1 1 Bioactive nanoscale assemblies of oligoelectrolytes and


1 1 Bioactive nanoscale assemblies of oligoelectrolytes and PEGylated oligomers with drugs and nucleic acids for targeted delivery systems A. Zaichenko [email protected] Lviv Polytechnic National University

2 2 The aim of the study: synthesis and testing oligoperoxide based functional synthetic polymer carriers as well as nanosized drug (gene) delivery systems for medicine and biotechnology

3 III. Conjugates of oligomer carriers with drugs and nucleic acids as well as nanoscale delivery systems. Structures and properties. Talk outline II. Functional surface-active oligoelectrolytes and nonionic surfactants as carriers for drug and gene delivery. Introduction. Why we need novel carriers and drug (gene) delivery systems? IY. Cellular study and biomedical application of developed drug and gene delivery systems

4 4 I. Why we need novel smart carriers and drug (gene) delivery systems?

5 Novel carriers for DRUG DELIVERY

6 Anticancer drug delivery Delivery of doxorubicin or other cytostatics to tumor cells by nanosized carriers to decrease efficient cytotoxic concentration of the drugs comparing with its concentration when acting in free form.

7 Application of novel drug delivery vehicles to decrease potential negative side effects of delivered drugs in the treated organism.

8 Major problems in development of novel drugs High price: Development and application of one novel drug of wide spread use cost up to 1 billion USD Low efficiency due to non-addressed action: Only 0.001-0.01% of drugs applied intravenously can reach their biological targets in the organism Development of drug resistance: During one year approximately 50% of cancer patients gain resistance to applied chemotherapeutic drugs

9 Most drug delivery carriers are nanosized. Some of them possess: Toxic actions towards cell structure and functions; Immunogenic (allergic) action; Teratogenic action; Other negative actions.

10 Novel carriers for GENE DELIVERY

11 Novel carriers for Antisense RNA (DNA) Delivery Overcoming Blood-Brain Barrier When Injected Intravenously.

12 The developed carriers must be applied for: delivery of any drug; delivery of water-insoluble drugs; delivery of nucleic acids (gene therapy and biotechnologies); attachment of any biomarker molecule for cell recognition and addressed drug delivery; imaging in diagnostics and monitoring treatment course; overcome biological barriers; lowering toxicity; prolongation of the action in the body.

13 II. Functional surface-active oligoelectrolytes and nonionic polymeric surfactants as carriers for drug and gene delivery: Oligoperoxide based strategy of the synthesis

