29267611171480181ea656cefda3aed8.ppt
- Количество слайдов: 56

ﺑﺴﻢ ﺍﻟﻠﻪ ﺍﻟﺮﺣﻤﻦ ﺍﻟﺮﺣﻴﻢ ﻭﺯﺍﺭﺓ ﺍﻟﺪﻓﺎﻉ ﺭﺋﺎﺳﺔ ﺍﻷﺮﻛﺎﻥ ﺍﻟﻤﺸﺘﺮﻛﺔ ﺍﻻﺩﺍﺭﺓ ﺍﻟﻌﺎﻣﺔ ﻟﻠﺨﺪﻣﺎﺕ ﺍﻟﻄﺒﻴﺔ ﻗﺴﻢ ﺍﻟﻌﻨﺎﻳﺔ ﺍﻟﻤﻜﺜﻔﺔ

Case Presentation: Prepared by : Dr. Altayeb M. Altayeb Abdalaziz ICU – Resident Supervised by : Dr. Kamal Osman Mergani Consultant Intensivist

Case Presentation : Mss. S. A. A. M is a 75 yrs old female who is known to be Hypertensive for 5 yrs on regular treatment, not known to be diabetic, referred to our department as a case of severe sepsis due to hospital acquired pneumonia. She was admitted on 10 th. 0 ct. 2012 & her examination on admission was as follow:

Ø Vital Signs GCS: 15/15, B. P: 110/60 , PR: 120/min , RR: 22/min, Temp. : 38 C Ø CNS: Unremarkable. Ø CVS: Unremarkable Ø Respiratory System : Fine crepes bilaterally Ø Abdominal & Renal system : Unremarkable What To Do NEXT ?

1. Patient Received (1)L of Normal Saline. 2. Blood sent for Investigations(CBC, RFTS). 3. Patient started antibiotics(Meropenem 1 g. TDS) 4. Observation of Vital signs & urine output hourly.

Patient Follow up: (11 th. Oct. 2012) General: Unwell , BP: 130/80 , PR: 100/min , RR: 20/min, Temp. 37. 6 C , U. O. P : Adequate at (0. 5 -1)ml/kg/hr Chest: Fine Crepes bilaterally CNS, CVS, Abdomen —> Unremarkable

Event Clock UOP NIL UOP: 50 ml/hr BP 110/60 1 L N. S UOP: 20 ml/hr B. P 100/50 • Investigations: • CBC: TWBCS(12. 2) , Hb(9) , Plt(400) • RFT&elect: Urea(40), Creat(2), Na+K Normal 11 12 1 Another Liter of N. S UOP Still NIL 9 3 B. P: 100/50(55) AT : 16: 15 Quartz 8 4 Confused. Tachypnic + N. E type II Resp. Failure on 7 5 0. 25 MCG/KG/MIN 6 ABG Dopamine 20 mcg/kg/min B. P: 100/50 • Connected To MV Hydrocortisone • BP: 110/60 UOP: NIL 50 mg. Q 6 hrs • Continue ABG: Met. 10 Mazen. K 2 Septic Shock acidosis Antibiotic • F/U via ABG & Vital signs

What to Do Next ?

1. Pt supported by Fluids & Inotropes (N. E+Dopamine Max. Dose) 2. Add Vancomycine (Renal dose). 3. Septic Screening Sent( Blood, Urine, Sputum) 4. Continue f/u of vital signs, UOP, DVT & Stress ulcer Prophylaxis.

1. 2. 3. 4. 5. 6. P. t condition continues to deteriorate in the next days despite maximum Inotropic & fluid support, Deterioration is Summarized as follows : Hypotensive on max. inotropes. Decrease LOC. Anuria + Impaired Renal profile. Progressive rising in TWBC. Chest crepes became worse. ABG: Metabolic Acidosis &Requiring more ventilator Support.

