radical.ppt
- Количество слайдов: 51
 Chapter 6. Free Radical Polymerization 6. 1 Introduction 6. 2 Free Radical Initiators. 6. 3 Techniques of Free Radical Polymerization. 6. 4 Kinetic and Mechanism of polymerization. 6. 5 Stereochemistry of polymerization. 6. 6 Polymerization of Dienes 6. 7 Monomer Reactivity 6. 8 Copolymerization. POLYMER CHEMISTRY
Chapter 6. Free Radical Polymerization 6. 1 Introduction 6. 2 Free Radical Initiators. 6. 3 Techniques of Free Radical Polymerization. 6. 4 Kinetic and Mechanism of polymerization. 6. 5 Stereochemistry of polymerization. 6. 6 Polymerization of Dienes 6. 7 Monomer Reactivity 6. 8 Copolymerization. POLYMER CHEMISTRY
 6. 1 Introduction A. Type of polymerization Addition polymerization Condensation polymerization 1. Free-radical polymerization 2. Ionic polymerization 3. Complex coordination polymerization POLYMER CHEMISTRY
6. 1 Introduction A. Type of polymerization Addition polymerization Condensation polymerization 1. Free-radical polymerization 2. Ionic polymerization 3. Complex coordination polymerization POLYMER CHEMISTRY
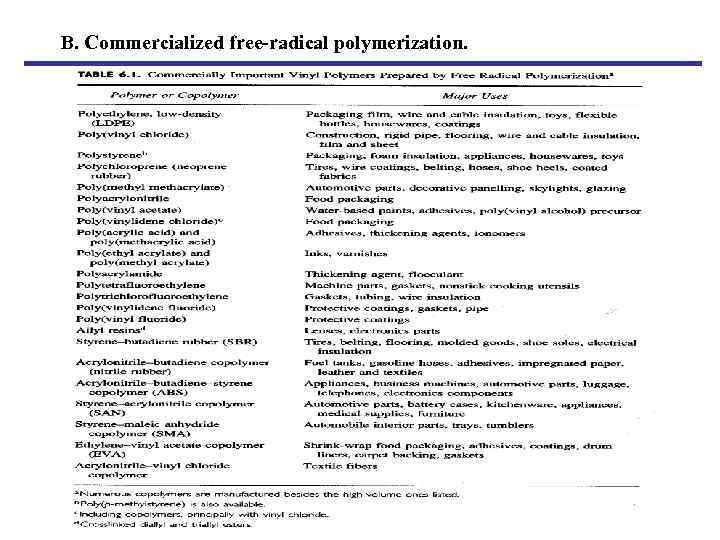 B. Commercialized free-radical polymerization.
B. Commercialized free-radical polymerization.
 6. 2 Free Radical Initiators. 6. 2. 1 Peroxides and Hydroperoxides A. Benzoly peroxide and other peroxides a. Thermal decomposition of BPO. b. Half-life of benzoyloxy radical : 30 min at 100℃ c. Cage effect : confining effect of solvent molecules. POLYMER CHEMISTRY
6. 2 Free Radical Initiators. 6. 2. 1 Peroxides and Hydroperoxides A. Benzoly peroxide and other peroxides a. Thermal decomposition of BPO. b. Half-life of benzoyloxy radical : 30 min at 100℃ c. Cage effect : confining effect of solvent molecules. POLYMER CHEMISTRY
 6. 2. 1 Peroxides and Hydroperoxides · (6. 5) · d. Other peroxides. Diacetyl peroxide Di-t-butyl peroxide (half-life: 10 hours at 120℃) · POLYMER CHEMISTRY
6. 2. 1 Peroxides and Hydroperoxides · (6. 5) · d. Other peroxides. Diacetyl peroxide Di-t-butyl peroxide (half-life: 10 hours at 120℃) · POLYMER CHEMISTRY
 6. 2. 1 Peroxides and Hydroperoxides e. Promoters : Inducing initiation at lower temperature. (6. 9) + + + - (6. 10) POLYMER CHEMISTRY
6. 2. 1 Peroxides and Hydroperoxides e. Promoters : Inducing initiation at lower temperature. (6. 9) + + + - (6. 10) POLYMER CHEMISTRY