14 14 The main approaches of synthesis of functional oligoperoxides and derived polymers 1. Copolymerization of unsaturated ditertiary peroxides with functional monomers in hydrocarbon media and aqueous medium. 2. Telomerization of functional monomers in the presence of peroxide-containing telogen in hydrocarbon media and aqueous medium. 3. Polymer analogous transformations using carboxyl, amino, epoxy, isocyanate, anhydride and other reactive functional groups of peroxide-containing oligoelectrolytes. I. Functional surface-active oligoperoxides, oligoelectrolytes and nonionic surfactants of telechelic, block, and comb-like structures.
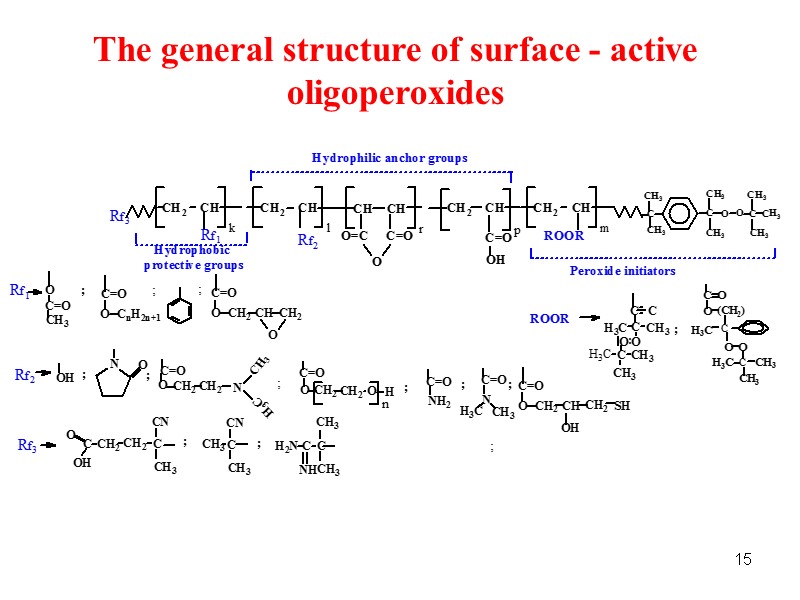
15 The general structure of surface - active oligoperoxides
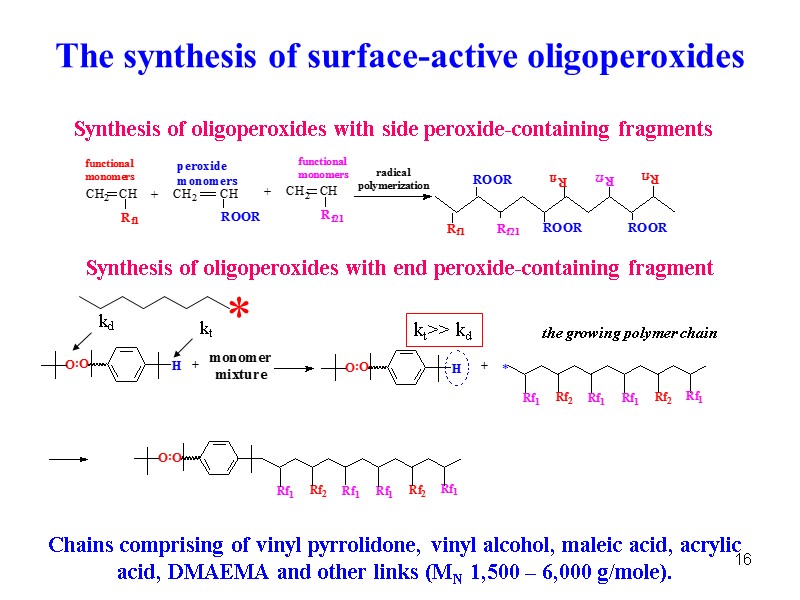
16 The synthesis of surface-active oligoperoxides Chains comprising of vinyl pyrrolidone, vinyl alcohol, maleic acid, acrylic acid, DMAEMA and other links (MN 1,500 – 6,000 g/mole). Synthesis of oligoperoxides with end peroxide-containing fragment Synthesis of oligoperoxides with side peroxide-containing fragments kd kt kt>> kd the growing polymer chain
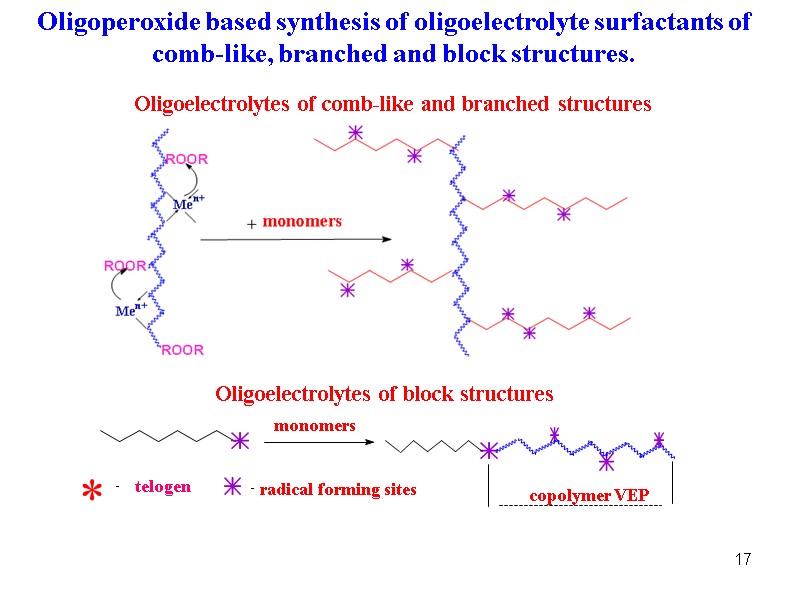
17 Oligoelectrolytes of comb-like and branched structures Oligoperoxide based synthesis of oligoelectrolyte surfactants of comb-like, branched and block structures. copolymer VEP monomers Oligoelectrolytes of block structures
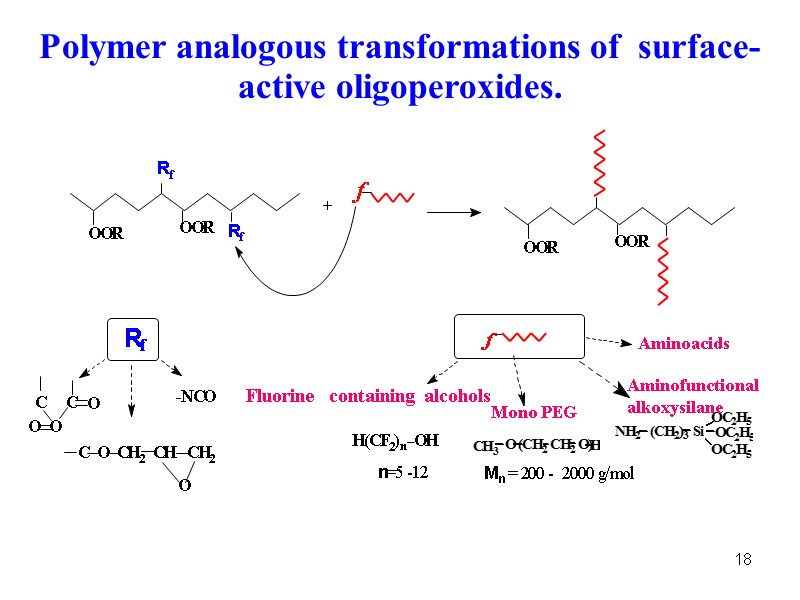
18 O Polymer analogous transformations of surface-active oligoperoxides.
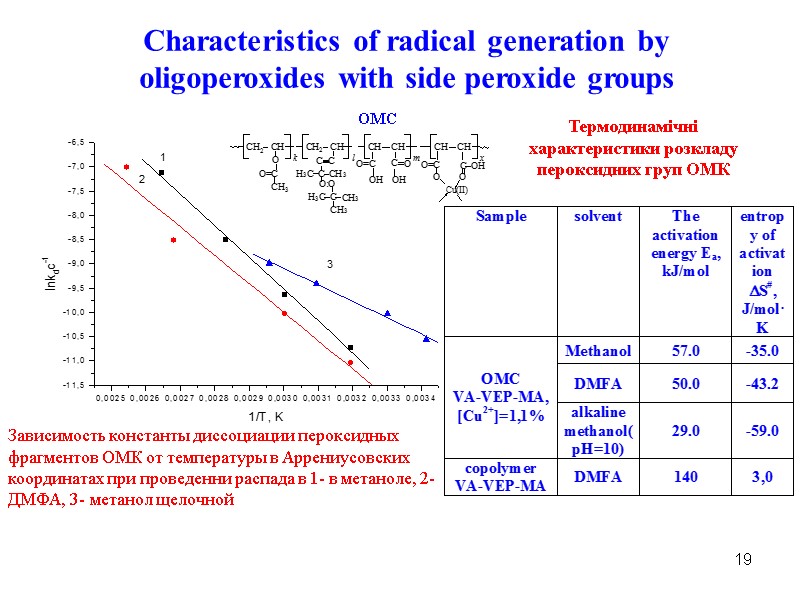
19 Characteristics of radical generation by oligoperoxides with side peroxide groups Зависимость константы диссоциации пероксидных фрагментов ОМК от температуры в Аррениусовских координатах при проведенни распада в 1- в метаноле, 2- ДМФА, 3- метанол щелочной OMC Термодинамічні характеристики розкладу пероксидних груп ОМК
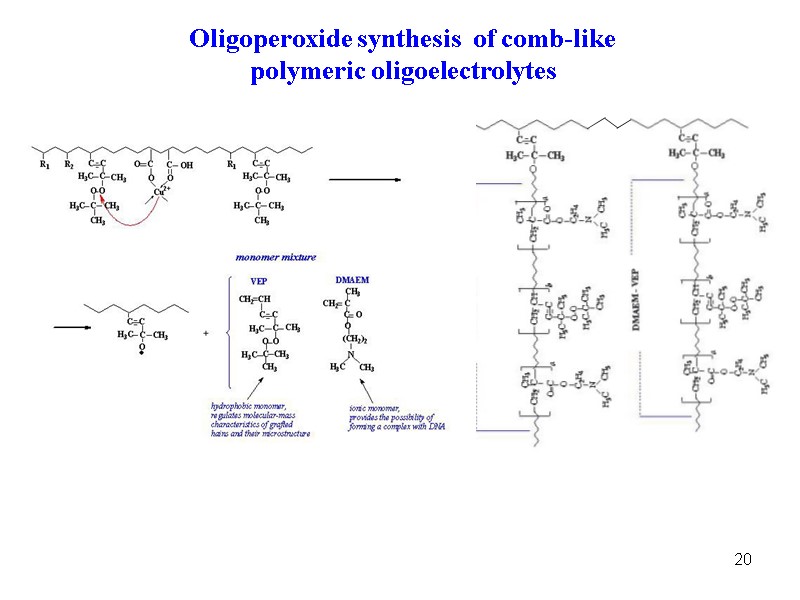
20 Oligoperoxide synthesis of comb-like polymeric oligoelectrolytes
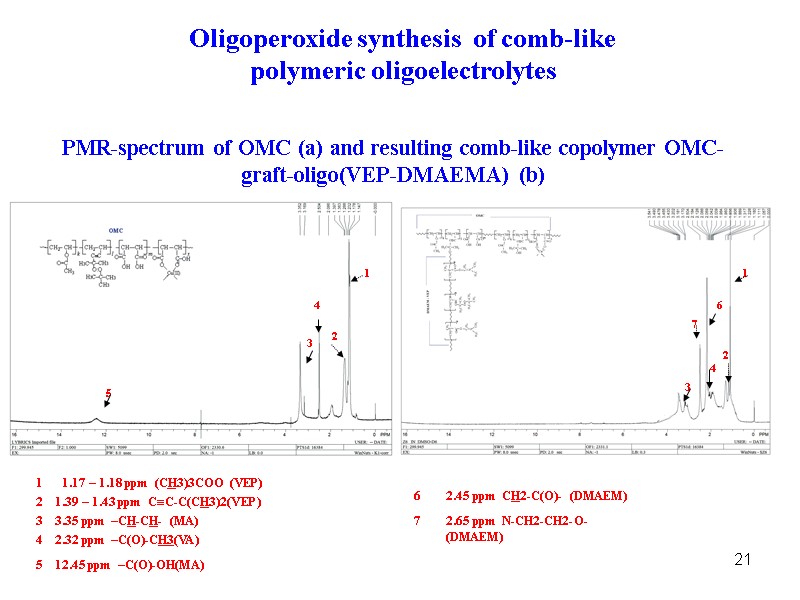
21 PMR-spectrum of OMC (a) and resulting comb-like copolymer OMC-graft-oligo(VEP-DMAEMA) (b) Oligoperoxide synthesis of comb-like polymeric oligoelectrolytes 1 1 2 2 3 3 6 4 4 7 5 1 1.17 – 1.18 ppm (CH3)3COO (VEP) 2 1.39 – 1.43 ppm CC-C(CH3)2(VEP) 3 3.35 ppm –CH-CH- (MA) 4 2.32 ppm –C(O)-CH3(VA) 5 12.45 ppm –C(O)-OH(MA) 6 2.45 ppm CH2-C(O)- (DMAEM) 7 2.65 ppm N-CH2-CH2-O- (DMAEM)
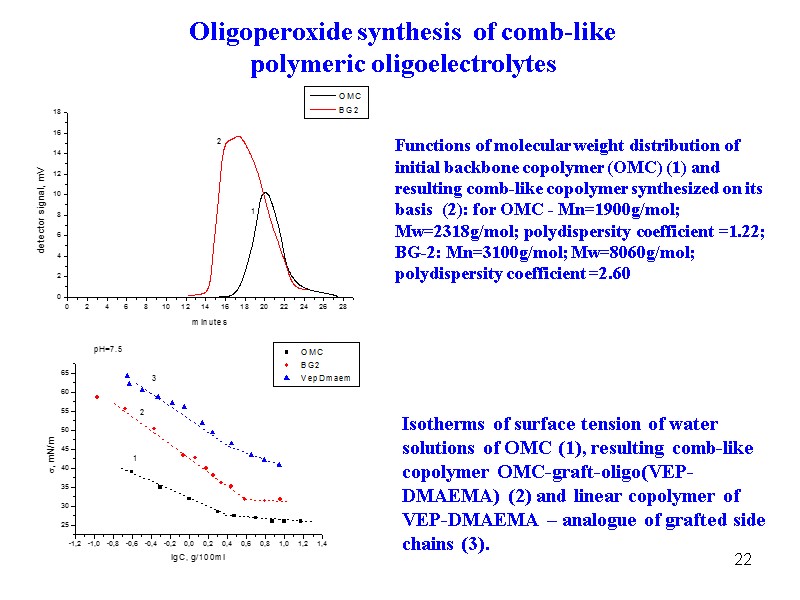
22 Functions of molecular weight distribution of initial backbone copolymer (OMC) (1) and resulting comb-like copolymer synthesized on its basis (2): for OMC - Mn=1900g/mol; Mw=2318g/mol; polydispersity coefficient =1.22; BG-2: Mn=3100g/mol; Mw=8060g/mol; polydispersity coefficient =2.60 Isotherms of surface tension of water solutions of OMC (1), resulting comb-like copolymer OMC-graft-oligo(VEP-DMAEMA) (2) and linear copolymer of VEP-DMAEMA – analogue of grafted side chains (3). Oligoperoxide synthesis of comb-like polymeric oligoelectrolytes
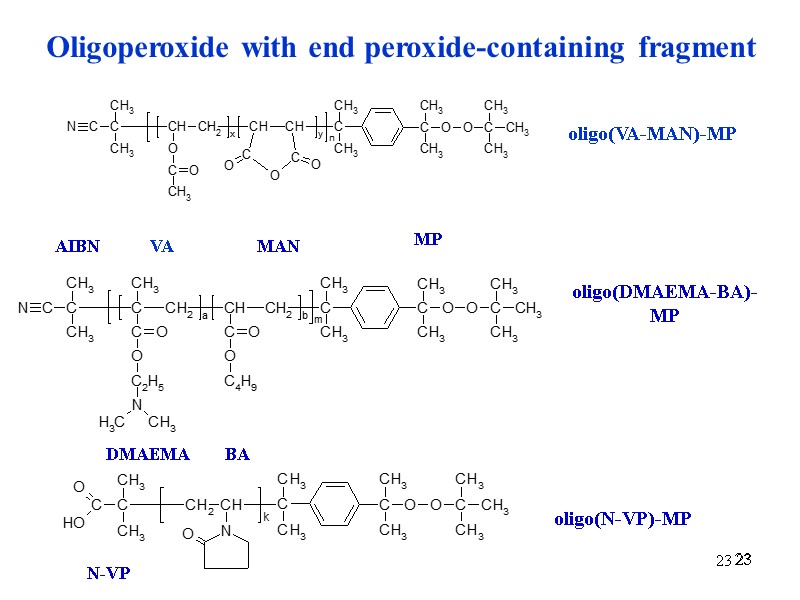
23 23 23 Oligoperoxide with end peroxide-containing fragment VA MAN MP oligo(VA-MAN)-MP AIBN DMAEMA BA oligo(DMAEMA-BA)-MP N-VP oligo(N-VP)-MP
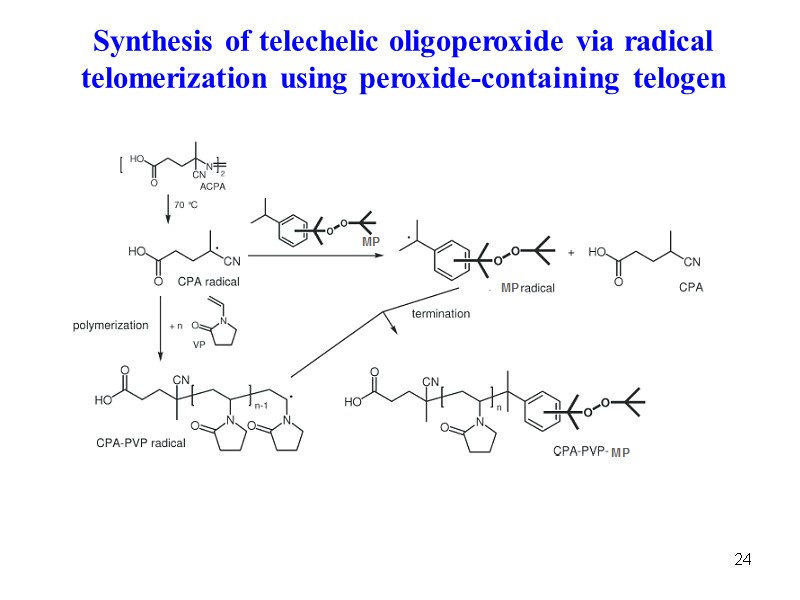
24 Synthesis of telechelic oligoperoxide via radical telomerization using peroxide-containing telogen
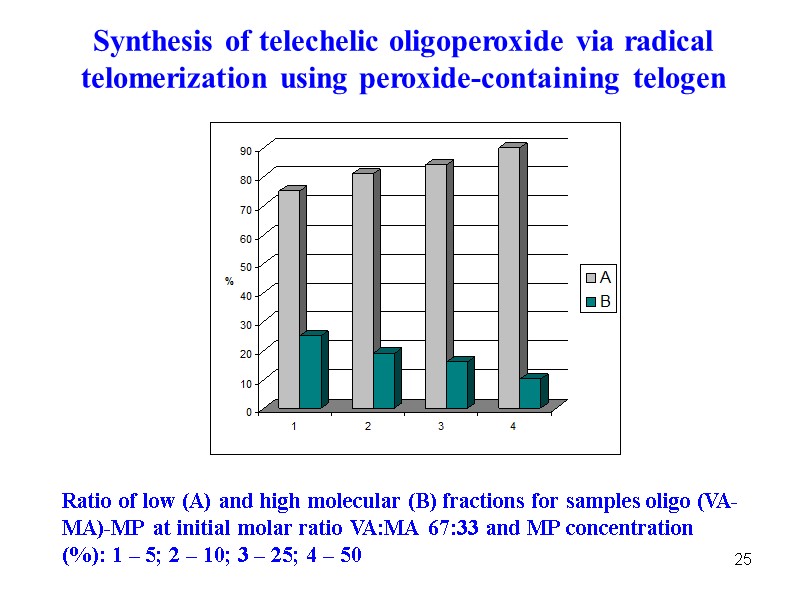
25 Ratio of low (A) and high molecular (B) fractions for samples oligo (VA-MA)-MP at initial molar ratio VA:MA 67:33 and MP concentration (%): 1 – 5; 2 – 10; 3 – 25; 4 – 50 Synthesis of telechelic oligoperoxide via radical telomerization using peroxide-containing telogen
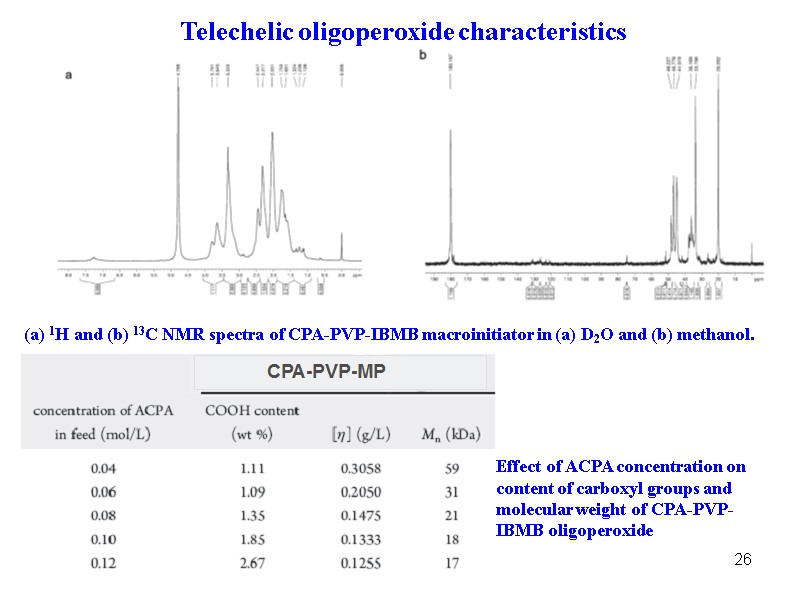
26 (a) 1H and (b) 13C NMR spectra of CPA-PVP-IBMB macroinitiator in (a) D2O and (b) methanol. Effect of ACPA concentration on content of carboxyl groups and molecular weight of CPA-PVP-IBMB oligoperoxide Telechelic oligoperoxide characteristics
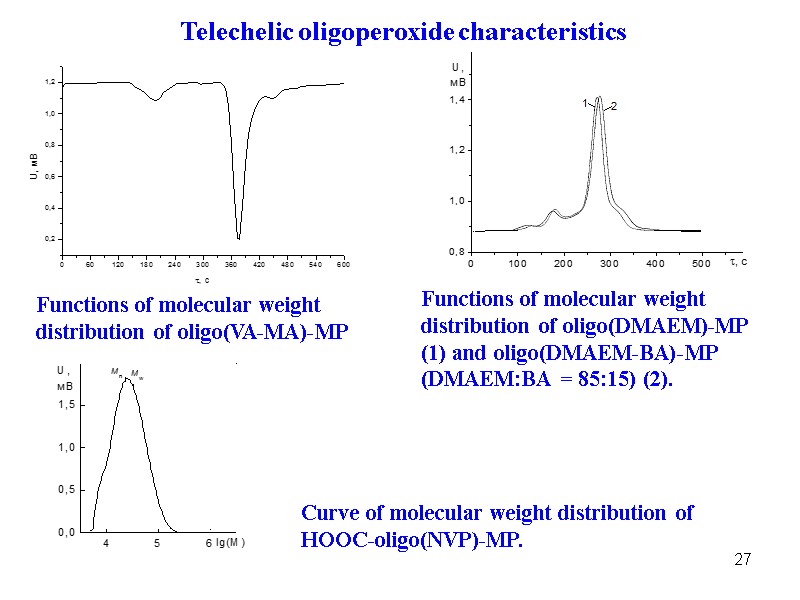
27 Functions of molecular weight distribution of oligo(VA-MA)-MP Functions of molecular weight distribution of oligo(DMAEM)-MP (1) and oligo(DMAEM-BA)-MP (DMAEM:BA = 85:15) (2). Curve of molecular weight distribution of НООС-oligo(NVP)-MP. Telechelic oligoperoxide characteristics
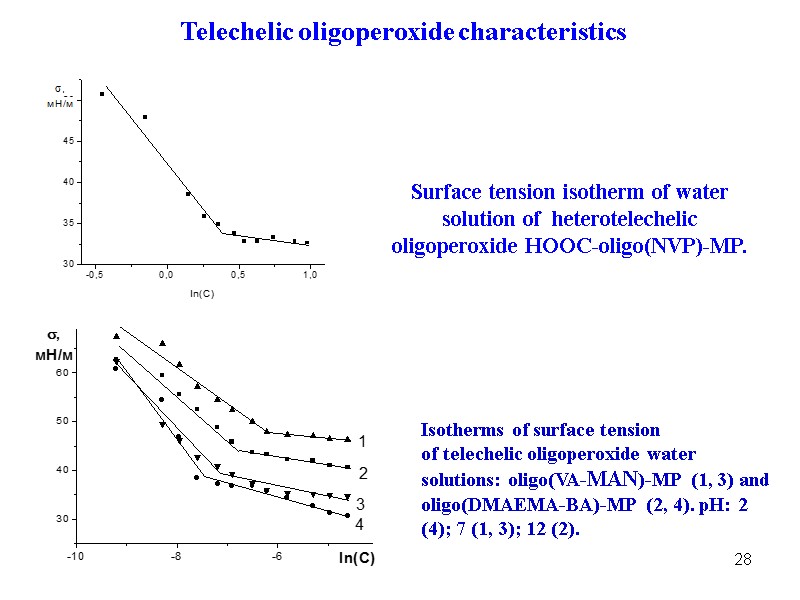
28 Isotherms of surface tension of telechelic oligoperoxide water solutions: oligo(VA-MAN)-MP (1, 3) and oligo(DMAEMA-BA)-MP (2, 4). pH: 2 (4); 7 (1, 3); 12 (2). Surface tension isotherm of water solution of heterotelechelic oligoperoxide НООС-oligo(NVP)-MP. Telechelic oligoperoxide characteristics
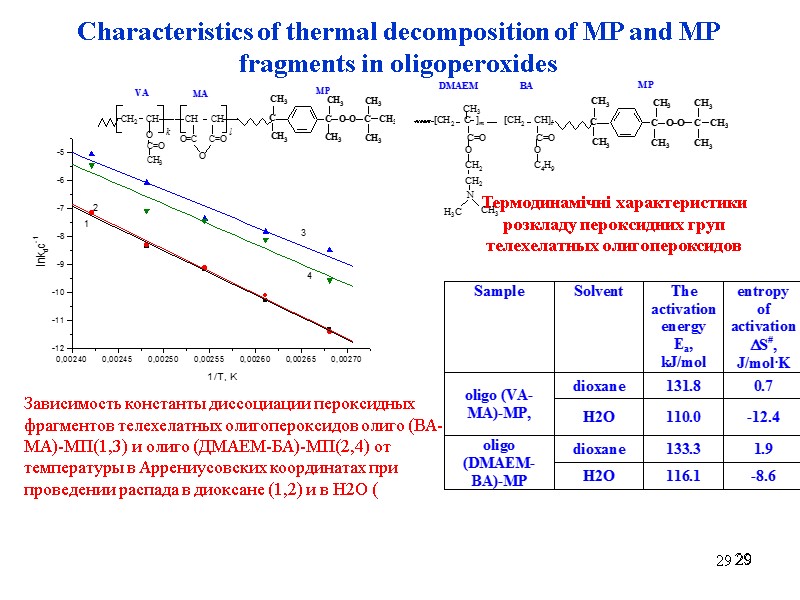
29 29 29 Characteristics of thermal decomposition of MP and MP fragments in oligoperoxides Зависимость константы диссоциации пероксидных фрагментов телехелатных олигопероксидов олиго (ВА-МА)-МП(1,3) и олиго (ДМАЕМ-БА)-МП(2,4) от температуры в Аррениусовских координатах при проведении распада в диоксане (1,2) и в Н2О ( Термодинамічні характеристики розкладу пероксидних груп телехелатных олигопероксидов
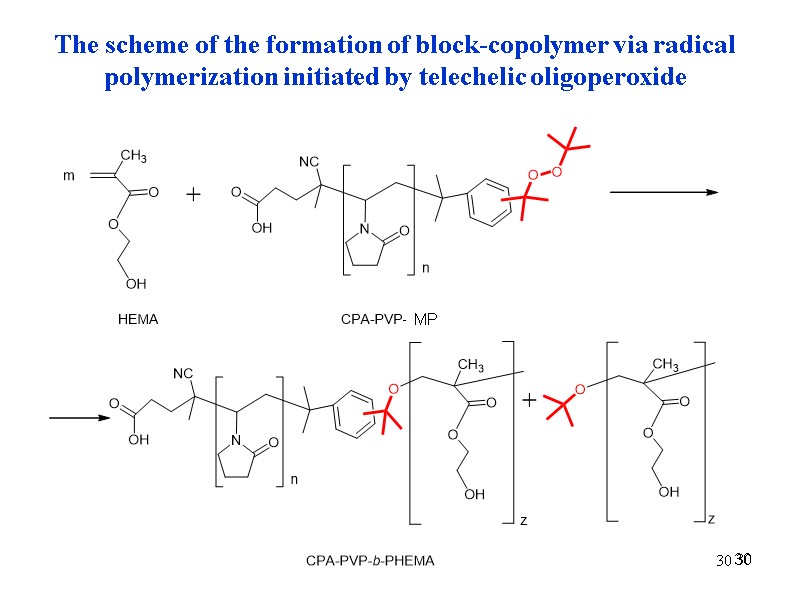
30 30 30 The scheme of the formation of block-copolymer via radical polymerization initiated by telechelic oligoperoxide
![31 Synthesis of block-copolymer via radical polymerization initiated by telechelic oligoperoxide [DMAEM]-links =95.5%, [MP]-fragments 31 Synthesis of block-copolymer via radical polymerization initiated by telechelic oligoperoxide [DMAEM]-links =95.5%, [MP]-fragments](https://present5.com/presentacii-2/20171211\24046-munich_nancy_zaichenko-last2.ppt\24046-munich_nancy_zaichenko-last2_31.jpg)
31 Synthesis of block-copolymer via radical polymerization initiated by telechelic oligoperoxide [DMAEM]-links =95.5%, [MP]-fragments =5.5%, Mn= 4500 g/mol (polydisper.=1.03) + Monomer mixture 2-aminoethyl 2-methylacrylate N-vinyl pyrrolidone Butyl acrylate Characterization of the block copolymer: [DMAEM]=12.6, [MP]=0.6%, [NVP]=70.2%, [BA]=12.4%, [AEM]=4.1%; Mn= 6500 g/mol; (polydispersity=1.29)
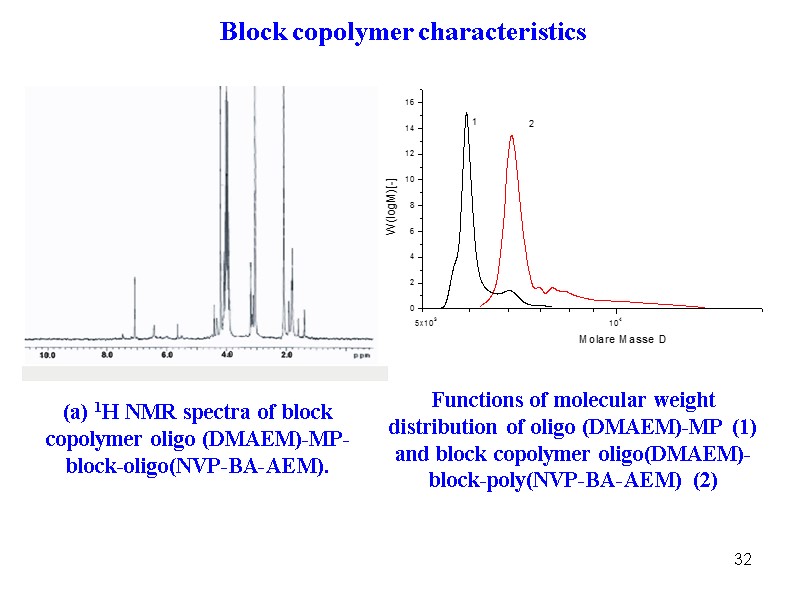
32 (a) 1H NMR spectra of block copolymer oligo (DMAEM)-MP- block-oligo(NVP-BA-AEM). Functions of molecular weight distribution of oligo (DMAEM)-MP (1) and block copolymer oligo(DMAEM)- block-poly(NVP-BA-AEM) (2) Block copolymer characteristics

