16525b40e740d5e96003aefd6e31ebf2.ppt
- Количество слайдов: 22

Chem 106: Class/ Lab Week 11 Pick a vial and a plastic dropper Using the vial number, sign-in on the Lab roster 1) TODAY: Fluid Exchange (Course/ Lab Manual pp. 89 -90) 2) Acid-Base Equilibrium Experiment (Course/ Lab Manual pp. 79 -82) Due & signed Today

Completed Report & Post Lab Questions Due Today: Course/ Lab Manual pg. 46 Plus Post Lab http: //chemconnections. org/general/chem 106/ethanol-ques-106. pdf Post Lab to turn in individually: [Each Partner turns in a completed form]

Chem 106: Class/ Lab Week 11 Follow Instructions http: //chemconnections. org/general/chem 120/fluid -ex. 106. html

Acid-Base Indicators
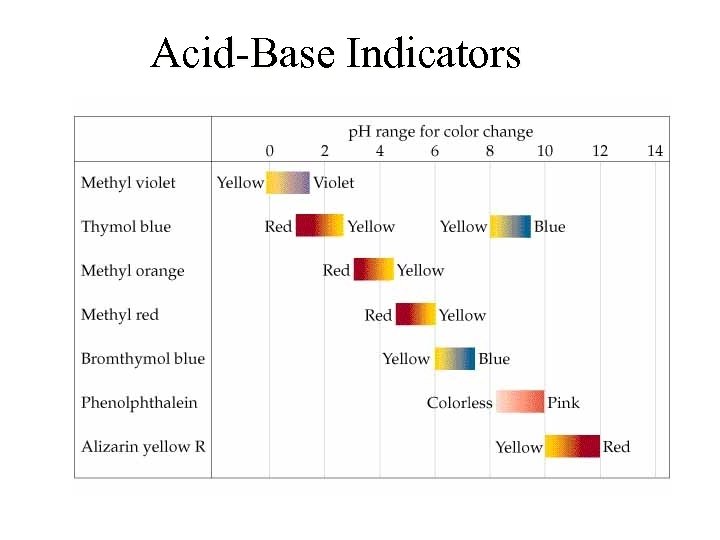
Acid-Base Indicators
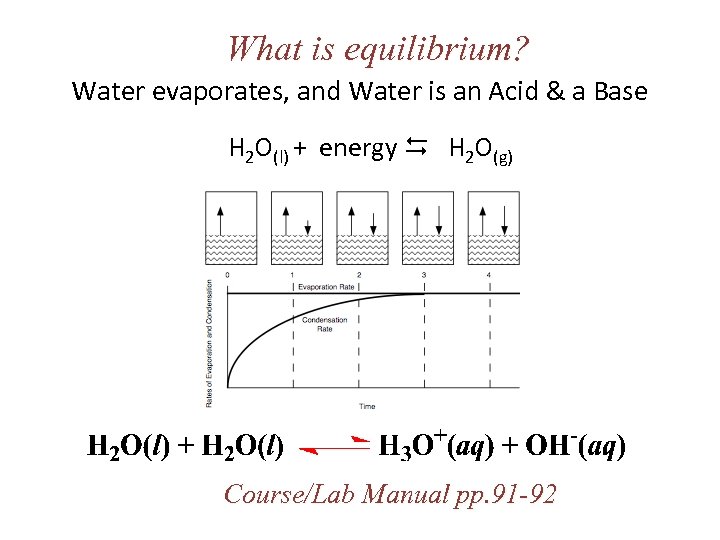
What is equilibrium? Water evaporates, and Water is an Acid & a Base H 2 O(l) + energy H 2 O(g) Course/Lab Manual pp. 91 -92
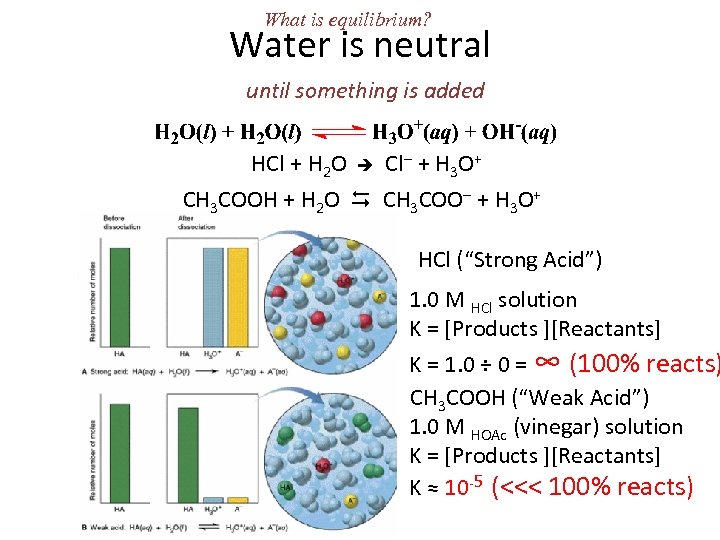
What is equilibrium? Water is neutral until something is added HCl + H 2 O Cl + H 3 O+ CH 3 COOH + H 2 O CH 3 COO + H 3 O+ HCl (“Strong Acid”) 1. 0 M HCl solution K = [Products ][Reactants] K = 1. 0 ÷ 0 = ∞ (100% reacts) CH 3 COOH (“Weak Acid”) 1. 0 M HOAc (vinegar) solution K = [Products ][Reactants] K ≈ 10 -5 (<<< 100% reacts)