 6. 2. 1 Peroxides and Hydroperoxides B. Hydroperoxide a. Thermal decomposition hydroperoxide b. Cumyl hydroperoxide. POLYMER CHEMISTRY
6. 2. 1 Peroxides and Hydroperoxides B. Hydroperoxide a. Thermal decomposition hydroperoxide b. Cumyl hydroperoxide. POLYMER CHEMISTRY
 6. 2. 2 Azo Compounds. A. α, α'-Azobis(isobutyronitrile) (AIBN). a. Decomposition of AIBN. b. Half-life of isobutyronitrile radical : 1. 3 hours at 80℃. POLYMER CHEMISTRY
6. 2. 2 Azo Compounds. A. α, α'-Azobis(isobutyronitrile) (AIBN). a. Decomposition of AIBN. b. Half-life of isobutyronitrile radical : 1. 3 hours at 80℃. POLYMER CHEMISTRY
 6. 2. 2 Azo Compounds. B. Side reaction : Cage effect. a. Tetramethylsuccinonitrile b. Ketenimine POLYMER CHEMISTRY
6. 2. 2 Azo Compounds. B. Side reaction : Cage effect. a. Tetramethylsuccinonitrile b. Ketenimine POLYMER CHEMISTRY
 6. 2. 3 Redox Initiators. A. One electron transfer reaction. a. Making free radical by one electron transfer by redox reaction. b. Low-temperature reaction. c. Emulsion polymerization. B. Example of redox system. POLYMER CHEMISTRY
6. 2. 3 Redox Initiators. A. One electron transfer reaction. a. Making free radical by one electron transfer by redox reaction. b. Low-temperature reaction. c. Emulsion polymerization. B. Example of redox system. POLYMER CHEMISTRY
 6. 2. 4 Photoinitiator A. Peroxide and Azo compound. Photolysis and thermalysis. B. Photolabile initiator. POLYMER CHEMISTRY
6. 2. 4 Photoinitiator A. Peroxide and Azo compound. Photolysis and thermalysis. B. Photolabile initiator. POLYMER CHEMISTRY
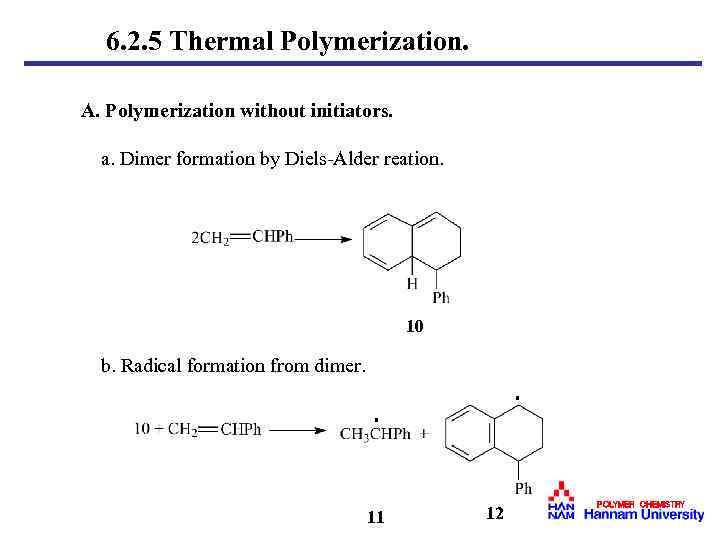 6. 2. 5 Thermal Polymerization. A. Polymerization without initiators. a. Dimer formation by Diels-Alder reation. 10 b. Radical formation from dimer. · · 11 12 POLYMER CHEMISTRY
6. 2. 5 Thermal Polymerization. A. Polymerization without initiators. a. Dimer formation by Diels-Alder reation. 10 b. Radical formation from dimer. · · 11 12 POLYMER CHEMISTRY
 6. 2. 6 Electrochemical Polymerization. A. Polymerization of electrolysis. a. Cathode reaction : electron transfer to monomer ion forming radical anion (6. 22) b. Anode reaction : electron transfer to anode forming radical cation (6. 23) B. Coating metal surfaces with polymers. POLYMER CHEMISTRY