33 Differential curves of molecular weight distribution of НООС-oligo(NVP)-MP (1) and HOOC-oligo(NVP)–block–poly(HEMA) (2). Characteristics of CPA-PVP-block-PHEMA obtained by CPA-PVP-IBMB-initiated polymerization of HEMA in the presence of MSA and DMSA (CPA-PVP-IBMB/HEMA ratio = 1/10 w/w) a CTA, chain transfer agent. b On the basis of nitrogen analysis. c Determined from SEC. d Dispersity. e Not determined. f DMSA,dimercaptosuccinic acid. g MSA, mercaptosuccinic acid. Block copolymer characteristics
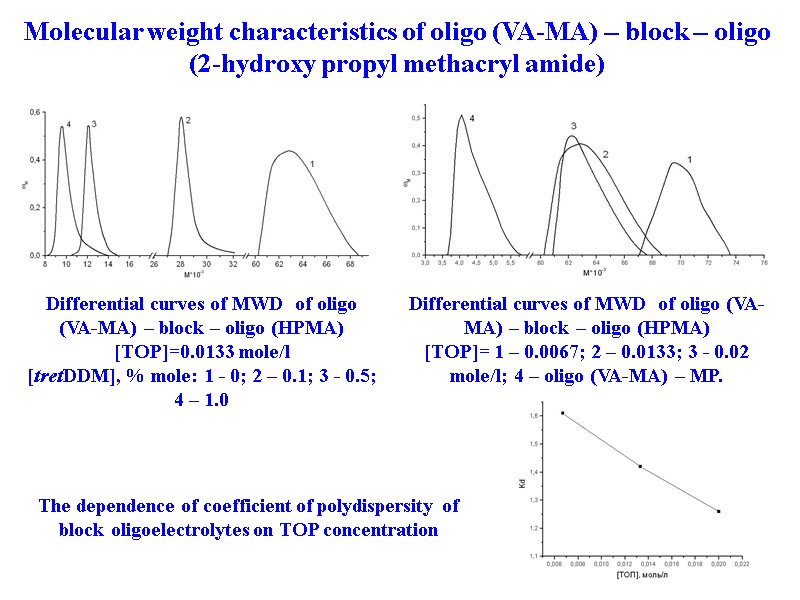
34 Differential curves of MWD of oligo (VA-MA) – block – oligo (HPMA) [TOP]= 1 – 0.0067; 2 – 0.0133; 3 - 0.02 mole/l; 4 – oligo (VA-MA) – MP. The dependence of coefficient of polydispersity of block oligoelectrolytes on TOP concentration Differential curves of MWD of oligo (VA-MA) – block – oligo (HPMA) [TOP]=0.0133 mole/l [tretDDM], % mole: 1 - 0; 2 – 0.1; 3 - 0.5; 4 – 1.0 Molecular weight characteristics of oligo (VA-MA) – block – oligo (2-hydroxy propyl methacryl amide)

