microbiol Infect, (2009), in press)

Serotyped isolates (%) Serotypes by rank order and cumulative serotype distribution Prevenar 13 targets serotypes responsible for most of the IPD burden in children < 5 years of age Serotype Pneumococcal Global Serotype Project (version 2), 30 November 2008. Prepared by GAVI’s Pneumo. ADIP *Weighted by regional disease burden Cumulative distribution (%) Global IPD serotype distribution among children <5 years – before pneumococcal conjugate vaccination
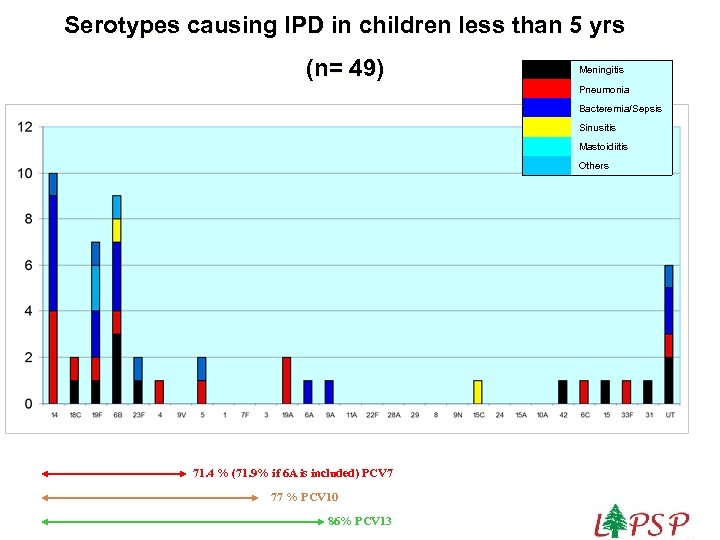
Serotypes causing IPD in children less than 5 yrs (n= 49) Bacteremia/Sepsis Sinusitis Mastoidiitis 86% PCV 13 Pneumonia 77 % PCV 10 Meningitis 71. 4 % (71. 9% if 6 A is included) PCV 7 Others
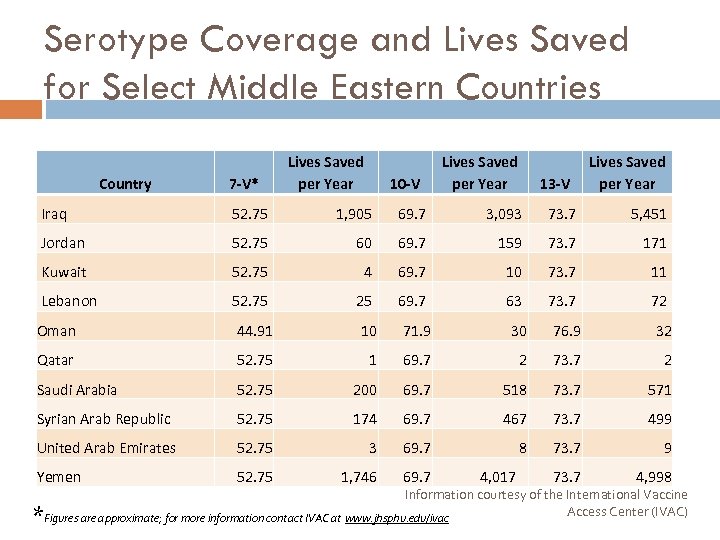
Serotype Coverage and Lives Saved for Select Middle Eastern Countries Country 7 -V* Lives Saved per Year 10 -V Lives Saved per Year 13 -V Lives Saved per Year Iraq 52. 75 1, 905 69. 7 3, 093 73. 7 5, 451 Jordan 52. 75 60 69. 7 159 73. 7 171 Kuwait 52. 75 4 69. 7 10 73. 7 11 Lebanon 52. 75 25 69. 7 63 73. 7 72 Oman 44. 91 10 71. 9 30 76. 9 32 Qatar 52. 75 1 69. 7 2 73. 7 2 Saudi Arabia 52. 75 200 69. 7 518 73. 7 571 Syrian Arab Republic 52. 75 174 69. 7 467 73. 7 499 United Arab Emirates 52. 75 3 69. 7 8 73. 7 9 Yemen 52. 75 1, 746 69. 7 4, 017 73. 7 4, 998 * Information courtesy of the International Vaccine Access Center (IVAC) Figures are approximate; for more information contact IVAC at www. jhsphu. edu/ivac

Prevention of Resistant Strains with Vaccination Strains Resistant to Erythromycin to Penicillin Strains Resistant to both PCV 7 75% 70% PCV 10 79% 75% 70 % PCV 13 79% 75% 70 % Dbaibo
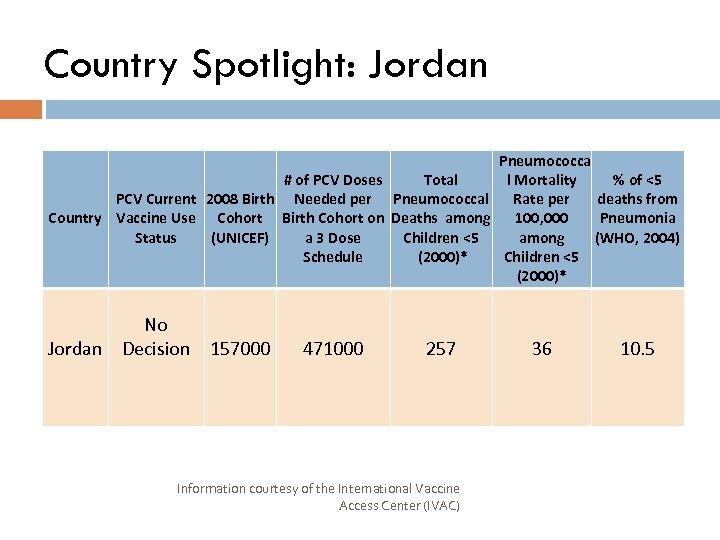
Country Spotlight: Jordan Pneumococca # of PCV Doses Total l Mortality % of <5 PCV Current 2008 Birth Needed per Pneumococcal Rate per deaths from Country Vaccine Use Cohort Birth Cohort on Deaths among 100, 000 Pneumonia Status (UNICEF) a 3 Dose Children <5 among (WHO, 2004) Schedule (2000)* Children <5 (2000)* No Jordan Decision 157000 471000 257 Information courtesy of the International Vaccine Access Center (IVAC) 36 10. 5

Proportion of pediatric pneumococcal disease prevented by PCV-13 89% 92% 73% 87% 86% Assumes cross protection within serogroup 6

Conclusions The pneumococccus is the leading cause of vaccine preventable disease worldwide Conjugate pneumococcal vaccines have proved efficacious and safe in infants as young as two months of age Conjugate vaccines have proved efficacious in preventing invasive pneumococcal infections in both children as well as the elderly due to decrease in the NP carraige rate

Conclusions Serotype coverage with the recently introduced PCV 13 covers more types that are of importance both in the westernized countries where non vaccine serotypes emerged as well as in developing countries There is currently enough evidence to suggest that the PCV be seriously considered for introduction into the Jordan EPI. 151
eee50c30a482b5cf25dcb477bc5fc09e.ppt