0edf9c43b3917f997f9a44913738a6c9.ppt
- Количество слайдов: 98

導管相關血流感染之感染管 制 台灣防疫學會 王任賢 理事長

Impact of Primary BSI
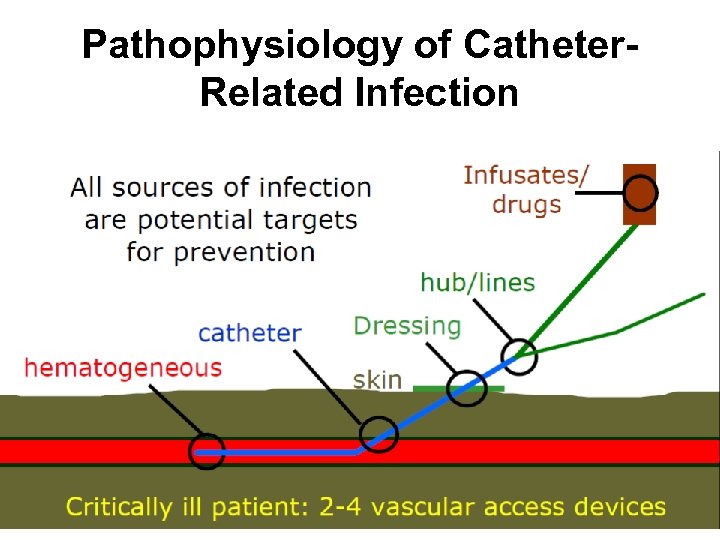
Pathophysiology of Catheter. Related Infection

Microbial Source of CRBSI • • Extraluminal biofilm is the major source of CRBSI within the first week of catheterization in short-term catheters. Extraluminal biofilm is the major source of tunnel infections (exit site infections) in long-term catheters. • Intraluminal biofilm is the major source of CRBSI after 1 week in both short- and long-term catheters. 1. Ryder, MA. Catheter-Related Infections: It's All About Biofilm. Topics in Advanced Practice Nursing e. Journal. 2005; 5(3) © 2005 Medscape. Posted 08/18/2005.

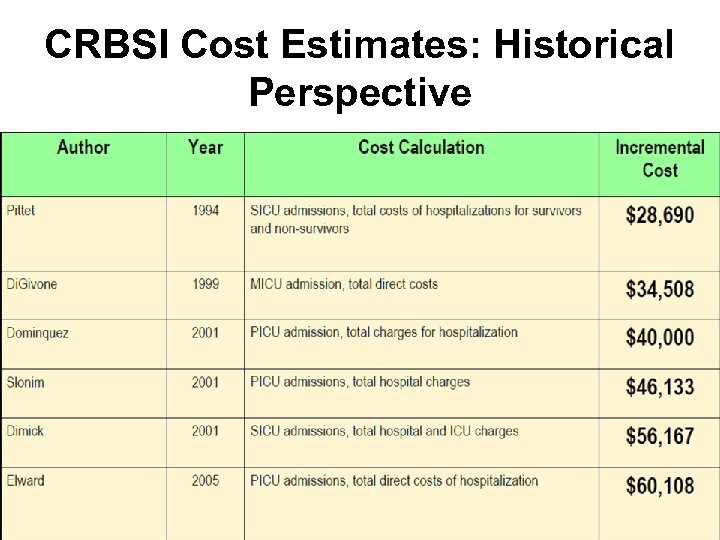
CRBSI Cost Estimates: Historical Perspective
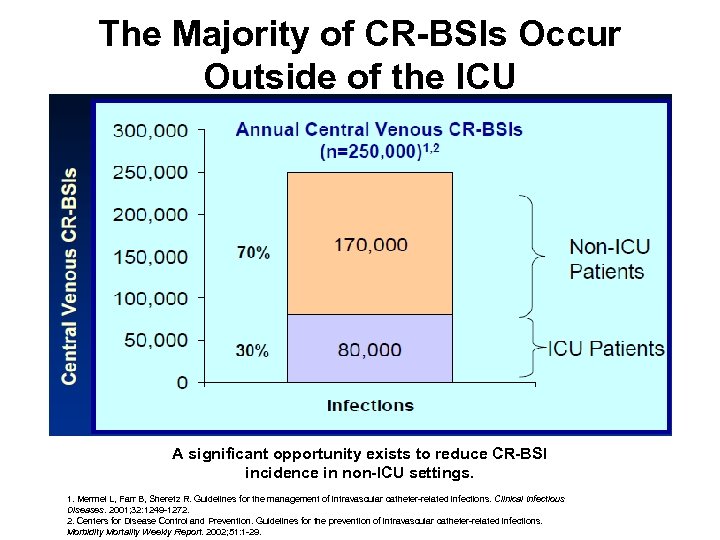
The Majority of CR-BSIs Occur Outside of the ICU A significant opportunity exists to reduce CR-BSI incidence in non-ICU settings. 1. Mermel L, Farr B, Sheretz R. Guidelines for the management of intravascular catheter-related infections. Clinical Infectious Diseases. 2001; 32: 1249 -1272. 2. Centers for Disease Control and Prevention. Guidelines for the prevention of intravascular catheter-related infections. Morbidity Mortality Weekly Report. 2002; 51: 1 -29.

The Institute for Healthcare Improvement (IHI) CVC-BSI Prevention Insertion Bundle----Evidence-Based Measures to Reduce Infections Associated with Catheter Insertion. • • Hand hygiene Maximal sterile barrier precautions Chlorhexidine skin antisepsis Optimal site care (device selection and site of insertion) Education Catheter removal Monitoring of practices Leadership

Basic vs. Special Approaches for the Prevention of CLA-BSIs Marschall J, et al. ICHE 2008; 29: S 22 -30.

導管置放過程

Basic Practices: Perform Hand Hygiene 1 Marschall J, et al. ICHE 2008; 29: S 22 -30.

Hand Hygiene: The Data Since 1977, 7 of 8 prospective studies have shown that improvement in hand hygiene significantly decreases infection rates Clin Infect Dis 1999; 29: 1287 -94

Effect of Hand Hygiene on Resistant Organisms* *All interventions included multifaceted intervention programs; Most quasiexperimental or observational, not randomized trials. No randomized trial of hand hygiene vs. no hand hygiene has been conducted. Source: Pittet D: Emerg Infect Dis 2001; 7: 234 -240
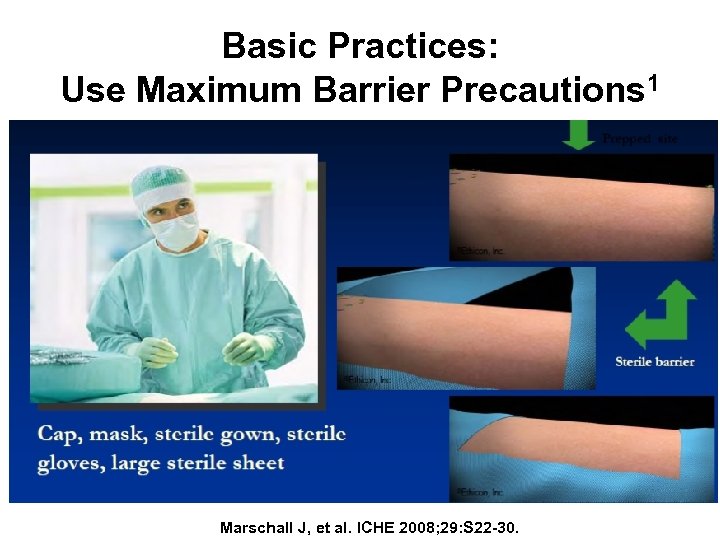
Basic Practices: Use Maximum Barrier Precautions 1 Marschall J, et al. ICHE 2008; 29: S 22 -30.
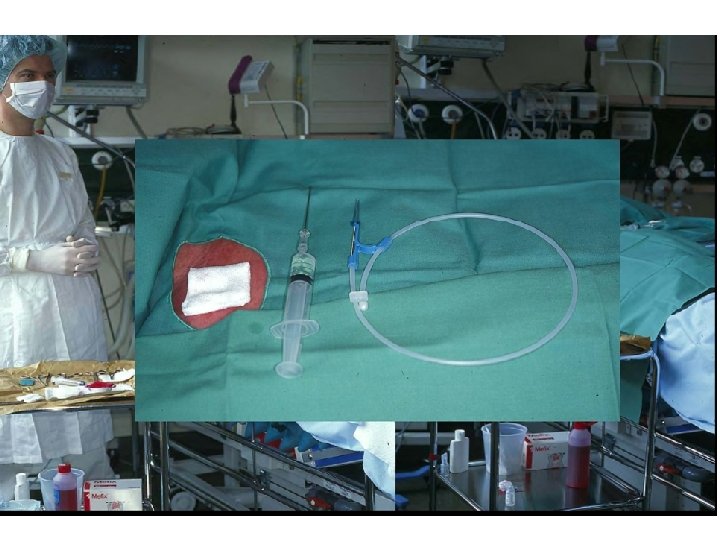