35 Design of comb-like polymeric carriers combining PEG and oligoelectrolyte side branches
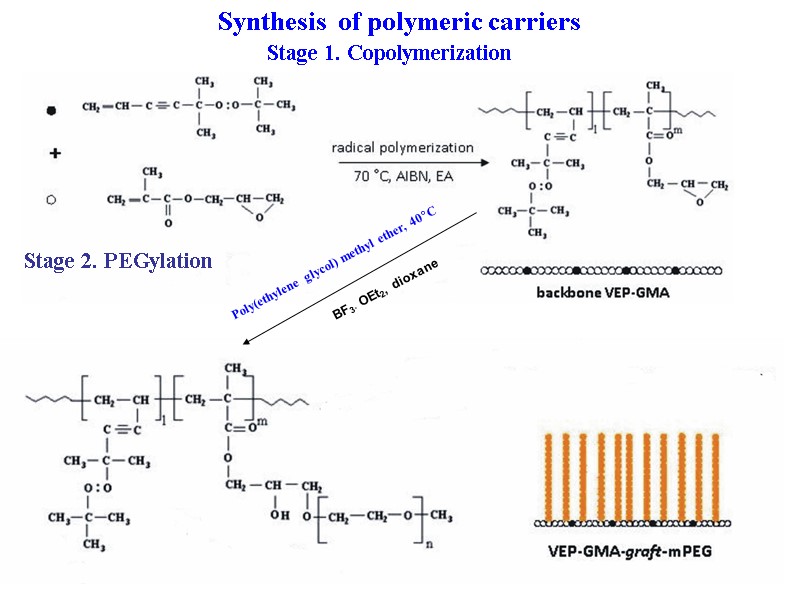
36 Stage 1. Copolymerization Poly(ethylene glycol) methyl ether, 40C BF3· OEt2, dioxane Stage 2. PEGylation Synthesis of polymeric carriers
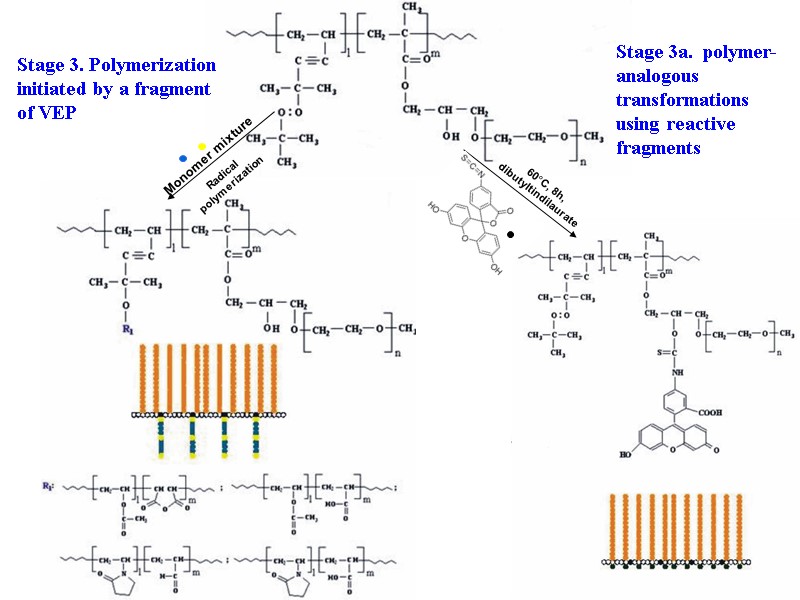
37 Stage 3a. polymer-analogous transformations using reactive fragments Stage 3. Polymerization initiated by a fragment of VEP Monomer mixture Radical polymerization 60C, 8h, dibutyltindilaurate
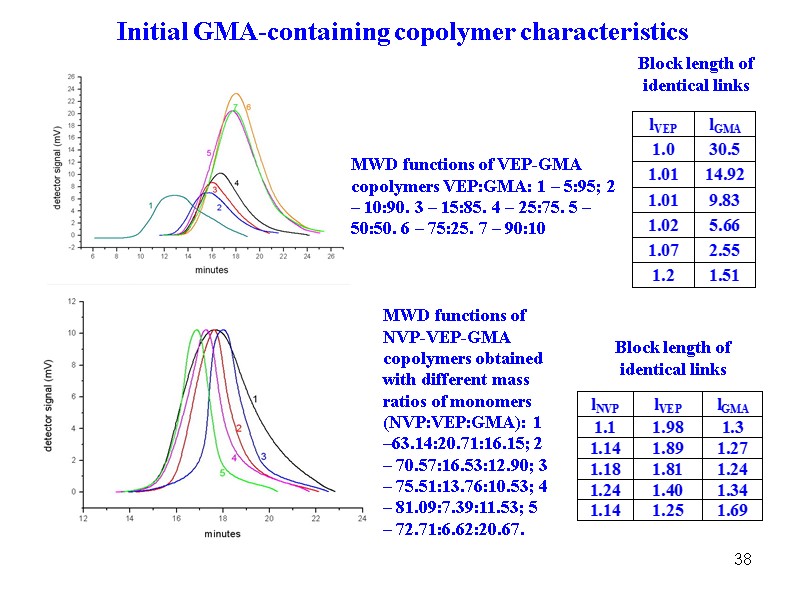
38 MWD functions of NVP-VEP-GMA copolymers obtained with different mass ratios of monomers (NVP:VEP:GMA): 1 –63.14:20.71:16.15; 2 – 70.57:16.53:12.90; 3 – 75.51:13.76:10.53; 4 – 81.09:7.39:11.53; 5 – 72.71:6.62:20.67. MWD functions of VEP-GMA copolymers VEP:GMA: 1 – 5:95; 2 – 10:90. 3 – 15:85. 4 – 25:75. 5 – 50:50. 6 – 75:25. 7 – 90:10 Block length of identical links Block length of identical links Initial GMA-containing copolymer characteristics
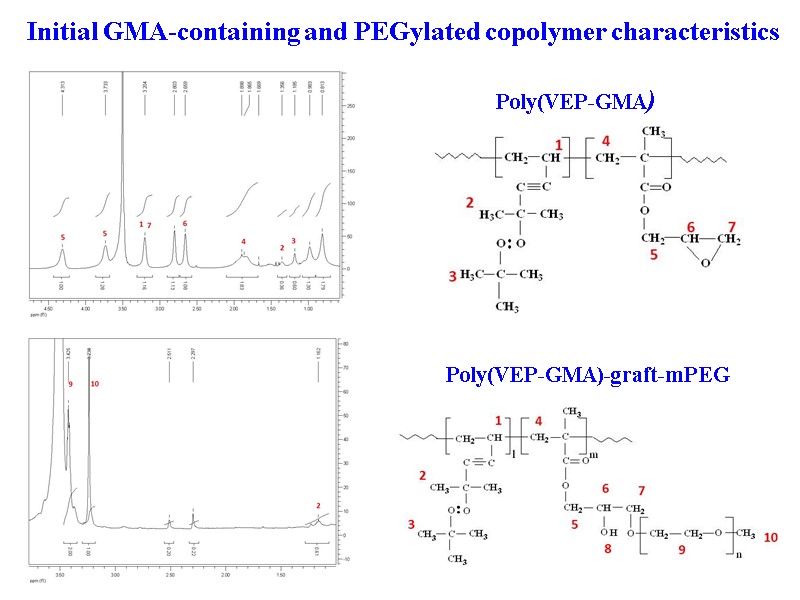
39 Poly(VEP-GMA)-graft-mPEG Poly(VEP-GMA) Initial GMA-containing and PEGylated copolymer characteristics
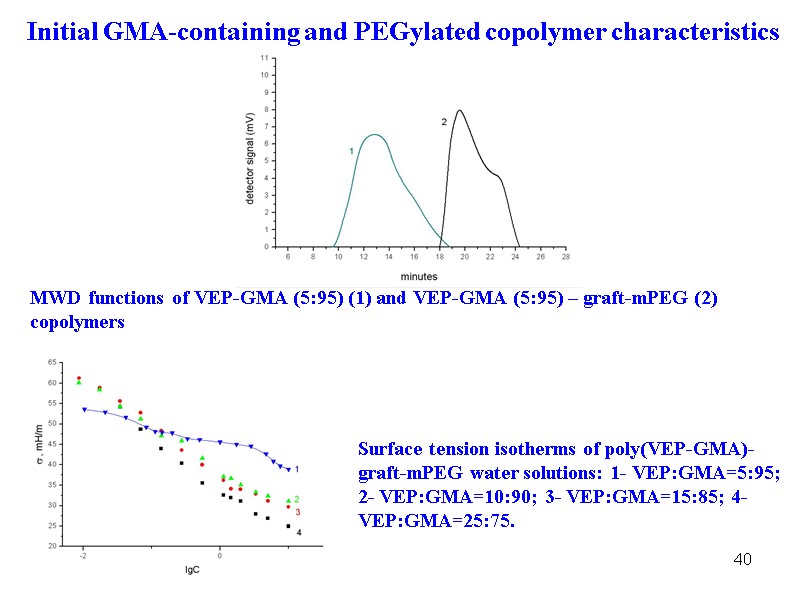
40 MWD functions of VEP-GMA (5:95) (1) and VEP-GMA (5:95) – graft-mPEG (2) copolymers Surface tension isotherms of poly(VEP-GMA)-graft-mPEG water solutions: 1- VEP:GMA=5:95; 2- VEP:GMA=10:90; 3- VEP:GMA=15:85; 4- VEP:GMA=25:75. Initial GMA-containing and PEGylated copolymer characteristics
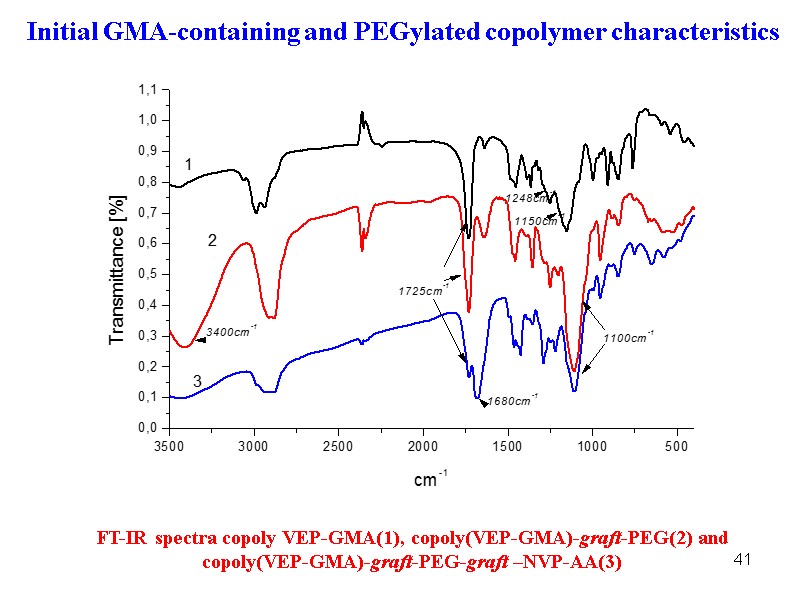
41 Initial GMA-containing and PEGylated copolymer characteristics FT-IR spectra copoly VEP-GMA(1), copoly(VEP-GMA)-graft-PEG(2) and copoly(VEP-GMA)-graft-PEG-graft –NVP-AA(3)