ANY Suggestions? A few observation and much reasoning leads to error, many observations and little reasoning to truth Alexis Carrel (1873 -1944)

Ketoconazole 15 th. 10. 012 Patient is added at condition continues to deteriorate clinically and investigation wise as follows:
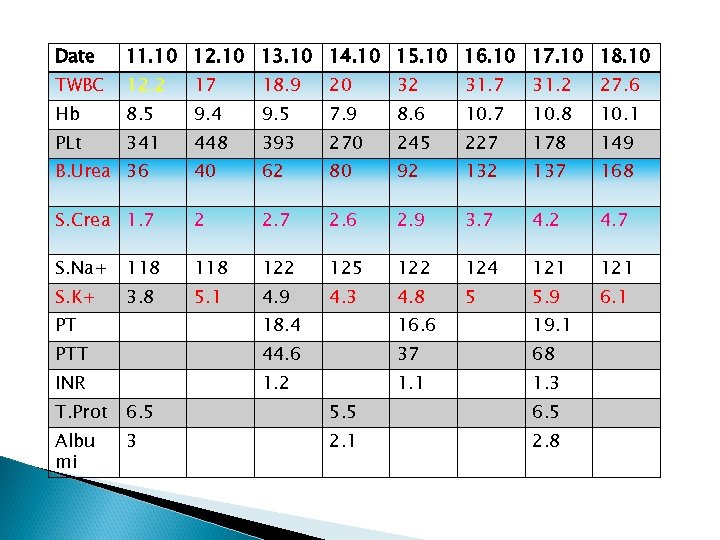
Date 11. 10 12. 10 13. 10 14. 10 15. 10 16. 10 17. 10 18. 10 TWBC 12. 2 17 18. 9 20 32 31. 7 31. 2 27. 6 Hb 8. 5 9. 4 9. 5 7. 9 8. 6 10. 7 10. 8 10. 1 PLt 341 448 393 270 245 227 178 149 B. Urea 36 40 62 80 92 137 168 S. Crea 1. 7 2 2. 7 2. 6 2. 9 3. 7 4. 2 4. 7 S. Na+ 118 122 125 122 124 121 S. K+ 5. 1 4. 9 4. 3 4. 8 5 5. 9 6. 1 3. 8 PT 18. 4 16. 6 19. 1 PTT 44. 6 37 68 INR 1. 2 1. 1 1. 3 T. Prot 6. 5 5. 5 6. 5 Albu mi 3 2. 1 2. 8
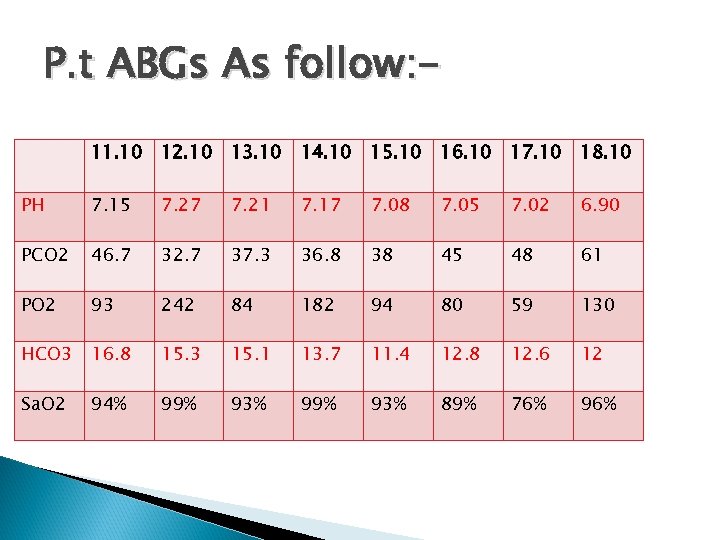
P. t ABGs As follow: 11. 10 12. 10 13. 10 14. 10 15. 10 16. 10 17. 10 18. 10 PH 7. 15 7. 27 7. 21 7. 17 7. 08 7. 05 7. 02 6. 90 PCO 2 46. 7 32. 7 37. 3 36. 8 38 45 48 61 PO 2 93 242 84 182 94 80 59 130 HCO 3 16. 8 15. 3 15. 1 13. 7 11. 4 12. 8 12. 6 12 Sa. O 2 94% 99% 93% 89% 76% 96%

Other Investigations: CT Abd. unremarkable. Echo : E. F=64%, PASP: 32 mmhg, No LV dysfunction or Valvular abnormality. CXR: Reticulonodular infiltration of both upper lobes, free costo/cardiophrenic angles with normal cardiac configuration.