What Are Maximal Barrier Precautions? • For You – Hand hygiene – Non-sterile cap and mask • All hair should be under cap • Mask should cover nose and mouth tightly – Sterile gown and gloves • For the Patient – Cover patient’s head and body with a large sterile drape

Who Needs To Be Dressed In MBP? • • The operator The assistant Anyone else who crosses the sterile field NOT people in the same room who are not involved with the procedure
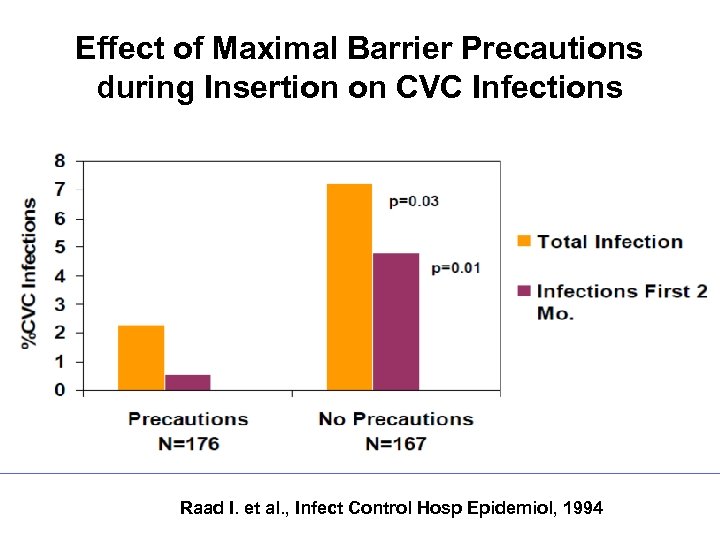
Effect of Maximal Barrier Precautions during Insertion on CVC Infections Raad I. et al. , Infect Control Hosp Epidemiol, 1994

Do I Really Need Maximal Barrier Precautions ? Author & Year Study Design Type of Line OR for infection without MBP Mermel 1991 Prospective Cross-sectional Swan. Ganz 2. 2 (p=0. 03) Raad 1994 Prospective Randomized Central 3. 3 (p=0. 03) Am J Med 1991; 91(3 B): 197 S-205 S Infect Control Hosp Epidemiol 1994; 15: 231 -8

Skin Prep • Chlorhexidine 2% is more effective than povidone iodine (Betadine) because it dries quickly and has longer residual action

Skin Prep • If you must use Betadine – Allow Betadine to dry completely (at least 2 minutes) – Do not blow on, fan, or blot the site to make it dry faster!

Basic Practices: Use CHG Skin Prep 1 • • Apply 30 seconds with friction Allow 30 seconds to dry Marschall J, et al. ICHE 2008; 29: S 22 -30.

Dressing The Line • Apply dressing immediately after placement when site is still sterile • Use transparent dressing (Sorbaview) unless site is oozing or pt is allergic

Maintaining The Line • Change transparent dressing weekly, gauze dressing daily, and any dressing that is damp, bloody, or non-occlusive • Do not use topical ointment or cream at insertion site • Do not leave a line undressed • Lines examined daily by medical staff

What Site Is Best? • “No randomized trial satisfactorily has compared infection rates for catheters placed in jugular, subclavian, and femoral sites. ” MMWR, 8/9/02

What Site Is Best? • The Hopkins Experience—retrospective analysis of SICU IJ & SC catheters that grew 15 cfu – IJ position was the only predictive factor of 15 cfu (OR 1. 83, p <. 001)

What Site Is Best? • RCT of femoral and SC lines in the ICU – 145 pts femoral/144 pts SC • Outcomes – Similar rates of mechanical complication: 17. 3% vs 18. 8% (p = NS) – Higher rate of infectious complications (colonization and BSI combined) in femoral grp: 19. 8% vs 4. 5% (p <. 001) – Higher rate of thrombotic complications in femoral grp: 21. 5% vs. 1. 9% (p <. 001); complete thrombosis 6% vs 0% JAMA; 2001, 286: 700 -7

What Site Is Best? • Based on these and other studies, the JHH VAD policy recommends that the preferred order of line placement is SC IJ F • Other factors to consider in site choice – Anatomic deformity – Coagulopathy – Operator experience

What Site Is Best For Children? • Traditionally femoral vein is site of first choice for all pediatric patients – Operator experience is determining factor for placement of lines elsewhere • Infectious data does not hold true in children – Site of insertion does not correlate with infectious complications • Same or fewer mechanical complications noted with femoral line placement • No thrombotic risk with femoral line placement
Basic Practices: Use Catheter Cart or Kit 1
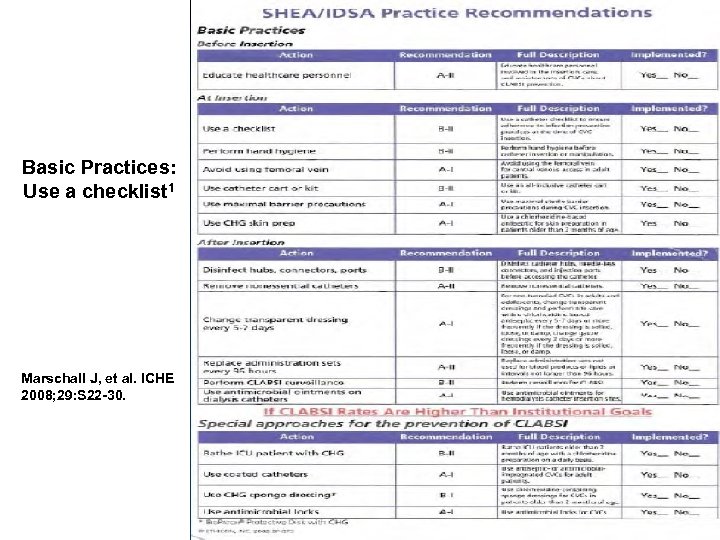
Basic Practices: Use a checklist 1 Marschall J, et al. ICHE 2008; 29: S 22 -30.

Education-based prevention of CVC-BSI Education-based strategy applied to the medical staff (students + residents + fellow) in of 6 U. S. ICUs Infection control (1 h): - Hand hygiene - Isolation precautions - IV guideline 1 h-training stations: - Blood draws through lines - Arterial punctures - Catheter insertion - Handling of patients Sherertz RJ et al. Ann Intern Med 2000; 132: 641 -8.
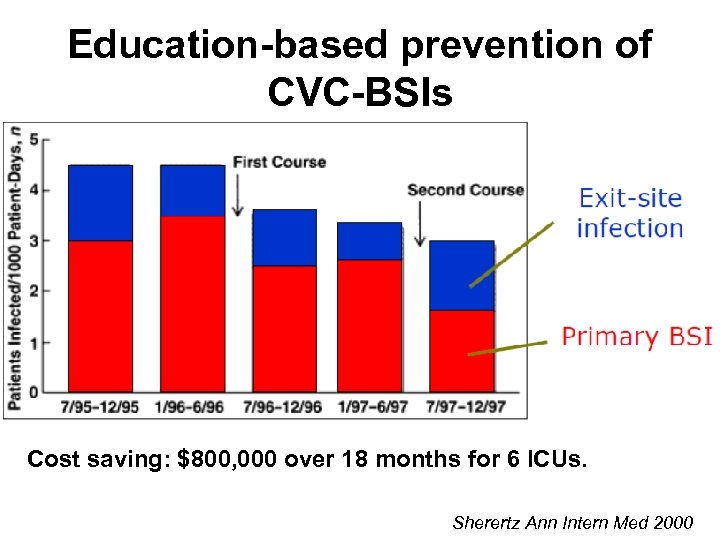
Education-based prevention of CVC-BSIs Cost saving: $800, 000 over 18 months for 6 ICUs. Sherertz Ann Intern Med 2000

導管維護

Other Evidence-Based Measures to Decrease the Risk of Infection During Maintenance of the Intravascular Catheter • Minimize catheter site skin bioburden. • Device selection • Aseptic manipulation of catheter connectors-Scrub the hub! • (Antibiotic/antiseptic lock) • (Impregnated-catheters)

