913aa237ef88329eaecdc106c534bbcc.ppt
- Количество слайдов: 87

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی WATER-The Substance of Life • Limits Kinds and Amounts of Vegetation on Earth • Limits Growth of Cities and Kinds of Industry Isfahan University of Technology

EARTH’S WATER SUPPLY Ø 70% of Earth’s Surface covered by water Ø 97% of Earth’s water supply in oceans ØIce at Polar Caps next most abundant supply (>2%) ØGroundwater next most abundant supply (~0. 5%); approximately 50% of ground water is > 0. 5 miles deep ØFresh water in lakes, ponds and streams ~0. 008% ØSoil and atmospheric water ~ 0. 001% ØBiological water ~0. 0001% ØAverage annual rainfall on land ~ 30 inches Isfahan University of Technology ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی
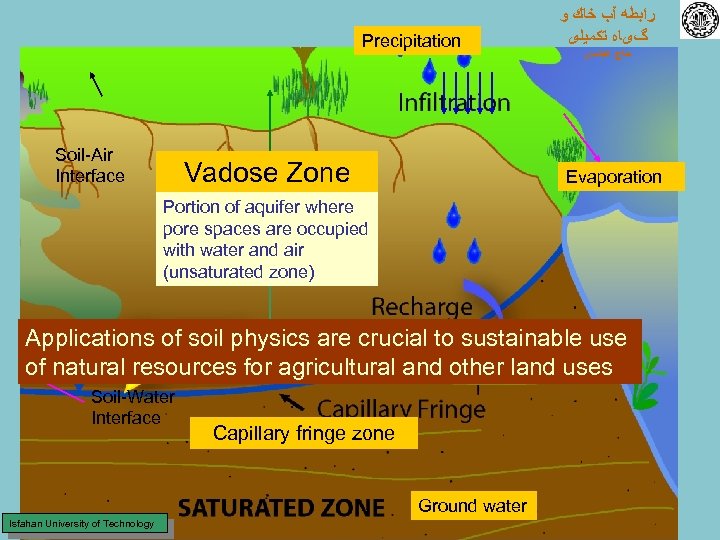
Precipitation Soil-Air Interface Vadose Zone ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Evaporation Portion of aquifer where pore spaces are occupied with water and air (unsaturated zone) Applications of soil physics are crucial to sustainable use of natural resources for agricultural and other land uses Soil-Water Interface Capillary fringe zone Ground water Isfahan University of Technology
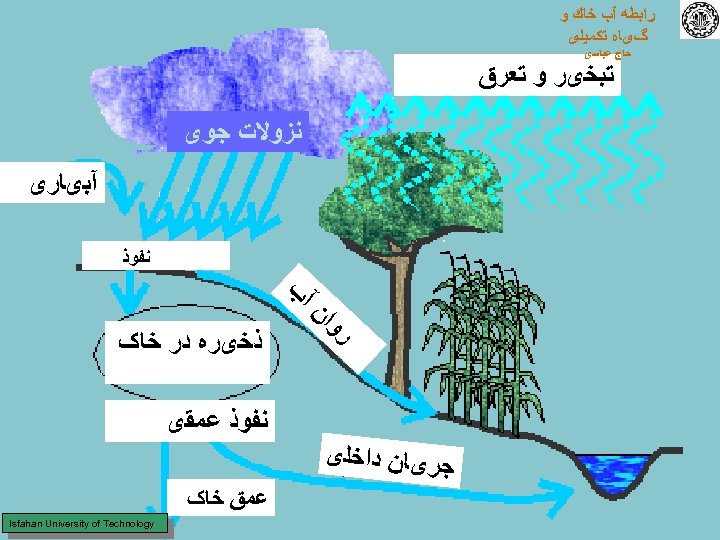
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی ﺗﺒﺨیﺮ ﻭ ﺗﻌﺮﻕ ﻧﺰﻭﻻﺕ ﺟﻮی آﺒیﺎﺭی ﺍﻥ ﺭﻭ ﻧﻔﻮﺫ آﺐ ﺫﺧیﺮﻩ ﺩﺭ ﺧﺎک ﻧﻔﻮﺫ ﻋﻤﻘی ﺟﺮیﺎﻥ ﺩﺍﺧﻠی ﻋﻤﻖ ﺧﺎک Isfahan University of Technology

Precipitation/Evaporation ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی • P/E>0. 75= Humid (Forests) • P/E >0. 5= Sub-Humid (Mixed Forest and Grasslands) • P/E >0. 25= Semi-Arid (Mixed Grasslands and Semi-Deserts) • P/E <0. 25= Arid (Deserts) Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Soil Water Relationships Isfahan University of Technology

• Bulk Density ( b) – b = soil bulk density, g/cm 3 – Ms = mass of dry soil, g – Vb = volume of soil sample, cm 3 • Typical values: 1. 1 - 1. 6 g/cm 3 • Particle Density ( p) – – – P = soil particle density, g/cm 3 Ms = mass of dry soil, g Vs = volume of solids, cm 3 • Typical values: 2. 6 - 2. 7 g/cm 3 Isfahan University of Technology ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی • Porosity ( ) • Typical values: 30 - 60% Isfahan University of Technology ﺣﺎﺝ ﻋﺒﺎﺳی

Water in Soils • Soil water content – Mass water content ( m) – m = mass water content (fraction) – Mw = mass of water evaporated, g ( 24 hours @ 105 o. C) – Ms = mass of dry soil, g Isfahan University of Technology ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی

• Volumetric water content ( v) ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی V = volumetric water content (fraction) Vw = volume of water Vb = volume of soil sample At saturation, V = As m As = apparent soil specific gravity = b/ w ( w = density of water = 1 g/cm 3) – As = b numerically when units of g/cm 3 are used – – – • Equivalent depth of water (d) – d = volume of water per unit land area = ( v A L) / A = v L – d = equivalent depth of water in a soil layer – L = depth (thickness) of the soil layer Isfahan University of Technology
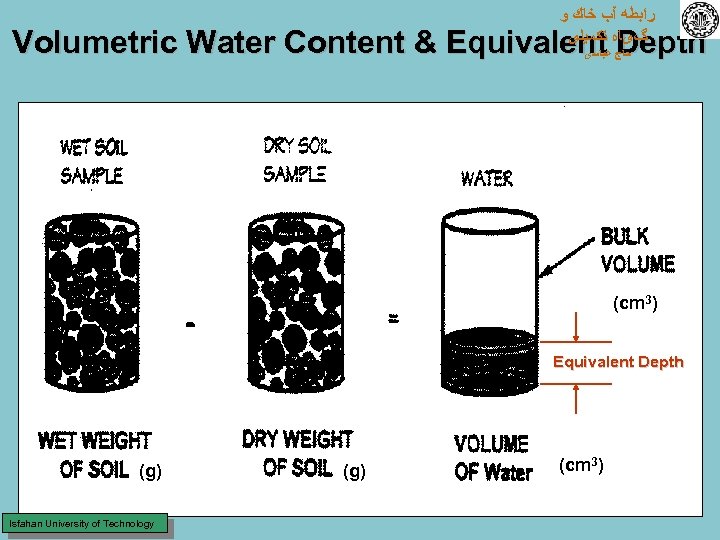
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Volumetric Water Content & Equivalent Depth ﺣﺎﺝ ﻋﺒﺎﺳی (cm 3) Equivalent Depth (g) Isfahan University of Technology (g) (cm 3)
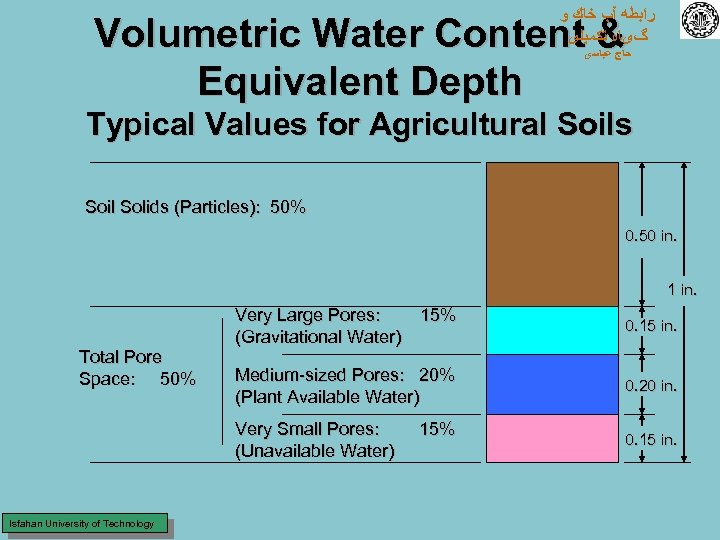
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Volumetric Water Content & Equivalent Depth ﺣﺎﺝ ﻋﺒﺎﺳی Typical Values for Agricultural Soils Soil Solids (Particles): 50% 0. 50 in. 1 in. Total Pore Space: 50% Very Large Pores: (Gravitational Water) 15% 0. 15 in. 0. 20 in. Very Small Pores: (Unavailable Water) Isfahan University of Technology Medium-sized Pores: 20% (Plant Available Water) 0. 15 in. 15%
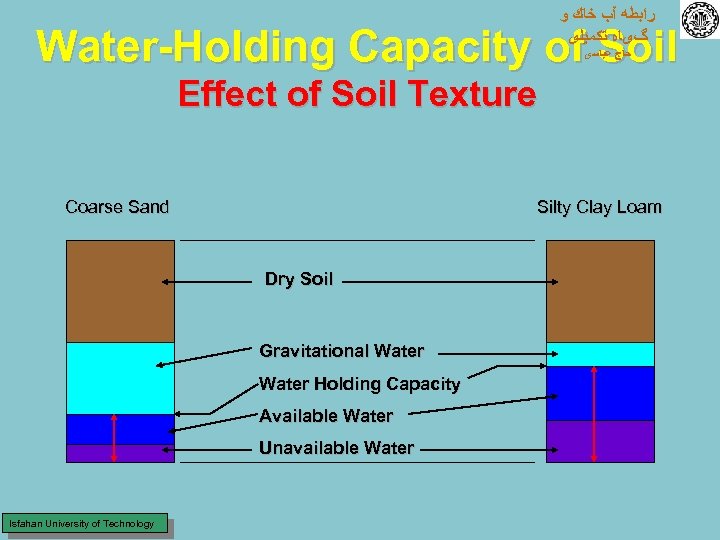
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Water-Holding Capacity of Soil ﺣﺎﺝ ﻋﺒﺎﺳی Effect of Soil Texture Coarse Sand Silty Clay Loam Dry Soil Gravitational Water Holding Capacity Available Water Unavailable Water Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Soil Water Content ﺣﺎﺝ ﻋﺒﺎﺳی Soil Moisture Content Water that may be evaporated from soil by heating at 1050 C to a constant weight Gravimetric moisture content (w) = Volumetric moisture content ( ) = mass of water evaporated (g) mass of dry soil (g) volume of water evaporated (cm 3) volume of soil (cm 3) = w* bulk density of soil density of water Bulk density of soil ( ) = mass of dry soil (g) volume of soil (cm 3) Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Soil Moisture Content: Methods of Measurement ﺣﺎﺝ ﻋﺒﺎﺳی 1. Difficulties encountered for accurate moisture measurement in the field: 2. Soils are highly variable 3. Soil moisture is highly dynamic (spatial temporal variability) 4. Plant water uptake is highly variable depending upon the stage of growth 5. State of growth is again dependent upon nutrient application, water availability, pests etc. 6. Chemicals present in the soil can make measurements unreliable 7. Costs involved Isfahan University of Technology
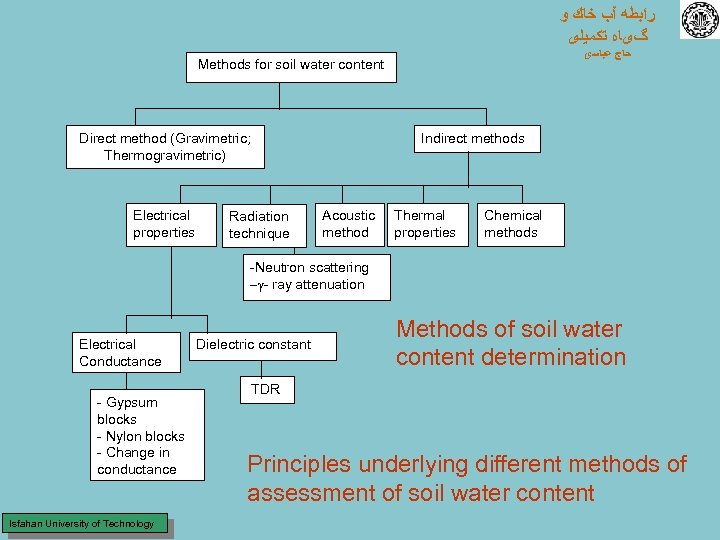
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Methods for soil water content Direct method (Gravimetric; Thermogravimetric) Electrical properties Indirect methods Radiation technique Acoustic method Thermal properties Chemical methods -Neutron scattering - - ray attenuation Electrical Conductance - Gypsum blocks - Nylon blocks - Change in conductance Isfahan University of Technology Dielectric constant Methods of soil water content determination TDR Principles underlying different methods of assessment of soil water content