6. 2. 6 Electrochemical Polymerization. A. Polymerization of electrolysis. a. Cathode reaction : electron transfer to monomer ion forming radical anion (6. 22) b. Anode reaction : electron transfer to anode forming radical cation (6. 23) B. Coating metal surfaces with polymers. POLYMER CHEMISTRY
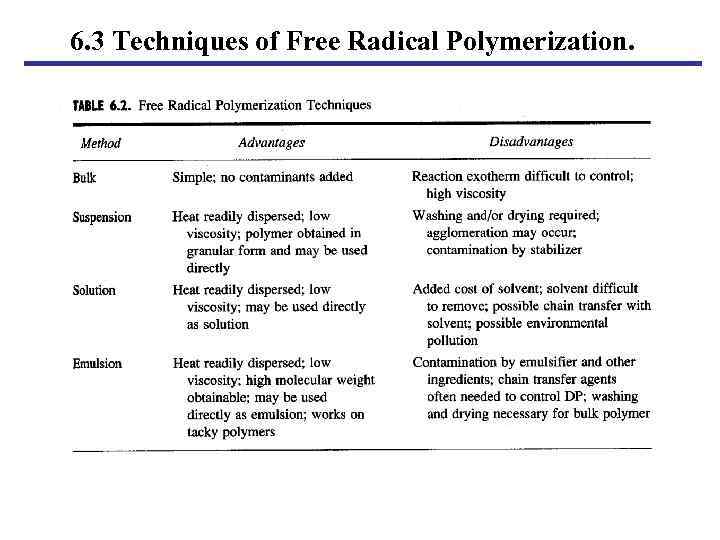 6. 3 Techniques of Free Radical Polymerization.
6. 3 Techniques of Free Radical Polymerization.
 6. 3 Techniques of Free Radical Polymerization. 6. 3. 1 Bulk A. Reactor charges. a. Monomer. b. Initiator (soluble in monomer). B. Problems. a. Heat transfer. b. Viscosity. c. Auto-acceleration. POLYMER CHEMISTRY
6. 3 Techniques of Free Radical Polymerization. 6. 3. 1 Bulk A. Reactor charges. a. Monomer. b. Initiator (soluble in monomer). B. Problems. a. Heat transfer. b. Viscosity. c. Auto-acceleration. POLYMER CHEMISTRY
 6. 3. 2 Suspension. A. Reactor charges. a. Monomer. b. Initiator (soluble in monomer). c. Water or other liquid. d. Stabilizer: Poly(vinyl alcohol), CMC B. Vigorously stirring to keep suspension. POLYMER CHEMISTRY
6. 3. 2 Suspension. A. Reactor charges. a. Monomer. b. Initiator (soluble in monomer). c. Water or other liquid. d. Stabilizer: Poly(vinyl alcohol), CMC B. Vigorously stirring to keep suspension. POLYMER CHEMISTRY
 6. 3. 3 Solution. A. Reactor charges. a. Monomer (soluble in solvent). b. Initiator (soluble in solvent). c. Solvent. B. Refluxing solution. POLYMER CHEMISTRY
6. 3. 3 Solution. A. Reactor charges. a. Monomer (soluble in solvent). b. Initiator (soluble in solvent). c. Solvent. B. Refluxing solution. POLYMER CHEMISTRY
 6. 3. 4 Emulsion. A. Reactor charges. a. Monomer. b. Redox initiator c. Soap or emulsifier. d. Water. e. Others (cf. Table 6. 3). B. Polymerization in swollen micelle. Latex products. POLYMER CHEMISTRY
6. 3. 4 Emulsion. A. Reactor charges. a. Monomer. b. Redox initiator c. Soap or emulsifier. d. Water. e. Others (cf. Table 6. 3). B. Polymerization in swollen micelle. Latex products. POLYMER CHEMISTRY
 6. 3. 4 Emulsion. TABLE 6. 3. Typical Emulsion Polymerization Recipesa Styrene-Buradiene Ingredients, Conditions Copolymer Polyacrylate Latex Ingredients (parts by weight) Water Butadiene Styrene Ethyl acrylate 2 -Chloroethyl vinyl ether p-Divinylbenzene Soap Potassium persulfate 1 -Dodecanethiol Sodium pyrophosphate 190 70 30 5 0. 3 0. 5 - 133 93 5 2 3 b 1 0. 7 Conditions Time Temperature Yield 12 hr 50 o. C 65% 8 hr 60 o. C 100% a. Recipes from Cooper. 23 b. Sodium lauryl sulfate.