Recommendations for Implementing Prevention and Monitoring Strategies: Special approaches for the prevention of CLA-BSIs Recommended for locations and/or populations within the hospital that have unacceptably high CLA-BSI rates despite implementation of the basic CLA-BSI prevention strategies. • Use antiseptic- or antimicrobial-impregnated CVCs for adult patients (A-I). • Use antimicrobial locks for CVCs (A-I). • Use CHG-containing sponge for CVCs in patients >2 months of age (B-I). • Bathe ICU patients >2 months of age with a CHG preparation on a daily basis (B-II). Marschall J. et al. , Infect Control Hosp Epidemiology 2008; 29: S 22 -30.
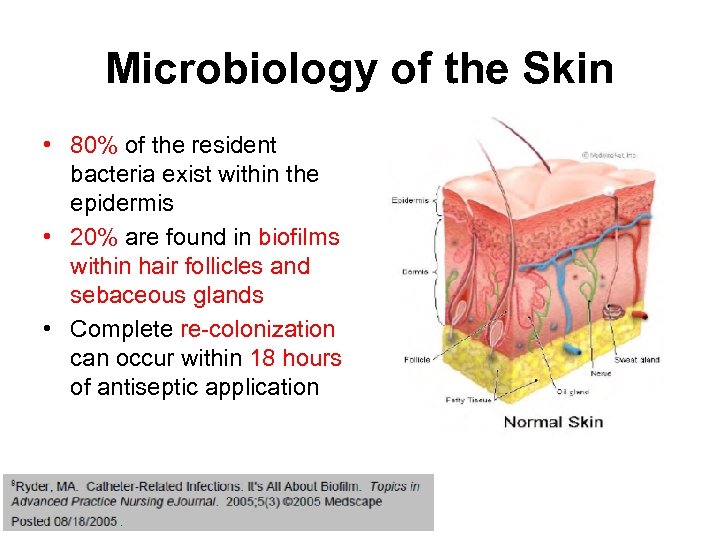
Microbiology of the Skin • 80% of the resident bacteria exist within the epidermis • 20% are found in biofilms within hair follicles and sebaceous glands • Complete re-colonization can occur within 18 hours of antiseptic application
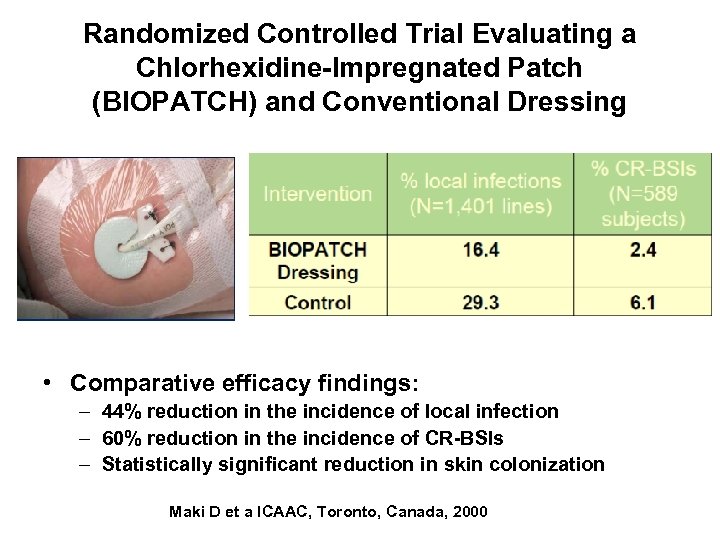
Randomized Controlled Trial Evaluating a Chlorhexidine-Impregnated Patch (BIOPATCH) and Conventional Dressing • Comparative efficacy findings: – 44% reduction in the incidence of local infection – 60% reduction in the incidence of CR-BSIs – Statistically significant reduction in skin colonization Maki D et a ICAAC, Toronto, Canada, 2000

Does the Bio. Patch Enhance CVC-BSI Prevention in Patients with Impregnated Catheters? • Study design: Prospective, randomized, open, controlled study in cancer chemotherapy patients requiring central venous catheters (CVC) for >5 days between January 2004 and January 2006. All patients had a chlorhexidine and silver sulfadiazine-impregnated triple lumen CVC. Randomized to CHG-sponge vs. standard dressing. Independent observation of site. • Results: 601 patients with 9, 731 CVC-days. Mean CVC duration: 16. 6 days (treatment) vs. 15. 8 days (control). Mean neutropenia: 7. 5 days (treatment) vs. 6. 9 days (control). CVC-related infections: 34/301 (11. 3%) in control vs. 19/300 (6. 3%) in CHG-sponge group (p=0. 016, RR=0. 54). CVC-related infections significantly reduced at internal jugular vein-inserted CVCs (P=0. 018). • Summary: The use of the CHG-sponge (Bio. Patch) reduced CVCrelated infections (54%) even when CHG-silver impregnated catheters were used. Reschulte H et al. Ann Hematol 2009; 88; 267 -72.

Timsit’s Randomized Controlled Trial:

Antibiotic Lock • Several trials suggest beneficial effect of antibiotic lock on catheter colonization or infection rate Studies include: Immunocompromised patients Neonatal intensive care unit patients • Negligible antibiotic reaches the bloodstream • Antibiotic resistance not documented yet, but studies have been small and relatively shortterm

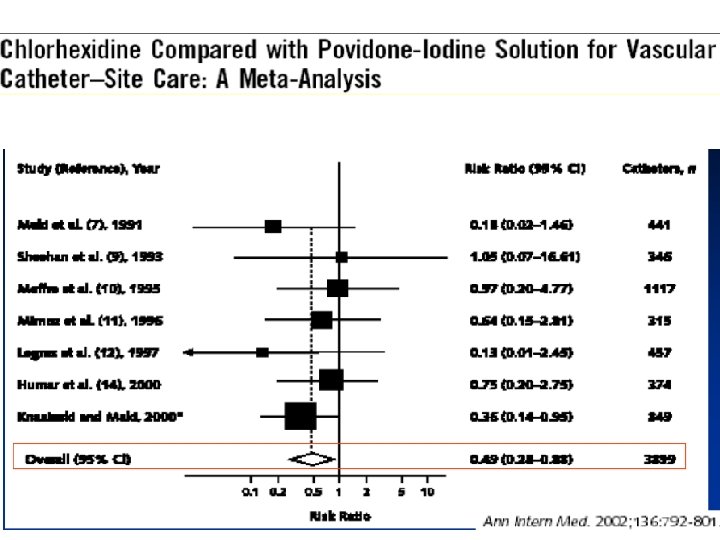
Does Vancomycin Lock or Flush Reduce CVC-BSIs? • Study design: Meta-analysis of prospective randomized studies, 1966 -2006. • Results: Seven studies with 463 patients; cancer (n=5), NICU (n=1), cancer/TPN (n=1). – Summary risk ratio for vancomycin-heparin lock or flush = 0. 49 (95% CI: 0. 26 -0. 95, P=0. 03). – Summary risk ratio for vancomycin-heparin lock: 0. 34 (95% CI: 0. 12 -0. 98, P=0. 04) • Conclusion: Use of vancomycin lock in high-risk patients with long-term IVDs reduces the risk of BSI. Sadfar N et al. CID 2006; 43: 474 -84
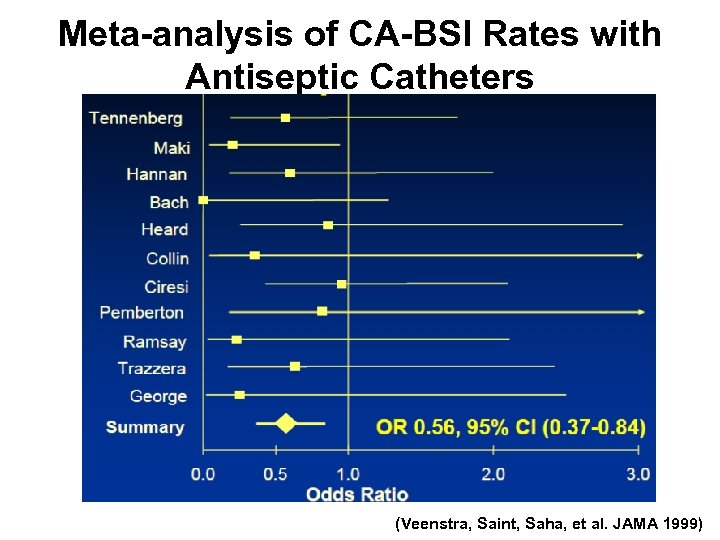
Meta-analysis of CA-BSI Rates with Antiseptic Catheters (Veenstra, Saint, Saha, et al. JAMA 1999)
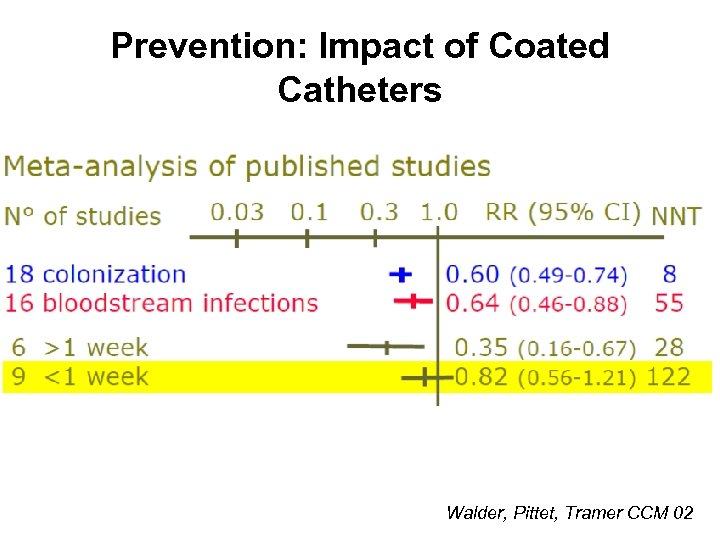
Prevention: Impact of Coated Catheters Walder, Pittet, Tramer CCM 02