42 42 controlled design of a structure controlled molecular weight (1,000 – 30,000g/mole) narrowed molecular weight distribution controlled macro and microstructure controlled functionality and reactivity controlled solubility, surface activity biocompatibility and non toxicity Why these oligoperoxide based oligoelectrolytes? Capability to form free radicals and initiate radical reactions

43 2. Study of drug and nucleic acid conjugation with oligomer carriers as well as water based nanosized delivery systems. Structural and colloidal-chemical characteristics of the conjugates.
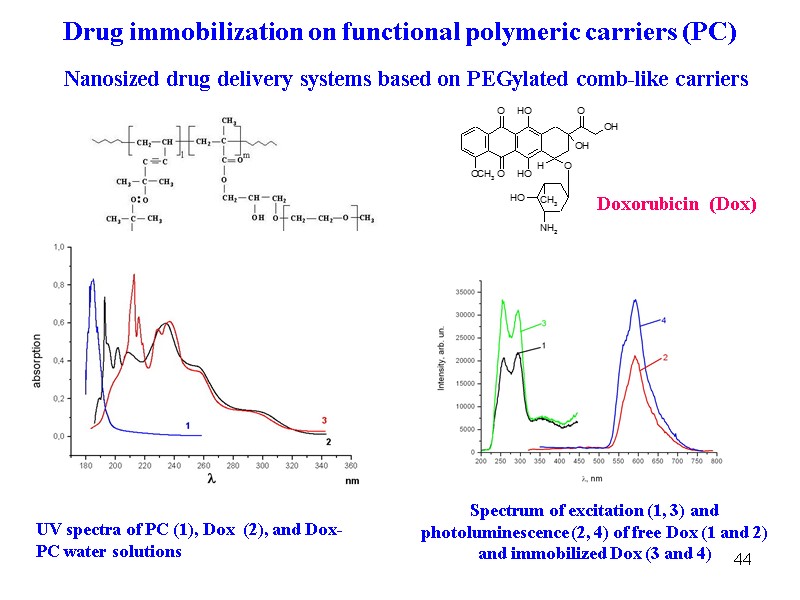
44 Nanosized drug delivery systems based on PEGylated comb-like carriers UV spectra of PC (1), Dox (2), and Dox-PC water solutions Spectrum of excitation (1, 3) and photoluminescence (2, 4) of free Dox (1 and 2) and immobilized Dox (3 and 4) Drug immobilization on functional polymeric carriers (PC) Doxorubicin (Dox)
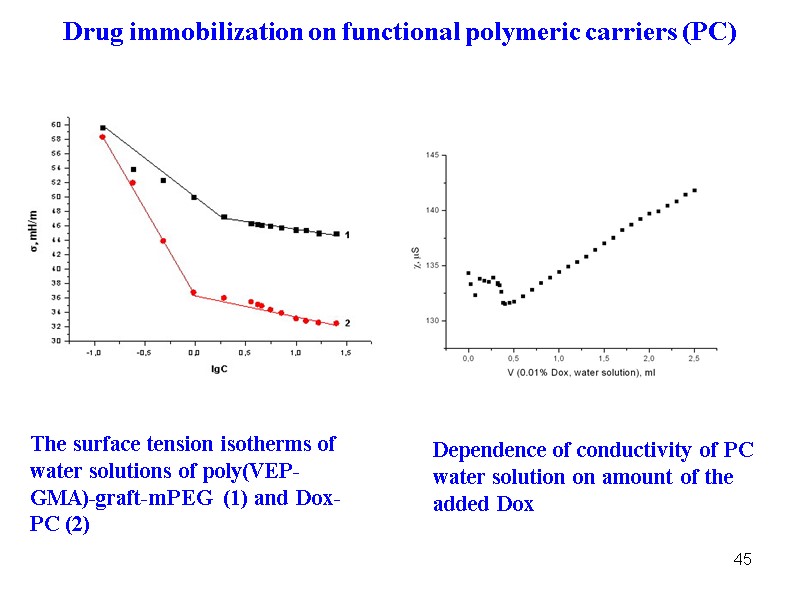
45 The surface tension isotherms of water solutions of poly(VEP-GMA)-graft-mPEG (1) and Dox-PC (2) Dependence of conductivity of PC water solution on amount of the added Dox Drug immobilization on functional polymeric carriers (PC)
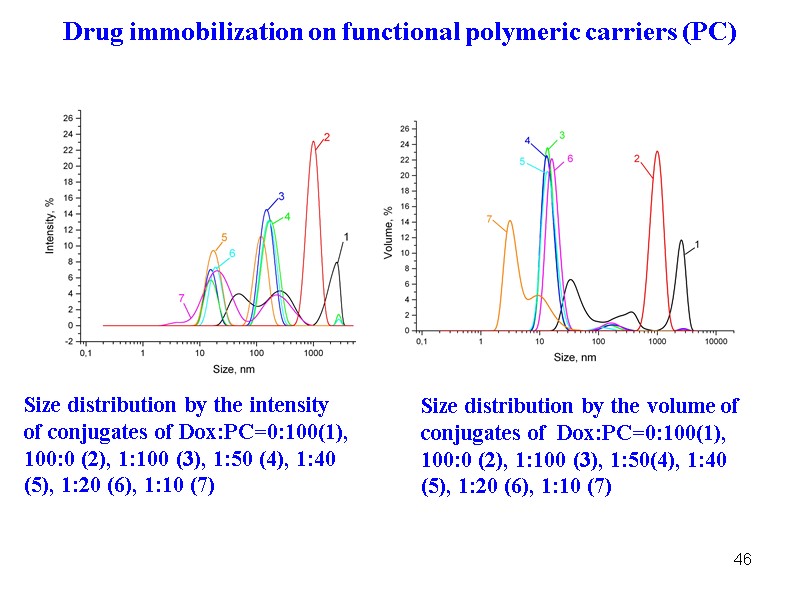
46 Size distribution by the intensity of conjugates of Dox:PC=0:100(1), 100:0 (2), 1:100 (3), 1:50 (4), 1:40 (5), 1:20 (6), 1:10 (7) Size distribution by the volume of conjugates of Dox:PC=0:100(1), 100:0 (2), 1:100 (3), 1:50(4), 1:40 (5), 1:20 (6), 1:10 (7) Drug immobilization on functional polymeric carriers (PC)
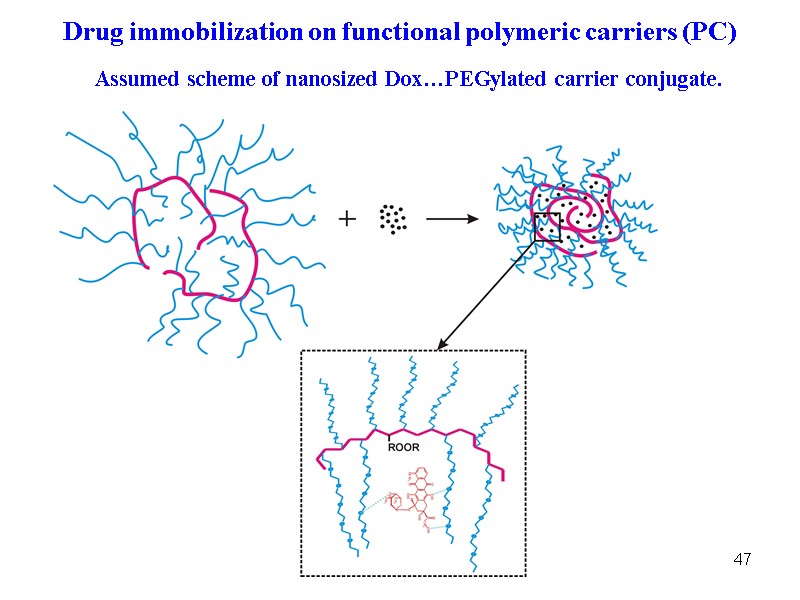
47 Assumed scheme of nanosized Dox…PEGylated carrier conjugate. Drug immobilization on functional polymeric carriers (PC)