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی DIRECT ﺣﺎﺝ ﻋﺒﺎﺳی Gravimetric: evaporating water at 1050 C. Thermogravimetric: Soil sample is weighted and saturated with alcohol and burned several times until a constant dry weight is obtained INDIRECT Electrical Conductance Isfahan University of Technology

DIRECT ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Gravimetric: evaporating water at 1050 C. Feel Method: Thermogravimetric: Soil sample is weighted and saturated with alcohol and burned several times until a constant dry weight is obtained Advantages: ensures accurate measurements, not dependent on salinity and soil type, easy to calculate Disadvantage: destructive test, time consuming, inapplicable to automatic control, must know dry bulk density to transform data to volume moisture content, inaccurate because of soil variability There are many classifications for soil types and major differences within each classification Soil management can have a major impact upon these soil properties. Compaction is the major cause of error in bulk density. Isfahan University of Technology http: //edis. ifas. ufl. edu/

Frequency Domain Reflectometry: radio frequency (RF) capacitance techniques ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Actually measures soil capacitance A pair of electrodes is inserted into the soil Soil acts as the dielectric completing a capacitance circuit, which is part of a feedback loop of a high frequency transistor oscillator As high frequency radio waves (about 150 MHz) are pulsed through the capacitance circuitry, a natural resonant frequency is established which is dependent on the soil capacitance, which is related to the dielectric constant by the geometry of the electric field established around the electrodes Two commercially available instruments using this technique: the Troxler Sentry 200 -AP probe and the Aquaterr probe Isfahan University of Technology

Time Domain Reflectometry (TDR): , 28 s ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی The soil bulk dielectric constant (K) is determined by measuring the time it takes for an electromagnetic pulse (wave) to propagate along a transmission line (L) that is surrounded by the soil Since the propagation velocity (v) is a function of K, the latter is therefore proportional to the square of the transit time (t, in seconds) down and back along the L K = (c/v)2 = ((c. t)/(2. L))2 where c is the velocity of electromagnetic waves in a vacuum (3 • 108 m/s or 186, 282 mile/s) and L is the length embedded in the soil (in m or ft) Isfahan University of Technology
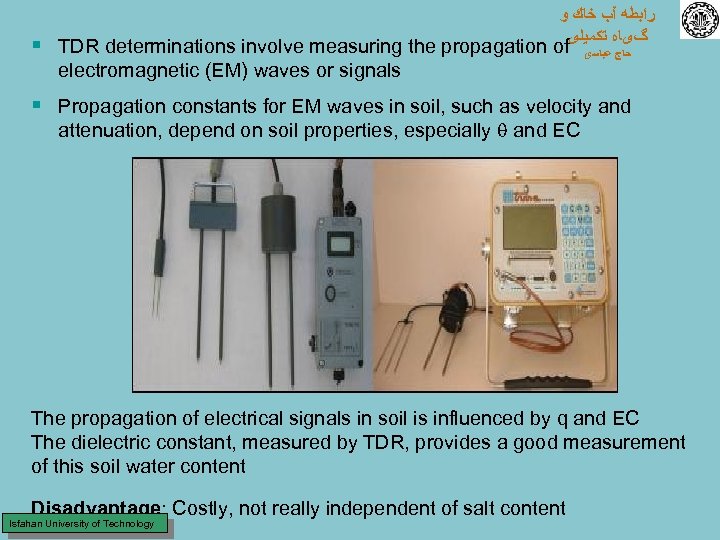
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی § TDR determinations involve measuring the propagation of electromagnetic (EM) waves or signals ﺣﺎﺝ ﻋﺒﺎﺳی § Propagation constants for EM waves in soil, such as velocity and attenuation, depend on soil properties, especially and EC The propagation of electrical signals in soil is influenced by q and EC The dielectric constant, measured by TDR, provides a good measurement of this soil water content Disadvantage: Costly, not really independent of salt content Isfahan University of Technology