Device Selection

输液方法 : I • 连续输液 – 固定连续输液 • 数小时 • 数天 • 数周 – 可以单种液 , 也可多种液

输液方法 : II • 连续输液合并间歇输液 – 固定输液 – 多次间歇输液 • 架接 30 -60分钟短期输液 • 直接由管路注射

输液方法 : III • 仅间歇输液 – 只在静脉给药时才用 – 每 6, 8, 12 或 24 小时 – 没有连续输液 – 行动比较方便 , 比较容易出院
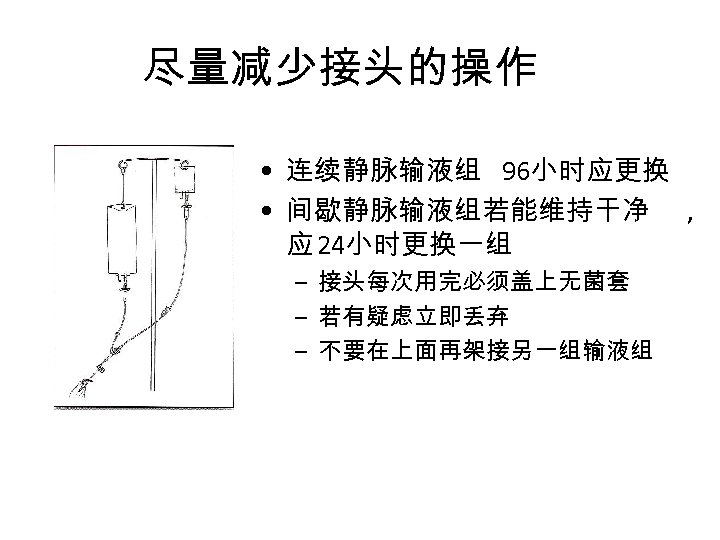
尽量减少接头的操作 • 连续静脉输液组 96小时应更换 • 间歇静脉输液组若能维持干净 , 应 24小时更换一组 – 接头每次用完必须盖上无菌套 – 若有疑虑立即丢弃 – 不要在上面再架接另一组输液组
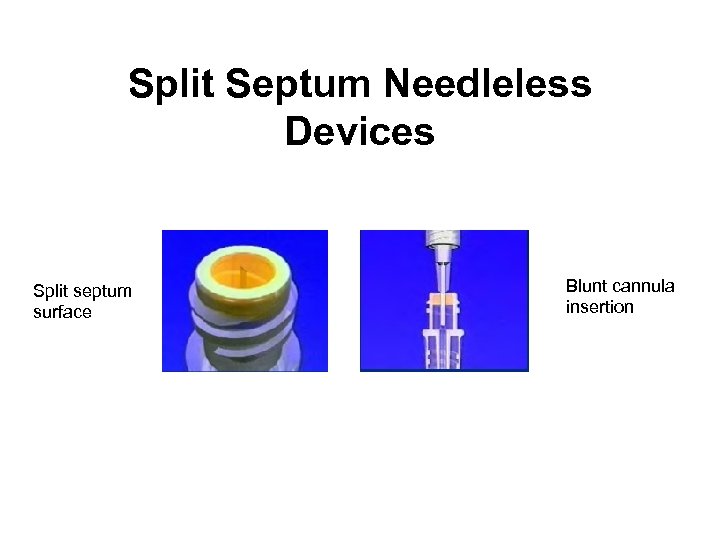
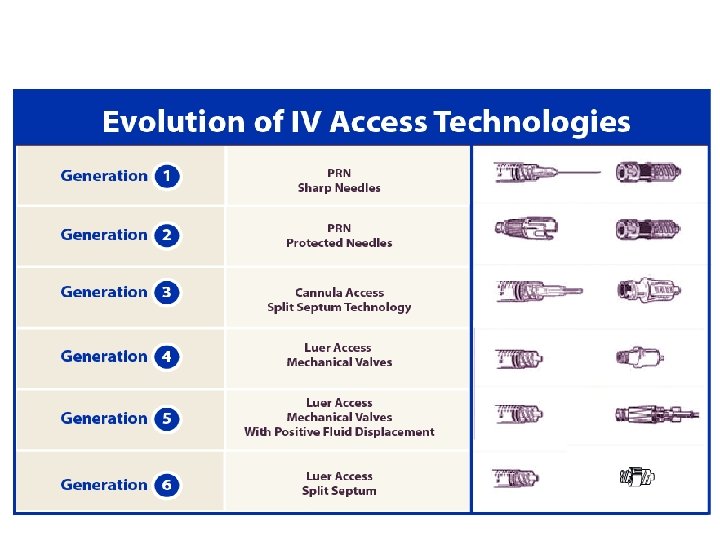
Split Septum Needleless Devices Split septum surface Blunt cannula insertion
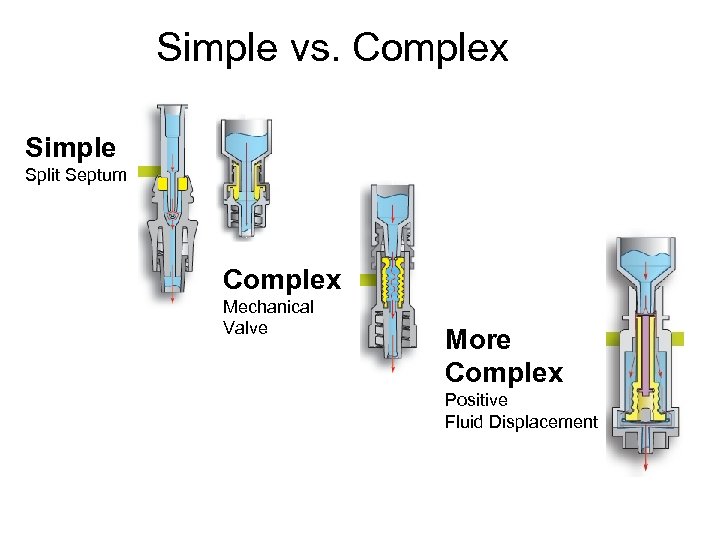
Simple vs. Complex Simple Split Septum Complex Mechanical Valve More Complex Positive Fluid Displacement
Mechanical Valves For illustrative purposes only. Does not imply association with increased BSIs.