What is equilibrium? Water is neutral It is an Acid and a Base until something is added (“Equilibrium Constant”) K = [Products ][Reactants] Reactants Products For example weak acids EXPERIMENT #1 TIME 1 2 3 4 5 6 7 8 9 10 11 12 13 EXPERIMENT #2 Amount Reactant 100 75 59 48 41 37 34 32 31 30 29 29 29 Amount Product 0 25 41 42 59 63 64 68 69 70 71 71 71 K = [71][29] K = 2. 4 TIME 1 2 3 4 5 6 7 8 9 10 11 12 13 Amount Reactant 0 10 16 20 22 24 25 26 27 28 29 29 29 Amount Product 100 90 84 80 78 76 75 74 73 72 71 71 71 K = [71][29] K = 2. 4
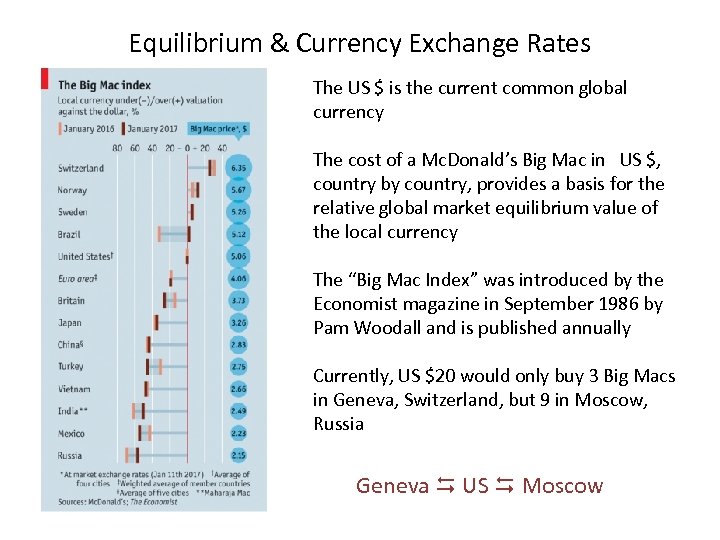
Equilibrium & Currency Exchange Rates The US $ is the current common global currency The cost of a Mc. Donald’s Big Mac in US $, country by country, provides a basis for the relative global market equilibrium value of the local currency The “Big Mac Index” was introduced by the Economist magazine in September 1986 by Pam Woodall and is published annually Currently, US $20 would only buy 3 Big Macs in Geneva, Switzerland, but 9 in Moscow, Russia Geneva US Moscow