48 Other Applications – Delivery of WATER-INSOLUBLE DRUGS
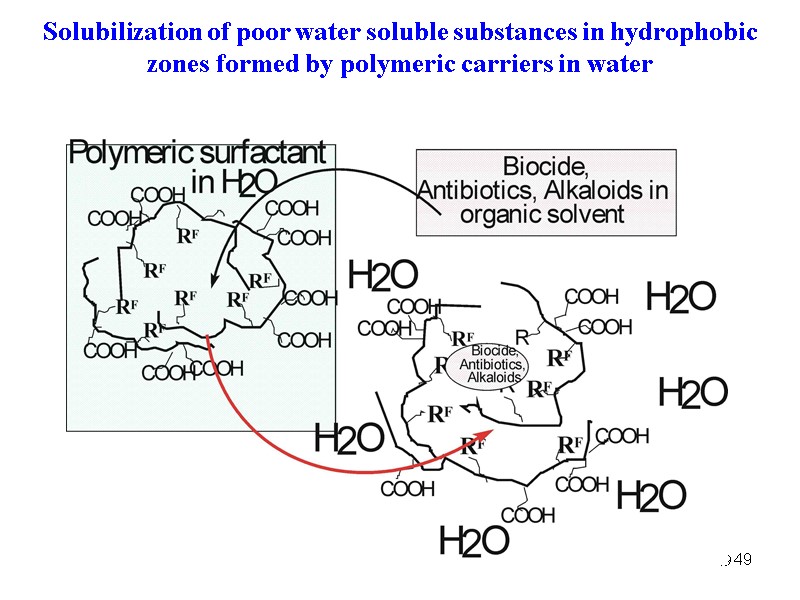
49 49 Solubilization of poor water soluble substances in hydrophobic zones formed by polymeric carriers in water
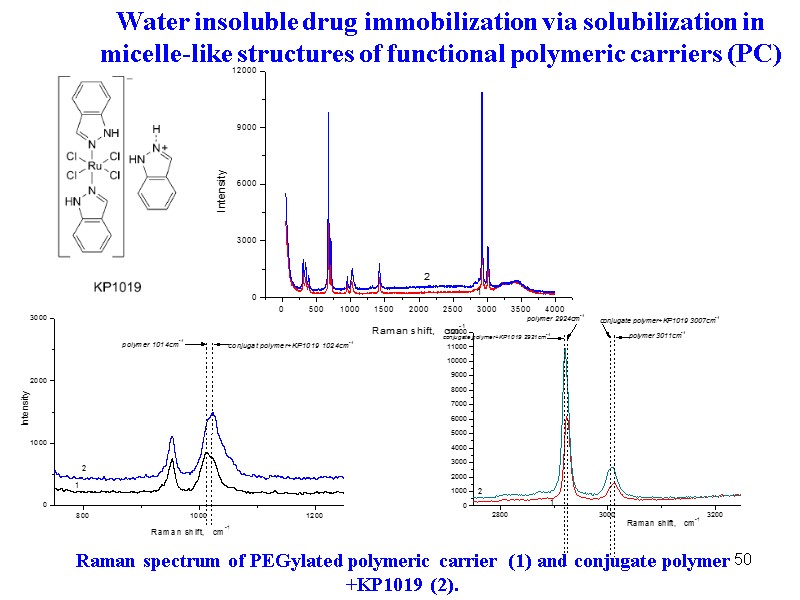
50 Raman spectrum of PEGylated polymeric carrier (1) and conjugate polymer +KP1019 (2). Water insoluble drug immobilization via solubilization in micelle-like structures of functional polymeric carriers (PC)
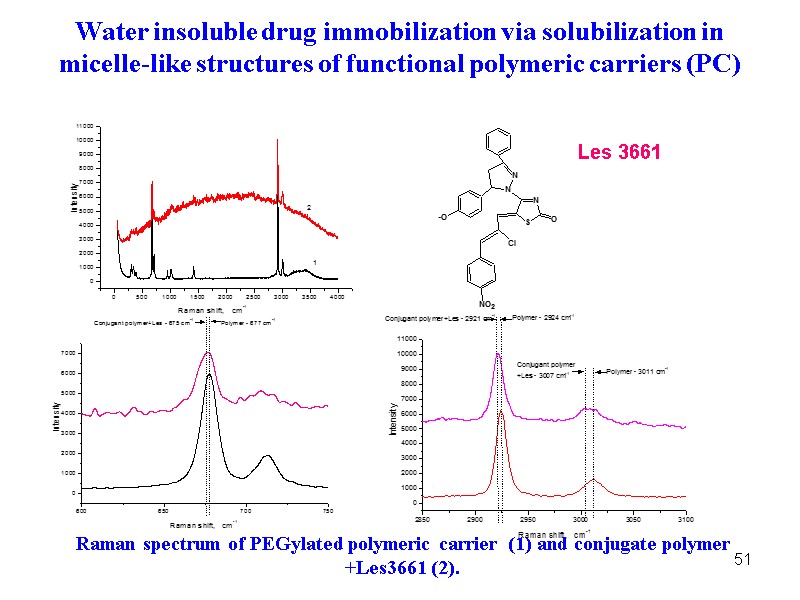
51 Raman spectrum of PEGylated polymeric carrier (1) and conjugate polymer +Les3661 (2). Les 3661 Water insoluble drug immobilization via solubilization in micelle-like structures of functional polymeric carriers (PC)
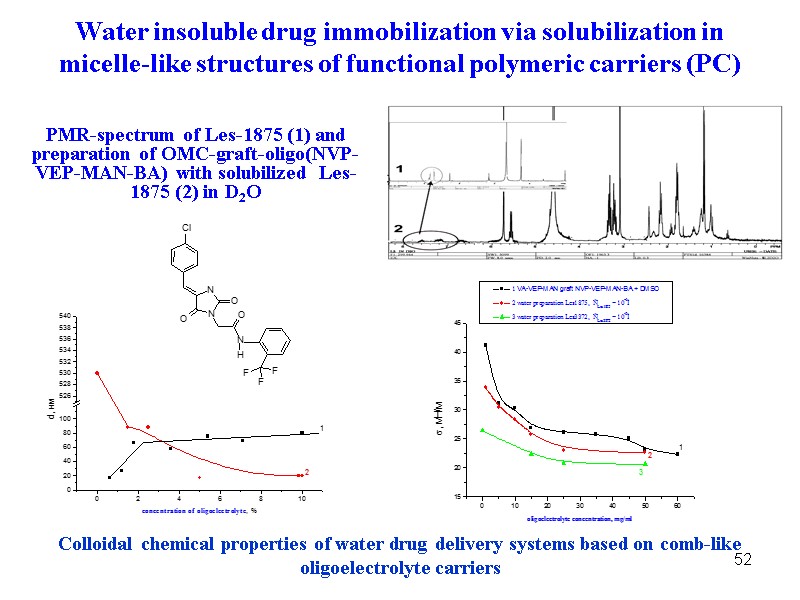
52 52 Colloidal chemical properties of water drug delivery systems based on comb-like oligoelectrolyte carriers PMR-spectrum of Les-1875 (1) and preparation of OMC-graft-oligo(NVP-VEP-MAN-BA) with solubilized Les-1875 (2) in D2O Water insoluble drug immobilization via solubilization in micelle-like structures of functional polymeric carriers (PC)
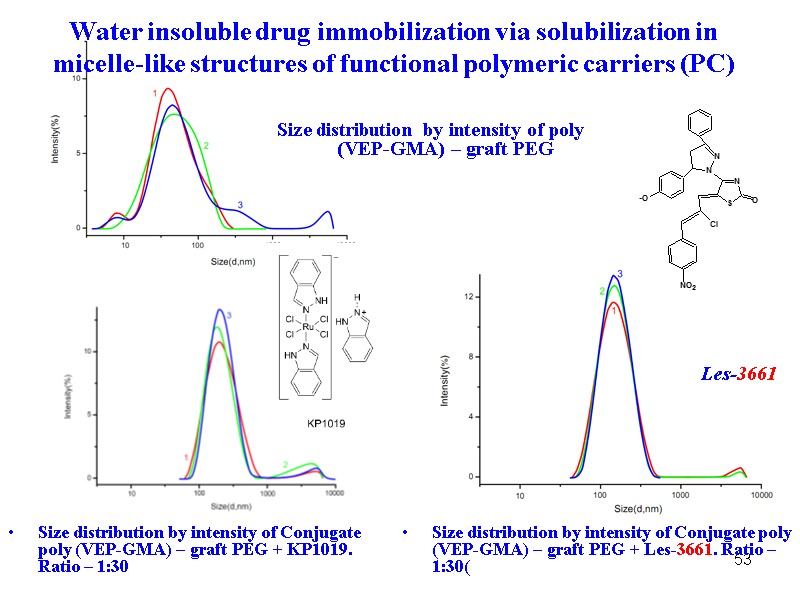
53 Size distribution by intensity of Conjugate poly (VEP-GMA) – graft PEG + Les-3661. Ratio – 1:30( Size distribution by intensity of poly (VEP-GMA) – graft PEG Size distribution by intensity of Conjugate poly (VEP-GMA) – graft PEG + KP1019. Ratio – 1:30 Les-3661 Water insoluble drug immobilization via solubilization in micelle-like structures of functional polymeric carriers (PC)
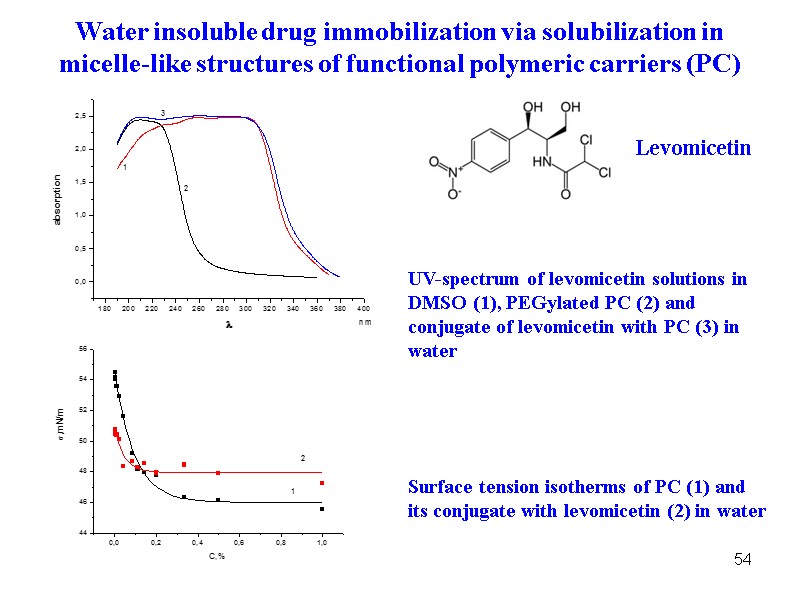
54 UV-spectrum of levomicetin solutions in DMSO (1), PEGylated PC (2) and conjugate of levomicetin with PC (3) in water Surface tension isotherms of PC (1) and its conjugate with levomicetin (2) in water Water insoluble drug immobilization via solubilization in micelle-like structures of functional polymeric carriers (PC) Levomicetin
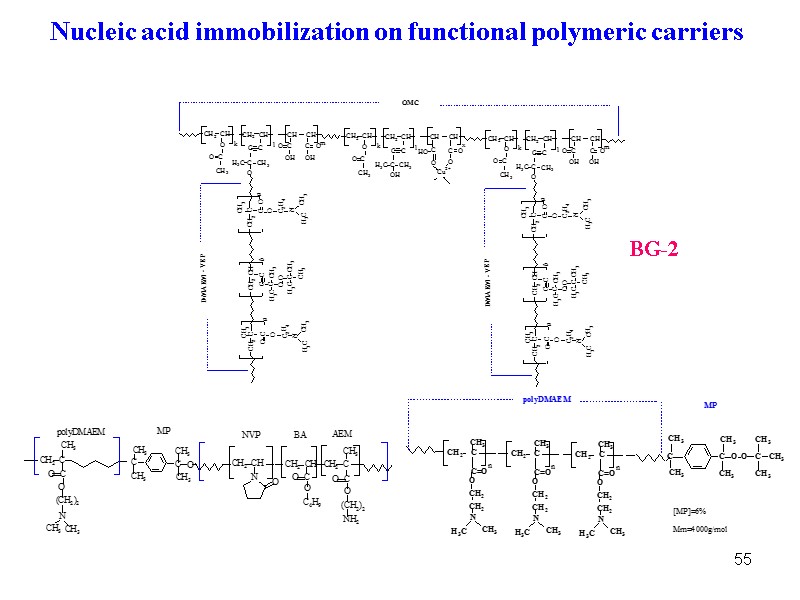
55 BG-2 Nucleic acid immobilization on functional polymeric carriers
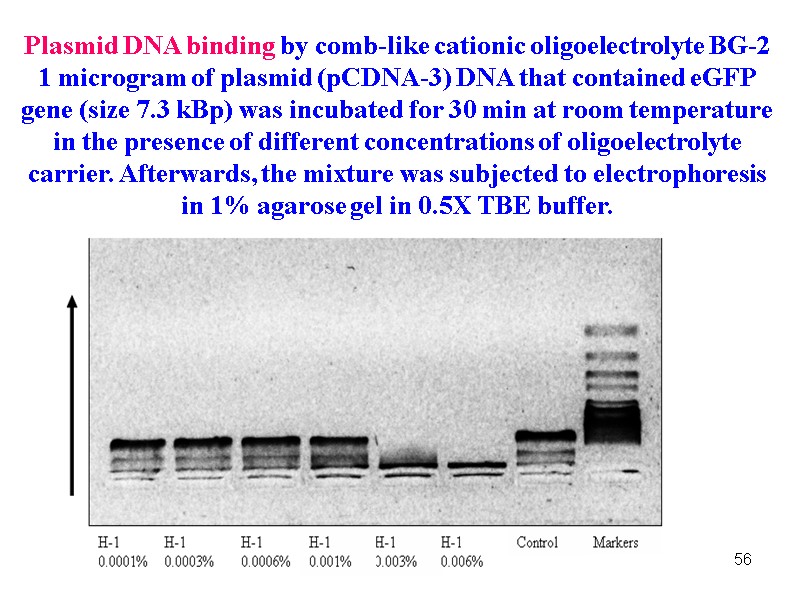
56 Plasmid DNA binding by comb-like cationic oligoelectrolyte BG-2 1 microgram of plasmid (pCDNA-3) DNA that contained eGFP gene (size 7.3 kBp) was incubated for 30 min at room temperature in the presence of different concentrations of oligoelectrolyte carrier. Afterwards, the mixture was subjected to electrophoresis in 1% agarose gel in 0.5Х TBE buffer.
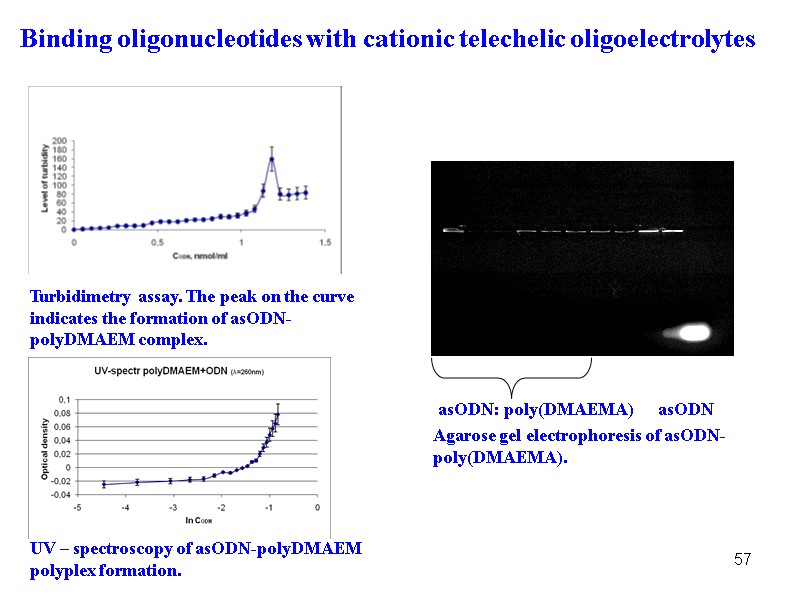
57 Turbidimetry assay. The peak on the curve indicates the formation of asODN-polyDMAEM complex. UV – sрectroscopy of asODN-polyDMAEM polyplex formation. Binding oligonucleotides with cationic telechelic oligoelectrolytes
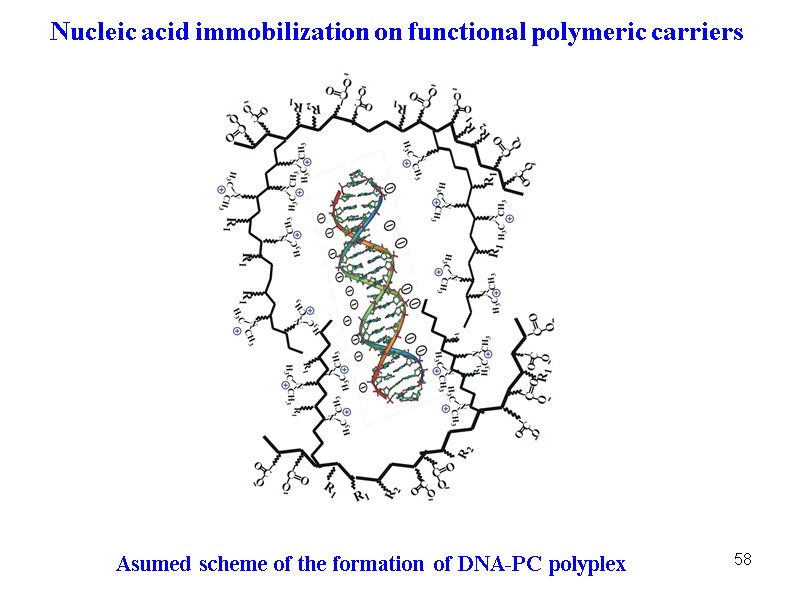
58 Asumed scheme of the formation of DNA-PC polyplex Nucleic acid immobilization on functional polymeric carriers