Increased BSIs Temporally Associated With the Introduction of A Mechanical Valve (MV) Needleless Device (ND) • Hospital: University of Virginia (hospital-wide) • Problem: Increased BSI rate after introduction of a MV ND in May 2002 – By June 2002, nosocomial BSI rate increased 61%; – January-May 2002 vs. May-December 2002: BSI rate: 2. 2 vs. 3. 5 per 1000 pt-days (RR=1. 6, p<. 0001) – 2. 9 -fold increase in CR-BSI with common skin organisms. – 1. 8 -fold increase in CR-BSI with non-skin organisms. Hall K et al, SHEA Annual Meeting 2004
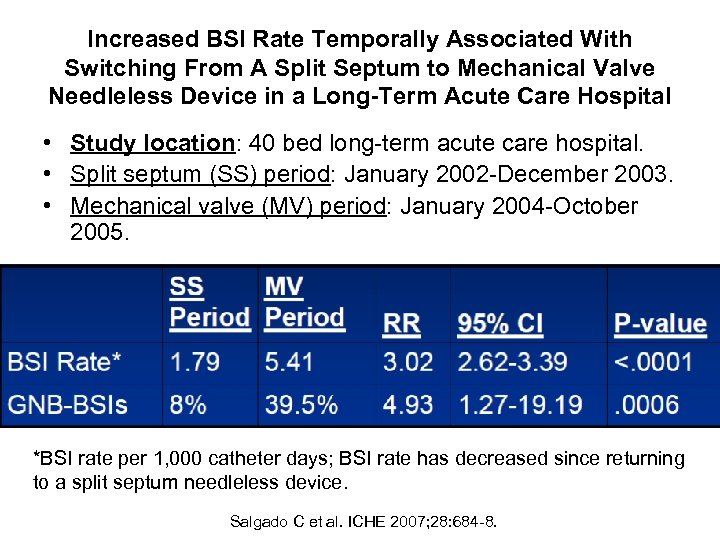
Increased BSI Rate Temporally Associated With Switching From A Split Septum to Mechanical Valve Needleless Device in a Long-Term Acute Care Hospital • Study location: 40 bed long-term acute care hospital. • Split septum (SS) period: January 2002 -December 2003. • Mechanical valve (MV) period: January 2004 -October 2005. *BSI rate per 1, 000 catheter days; BSI rate has decreased since returning to a split septum needleless device. Salgado C et al. ICHE 2007; 28: 684 -8.
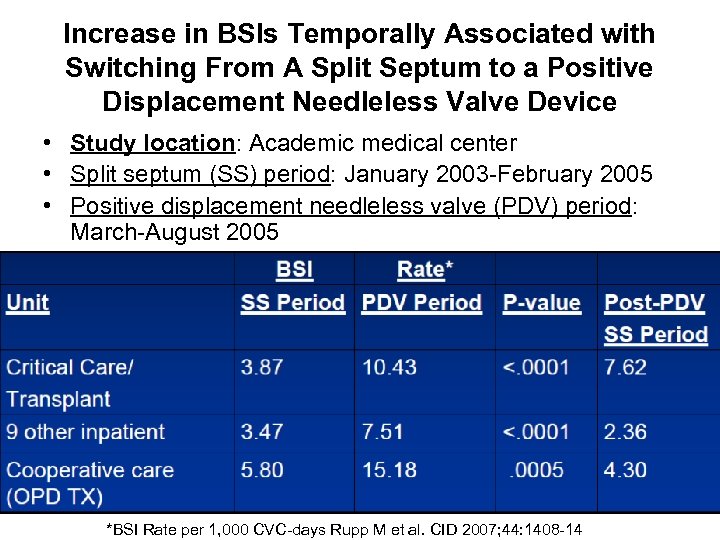
Increase in BSIs Temporally Associated with Switching From A Split Septum to a Positive Displacement Needleless Valve Device • Study location: Academic medical center • Split septum (SS) period: January 2003 -February 2005 • Positive displacement needleless valve (PDV) period: March-August 2005 *BSI Rate per 1, 000 CVC-days Rupp M et al. CID 2007; 44: 1408 -14

How May the Mechanical Valves Lead to BSIs? • Location: Wake Forest University School of Medicine. • Study Design: Quantitative cultures of blood from ICU patients drawn through MV ND from December 12, 2004 to January 21, 2005 (initial syringe pull back of morning blood draw). • Results: – – 226 “discards” obtained from 83 patients. 39/226 (17%; range 8% to 50%, by unit) culture positive. Colony forming units (CFU/ml): median=0. 3, range 0. 1 ->100. Pathogens: 25 CNS, 5 yeast, 2 S. aureus, 2 each Serratia or Enterococcus spp. , 1 each S. maltophilia or Acinetobacter spp. ; 31% would be considered pathogens in a blood culture. – 31% of nurses did not disinfect the MV before accessing system. Karchmer TB et al. SHEA 2005, Abstract #307

Disinfection of Needleless Catheter Connectors • Study design: In vitro study. – 3 luer-activated valved connectors (Clearlink [Baxter Healthcare], Posi. Flow [Becton-Dickinson], and Micro CLAVE [ICU Medical]) were studied. – 36 connectors from each tested concurrently. – One device as control, the rest inoculated by immersing the membranous surface in a suspension of E. faecalis containing >108 colony forming units (CFUs) per ml. Septum allowed to dry for 24 hours (final inoculum 105 CFU/ml). – Accessed by sterile syringe containing 3 ml of sterile tryptocase soy broth and flushed with broth. Menyhay and Maki ICHE 2006; 27: 23 -27
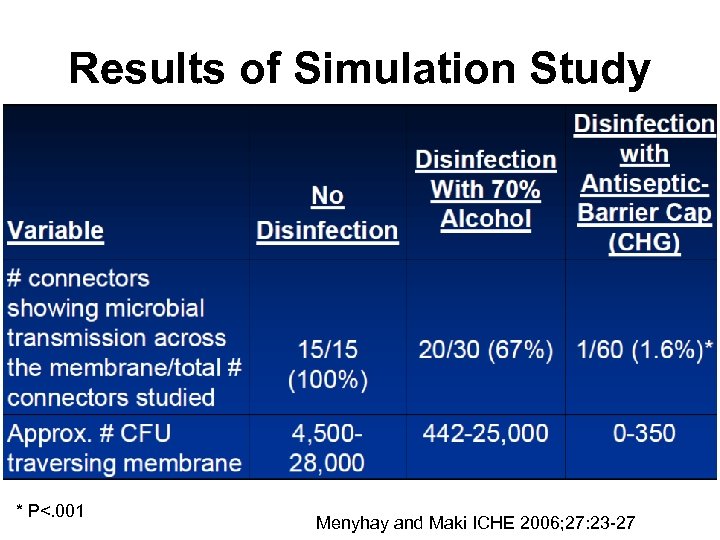
Results of Simulation Study * P<. 001 Menyhay and Maki ICHE 2006; 27: 23 -27

Disinfection of Mechanical Valves • Study design: 300 MVs (4 types from 3 manufacturers) were tested. Each septum inoculated with 105 CFUs/ml of S. epidermidis, S. aureus, P. aeruginosa, and/or C. albicans. Membranous septum disinfected for 15 seconds with friction, using 70% alcohol or 3. 15% chlorhexidine/70% alcohol (Chlorascrub™). 0. 9% nonbacteriostatic saline flush solutions were collected downstream and quantitatively cultured. • Results: Disinfection of the membranous septum for 15 seconds with friction, using either 70% alcohol alone or 3. 15% chlorhexidine/70% alcohol (Chlorascrub™) was equally effective in preventing the transfer from the membranous septum downstream in the process of accessing the ports. Kaler W et al. JAVA 2007
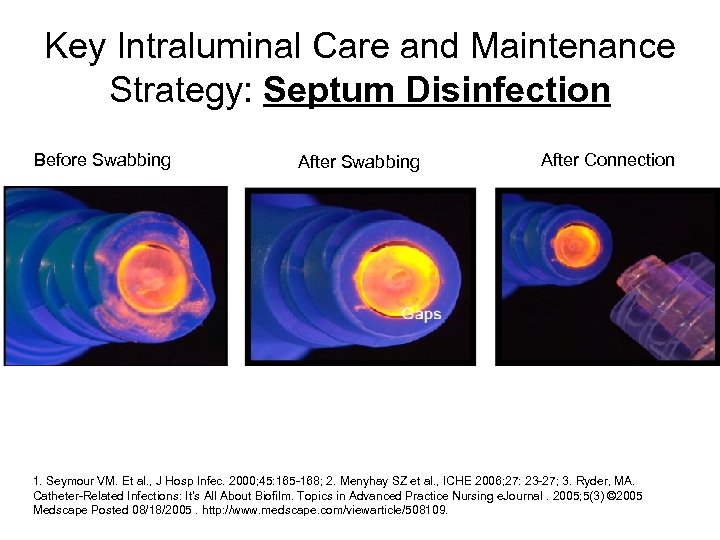
Key Intraluminal Care and Maintenance Strategy: Septum Disinfection Before Swabbing After Connection 1. Seymour VM. Et al. , J Hosp Infec. 2000; 45: 165 -168; 2. Menyhay SZ et al. , ICHE 2006; 27: 23 -27; 3. Ryder, MA. Catheter-Related Infections: It's All About Biofilm. Topics in Advanced Practice Nursing e. Journal. 2005; 5(3) © 2005 Medscape Posted 08/18/2005. http: //www. medscape. com/viewarticle/508109.

