19a7062b6f1490dabdf6f6512464b1d6.ppt
- Количество слайдов: 23

FEDERAL INSTITUTE FOR RISK ASSESSMENT Good Laboratory Practice GLP in practice Dr. Wolf Burchard Bulling Federal Institute for Risk Assessment Thielallee 88 -92 D-14195 Berlin Tel. +49 30 -8412 -3936 Fax +49 30 -8412 -3970 w. bulling@bfr. bund. de www. bfr. bund. de

OECD Council Decision 1981 OECD 1981 Decision of the OECD Council concerning the Mutual Acceptance of Data in the Assessment of Chemicals C(81)30(Final) ted lida ds Va tho Me Annex 1: OECD Test Guidelines LP G Annex 2: OECD GLP Principles Data accepted Wolf Burchard Bulling, Workshop on GLP, 17 December 2010, Zagreb Page 2

Safety Studies Scientific Technical Test Guidelines (scientific method) GLP-Principles (organisational framework) Use of the current state of the art Reliability of test facility and data Reconstruction of study conduct Quality assurance Report/Archiving Interpretation of data Regulatory Authority Monitoring Authority Recognition of Data Wolf Burchard Bulling, Workshop on GLP, 17 December 2010, Zagreb Page 3
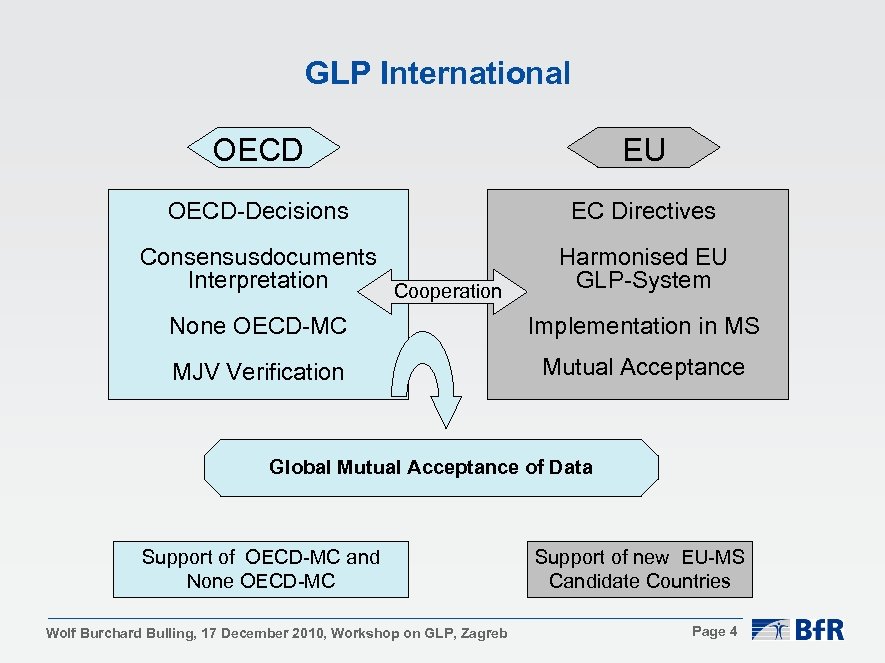
GLP International OECD EU OECD-Decisions EC Directives Consensusdocuments Interpretation Cooperation Harmonised EU GLP-System None OECD-MC Implementation in MS MJV Verification Mutual Acceptance Global Mutual Acceptance of Data Support of OECD-MC and None OECD-MC Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Support of new EU-MS Candidate Countries Page 4

Legal aspects Principles Monitoring OECD Council Decision 1981 Mutual Acceptance of Data Annex 2: GLP Principles (rev. 1997) OECD Decision-Recommendation 1989, - Monitoring Procedures rev. 1995 - Conduct of Inspections - Exchange of Information EU Council Directive 2004/10/EC (87/18/EEC 99/11/EC) EU Council Directive 2004/9/EC (88/320/EEC 90/18/EEC, 99/12/EC) D: Chemicals Act (Chem. G) 2008 (1990, 1994, 2002) D: GLP General Administrative Provision (Chem. Vw. V-GLP) 1997 (1990) Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 5