Time Domain Transmission (TDT) ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی This method measures the one-way time for an electromagnetic pulse to propagate along a transmission line (L). Thus, it is similar to TDR, but requires an electrical connection at the beginning and ending of the length. Notwithstanding, the circuit is simple compared with TDR instruments. Disadvantages: Reduced precision, because the generated pulse is distorted during transmission; soil disturbance during installation; needs to be permanently installed in the field Isfahan University of Technology
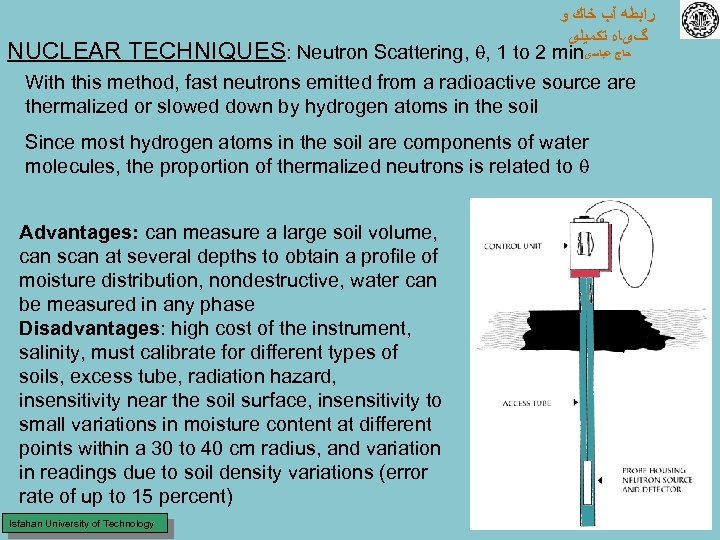
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی NUCLEAR TECHNIQUES: Neutron Scattering, , 1 to 2 min ﺣﺎﺝ ﻋﺒﺎﺳی With this method, fast neutrons emitted from a radioactive source are thermalized or slowed down by hydrogen atoms in the soil Since most hydrogen atoms in the soil are components of water molecules, the proportion of thermalized neutrons is related to Advantages: can measure a large soil volume, can scan at several depths to obtain a profile of moisture distribution, nondestructive, water can be measured in any phase Disadvantages: high cost of the instrument, salinity, must calibrate for different types of soils, excess tube, radiation hazard, insensitivity near the soil surface, insensitivity to small variations in moisture content at different points within a 30 to 40 cm radius, and variation in readings due to soil density variations (error rate of up to 15 percent) Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Gamma Attenuation: volumetric water content, < 1 min ﺣﺎﺝ ﻋﺒﺎﺳی v This method assumes that the scattering and absorption of gamma rays are related to the density of matter in their path v The specific gravity of a soil remains relatively constant as the wet density changes with increases or decreases in moisture v Changes in wet density are measured by the gamma transmission technique and the moisture content is determined from this density change Advantages: can determine mean water content with depth, can be automated for automatic measurements and recording, can measure temporal changes in soil water, nondestructive measurement Disadvantages: restricted to soil thickness of 1 inch or less, but with high resolution, affected by soil bulk density changes, costly and difficult to use, large errors possible when used in highly stratified soils Isfahan University of Technology
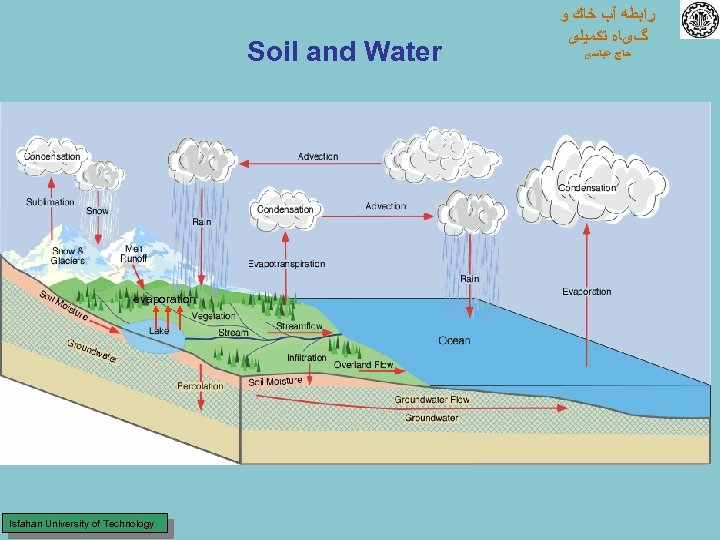
Soil and Water evaporation Isfahan University of Technology ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی

WATER CONTENT Ø LIQUID • Gravimetric (Mass/Mass) • Volumetric (Volume/Volume) • Relative Ø VAPOR • Concentration (Mass/ volume air) • Pressure (KPa) • Relative Humidity

USES of WATER in PLANTS • • • 1. Constituent 2. Solvent 3. Reactant-product 4. Turgidity 5. Temperature Control

CONSTITUENT • Water constitutes more than 70% of fresh weight of most plants (Seeds are exception) • Between 60 -90% of the water is contained in the plant cell providing both biological and physical functions. • The remaining 10 -40% is contained as liquid in cell walls providing a continuum between the soil supply and the living cell.

SOLVENT • Dissolves both organic and inorganic constituents essential for life • Dissolves gases-CO 2

REACTANT-PRODUCT • Photosynthesis CO 2+ H 2 O = SUGAR • Respiration CH 2 O + O 2 = CO 2 + H 2 O

ENERGY BALANCE • Evaporation dissipates heat • Condensation-precipitation releases heat • Amount of water passing through plants as transpiration depends on environment and species Wheat 1000 kg H 2 O per kg dry matter Cotton 5000 kg H 2 O per kg lint

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ 1 mole water ~ 18 cm 3 contains 6. 02 x 1023 molecules ﺗﻜﻤﻴﻠی 1 cm 3 ~ 3. 3 x 1022 (33 thousand billion) ﺣﺎﺝ ﻋﺒﺎﺳی Consider a beach 1. 6 x 106 m (1000 miles) long 200 m (656 feet) wide 100 m (328 feet) deep Volume beach = 3. 2 x 1010 m 3 sand Assume that each grain = a sphere 1 mm diameter With loose packing 109 (one billion) grains in 1 m 3 Entire beach 3. 2 x 1019 sand grains, 1000 < number of molecules in 1 cm 3 water It would take 1000 beaches to contain as many sand grains as molecules in 1 cm 3 of water Each water molecule is ~ 3 A (3 x 10 -10 m) It would take 33 x 106 layers to form a layer of water 1 cm deep.
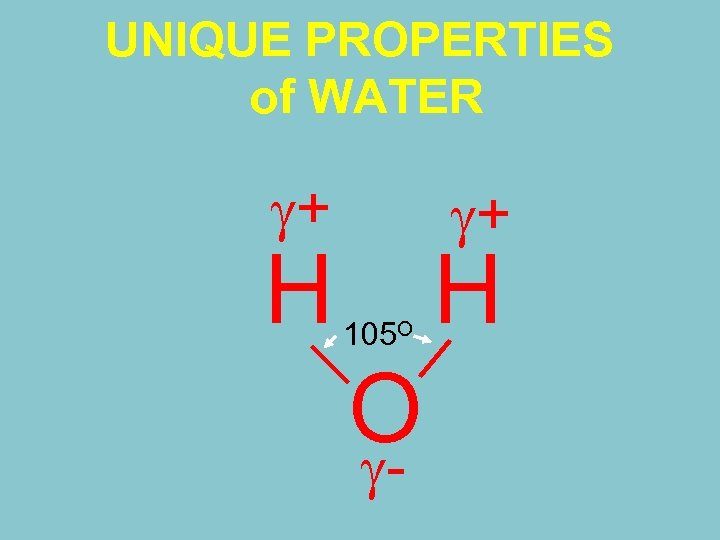
UNIQUE PROPERTIES of WATER + H + 105 O O - H
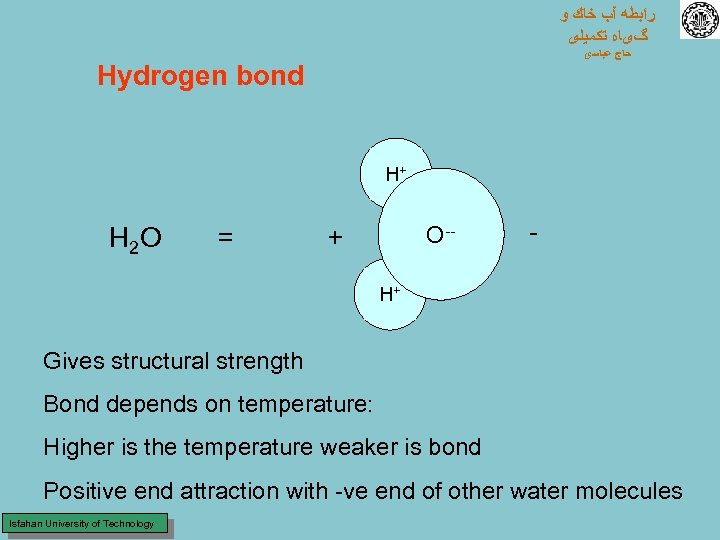
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Hydrogen bond H+ H 2 O = O-- + - H+ Gives structural strength Bond depends on temperature: Higher is the temperature weaker is bond Positive end attraction with -ve end of other water molecules Isfahan University of Technology
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی
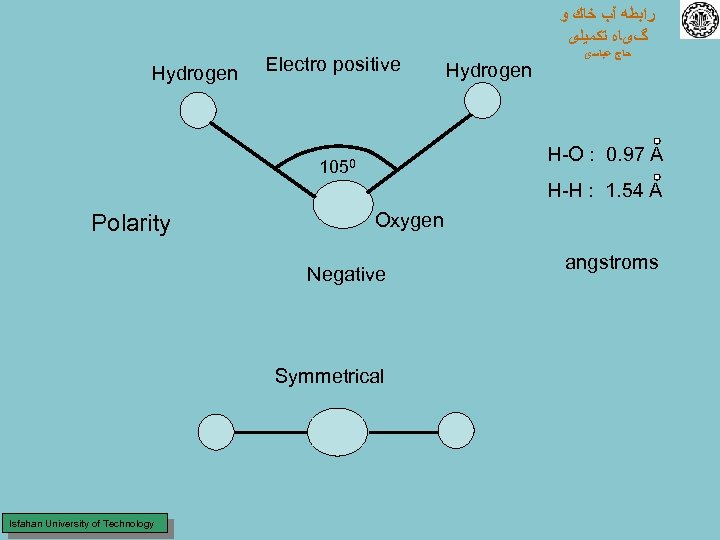
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Hydrogen Electro positive Hydrogen ﺣﺎﺝ ﻋﺒﺎﺳی H-O : 0. 97 A 1050 H-H : 1. 54 A Polarity Oxygen Negative Symmetrical Isfahan University of Technology angstroms
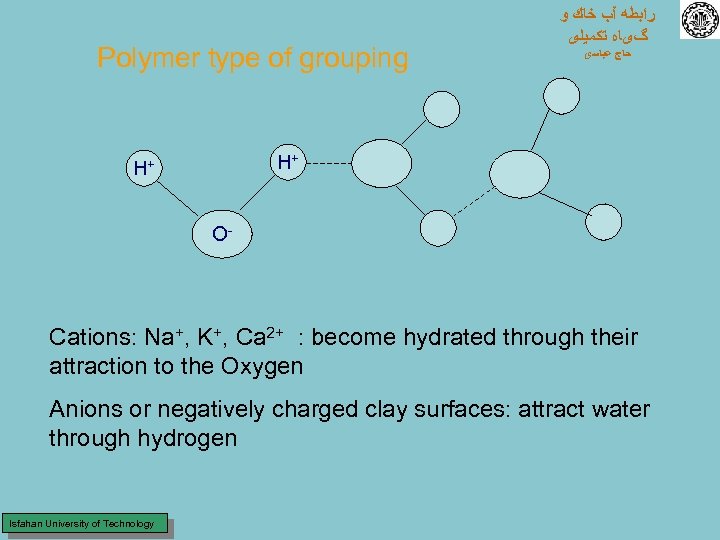
Polymer type of grouping ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی H+ H+ O- Cations: Na+, K+, Ca 2+ : become hydrated through their attraction to the Oxygen Anions or negatively charged clay surfaces: attract water through hydrogen Isfahan University of Technology
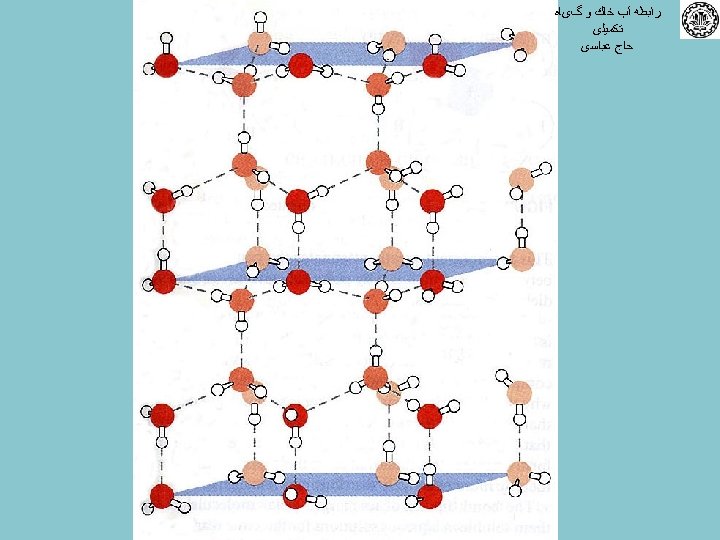
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی

Some definitions ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Adhesion - the attraction or clinging together of unlike substances Cohesion - the attraction of a substance for itself; the mutual attraction among molecules or particles comprising a substance tat allows it to cling together as a continuous mass. absorption - the process by which one substance is taken into and included within another substance, as the absorption of water by soil or nutrients by plants. adsorption - the increased concentration of molecules or ions at a surface, including exchangeable cations and anions on soil particles. flickering clusters refers to the quasicrystalline state of water molecules while in a liquid state. The molecules associate and dissociate repeatedly in transitory or flickering polymer groups. Water molecules are attracted to one another due to the hydrogen bonding that takes place between the negatively charged end of the oxygen atom in the molecule and the positively charged ends of hydrogen atoms in adjacent water molecules. Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی heat of fusion (ice) The amount of energy required to turn a liquid ﺣﺎﺝ ﻋﺒﺎﺳی a solid. into heat of vaporization The amount of energy required to turn a liquid to a vapour (to overcome the attractive forces between adjacent molecules in a liquid). dipole moment a measure of the tendency of a polar molecule to be affected by an electrical or magnetic field (i. e. , NMR - Nuclear magnetic resonance) Volumetric heat capacity is the change in the heat content of a unit volume per unit change in temperature. Specific heat is the change in the heat content of a unit mass per unit change in temperature. surface tension A molecule at the surface of a liquid is not completely surrounded by other molecules of the liquid. The forces acting upon it are unbalanced, with the result that it experiences a stronger attraction into the body of the liquid (cohesion) rather than into the less dense gaseous phase. This unbalanced force draws the surface molecules inward with and results in the tendency for the surface to contract and the molecules to be slightly denser Isfahan University of Technology at the surface.

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی sublimation the direct transition from the solid state to the vapor state hydrophobic water repellent capillary attraction - a liquid's movement over or retention by a solid surface due to the interaction of adhesive and cohesive forces. capillary fringe a zone just above the water table that is maintained in an essentially saturated state by capillary forces of lift. viscosity (centipose, c. P, N s m-2 x 10 -3, kg m-1 s-1). When a fluid is moved in shear (that is to say, when adjacent layers are fluid are made to slide over each other), the force required is proportional to the velocity of shear. Viscosity is the proportionality factor. It is the property of the fluid to resist the rate of shearing and can be visualized as an internal friction. Fluids of lower viscosity flow more readily. Thus oil has a higher viscosity than water. Fluidity is the reciprocal of viscosity. Viscosity is the preferred term. Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Cohesive – forces of attraction between like molecules. At an air-water interface – surface tension. Adhesion – attraction of one substance for a substance of another kind. Tensile strength – work that must be done to create or extend a new or larger surface. Viscosity – resistance to flow.

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی If water were an ordinary compound whose molecules are subject to weak forces, its boiling and freezing point would fall below hydrogen sulfide Strong hydrogen bonding between water molecules prevents this Water occurs in all three states (solid, liquid, and gaseous) at prevailing temperatures on the earth’s surface Example: Ice cubes in a glass at room temperature Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Why water wets clean glass? ﺣﺎﺝ ﻋﺒﺎﺳی § Surface of glass has O and unpaired electrons § Water molecules form hydrogen bond § Force stronger than gravity Why water does not stick to glass surface coated with grease? § Surface of grease has no O and free electrons § Water molecules cannot form hydrogen bond § Therefore, water do not stick Isfahan University of Technology
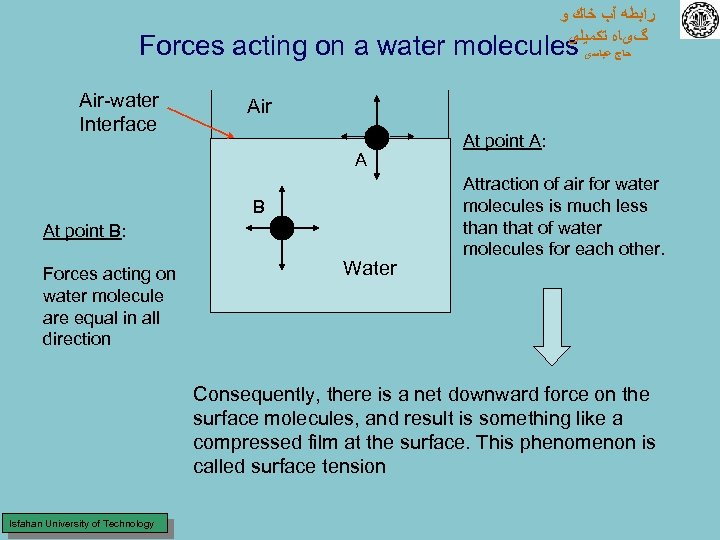
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Forces acting on a water molecules ﺣﺎﺝ ﻋﺒﺎﺳی Air-water Interface Air A B At point B: Forces acting on water molecule are equal in all direction Water At point A: Attraction of air for water molecules is much less than that of water molecules for each other. Consequently, there is a net downward force on the surface molecules, and result is something like a compressed film at the surface. This phenomenon is called surface tension Isfahan University of Technology
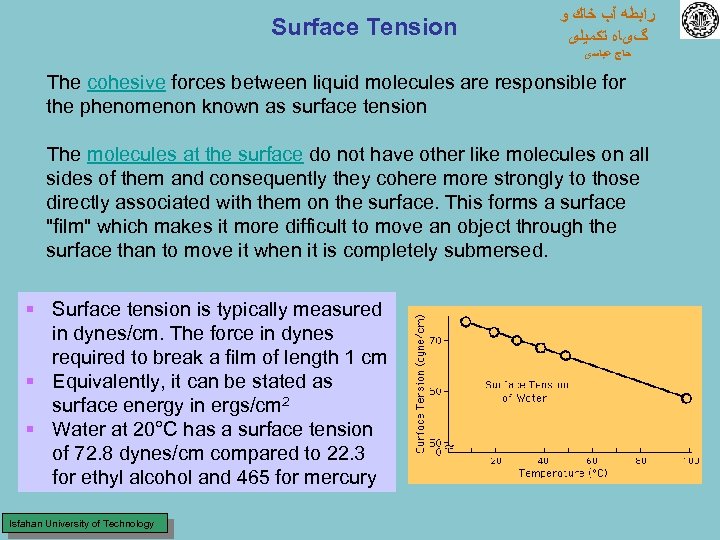
Surface Tension ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی The cohesive forces between liquid molecules are responsible for the phenomenon known as surface tension The molecules at the surface do not have other like molecules on all sides of them and consequently they cohere more strongly to those directly associated with them on the surface. This forms a surface "film" which makes it more difficult to move an object through the surface than to move it when it is completely submersed. § Surface tension is typically measured in dynes/cm. The force in dynes required to break a film of length 1 cm § Equivalently, it can be stated as surface energy in ergs/cm 2 § Water at 20°C has a surface tension of 72. 8 dynes/cm compared to 22. 3 for ethyl alcohol and 465 for mercury Isfahan University of Technology
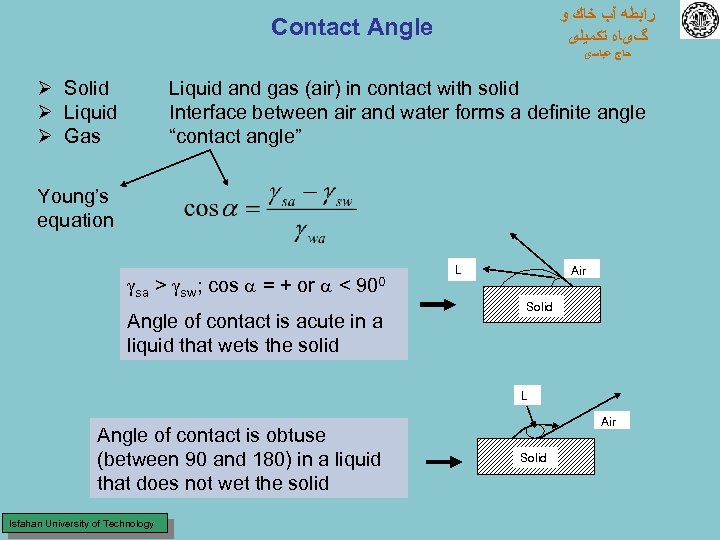
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Contact Angle ﺣﺎﺝ ﻋﺒﺎﺳی Ø Solid Ø Liquid Ø Gas Liquid and gas (air) in contact with solid Interface between air and water forms a definite angle “contact angle” Young’s equation sa > sw; cos a = + or a < 900 Angle of contact is acute in a liquid that wets the solid L Air Solid L Angle of contact is obtuse (between 90 and 180) in a liquid that does not wet the solid Isfahan University of Technology Air Solid