Change Mechanical Valves and Bloodstream Infections • Study design: Intervention study. Changed from an opaque to clear mechanical valve. Change the mechanical valve whenever blood is visible (e. g. , after blood infusion or withdrawal). VA Hospital, Seattle, WA. • Results: CR-BSI rate: 7. 4 to 1. 5 per 1, 000 linedays (p<0. 05). Blood culture contamination: 4. 4 to 1. 8 per 1, 000 line-days. Cost savings: $241, 000. Valve replacement increased 52%. • Conclusion: Changing to a clear valve and changing it whenever blood contamination visible reduced CR-BSIs, blood culture contamination, and saved money. 2007 VHA MRSA Prevention Forum, Orlando, FL, Nov 2007
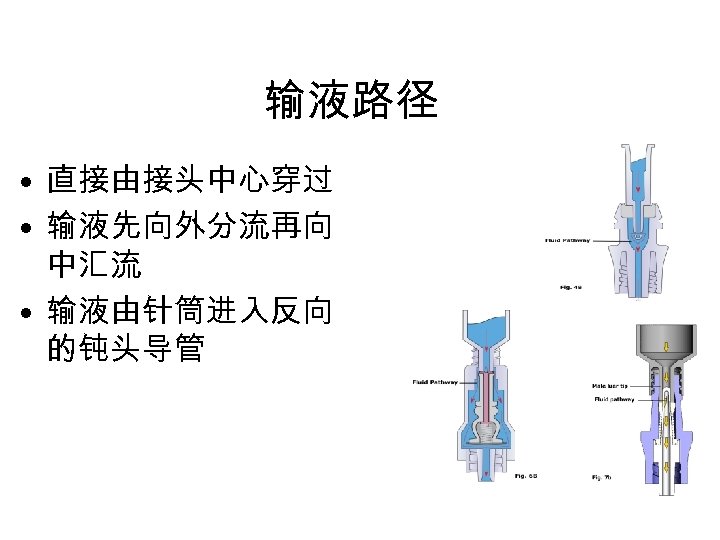
输液路径 • 直接由接头中心穿过 • 输液先向外分流再向 中汇流 • 输液由针筒进入反向 的钝头导管
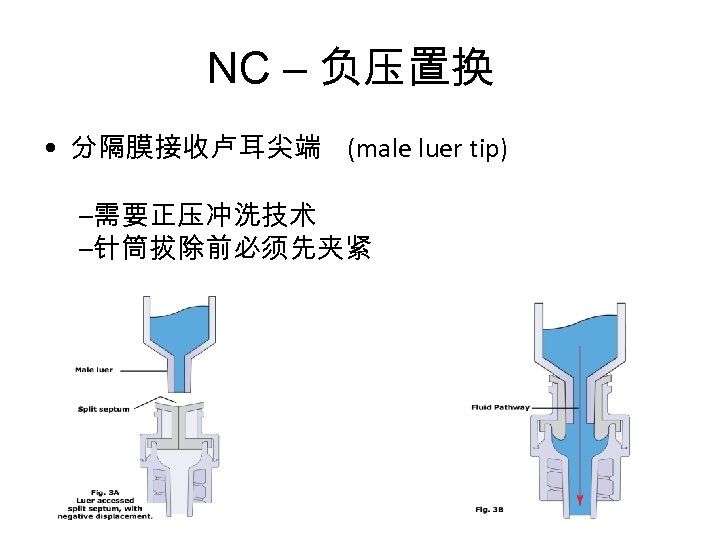
NC – 负压置换 • 分隔膜接收卢耳尖端 (male luer tip) –需要正压冲洗技术 –针筒拔除前必须先夹紧
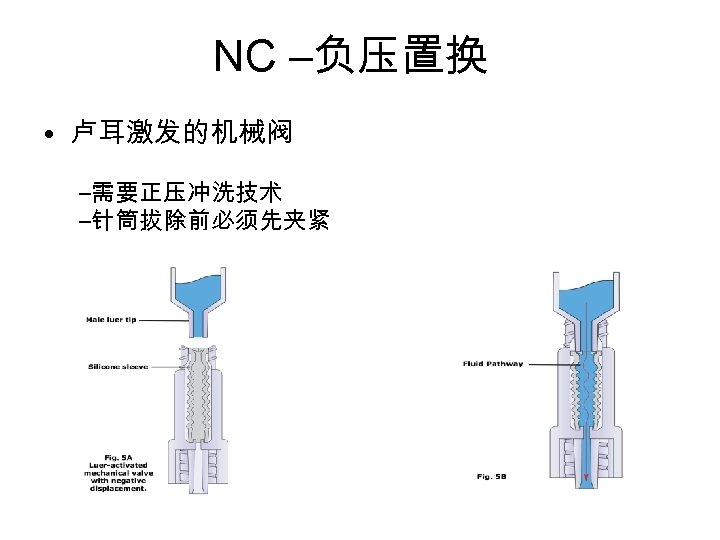
NC –负压置换 • 卢耳激发的机械阀 –需要正压冲洗技术 –针筒拔除前必须先夹紧
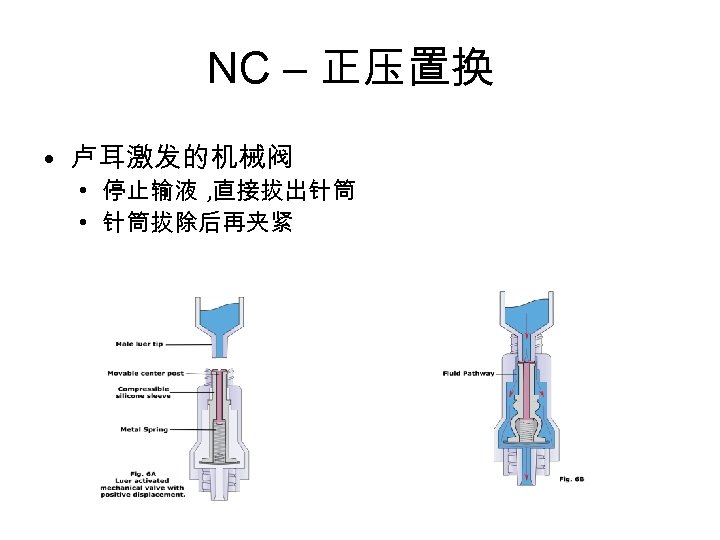
NC – 正压置换 • 卢耳激发的机械阀 • 停止输液 , 直接拔出针筒 • 针筒拔除后再夹紧
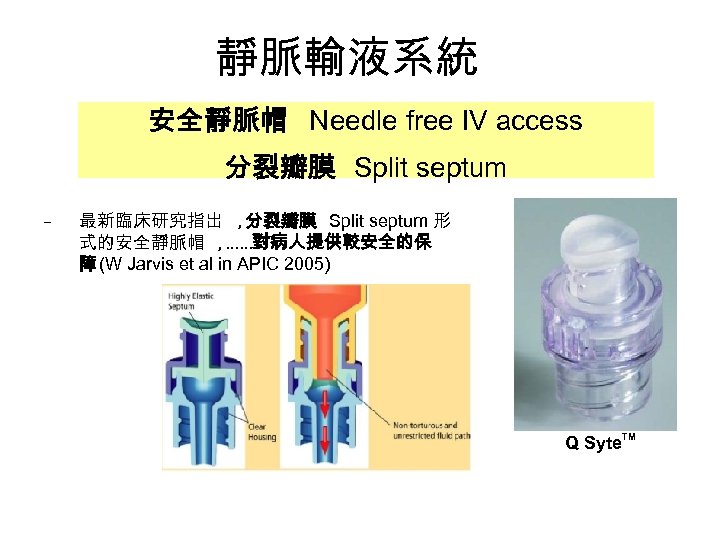
靜脈輸液系統 安全靜脈帽 Needle free IV access 分裂瓣膜 Split septum – 最新臨床研究指出 , 分裂瓣膜 Split septum 形 式的安全靜脈帽 , ……對病人提供較安全的保 障 (W Jarvis et al in APIC 2005) Q Syte TM

Flushing is Important • Flushing removes blood, glucose or other contaminants that may support microbial growth. • Use of pre-filled syringes for flushing reduces the risk of extrinsic contamination. • Use of pre-filled syringes avoids outbreaks associated with vial contamination/re-use.

導管 維護與沖管 • 放置導管是為了因應 臨床治療 之 需 求 , 但導管維護不良 將影響治療 成效 , 進而可能造成 : ü治療週期 延長 , 如 : 因導管相關併 發症產生的醫療需求 ü住院天數增加 ü增加病患因 更換導管的 疼痛不適 及 風險 ü放置 新導管的成本 導管沖洗 是 評估 並保持 導管 暢通 的重要 管路維護技術之一