Documents OECD Series on GLP – OECD GLP Principles (1) – Compliance Monitoring (2, 3, 9) – Consensus Documents (4, 5, 6, 7, 8, 10, 13) – Advisory Documents (11, 12, 14, 15) Documents are available on the OECD GLP Website www. oecd. org/env/glp Additional National Consensus Documents (e. g. Germany): - Archiving, Computerised systems, Multi-Site Studies, GLP Inspectors Manual (www. bfr. bund. de) Checklists for Inspection (not in Germany) Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 6
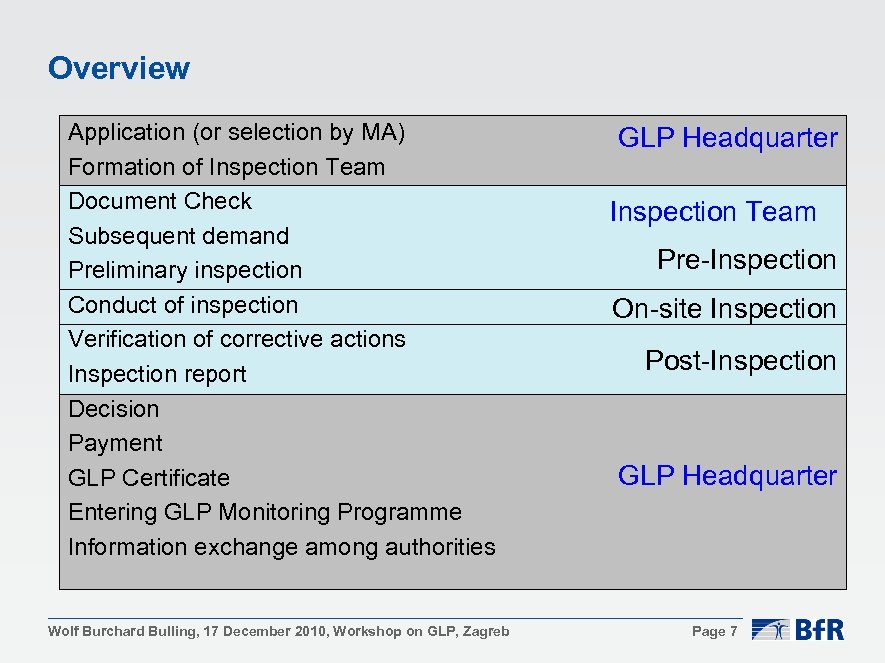
Overview Application (or selection by MA) Formation of Inspection Team Document Check Subsequent demand Preliminary inspection Conduct of inspection Verification of corrective actions Inspection report Decision Payment GLP Certificate Entering GLP Monitoring Programme Information exchange among authorities Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb GLP Headquarter Inspection Team Pre-Inspection On-site Inspection Post-Inspection GLP Headquarter Page 7

GLP Inspectors University degree At least two inspectors Regular training activities External experts GLP training courses Communication skills Exchanging experience Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Relevant experience Page 8

GLP Area of Expertise Studies required by regulations for the purpose of licensing of pharmaceuticals, pesticides, food and feed additives, cosmetic products, veterinary drugs and industrial chemicals GLP Study (Definition OECD Principles, 97) Non-clinical health and environmental safety study, means an experiment or set of experiments in which a test item is examined under laboratory conditions or in the environment to obtain data on its properties and/or its safety, intended for submission to appropriate regulatory authorities. GLP Studies are summarized in 9 OECD Test Categories Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 9

GLP Area of Expertise Activities not covered by GLP (Scope of GLP) Ø Studies not required by Regulatory Authorities for risk assessment Ø Test Guidelines Ø Suitability of the design of studies or the studies objectives Ø Suitability of the test system used Ø Scientific issues Ø Interpretation of the findings of the studies Ø Compliance with animal welfare legislation Ø Compliance with health and safety legislation Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb If study results are not influenced Page 10

Key Elements of GLP ORGANISATION adequate environment for the experimental work PLAN - PERFORM - REPORT a study in a proper way and while doing so RECORD (for traceability) MONITOR (for quality) ARCHIVE (for reconstruction) Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 11