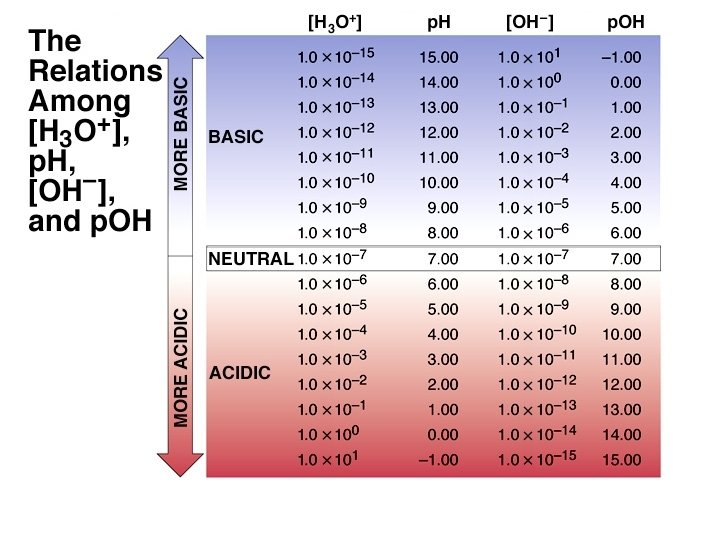
Water is neutral It is an Acid and a Base What is equilibrium? Reactants Products (“Equilibrium Constant”) K = [Products ][Reactants]

Water is neutral It is an Acid and a Base What is equilibrium? ¥ 2 O + H 2 O H 3 O+ + OH H acid 1 base 2 conj acid 2 conj base 1 ¥ w = 1 10 14 at 25°C K ¥ w = [H 3 O+ ][OH ] = [1 10 7 M] K
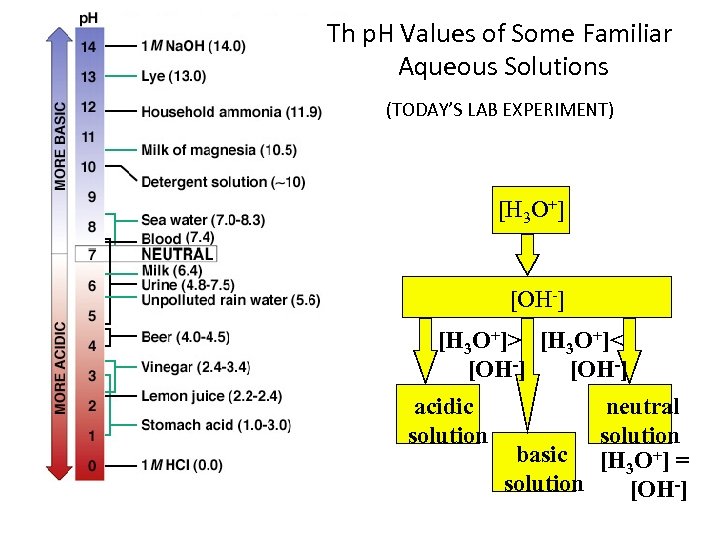
Th p. H Values of Some Familiar Aqueous Solutions (TODAY’S LAB EXPERIMENT) [H 3 O+] [OH-] [H 3 O+]> [H 3 O+]< [OH-] acidic solution neutral solution basic [H 3 O+] = solution [OH-]

BUFFERS Weak Acid- Weak Base Systems Example: H 2 CO 3(aq) / HCO 3 -1(aq) / CO 3 -2(aq) CO 2(g) + H 2 O (l) HCO 3 -1(aq) + H+1(aq) CO 3 -2(aq) + H+1(aq) https: //www. youtube. com/watch? v=XR_0 k 8 JIaw. Y https: //www. youtube. com/watch? v=ZLKEj. Xb. CU 30

H 2 CO 3(aq) / HCO 3 -1(aq) / CO 3 -2(aq) Two VERY IMPORTANT Acid-Base: Bicarbonate Buffer Systems CO 2(g) + H 2 O (l) HCO 3 -1(aq) + H+1(aq) CO 3 -2(aq) + H+1(aq) 1. Blood: a human’s blood serum volume is relatively small, 4 -6 Liters with a narrow p. H range, p. H = 7. 35 – 7. 45; p. H is maintained through buffering (homeostasis) Have you ever had respiratory alkalosis during an exam? 2. Oceans: an extraordinarily large volume of a “salt water” solution with a p. H ~ 8. 1; maintained through buffering

Human & Oceanic Bicarbonate Buffer Systems http: //chemconnections. org/general/chem 121/Buffers-Med. Pres. htm http: //chemconnections. org/general/chem 121/Buffers-CO 2 Oceans-2011. htm