59 4. Cellular study and biomedical application of developed drug and gene delivery systems *study was fulfilled by team of Institute of Cell Biology of NASU under guidance of professor R. Stoika
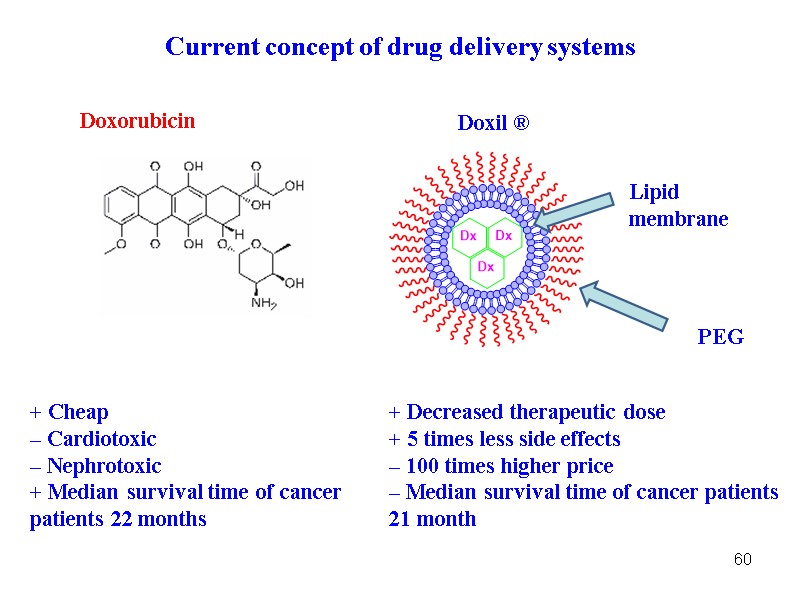
60 Current concept of drug delivery systems Lipid membrane PEG Doxorubicin Doxil ® + Cheap – Cardiotoxic – Nephrotoxic + Median survival time of cancer patients 22 months + Decreased therapeutic dose + 5 times less side effects – 100 times higher price – Median survival time of cancer patients 21 month

61 How to enhance therapeutic efficiency of drug delivery systems? 1) Increase of maximum tolerated dose of drug for more successful killing of non-proliferating tumor cells 2) Usage of cytoprotector to protect normal tissues from toxic side effects of Dx 3) Stable and effective platform for drug delivery polymeric carrier containing monoperoxine, vinylacetate and maleic anhydride residues

62 In vitro study of cellular and molecular mechanisms of anticancer activity of novel Dx nanocomposites, functionalized with NAE
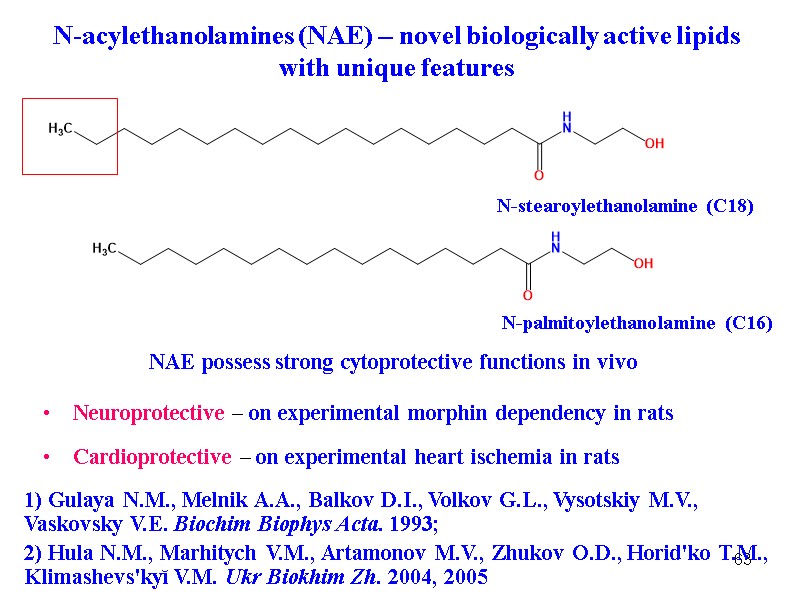
63 N-acylethanolamines (NAE) – novel biologically active lipids with unique features 1) Gulaya N.M., Melnik A.A., Balkov D.I., Volkov G.L., Vysotskiy M.V., Vaskovsky V.E. Biochim Biophys Acta. 1993; 2) Hula N.M., Marhitych V.M., Artamonov M.V., Zhukov O.D., Horid'ko T.M., Klimashevs'kyĭ V.M. Ukr Biokhim Zh. 2004, 2005 N-stearoylethanolamine (C18) N-palmitoylethanolamine (C16) NAE possess strong cytoprotective functions in vivo Neuroprotective – on experimental morphin dependency in rats Cardioprotective – on experimental heart ischemia in rats
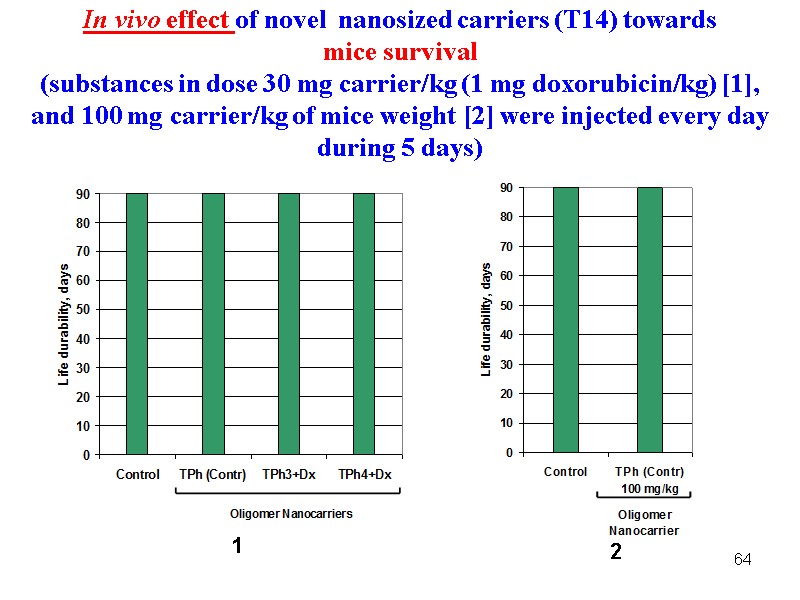
64 In vivo effect of novel nanosized carriers (Т14) towards mice survival (substances in dose 30 mg carrier/kg (1 mg doxorubicin/kg) [1], and 100 mg carrier/kg of mice weight [2] were injected every day during 5 days) 1 2
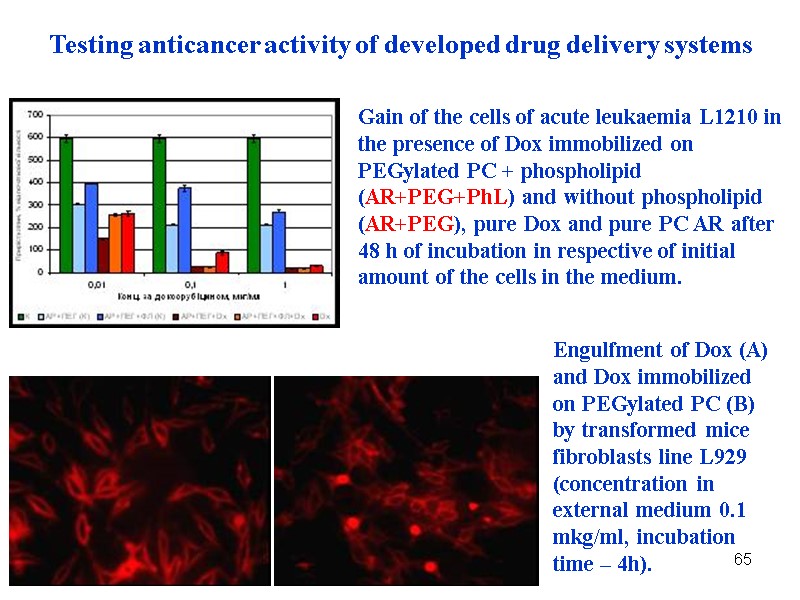
65 Gain of the cells of acute leukaemia L1210 in the presence of Dox immobilized on PEGylated PC + phospholipid (AR+PEG+PhL) and without phospholipid (AR+PEG), pure Dox and pure PC AR after 48 h of incubation in respective of initial amount of the cells in the medium. Engulfment of Dox (A) and Dox immobilized on PEGylated PC (B) by transformed mice fibroblasts line L929 (concentration in external medium 0.1 mkg/ml, incubation time – 4h). Testing anticancer activity of developed drug delivery systems
66
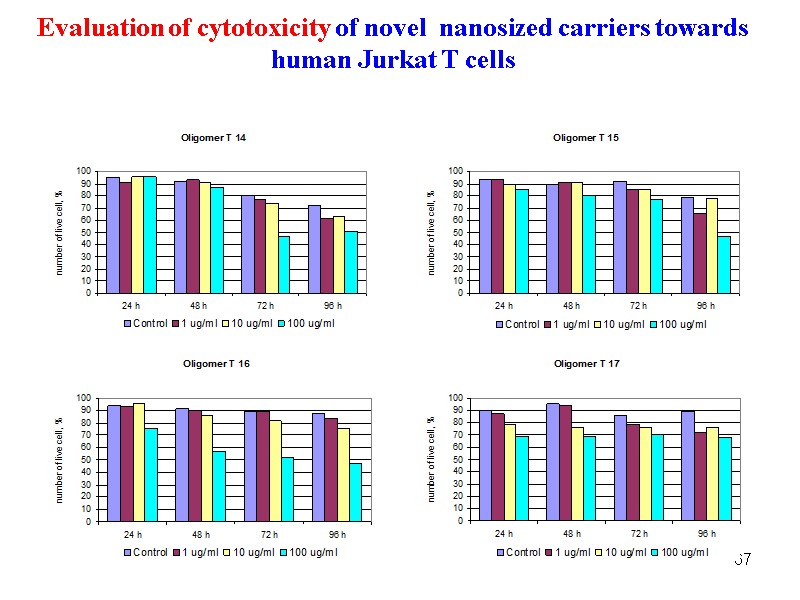
67 Evaluation of cytotoxicity of novel nanosized carriers towards human Jurkat T cells
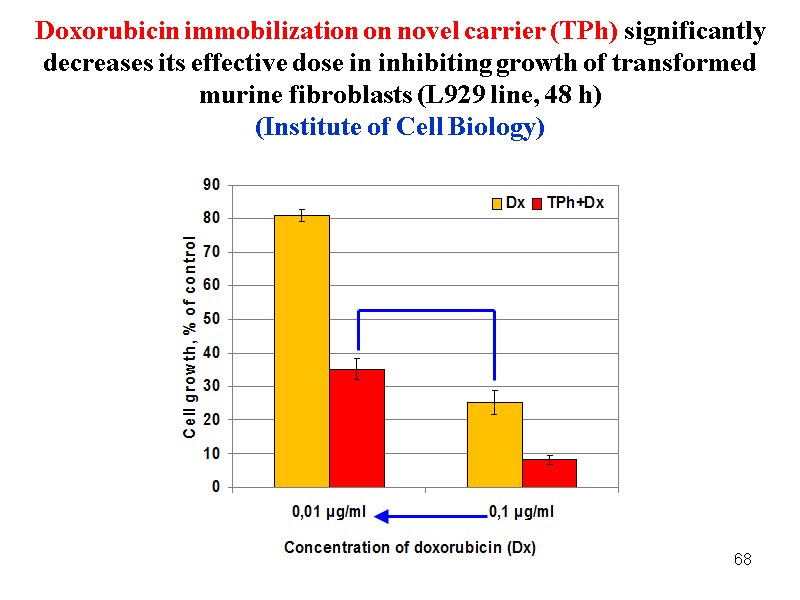
68 Doxorubicin immobilization on novel carrier (TPh) significantly decreases its effective dose in inhibiting growth of transformed murine fibroblasts (L929 line, 48 h) (Institute of Cell Biology)
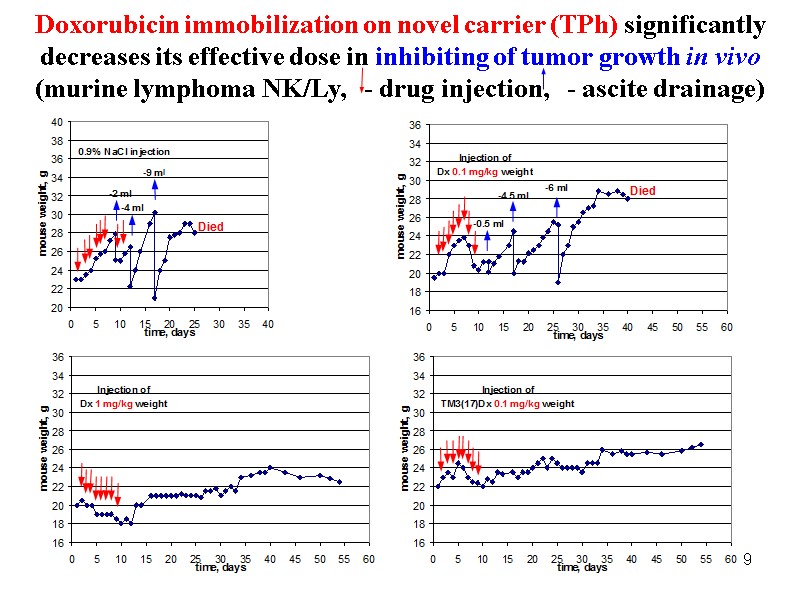
69 Doxorubicin immobilization on novel carrier (TPh) significantly decreases its effective dose in inhibiting of tumor growth in vivo (murine lymphoma NK/Ly, - drug injection, - ascite drainage)
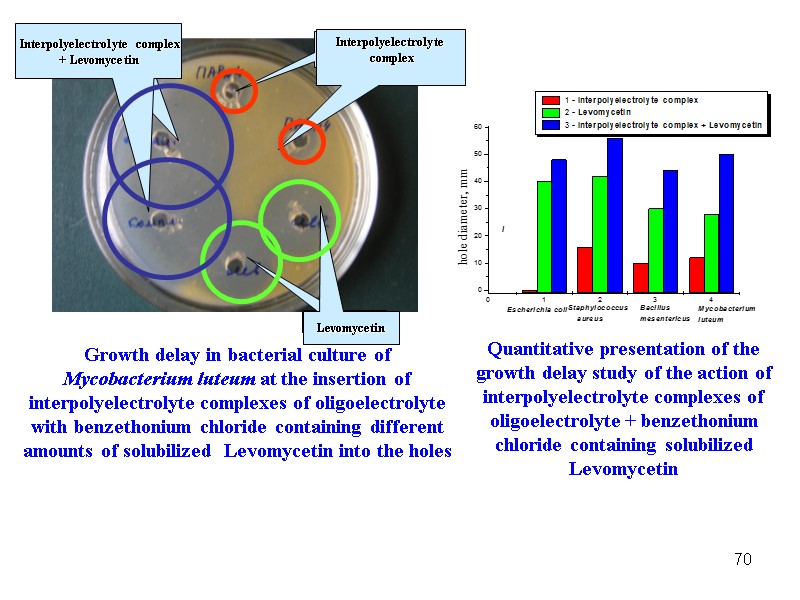
70 Growth delay in bacterial culture of Mycobacterium luteum at the insertion of interpolyelectrolyte complexes of oligoelectrolyte with benzethonium chloride containing different amounts of solubilized Levomycetin into the holes Quantitative presentation of the growth delay study of the action of interpolyelectrolyte complexes of oligoelectrolyte + benzethonium chloride containing solubilized Levomycetin