Culture result: - (16 th. 10. 012) Blood & urine: ü No Growth. Ø Sputum culture: ü Acenitobacter baumanii [Aerobic Gm –ve coccobacilli]. ü Multidrug o o resistance to: Meropenem Vancomycine. Amikacine Cephalosporine. Gentamicine Piperacilline Ciprofloxacine


What NEXT ?

1. Stop All antibiotics. 2. Put patient in a contact isolation 3. Continue with the other supportive therapy. 4. Search for : (Colistine OR Tigicycline). =Unfortunately BOTH are not available in Sudan =
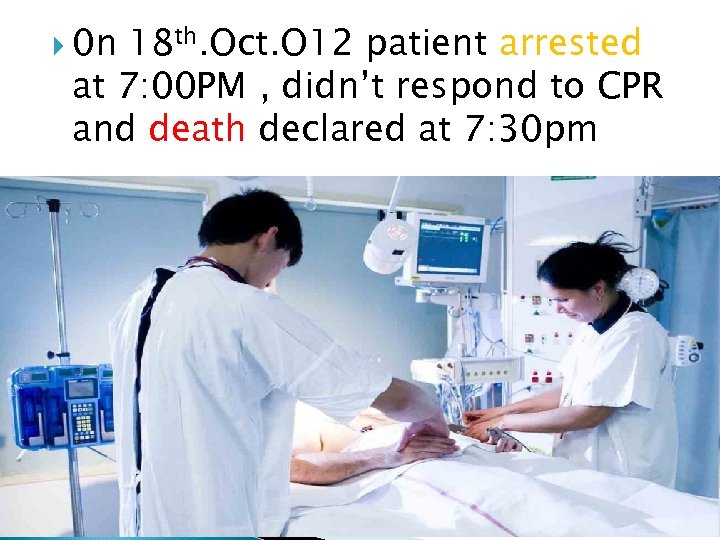
0 n 18 th. Oct. O 12 patient arrested at 7: 00 PM , didn’t respond to CPR and death declared at 7: 30 pm

Faculty of medicine U of G in Autumn ( wadmadani )SUDAN

Acenitobacter Baumannii: “We are born to see, but have to train ourselves to observe”

Background: Acinetobacter baumannii Gram negative Coccobacillus. Strictly Aerobic, Non motile. Water and soil. Associated with antibiotic resistance. Not part of normal human flora Infections and outbreaks ◦ Intensive care unit and healthcare settings ◦ Compromised immune systems and risk ◦ Colonized and infected patients as point sources

Acinetobacter baumannii Infections in Iraq war Since Operation Iraqi Freedom began in 2003, more than 700 US soldiers have been infected or colonized with Acinetobacter baumannii. A significant number of additional cases have been found in the Canadian and British armed forces, and among wounded Iraqi civilians.

Global pockets of Acinetobacter baumanii infections

Risk factors include: hospitalization significant comorbidity mechanical ventilation cardio respiratory failure previous infection antimicrobial therapy CVP lines urinary catheters
Hospitalized patients are highly venerable to Acinetobacter Infections Acinetobacter infections are uncommon and occur almost exclusively in hospitalized patients

• • • “A doctor is a person who kills your ills with pills, & then kills you with his bills” Anonymous

"It is nice to have money and the things that money can buy, but it's important to make sure you haven't lost the things money can't buy. " George Lorimer 1867 -1937, Editor of "Saturday Evening Post"
Diagnosis of Acinetobacter Infections Infection or colonization with Acinetobacter is usually diagnosed by clinical culture of blood, sputum, urine, wound, sterile body fluid, etc. Microbiologic cultures can be processed by standard methods on routine media.