6. 3. 4 Emulsion. TABLE 6. 3. Typical Emulsion Polymerization Recipesa Styrene-Buradiene Ingredients, Conditions Copolymer Polyacrylate Latex Ingredients (parts by weight) Water Butadiene Styrene Ethyl acrylate 2 -Chloroethyl vinyl ether p-Divinylbenzene Soap Potassium persulfate 1 -Dodecanethiol Sodium pyrophosphate 190 70 30 5 0. 3 0. 5 - 133 93 5 2 3 b 1 0. 7 Conditions Time Temperature Yield 12 hr 50 o. C 65% 8 hr 60 o. C 100% a. Recipes from Cooper. 23 b. Sodium lauryl sulfate.
 6. 4 Kinetic and Mechanism of polymerization. A. Mechanism of free-radical polymerization. a. Initiation. 1) Decomposition. Initiator → 2 R․ 2) Addition. (6. 25) POLYMER CHEMISTRY
6. 4 Kinetic and Mechanism of polymerization. A. Mechanism of free-radical polymerization. a. Initiation. 1) Decomposition. Initiator → 2 R․ 2) Addition. (6. 25) POLYMER CHEMISTRY
 6. 4 Kinetic and Mechanism of polymerization. b. Propagation. (6. 26) 1) Head-to-tail orientation : predominant reaction. Steric and electronic effects. 2) Examples of not exclusively head-to-tail orientation. (13 -17% of head to head) (5 -6% of head to head) (19% of head to head) POLYMER CHEMISTRY
6. 4 Kinetic and Mechanism of polymerization. b. Propagation. (6. 26) 1) Head-to-tail orientation : predominant reaction. Steric and electronic effects. 2) Examples of not exclusively head-to-tail orientation. (13 -17% of head to head) (5 -6% of head to head) (19% of head to head) POLYMER CHEMISTRY
 6. 4 Kinetic and Mechanism of polymerization. c. Termination. 1) Combination. (6. 27) Polystyrene radical. (6. 29) POLYMER CHEMISTRY
6. 4 Kinetic and Mechanism of polymerization. c. Termination. 1) Combination. (6. 27) Polystyrene radical. (6. 29) POLYMER CHEMISTRY
 6. 4 Kinetic and Mechanism of polymerization. 2) Disproportionation. (6. 28) Poly(methyl methacrylate) radical. (6. 30) ① Repulsion of ester group. ② Easy alpha hydrogen abstraction. 3) Acrylonitrile : Combination virtually exclusively at 60℃. 4) Poly(vinyl acetate) : Disproportionation. POLYMER CHEMISTRY
6. 4 Kinetic and Mechanism of polymerization. 2) Disproportionation. (6. 28) Poly(methyl methacrylate) radical. (6. 30) ① Repulsion of ester group. ② Easy alpha hydrogen abstraction. 3) Acrylonitrile : Combination virtually exclusively at 60℃. 4) Poly(vinyl acetate) : Disproportionation. POLYMER CHEMISTRY