Key Elements of GLP Ø Ø Ø Ø Ø Organisation and Personnel Quality Assurance Programme Facilities Apparatus, Materials, Reagents and Specimens Test Systems Test and Reference Substances Standard Operating Procedures Performance of the Study Reporting of Study Results Storage and Retention of Records (Headlines of the GLP Principles) Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 12

Outline of an on-site GLP Inspection Structure Ø Pre-Inspection Ø Starting Conference Ø Laboratory Walk Ø Study Audits (on-going, completed) Ø Closing Conference Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 13

Practical auditing Audit strategy All items from the Principles should be covered Ø Discussion of submitted documents Ø Selection and detailed check of specific examples (Personnel, Equipment, SOPs, Studies …) Ø Systematic approach to problematic areas Ø Procedures verified by documentation Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 14

Practical auditing The W rule for documentation Who did What, When, Where, What with, and Why The fundamental principle of GLP: not recorded = not done Giant Lot of Paper Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 15

Practical auditing GLP Inspector Ø Good working atmosphere Ø Be friendly, helpful and diplomatic Ø Be open and really Listen Ø Try to understand Ø Sharing information Ø Lead when you must Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 16

Practical auditing Each test facility may find their own way to implement all the requirements of the GLP Principles, because room is left in GLP for flexibility Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 17