Biochemical Reactions Oxidase negative (opposite to Neisseria spp. or Moraxella spp. ) Haemolytic Indole negative. Catalase positive. Nonmotile Strictly aerobic Gram negative coccobacillus Highly antibiotic resistant
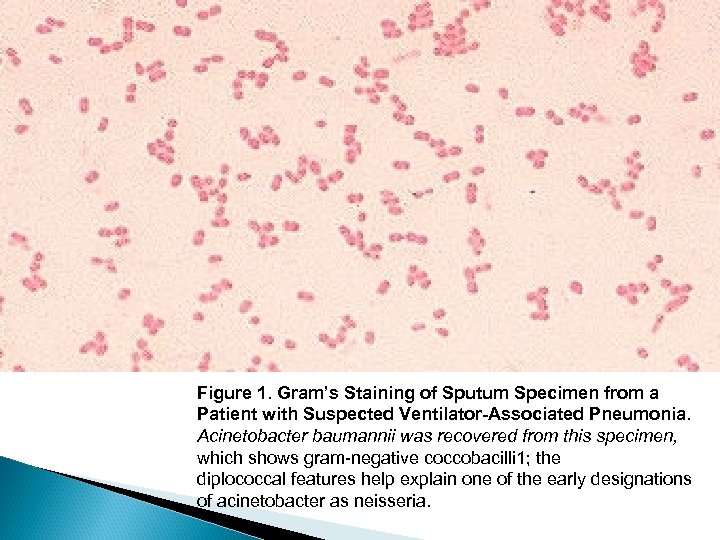
Figure 1. Gram’s Staining of Sputum Specimen from a Patient with Suspected Ventilator-Associated Pneumonia. Acinetobacter baumannii was recovered from this specimen, which shows gram-negative coccobacilli 1; the diplococcal features help explain one of the early designations of acinetobacter as neisseria.

Acinetobacter outbreaks 19772000 Extensive Literature review and summary of 51 Acinetobacter outbreaks Characteristic Number of reports Publication year: 1977 -1990 1991 -2000 24 27 ICU setting 38 Patient age category: Adult Pediatric 45 6 Comment The majority of the reports occurred over the last 9 years 75 percent of reports were exclusively or predominantly ICU related outbreaks or clusters 88 percent of all outbreaks were in an adult population Villegas M, Hartstein A. Infect Control Hosp Epidemiol. 2003; 24: 284 -295
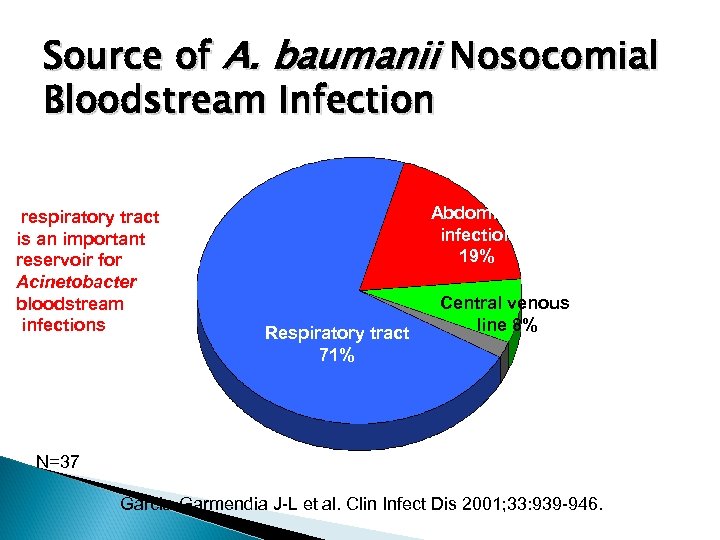
Source of A. baumanii Nosocomial Bloodstream Infection respiratory tract is an important reservoir for Acinetobacter bloodstream infections Abdominal infection 19% Respiratory tract 71% Central venous line 8% N=37 Garcia-Garmendia J-L et al. Clin Infect Dis 2001; 33: 939 -946.
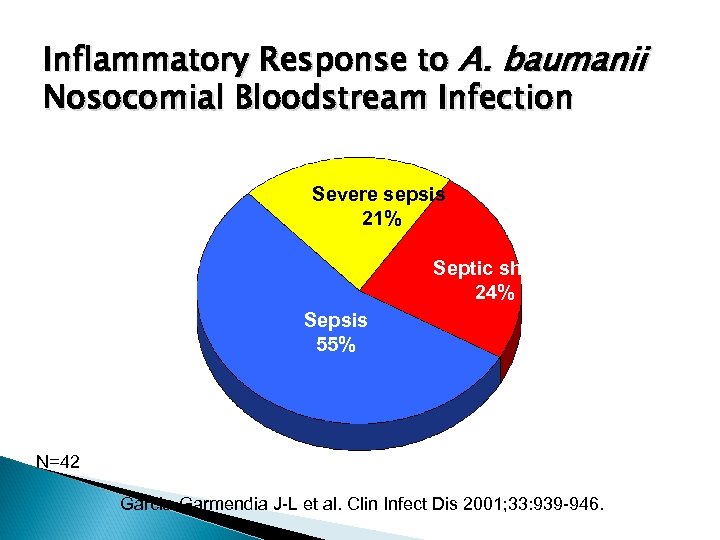
Inflammatory Response to A. baumanii Nosocomial Bloodstream Infection Severe sepsis 21% Septic shock 24% Sepsis 55% N=42 Garcia-Garmendia J-L et al. Clin Infect Dis 2001; 33: 939 -946.
Complex mechanisms of Drug Resitance makes treatment options difficult