 B. Kinetic of free radical polymerization. a. Assumption. 1) The rates of initiation, propagation, and termination are all different. 2) Independent of chain length. 3) Negligible end group. 4) At steady state, constant radical concentration. (steady state assumption) b. Initiation (Ri) Ri = -d[M ·] dt = 2 fkd[I] f : Initiator efficiency. radicals that initiate a polymer chain f = radicals formed from initiator kd : Decomposition rate constant. [I] : molar concentration of initiator. [M ·] : molar concentration of radical. POLYMER CHEMISTRY
B. Kinetic of free radical polymerization. a. Assumption. 1) The rates of initiation, propagation, and termination are all different. 2) Independent of chain length. 3) Negligible end group. 4) At steady state, constant radical concentration. (steady state assumption) b. Initiation (Ri) Ri = -d[M ·] dt = 2 fkd[I] f : Initiator efficiency. radicals that initiate a polymer chain f = radicals formed from initiator kd : Decomposition rate constant. [I] : molar concentration of initiator. [M ·] : molar concentration of radical. POLYMER CHEMISTRY
![B. Kinetic of free radical polymerization. c. Termination rate ( Rt ) -d[M·] Ri= B. Kinetic of free radical polymerization. c. Termination rate ( Rt ) -d[M·] Ri=](https://present5.com/presentation/3/233991529_423267403.pdf-img/233991529_423267403.pdf-25.jpg) B. Kinetic of free radical polymerization. c. Termination rate ( Rt ) -d[M·] Ri= = 2 kt[M·]2 dt kt = ktc+ ktd d. Propagation rate ( Rp ) Steady state assumption. Ri=Rt 2 [M·]= -d[M] = kp[M][M·] Rp = dt -d[M] = kp[M] Rp = dt POLYMER CHEMISTRY
B. Kinetic of free radical polymerization. c. Termination rate ( Rt ) -d[M·] Ri= = 2 kt[M·]2 dt kt = ktc+ ktd d. Propagation rate ( Rp ) Steady state assumption. Ri=Rt 2 [M·]= -d[M] = kp[M][M·] Rp = dt -d[M] = kp[M] Rp = dt POLYMER CHEMISTRY
 B. Kinetic of free radical polymerization. e. Average kinetic chain length ( ) = = kp[M][M·] = 2 kt[M·]2 Rp = Rp Rt kp[M] 2 kt[M·] = Ri kp[M] Disproportionation : DP = Combination : DP = 2 POLYMER CHEMISTRY
B. Kinetic of free radical polymerization. e. Average kinetic chain length ( ) = = kp[M][M·] = 2 kt[M·]2 Rp = Rp Rt kp[M] 2 kt[M·] = Ri kp[M] Disproportionation : DP = Combination : DP = 2 POLYMER CHEMISTRY