71 Conclusions coming from drug delivery experiments

72 Application of the developed polymeric drug delivery platforms permitted: to decrease acting concentrations of highly toxic anticancer drugs (ex. Doxorubicin) from 50% to 100 times (in vitro and in vivo, depending on specific target tumor cells and drugs); this result predicts the corresponding decrease of potential negative side effects of these drugs in the treated organism.
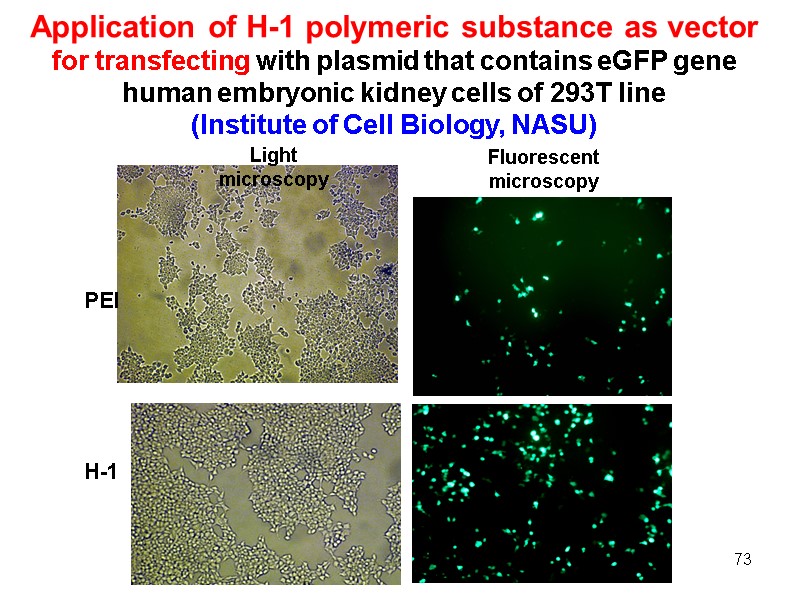
73 Application of H-1 polymeric substance as vector for transfecting with plasmid that contains eGFP gene human embryonic kidney cells of 293T line (Institute of Cell Biology, NASU) Light microscopy Fluorescent microscopy РЕІ H-1
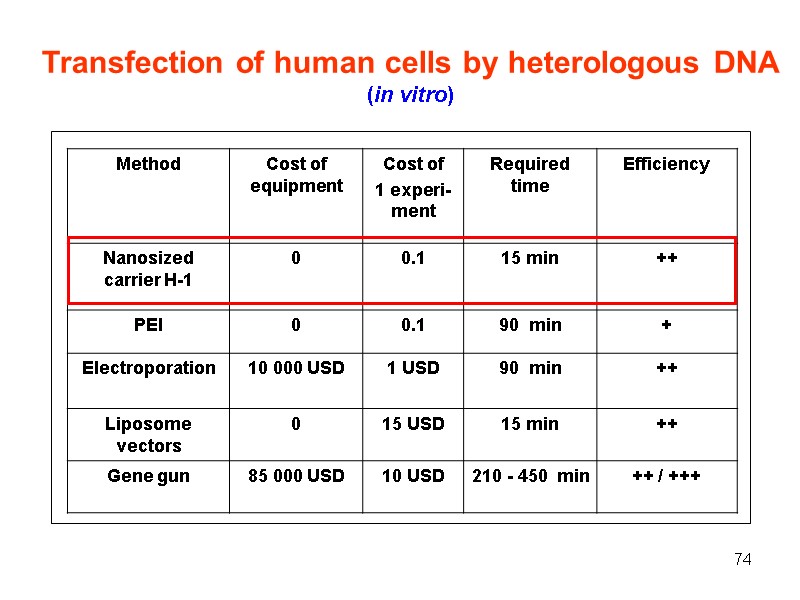
74 Transfection of human cells by heterologous DNA (in vitro)

75 Intelectual Property PCT Application No IB2010/001538 USA Provisional Patent Highly efficient systems for delivery of nucleic acids Rostyslav Stoika, Yevhen Filyak, Oleksandr Zaichenko, Nataliya Mitina
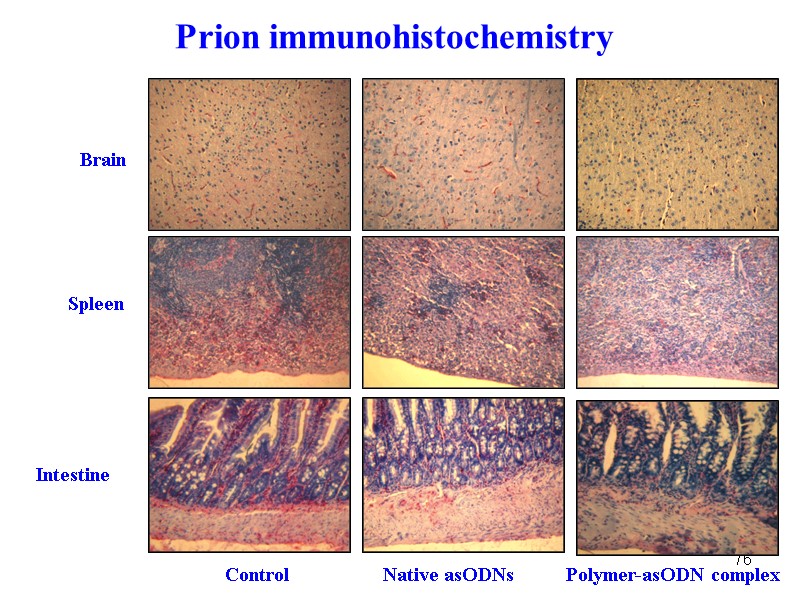
76 Prion immunohistochemistry Brain Spleen Intestine Control Native asODNs Polymer-asODN complex
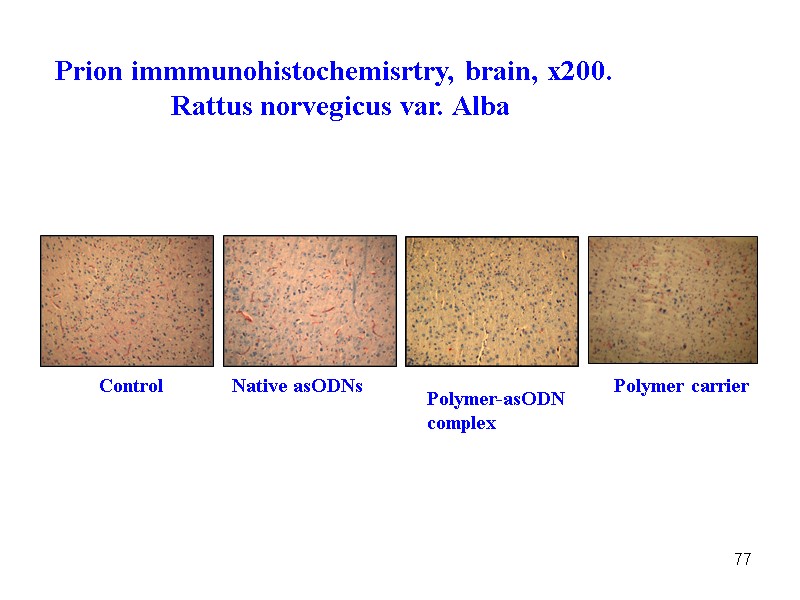
77 Control Native asODNs Polymer-asODN complex Polymer carrier Prion immmunohistochemisrtry, brain, x200. Rattus norvegicus var. Alba

78 78 Many thanks to my team and partners Dr. N. Mitina, Dr. K. Rayevska, Dr. T. Skorokhoda, PhD student A. Riabtseva; Professor R. Stoika, Dr. N. Boiko, Dr. E. Filyak and team from Cell Biology Institute of NASU, PhD students L. Ivanitska, N. Finyuk; for the experimental work as well as for the collaboration, ideas and discussion Acknowledgements
24046-munich_nancy_zaichenko-last2.ppt
- Количество слайдов: 78