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Hydrophilic Versus Hydrophobic Soils ﺣﺎﺝ ﻋﺒﺎﺳی When the adhesive forces between water molecules and an object are weaker than the cohesive forces between water molecules, the surface repels water and is said to be hydrophobic. Hydrophobic soils restrict the entry of water, which 'balls up' or sits on the soil in beads rather than infiltrating the soil. Hydrophobic soils exhibit an obtuse (greater than or equal to 90 o) wetting angle that causes capillary repulsion, so preventing water from entering soil pores Hydrophilic or normally wettable soils display an acute (less than 90 o) angle of contact with water, allowing infiltration. adhesive forces between water molecules and an object are stronger than the cohesive forces between water molecules Isfahan University of Technology
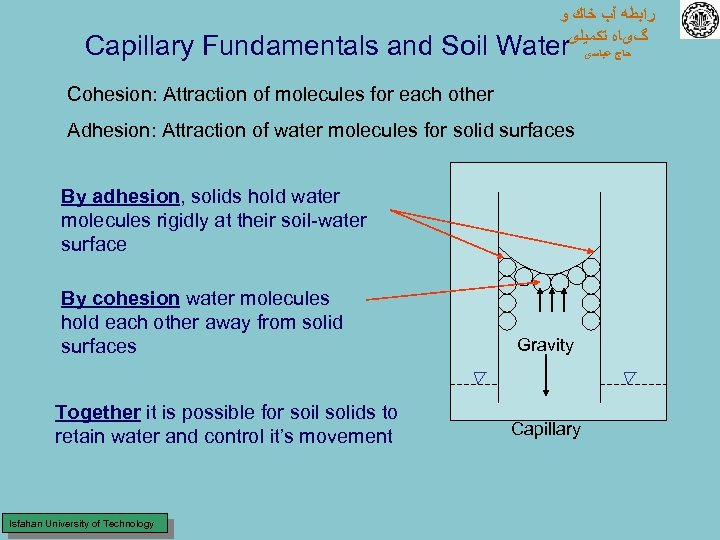
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Capillary Fundamentals and Soil Water Cohesion: Attraction of molecules for each other Adhesion: Attraction of water molecules for solid surfaces By adhesion, solids hold water molecules rigidly at their soil-water surface By cohesion water molecules hold each other away from solid surfaces Together it is possible for soil solids to retain water and control it’s movement Isfahan University of Technology Gravity Capillary ﺣﺎﺝ ﻋﺒﺎﺳی
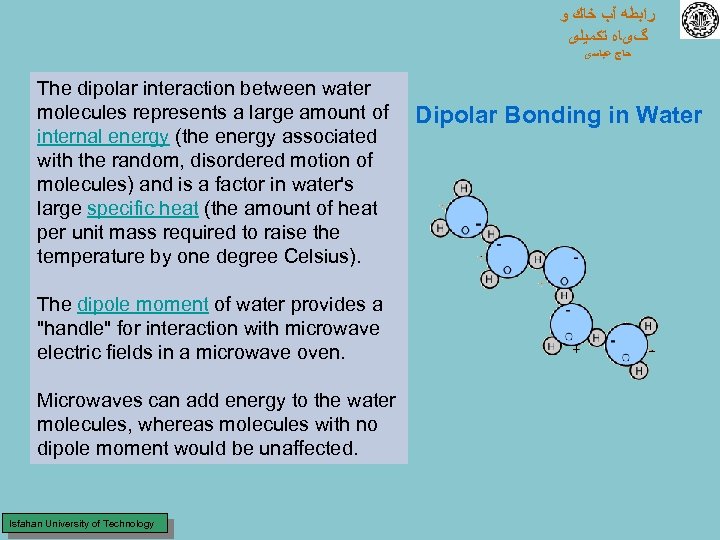
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی The dipolar interaction between water molecules represents a large amount of internal energy (the energy associated with the random, disordered motion of molecules) and is a factor in water's large specific heat (the amount of heat per unit mass required to raise the temperature by one degree Celsius). The dipole moment of water provides a "handle" for interaction with microwave electric fields in a microwave oven. Microwaves can add energy to the water molecules, whereas molecules with no dipole moment would be unaffected. Isfahan University of Technology Dipolar Bonding in Water

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Water rises in the capillary against the force of gravity ﺣﺎﺝ ﻋﺒﺎﺳی !!!! What happens if there is no force of gravity !!!!! Water Isfahan University of Technology Water

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Capillary Mechanism ﺣﺎﺝ ﻋﺒﺎﺳی 2 r 1 Rise continues till: Weight of water in the tube (force of gravity) = Total cohesive and adhesive forces h 1 2 r 2 h 2 Water Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی 2 r ﺣﺎﺝ ﻋﺒﺎﺳی Force of gravity = Mass of water column * Acceleration = (volume of water * density) * g = (p * r 2* h) *dw * g …………(A) Total cohesive and adhesive forces h = (perimeter) * surface tension =2*p*r* At equilibrium: …………(B) Water A=B (p * r 2* h) *dw * g = 2 * p * r * use Show = 72. 75 dynes/cm dw= 0. 9982 g/cm 3 g = 980 cm/s 2 Isfahan University of Technology
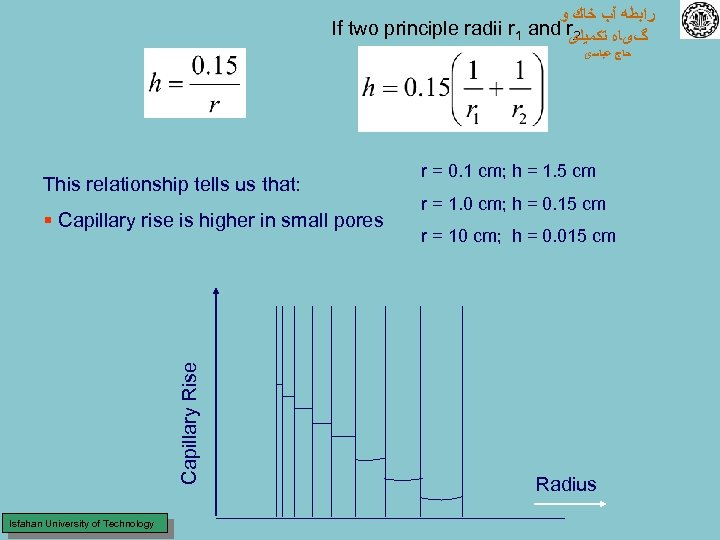
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ If two principle radii r 1 and r 2 گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی This relationship tells us that: Capillary Rise § Capillary rise is higher in small pores Isfahan University of Technology r = 0. 1 cm; h = 1. 5 cm r = 1. 0 cm; h = 0. 15 cm r = 10 cm; h = 0. 015 cm Radius
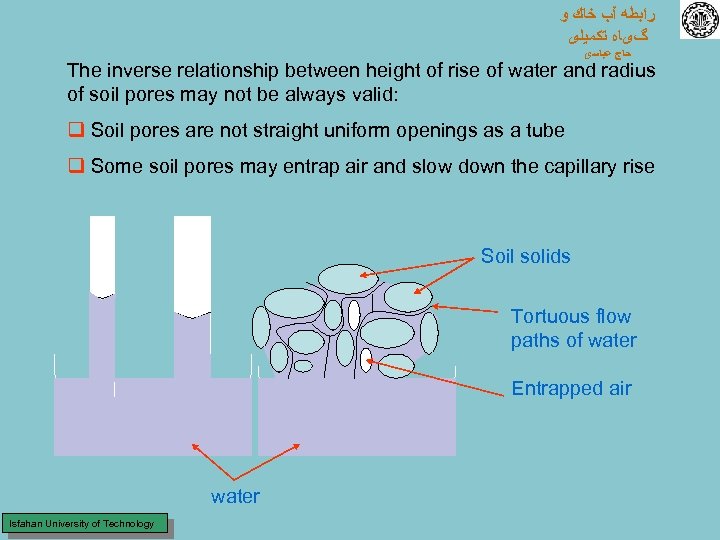
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی The inverse relationship between height of rise of water and radius of soil pores may not be always valid: q Soil pores are not straight uniform openings as a tube q Some soil pores may entrap air and slow down the capillary rise Soil solids Tortuous flow paths of water Entrapped air water Isfahan University of Technology
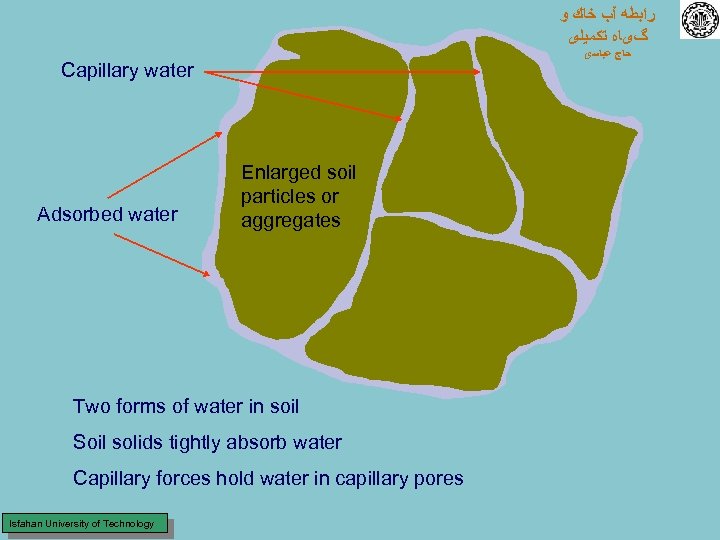
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Capillary water Adsorbed water Enlarged soil particles or aggregates Two forms of water in soil Soil solids tightly absorb water Capillary forces hold water in capillary pores Isfahan University of Technology
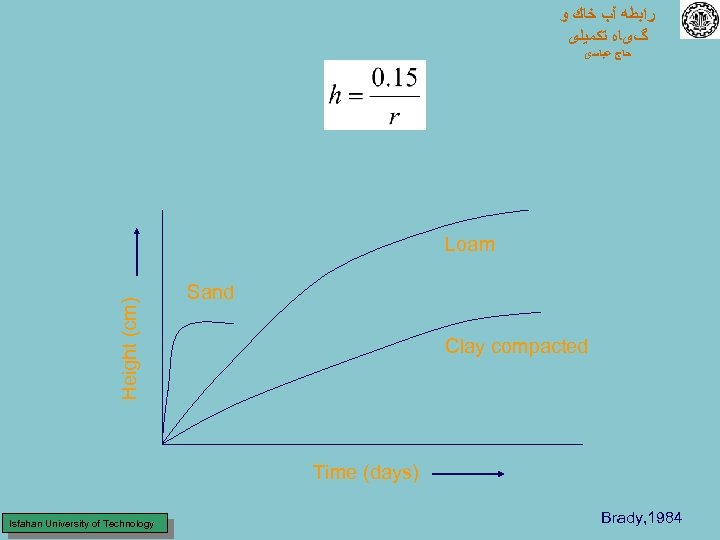
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Height (cm) Loam Sand Clay compacted Time (days) Isfahan University of Technology Brady, 1984