Inspection Report Compulsary part of each GLP inspection Finalised in a timely manner after the inspection Confidential OECD Guidance (No. 9) for the Preparation Summary Introduction Narrative Exit Discussion Annexes Other Information Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 18
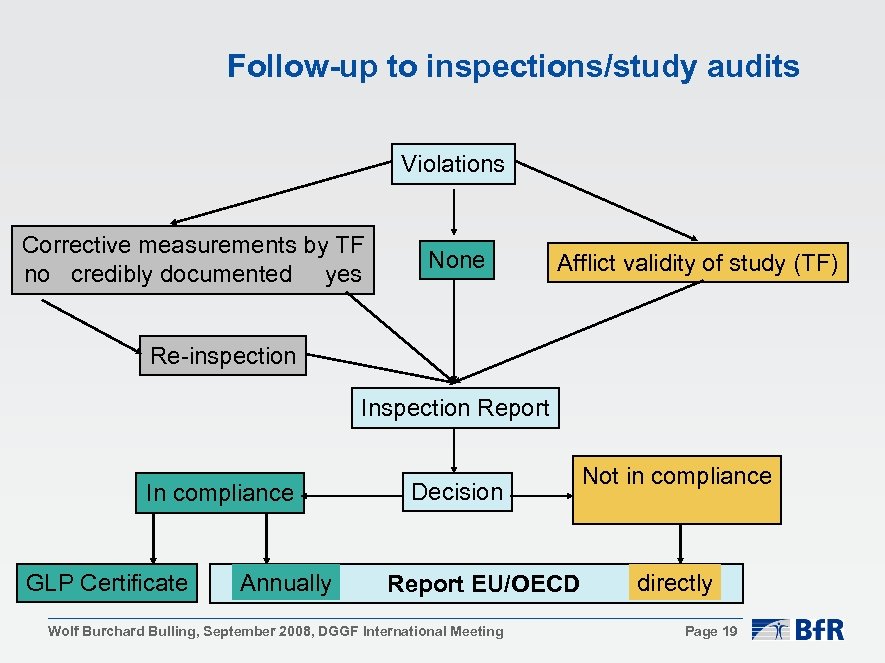
Follow-up to inspections/study audits Violations Corrective measurements by TF no credibly documented yes None Afflict validity of study (TF) Re-inspection Inspection Report In compliance GLP Certificate Annually Decision Report EU/OECD Wolf Burchard Bulling, September 2008, DGGF International Meeting Not in compliance directly Page 19
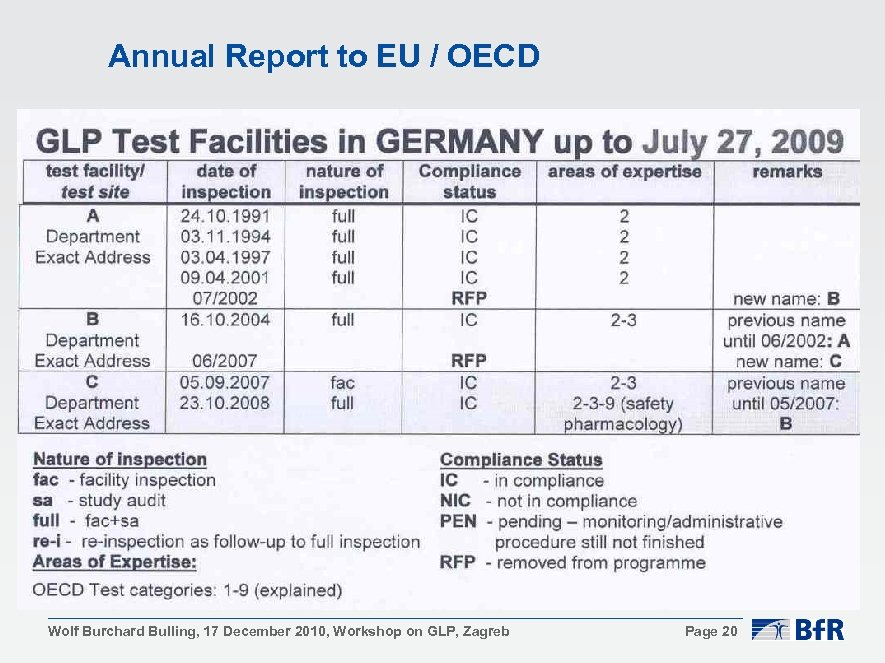
Annual Report to EU / OECD Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 20
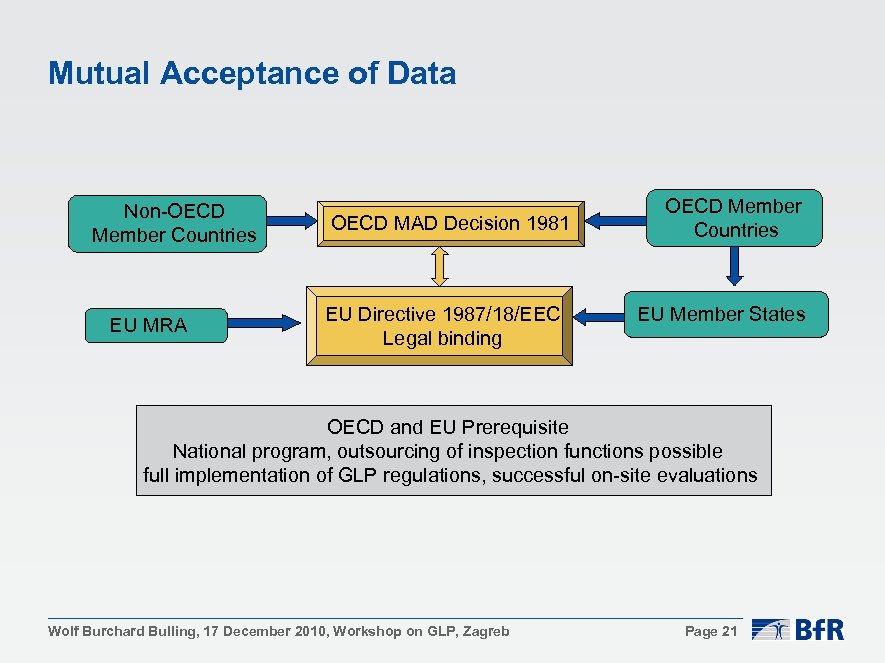
Mutual Acceptance of Data Non-OECD Member Countries EU MRA OECD MAD Decision 1981 EU Directive 1987/18/EEC Legal binding OECD Member Countries EU Member States OECD and EU Prerequisite National program, outsourcing of inspection functions possible full implementation of GLP regulations, successful on-site evaluations Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 21

GLP can not always prevent poor quality or falsification of data, but a robust GLP quality system makes it difficult and acts as a good deterrent. Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 22

Thank you for your attention! ANY QUESTIONS? Wolf Burchard Bulling, 17 December 2010, Workshop on GLP, Zagreb Page 23
19a7062b6f1490dabdf6f6512464b1d6.ppt