沖洗導管的目的 沖管是透過針筒約 0. 9%生理鹽水注入導管內 , 以達到 : – – – – 確認導管在首次插入時的正確位置 確保導管在輸藥前的正確位置 確保藥物已無殘留在導管內 , 完全投與病人 確保將兩種不相容的藥物分隔開 , 避免不良反應 在中央靜脈導管抽血前後或輸血後保持導管通暢 在輸送營養液後沖管確保導管無乳脂殘留 保持間歇輸藥的導管孔道通暢 減少管路 流量 異常
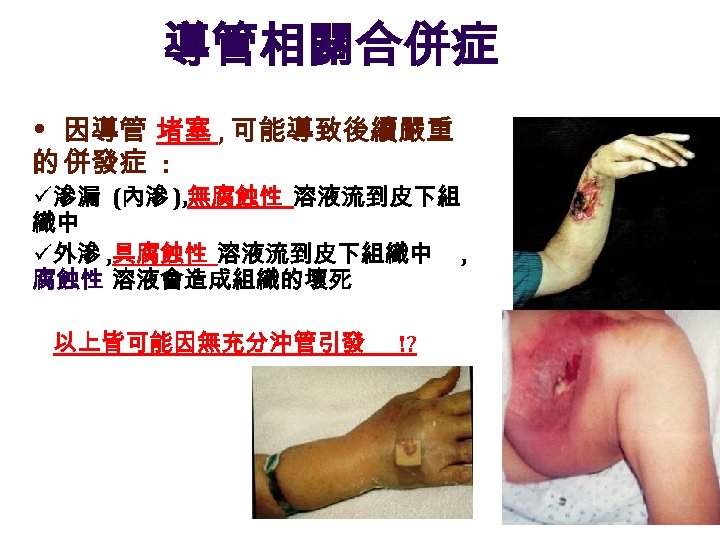
導管相關合併症 • 因導管 堵塞 , 可能導致後續嚴重 的 併發症 : ü 滲漏 (內滲 ), 無腐蝕性 溶液流到皮下組 織中 ü 外滲 , 具腐蝕性 溶液流到皮下組織中 , 腐蝕性 溶液會造成組織的壞死 以上皆可能因無充分沖管引發 !?
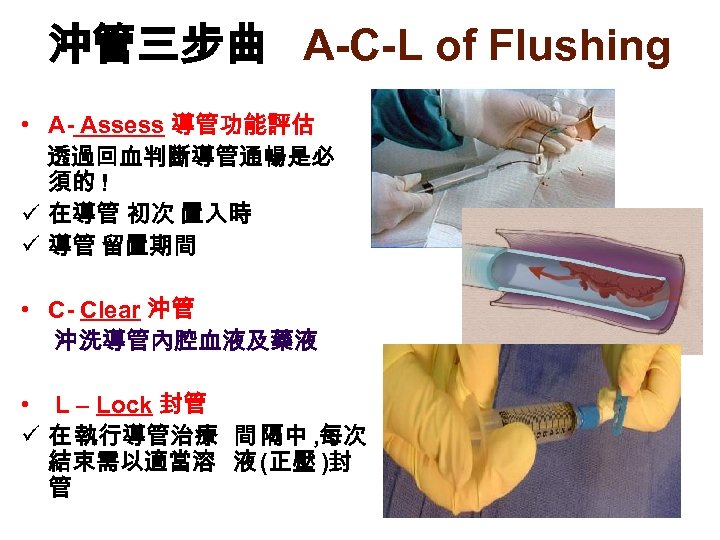
沖管三步曲 A-C-L of Flushing • A- Assess 導管功能評估 透過回血判斷導管通暢是必 須的 ! ü 在導管 初次 置入時 ü 導管 留置期間 • C- Clear 沖管 沖洗導管內腔血液及藥液 • L – Lock 封管 ü 在 執行導管治療 間 隔中 , 每次 結束需以適當溶 液 (正壓 )封 管
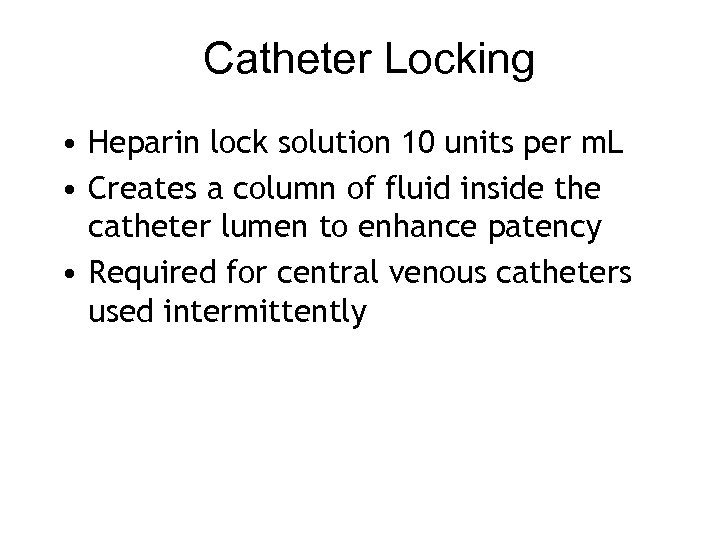
Catheter Locking • Heparin lock solution 10 units per m. L • Creates a column of fluid inside the catheter lumen to enhance patency • Required for central venous catheters used intermittently

沖封管的 方法 在中央靜脈導管 • SASH üS =鹽水 Saline üA=給藥 Administer medication üS=鹽水 Saline üH=肝素鹽水 Heparin 在外周短靜脈導 管 • SAS üS =鹽水 Saline üA=給藥 Administer medication üS=鹽水 Saline

Peripheral Catheters • 2 to 3 m. Ls Normal saline • Flushing & locking • Heparin is not used • No difference in patency of peripheral catheters with saline vs heparin

沖管溶液的種類 • 多劑量的沖管液 – 大瓶裝,如 : 30 ml 0. 9% 生 理鹽水 – 大 輸液袋,如 : 250 ml、 500 ml或者 1000 ml 的 0. 9% 生理鹽水 多次抽取使用 -----高 風險!

Multiple Dose Vials (MDV) • RISK far outweighs their benefits • Bacterial contamination rates from 0% to 27% • Contains benzyl alcohol as preservative – Limit volume of bacteriostatic normal saline to no more than 30 m. L in 24 hours in adults – Contraindicated in neonates • Bacteriostatic, not bacteriocidal

多劑量輸液袋 / 瓶 • 由護 理人員手 準備的沖洗注射器 易 產生污染 – 根據 英國醫院腫瘤、重症監護室和透析室 研究發現 , 以 無菌 空針從 單劑量瓶 溶液由 護士手 配置的沖洗液 中,污染機率高達 8%
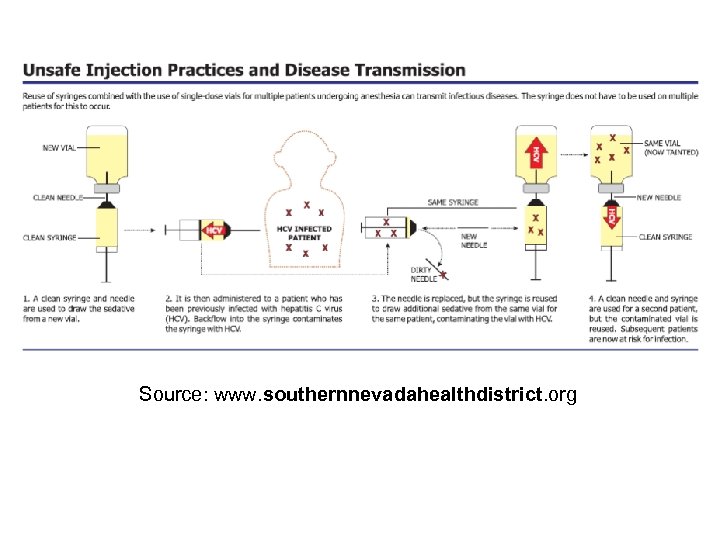
Source: www. southernnevadahealthdistrict. org

傳統配製與預充式的對 研究 比 • 回顧從 25個中度急症 的護理 單位收集 的 研 究 資料 (Rosenthal, 1999) – 到院 患者 均 使用中心靜脈導管 – 62位 患者使用 2 個月 以上的 預充式 針筒 沖洗 靜 脈管路 • 發現 3 例併發症 : 1例導管相關性感染, 1例靜 脈炎,1例 導管 堵塞 – 64位 患者使用 2 個月 以上 護士手 配置 的 沖洗 液進行沖洗 靜脈管路 • 發現 15例併發症 : 10 例感染,5 例堵塞,3 例 靜脈炎

Worthington Study (2001) • 研究使用無菌空針由 單次使用的安瓶抽取生 理鹽水作為靜脈導管沖洗 • 研究單位包括 : 加護病房 , 腫瘤科 , 洗腎中心及 外科病房等 • 研究發現 : – 8%污染 來自針筒尖端及生理鹽水 , 且延伸污染到中 央靜脈導管及導管接頭 – 護士沒有在打開前先消毒生理鹽水安瓶外周表面 • 後繼研究用預充式生理鹽水 , 發現 可節省 68% 時 • 建議 : 應盡早採納最新沖管基準 Worthington et al, “ Are contaminated flush solutions an overlooked source for catheter related sepsis? ” in The Hospital Infection Society. 2001, p. 81 -83

Calop Study (2000) • 手 配置預充式生理鹽水來進行外周靜脈導管 及中央導管沖管 – 8% 手 預充的生理鹽水受污染 – 對照研究 : Trautmann 等人也觀察到有 7. 8% 生理鹽水受污染 Calop J, “Maintenance of peripheral and central intravenous infusion devices by 0. 9% sodium chloride with or without heparin as a potential source of catheter microbial contamination” in Journal of Hospital Infection. 46: 161 -162則