UNIQUE PROPERTIES of WATER Ø TEMPERATURE RELATIONS – Heat of Vaporization = 540 cal/g@100 C 2. 45 MJ/kg – Heat of Fusion = 80 cal/g@0 C – Heat Capacity = 1. 0 cal/g – Thermal Conductivity = High
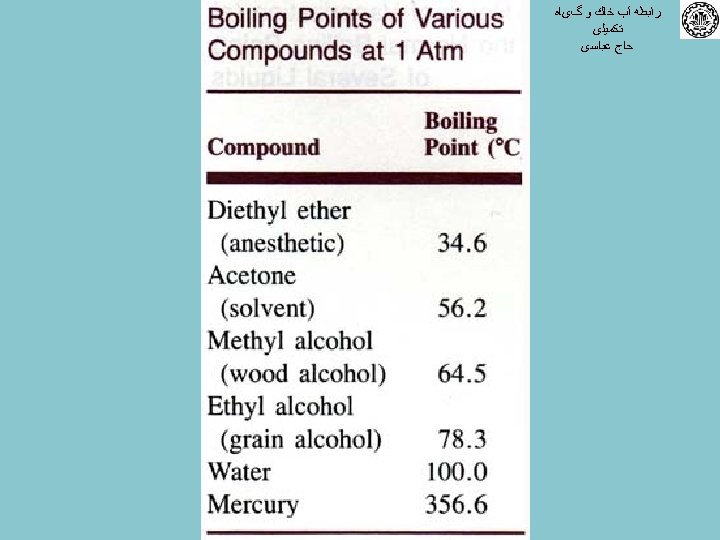
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی
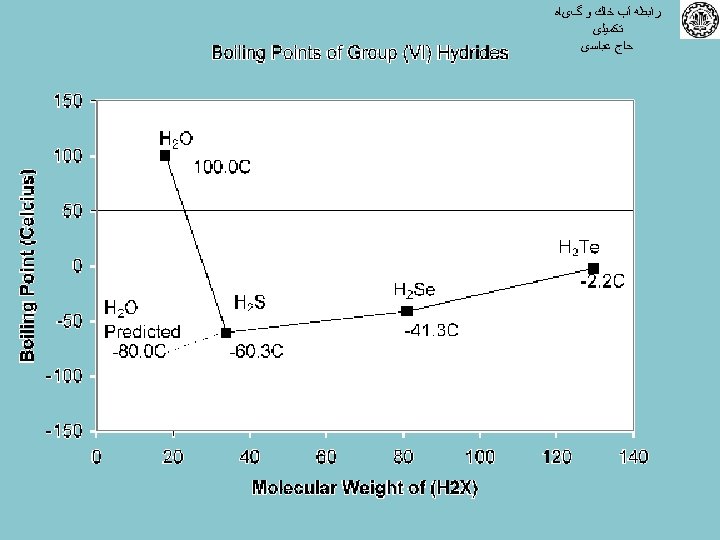
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی

UNIQUE PROPERTIES of WATER Ø DENSITY – 0. 9998 g/cm 3 at 0 o. C – 1. 0 g/cm 3 at 4 o. C – 0. 9956 g/cm 3 at 30 o. C
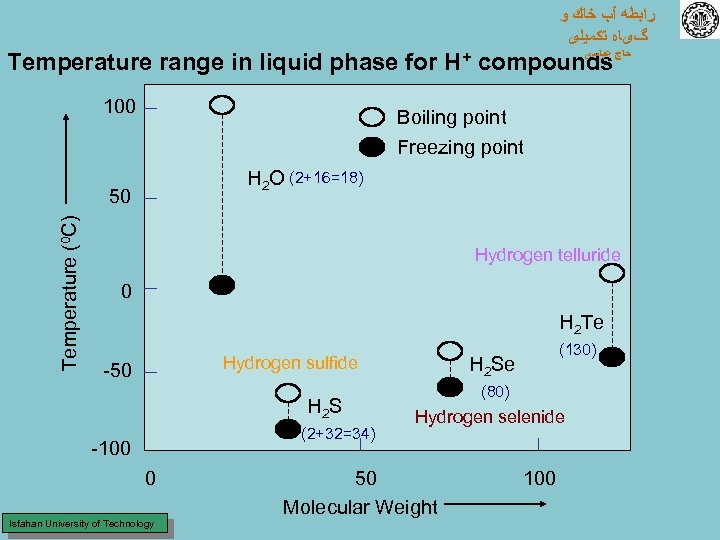
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﻋﺒﺎﺳی Temperature range in liquid phase for H+ compounds ﺣﺎﺝ 100 Boiling point Freezing point H 2 O (2+16=18) Temperature (0 C) 50 Hydrogen telluride 0 H 2 Te Hydrogen sulfide -50 H 2 S (2+32=34) -100 0 Isfahan University of Technology (130) H 2 Se (80) Hydrogen selenide 50 Molecular Weight 100
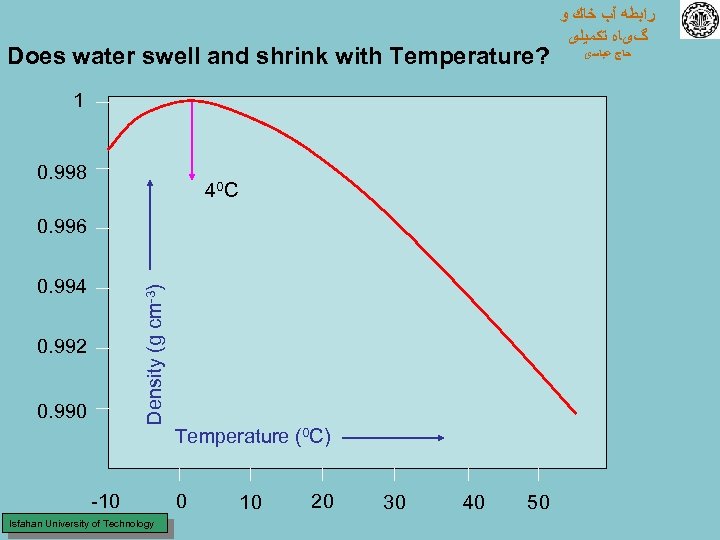
Does water swell and shrink with Temperature? 1 0. 998 40 C 0. 996 Density (g cm-3) 0. 994 0. 992 0. 990 Temperature (0 C) -10 Isfahan University of Technology 0 10 20 30 40 50 ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی
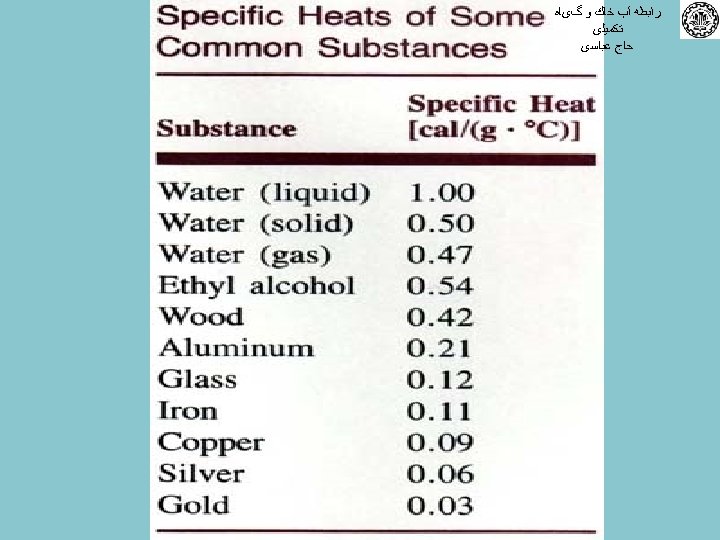
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی
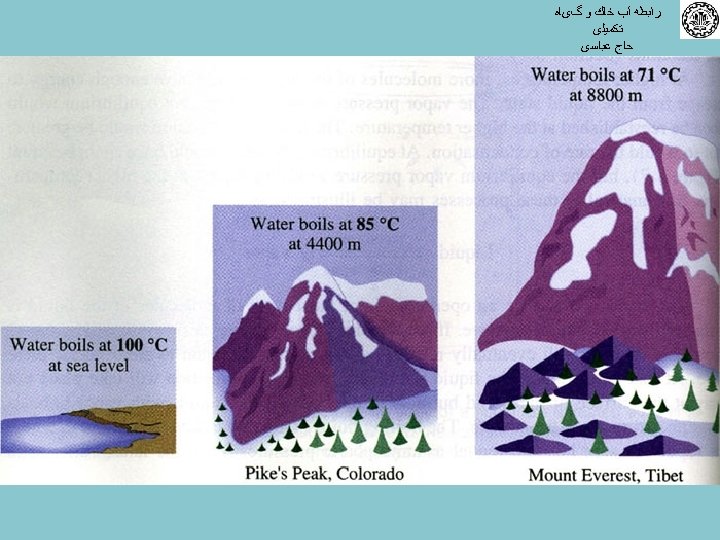
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی

UNIQUE PROPERTIES of WATER Ø SOLVENT – Small Size – Polar characteristics – High Dielectric Constant

UNIQUE PROPERTIES of WATER Ø Transparent to Visible Radiation (400 -700 nm) Ø Highly Absorbent in Infra-Red (>1000 nm)

UNIQUE PROPERTIES of WATER Ø HIGH SURFACE TENSION – Adhesion – Cohesion – Tensile Strength ( >30 MPa ~4000#/in 2)

UNIQUE PROPERTIES of WATER Ø IONIZATION – Only 1 molecule out of 55 x 107 is ionized p. H ~ - Log (H+)
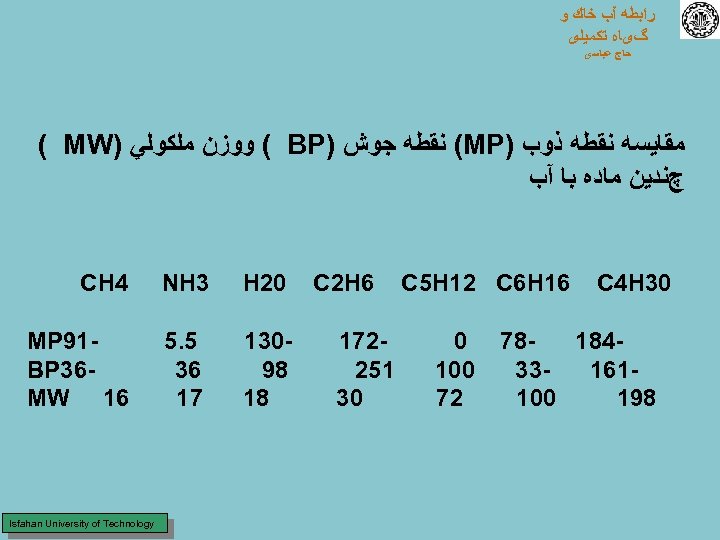
ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی ﻣﻘﺎﻳﺴﻪ ﻧﻘﻄﻪ ﺫﻭﺏ ) (MP ﻧﻘﻄﻪ ﺟﻮﺵ ) ( BP ﻭﻭﺯﻥ ﻣﻠﻜﻮﻟﻲ ) ( MW چﻨﺪﻳﻦ ﻣﺎﺩﻩ ﺑﺎ آﺐ 03 C 4 H 61 C 5 H 12 C 6 H 87 481 33 161 001 891 0 001 27 6 C 2 H 271 152 03 02 H 3 NH 4 CH 031 89 81 5. 5 63 71 19 MP 63 BP 61 MW Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Properties of Water Fig. 1. Temperature range of various hydride groups. Isfahan University of Technology

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی ﺑﺎﻭﺟﻮﺩﺍﻳﻨﻜﻪ ﻭﺯﻥ ﻣﻠﻜﻮﻟﻲ آﺐ ﻧﺴﺒﺘﺎ ﻛﻢ ﺍﺳﺖ ﻭﻟﻲ ﻣﻘﺎﻭﻣﺖ پﻴﻮﻧﺪ ﻫﻴﺪﺭﻭژﻨﻲ ﻭﺳﺎﺧﺘﻤﺎﻥ ﺩﺭﻭﻧﻲ آﻦ ﺑﺎﻋﺚ ﺷﺪﻩ ﺗﺎ ﺩﺭﺩﺭﺟﻪ ﺣﺮﺍﺭﺕ ﻃﺒﻴﻌﻲ ﺑﺼﻮﺭﺕ ﻣﺎﻳﻊ ﺑﺎﺷﺪ ﻧﻪ ﺑﺼﻮﺭﺕ ﺑﺨﺎﺭﺑﺪﻳﻦ ﻣﻌﻨﻲ ﻛﻪﻋﻨﺎﺻﺮﻱ ﺑﺎ ﻭﺯﻥ ﻣﻠﻜﻮﻟﻲ ﺗﻘﺮﻳﺒﺎ ﺷﺒﻴﻪ ﺑﻪ آﺐ ﺩﺭ ﺩﺭﺟﻪ ﺣﺮﺍﺭﺗﻬﺎﻱ ﺑﺴﻴﺎﺭپﺎﻳﻴﻦ ﺫﻭﺏ ﻣﻲ ﺷﻮﻧﺪ ﻭﻳﺎﺑﺠﻮﺵ ﻣﻲ آﻴﻨﺪ ﺍﺯ ﻃﺮﻑ ﺩﻳگﺮ ﻋﻨﺎﺻﺮﺩﻳگﺮﺑﺎﻳﺪ ﻭﺯﻥ ﻣﻠﻜﻮﻟﻲ ﺑﺴﻴﺎﺭﺑﺎﻻﻳﻲ ﺩﺍﺷﺘﻪ ﺑﺎﺷﻨﺪ ﺗﺎ ﺑﺘﻮﺍﻧﻨﺪ ﺩﺭﺩﺭﺟﻪ ﺣﺮﺍﺭﺗﻬﺎﻱ ﺫﻭﺏ ﻳﺎﺟﻮﺵ آﺐ ﺫﻭﺏ ﻳﺎ ﺑﺤﺎﻟﺖ ﺟﻮﺵ ﺩﺭآﻴﻨﺪ ﻳﻌﻨﻲ آﺐ ﺩﺭﺣﺎﻟﺖ ﻣﺎﻳﻊ ﻣﺎﻧﻨﺪ ﻣﻠﻜﻮﻟﻬﺎﻱ ﺑﺴﻴﺎﺭﺳﻨگﻴﻦ ﻋﻤﻞ ﻣﻲ ﻛﻨﺪ. Isfahan University of Technology