Problems in treating Acinetobacter baumannii One of the biggest issues with treating Acinetobacter baumannii is that the bacterium is naturally resistant to a number of antibiotics, making it challenging to find a drug regimen which will effectively attack it in an infected patien

Documented mechanisms of resistance in Acinetobacter baumannii Quantitative and/or qualitative changes in outer membrane porins. Altered penicillin-binding proteins. Aminoglycosides-modifying enzymes. Broad-spectrum β-lactamases. Efflux pumps.

Treatment Carbapenems (Imipenem and Meropenem) are the mainstay of treatment for antimicrobial-resistant gram-negative infections, though Carbapenems-resistant Acinetobacter is increasingly reported. Resistance to the Carbapenems class of antibiotics makes multidrug-resistant Acinetobacter infections difficult, if not impossible, to treat.

Multidrug Resistant strains a Global Concern Multidrug-resistant A. baumannii is a common problem in many hospitals in the US and Europe. First line treatment is with a Carbapenems antibiotic such as imipenem, but carbapenem resistance is increasingly common. Other treatment options include Colistin, tigecycline and Aminoglycosides.
Treating the Resistant Infections Colistin and Polymyxin B have been used to treat highly resistant Acinetobacter infections. The choice of appropriate therapy is further complicated by the toxicity of colistin which is mainly renal. Acinetobacter isolates resistant to colistin and Polymyxin B have also been reported.

Polymyxin -E antibiotics (Colistin) History ◦ Used extensively worldwide in topical otic and ophthalmic solutions for decades ◦ Intravenous Colistin was initially used in Japan and in Europe during the 1950 s, and in the United States in the form of colistimethate sodium in 1959 ◦ The intravenous formulations of colistin and polymyxin B were gradually abandoned in most parts of the world in the early 1980 s because of the reported high incidence of nephrotoxicity ◦ Colistin was mainly restricted during the past 2 decades for the treatment of lung infections due to multidrug-resistant (MDR), gram-negative bacteria in patients with cystic fibrosis

Numerous recent clinical studies have confirmed that colistin is an efficient antimicrobial agent against nosocomial infections, including bacteremia, ventilatorassociated pneumonia, urinary tract infection, and meningitis due to MDR GNB, such as P. aeruginosa, A. baumannii, and K. pneumonia, with an acceptable safety profile. Whereas colistin is mainly administered i. v. incritically ill patients, it can be safely be administered by inhalation in patients with pneumonia/VAP or intrathecally in patients with meningitis due to MDR GNB.

Polymyxin antibiotics (Colistin): Mechanism of action: ◦ Target: Bacterial cell membrane( Bactericidal). Colistin binding with the bacterial membrane occurs through electrostatic interactions between the cationic polypeptide (colistin) and anionic lipopolysaccharide (LPS) molecules in the outer membrane of the gram-negative bacteria leads to a derangement of the cell membrane The result of this is an increase in the permeability of the cell envelope, leakage of cell contents, and, subsequently, cell death.