預充式生理食鹽水特點
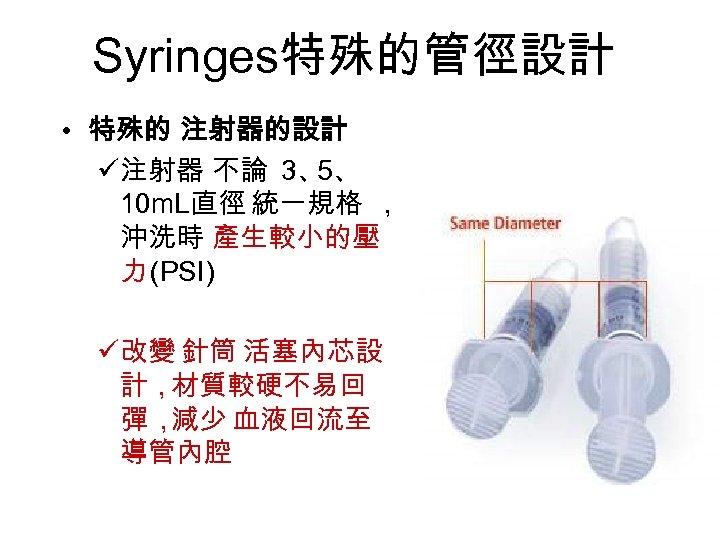
Syringes特殊的管徑設計 • 特殊的 注射器的設計 ü注射器 不論 3、 5、 10 m. L直徑 統一規格 , 沖洗時 產生較小的壓 力 (PSI) ü改變 針筒 活塞內芯設 計,材質較硬不易回 彈, 減少 血液回流至 導管內腔
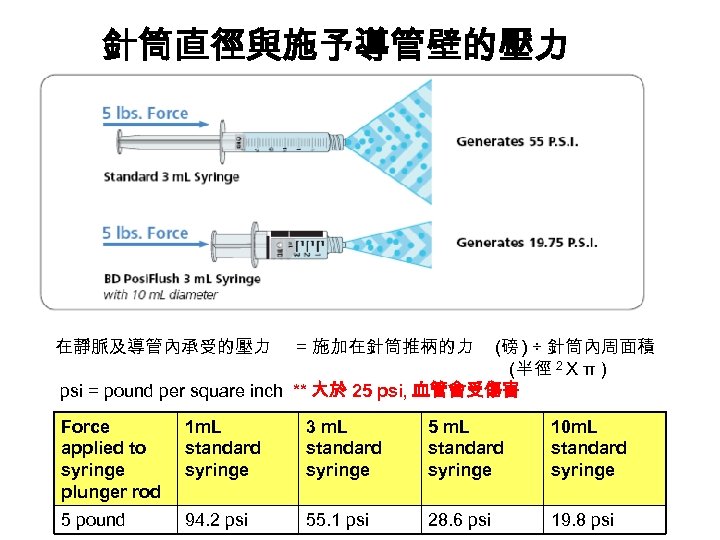
針筒直徑與施予導管壁的壓力 在靜脈及導管內承受的壓力 = 施加在針筒推柄的力 (磅 ) ÷ 針筒內周面積 (半徑 2 X π) psi = pound per square inch ** 大於 25 psi, 血管會受傷害 Force applied to syringe plunger rod 1 m. L standard syringe 3 m. L standard syringe 5 m. L standard syringe 10 m. L standard syringe 5 pound 94. 2 psi 55. 1 psi 28. 6 psi 19. 8 psi

BD Posi. Flush. TM 沖管過程零回血 (針對 PICC的實驗 ) 獨特的內塞設計,減少血液迴流,差異比較 : 是 0 cm 與 14. 2 cm 的
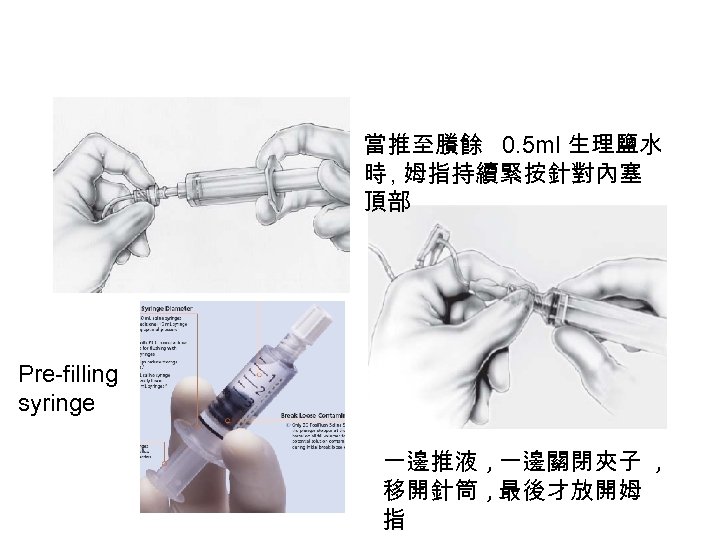
當推至賸餘 0. 5 ml 生理鹽水 時 , 姆指持續緊按針對內塞 頂部 Pre-filling syringe 一邊推液 , 一邊關閉夾子 , 移開針筒 , 最後才放開姆 指

Recommendations for Implementing Prevention and Monitoring Strategies: Approaches that should not be considered a routine part of CLA-BSI prevention • Do not use antimicrobial prophylaxis for shortterm or tunneled catheter insertion or while catheters are in situ (A-I) • Do not routinely replace CVCs or arterial catheters (A-I). • Do not routinely use positive-pressure needleless connectors with mechanical valves before a thorough assessment of risks, benefits, and education regarding proper use (B-II). Marschall J. et al. , Infect Control Hosp Epidemiology 2008; 29: S 22 -30.

Interventions That Prove That Implementation of Evidence. Based CVC-BSI Prevention Measures Can Prevent Infections, Save Lives, and Save Money

Central Line Insertion Checklist -Adults QUALITY IMPROVEMENT THIS FORM IS NOT PART OF THE PATIENT'S PERMANENT RECORD. Please return the form to your Nurse Manager. If a step has was not followed, please note and the Nurse Manager will follow up with the physician.
Developing a Physician Champion—Prevention Should Be The Focus of Clinicians, Not Just Infection Control Personnel.

Keys to Success • • • Senior leader support Clinical Champion Day to day leader A multidisciplinary team Staff buy-in Project sustainability

Eliminating Catheter-Associated Bloodstream Infections in an Intensive Care Unit • Study design: Prospective cohort study in a surgical ICU. Johns Hopkins Hospital. • Intervention: Staff education, catheter insertion cart, daily removal reminders, evidence-based guideline checklist, nurse empowerment. Jan 1998 -Dec 31, 2002. • Measurement: CVC-BSIs and guideline adherence. • Results: CVC-BSI rate decreased from 11. 3 to 0 per 1, 000 CVC-days. 43 CVC-BSIs, 8 deaths, and $1, 945, 922 saved. Berenholtz SM et al Crit Care Med 2004; 32: 2014 -20.

Keystone Project • Michigan Hospital Association • 127 intensive care units (ICUs) in Michigan and five other states. • 68 ICUs totally eliminated CVC-BSIs. • For 6 months, they eliminated VAP. • Estimates that they saved >1, 578 lives, reduced 81, 000 hospital days, and saved $165 million. • Hospitals in Rhode Island, New Jersey and Maryland are replicating the Keystone Project locally and others will follow. Pronovost P. et al NEJM 2006; 355: 2725 -32.

Keystone Project • Study design: Intervention cohort study in 108 Michigan Intensive care units (ICUs) over 18 months. Comparison of CVC-BSI rates before, during, and after intervention. • Results: 103 ICUs. 1, 981 months of ICU data and 375, 757 catheter-days. Conclusion: An evidence-based intervention resulted in a large and sustainable decrease (up to 66%) in CVCBSI rates that was maintained for 18 months. Pronovost P. et al NEJM 2006; 355: 2725 -32.

Conclusions • CVC-Related BSIs are a major cause of patient morbidity and mortality. • Prevention of CVC-Related BSIs requires a multi-factorial approach, including: – Implementation of CDC CVC-BSI Prevention Guideline (2002) or SHEA Compendium Recommendations (2008) – Implementing new prevention evidence (> October 2008). – Implementation of insertion and maintenance bundles. – Educating staff; Insuring adequate and properly trained staff – Insuring that policy = practice (clinician accountability) – Monitoring CVC insertion and maintenance processes and CVC-related BSI rates (outcomes). • A comprehensive CVC-related BSI prevention program can dramatically reduce infection rates and improve patient safety. • A rate of ZERO CVC-BSIs in ICU patients is a reality and should be our goal. If “Prevention is Primary”, then action is essential!

懇請賜教
0edf9c43b3917f997f9a44913738a6c9.ppt