COLLIGATIVE PROPERTIES of AQUEOUS SOLUTIONS PROPERTY PURE WATER 1. O M SOLUTION 1. Vapor Pressure 0. 61 KPa@Oo Decreased according 101. 3 KPa@100 o 2. Boiling Point 100 o. C 3. Freezing Point 0 o. C 4. Osmotic Potential 0 5. Chemical Potential 0 to Raoult’s Law 100. 52 o. C -1. 86 o. C -2. 27 MPa Decreased
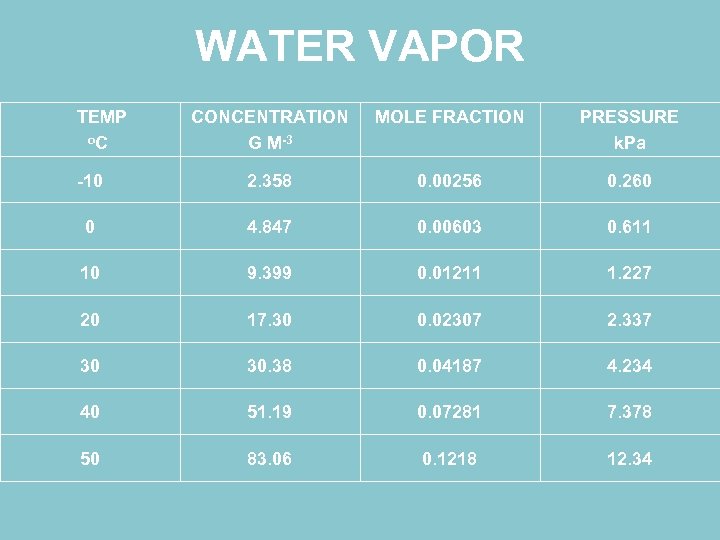
WATER VAPOR TEMP o. C CONCENTRATION G M-3 MOLE FRACTION PRESSURE k. Pa -10 2. 358 0. 00256 0. 260 0 4. 847 0. 00603 0. 611 10 9. 399 0. 01211 1. 227 20 17. 30 0. 02307 2. 337 30 30. 38 0. 04187 4. 234 40 51. 19 0. 07281 7. 378 50 83. 06 0. 1218 12. 34
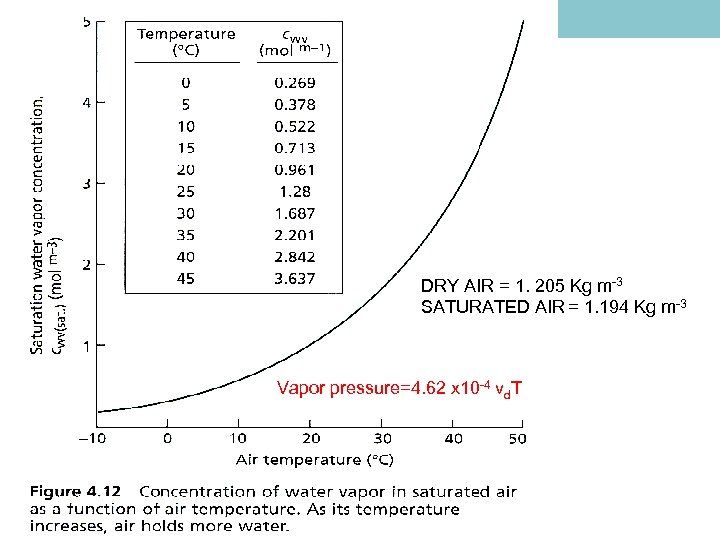
DRY AIR = 1. 205 Kg m-3 SATURATED AIR = 1. 194 Kg m-3 Vapor pressure=4. 62 x 10 -4 vd. T
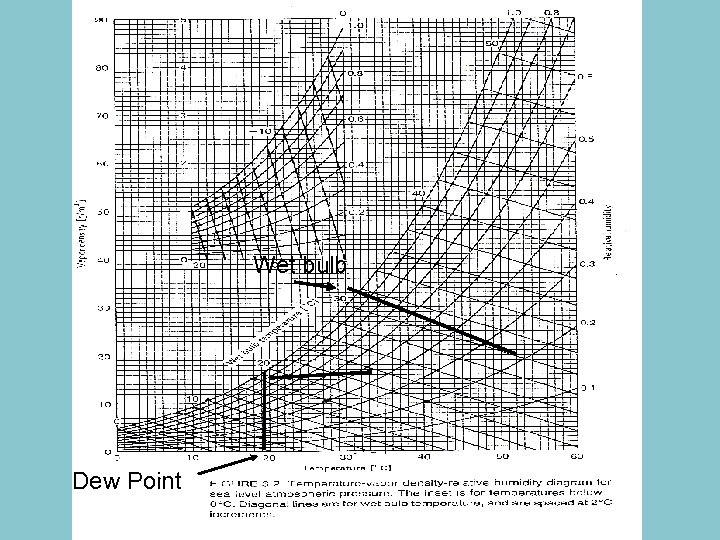
Wet bulb Dew Point
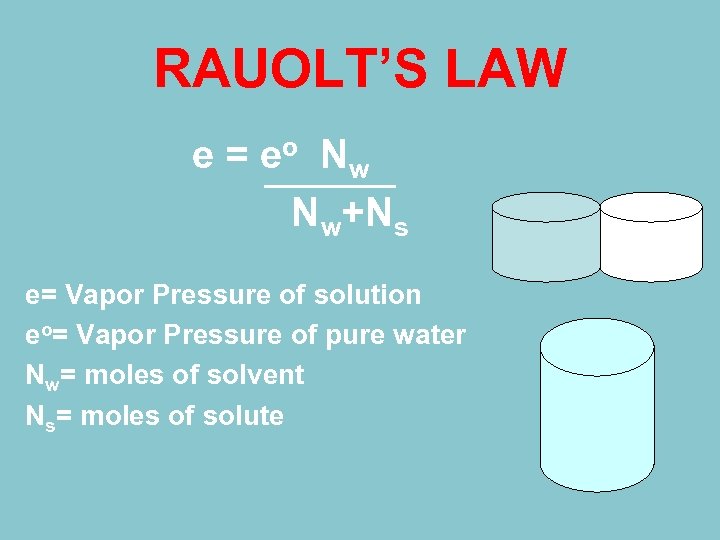
RAUOLT’S LAW e = e o Nw Nw+Ns e= Vapor Pressure of solution eo= Vapor Pressure of pure water Nw= moles of solvent Ns= moles of solute

OSMOTIC PRESSURE Van’t Hoff equation = Ns. RT V = osmotic pressure in Mega Pascals Ns= moles of solute R= gas constant (0. 0083 L MPa /mol @273 o) T= absolute temperature V= volume of solvent in Liters (RT= 2. 272 @) 0 o. C &2. 437 @ 20 o. C liter. MPamol-1)

CHEMICAL POTENTIAL of WATER in a SOLUTION A measure of the ability of WATER to do work Go = partial molal Gibb’s free energy Go = -RTln. K (K = equilibrium constant) A B G Negative = Spontaneous G Positive = Additional Energy Required

CHEMICAL POTENTIAL of WATER µw-µwo = RT ln. Nw µw= chemical potential of water in solution (Jmol-1) µwo= chemical potential of pure water R= Gas constant = 8. 314 J mol-1 K-1 T= Absolute temperature Nw= mole fraction of water in solution-can be replaced by ln e/eo

WATER POTENTIAL (PRESSURE UNITS) w = RT ln e/eo V w The water potential of a solution is decreased by those factors which reduce the vapor pressure 1. Addition of solutes (Osmotic) 2. Matric forces ( Interfacial Adhesive Forces) 3. Reduction in Temperature 4. Tension

WATER MOVEMENT Flux Jw (moles per meter 2 per second) = Concentration Gradient ( - D mols/DDistance; DY) * Diffusion Coefficient (DW) Ø LIQUID STATE • VAPOR STATE • Potential Gradient • Hydraulic Conductivity (Cells 7 x 10 -13 ms-1) (Soil 10 -2 – 10 -10 m 2 sec-1) • Vapor Pressure Gradient • Diffusion Coefficient (2. 4 x 10 -5 m-2 s-1) • Resistance

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی Forces that affect movement of water into the soil ﺣﺎﺝ ﻋﺒﺎﺳی Gravity: a constant force that pulls the water downward Cohesion: attraction of water molecules for each other. It is the force that holds a droplet of water together Adhesion: attraction of water molecules to other substances. This force causes water molecules to adhere to other objects, such as soil particles Placing a drop of water on a piece of newsprint paper Force of adhesion between the water molecules and the paper molecules is greater than the force of cohesion that holds the water molecules together The water droplet spreads out and soaks into the paper Placing a drop of water on a piece of waxed paper Force of adhesion between the water molecules and the paper molecules is lower than the force of cohesion that holds the water molecules together The water droplet remains intact Isfahan University of Technology

TOTAL WATER POTENTIAL = – ( W p + m W = Total Water Potential p = Pressure Potential = Osmotic Potential m = Matric Potential g = Gravitational Potential + ) g

WATER POTENTIAL Ø SOIL SYSTEM • Matric Forces Surface Tension Electrostatic Association • Osmotic Ø PLANT SYSTEM • Surface Tension • Cohesion • Osmotic • Pressure
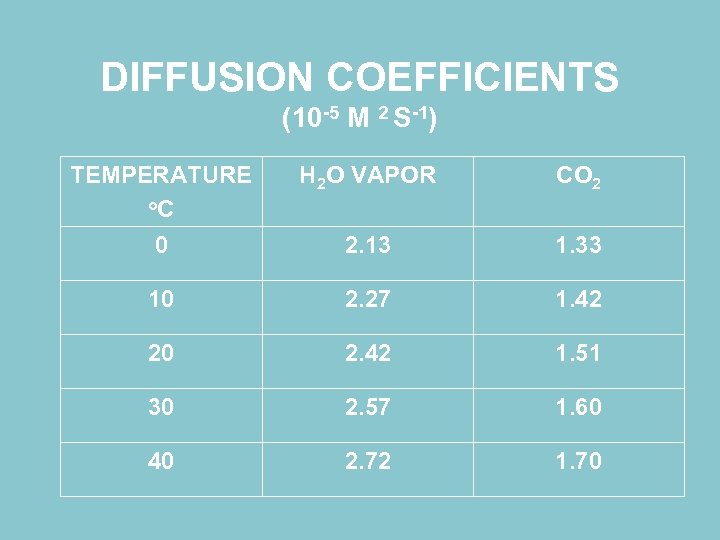
DIFFUSION COEFFICIENTS (10 -5 M 2 S-1) TEMPERATURE o. C 0 H 2 O VAPOR CO 2 2. 13 1. 33 10 2. 27 1. 42 20 2. 42 1. 51 30 2. 57 1. 60 40 2. 72 1. 70

ﺭﺍﺑﻄﻪ آﺐ ﺧﺎﻙ ﻭ گیﺎﻩ ﺗﻜﻤﻴﻠی ﺣﺎﺝ ﻋﺒﺎﺳی Physical Properties of Water l Liquid phases in soil and plant are similar l In both systems the liquid is a solution of water and dissolved substances l Physical properties of water
913aa237ef88329eaecdc106c534bbcc.ppt