Polymyxin antibiotics Important pharmacokinetic parameters: ◦ Colistin sulfate and colistimethate sodium are not absorbed by the gastrointestinal tract with oral administration ◦ Primary route of excretion is through glomerular filtration ◦ Experimental studies have shown that colistin is tightly bound to membrane lipids of tissues, including liver, lung, kidney, brain, heart, and muscles ◦ Concentration of colistin in the CSF is 25% of the serum concentration

Polymyxin antibiotics (Colistin) Route Dosage Intravenous • 2. 5 -5 mg/kg (31, 250 -62, 500 IU/kg) per day, divided into 2 -4 equal doses • (1 mg of colistin equals 12, 500 IU). Modification of the total daily dose is required in the presence of renal impairment Intramuscular • Same as IV Inhalation • 40 mg (500, 000 IU) every 12 h for patients <40 kg and 80 mg (1 million IU) every 12 h for patients >40 kg • For recurrent pulmonary infections, the dose can be increased to 160 mg (2 million IU) every 8 h Intraventricular/ intrathecal (not FDA approved) • Intrathecal dosage ranged from 3. 2 mg (40, 000 IU) to 10 mg (125, 000 IU) given once per day • Intraventricular dosage ranged from 10 mg (125, 000 IU) to 20 mg (250, 000 IU) per day (divided into 2 doses) Limited data based on case reports

ADMINISTRATION AND PHARMACOKINETICS: v Loading dose should be given as an IV infusion over 2 hours. v Maintenance dose should be given as IV infusion can be given over 30 minutes. v First maintenance dose should be given 24 hours after loading dose. v IM administration is not recommended. v Half-life is 3 -4 hours in patients with normal renal function, and up to 2 -3 days in v patients with renal impairment.

Polymixin adverse effects Nephrotoxicity • The majority of nephrotoxic events are reversible • 1970’s- incidence of nephrotoxicity was 20. 2% • More recent studies- incidence of nephrotoxicity ranged from 8%-18%. • Lower incidence of Nephrotoxicity at present: –Greater supportive treatment to critically ill patients –Close monitoring of renal function –Avoidance of co-administered nephrotoxic agents –Older formulations of Colistin contained a greater proportion of colistin sulfate (greater nephrotoxicity)


Can Acinetobacter Infect Health care Workers ?

Acinetobacter rarely causes serious infection in otherwise healthy people and therefore poses minimal threat to healthcare workers or patients’ family members. Pregnant healthcare workers are not at increased risk from this organism and can therefore care for patients infected or colonized with the organism.

Preventing Acinetobacter Transmission in the ICU General Measures Hand hygiene ◦ Use of alcohol-based hand sanitizers Contact precautions ◦ Gowns/gloves ◦ Sterilization and hand wash Environmental decontamination Prudent use of antibiotics

Early Recognition & Control of the source. Sterilization of the equipments ( MV, Circuits… etc). The organism is highly susceptible to antiseptic & disinfectants. Hand wash ( Generally the hardest measure to implement).

Efficacy of Handwashing Agents against Acinetobacter Experimental study to access removal of A. baumanii from the hands of volunteers ◦ Fingertips inoculated with either 103 CFU (light contamination) or 106 CFU (heavy contamination) Removal Rate Agent Light contamination Heavy contamination Plain soap 99. 97% 92. 40% 70% Ethyl alcohol 99. 98% 98. 94% 10% Povidone-iodine 99. 98% 98. 48% 4% Chlorhexidine 99. 81% 91. 39% Cardoso CL et al. Am J Infect Control 1999; 27: 327 -331.

Summary: Although commonly found on the skin of healthy humans, Acinetobacter plays the role of an opportunistic pathogen in the critically ill patient High level of antibiotic resistance makes it well suited as a pathogen in areas with high use of antibiotics (e. g. , ICU setting) Control requires good hand hygiene, barrier precautions & environmental decontamination ◦ Alcohol-based products containing chlorhexidine should be considered the hand hygiene agents of choice
29267611171480181ea656cefda3aed8.ppt