EQUILIBRIUM CO 2 & Oceanic Bicarbonate Buffering CO 2(g) + H 2 O (l) HCO 3 -1(aq) + H+1(aq) CO 3 -2(aq) + H+1(aq) Oceans: p. H ~ 8. 1 and falling http: //www. tos. org/oceanography/issues/issue_archive/22_4. html Increasing CO 2 is decreasing ocean p. H; long term effects? http: //sos. noaa. gov/datasets/Ocean/ocean_acidification. html

Completed Report & Post Lab Questions Due Today: Course/ Lab Manual pg. 46 Plus Post Lab http: //chemconnections. org/general/chem 106/ethanol-ques-106. pdf Post Lab to turn in individually: [Each Partner turns in a completed form]
Theoretical & Percent Yield Calculations Distillation (Course/ Lab Manual pg. 46)
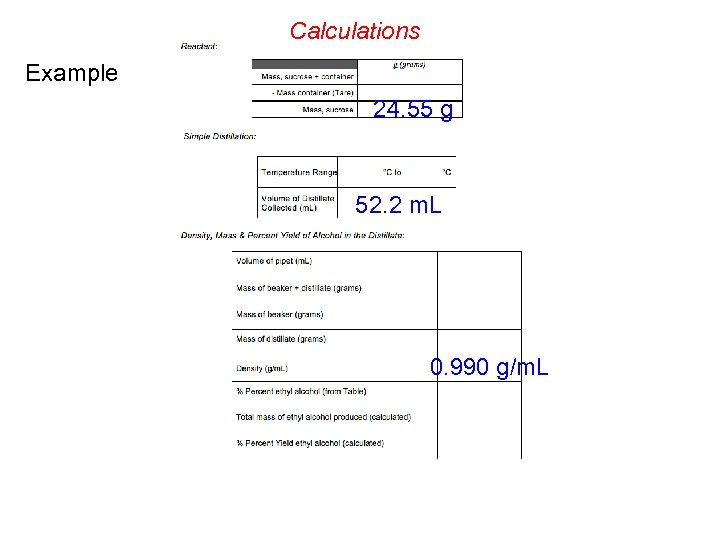
Calculations Example 24. 55 g 52. 2 m. L 0. 990 g/m. L
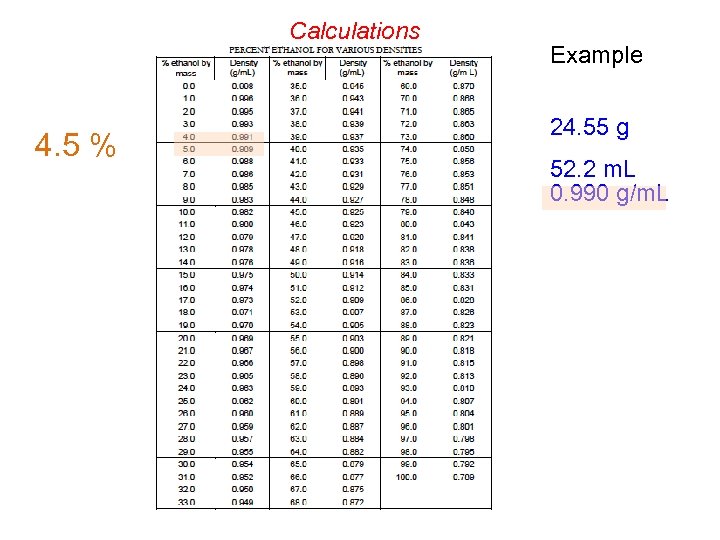
Calculations 4. 5 % Example 24. 55 g 52. 2 m. L 0. 990 g/m. L

Theoretical Yield Calculation 24. 55 g ? g (theoretical) 4 Molar mass = 342. 3 g/mol ? mol sucrose = 24. 55 g / 342. 3 g/mol = 0. 07172 mol Molar mass = 46. 07 g/mol ? mol C 2 H 5 OH = 4 x mol sucrose = 0. 2869 mol ? g (theoretical) = mol C 2 H 5 OH x 46. 07 g/mol = 13. 22 g _________ ? g (actual) = [4. 5 % , that is: 4. 5/100] x 52. 2 m. L x 0. 990 g/m. L = 2. 33 g % Yield = g (actual) / g (theoretical) x 100 = 17. 6 %
16525b40e740d5e96003aefd6e31ebf2.ppt