 B. Kinetic of free radical polymerization. f. Gel effect : Trommsdorff effect, Norris-smith effect. 1) Difficult termination reaction because of viscosity. 2) Ease propagation reaction because monomer size is small, even though high viscosity. 3) Autoacceleration by exotherm of propagation reaction. 4) To obtain extraordinary high molecular weight polymer like gel. POLYMER CHEMISTRY
B. Kinetic of free radical polymerization. f. Gel effect : Trommsdorff effect, Norris-smith effect. 1) Difficult termination reaction because of viscosity. 2) Ease propagation reaction because monomer size is small, even though high viscosity. 3) Autoacceleration by exotherm of propagation reaction. 4) To obtain extraordinary high molecular weight polymer like gel. POLYMER CHEMISTRY
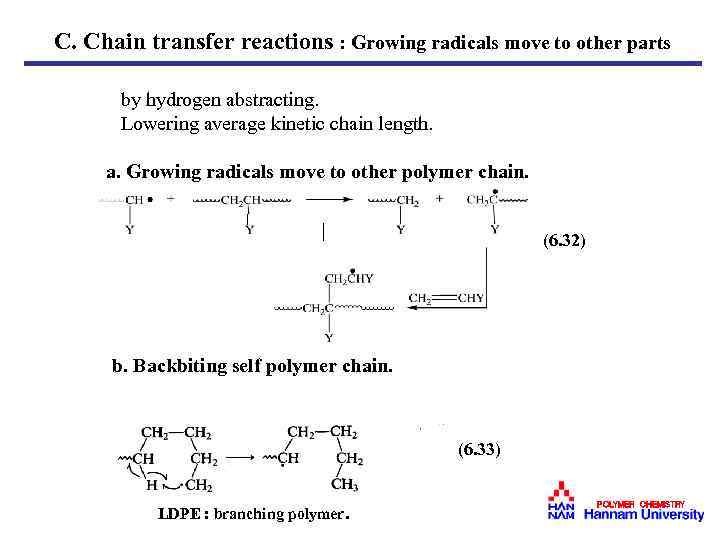 C. Chain transfer reactions : Growing radicals move to other parts by hydrogen abstracting. Lowering average kinetic chain length. a. Growing radicals move to other polymer chain. (6. 32) b. Backbiting self polymer chain. (6. 33) LDPE : branching polymer. POLYMER CHEMISTRY
C. Chain transfer reactions : Growing radicals move to other parts by hydrogen abstracting. Lowering average kinetic chain length. a. Growing radicals move to other polymer chain. (6. 32) b. Backbiting self polymer chain. (6. 33) LDPE : branching polymer. POLYMER CHEMISTRY
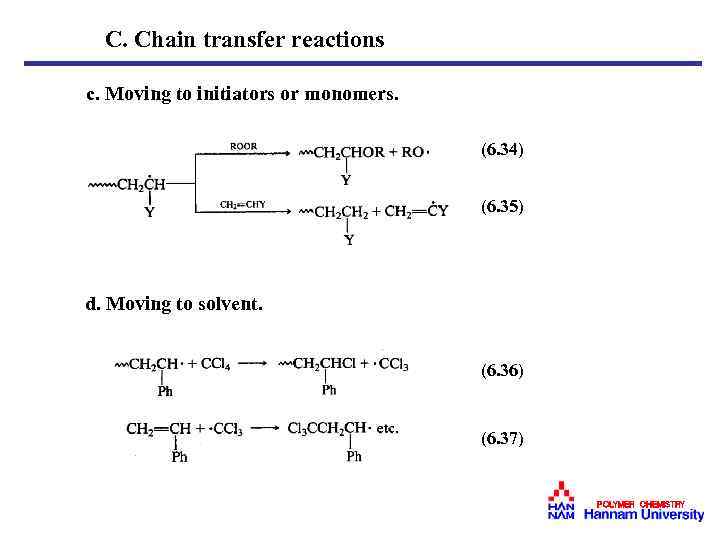 C. Chain transfer reactions c. Moving to initiators or monomers. (6. 34) (6. 35) d. Moving to solvent. (6. 36) (6. 37) POLYMER CHEMISTRY
C. Chain transfer reactions c. Moving to initiators or monomers. (6. 34) (6. 35) d. Moving to solvent. (6. 36) (6. 37) POLYMER CHEMISTRY
 C. Chain transfer reactions e. Moving to chain transfer agent. (6. 39) 1 tr = 1 + [M} Ct : Chain transfer constant. [T] : Concentration of chain transfer agent. f. Telomerization : At high concentration of transfer agent, ktr>kp. Low-molecular-weight polymers are obtained. (Telomer) POLYMER CHEMISTRY
C. Chain transfer reactions e. Moving to chain transfer agent. (6. 39) 1 tr = 1 + [M} Ct : Chain transfer constant. [T] : Concentration of chain transfer agent. f. Telomerization : At high concentration of transfer agent, ktr>kp. Low-molecular-weight polymers are obtained. (Telomer) POLYMER CHEMISTRY
 D. Leaving free radical polymerization : Atom transfer polymerization. a. Copper(I) bypyridyl(bpy) complex: (6. 42) (6. 43) (6. 44) b. TEMPO (18) : 2, 2, 6, 6 -tetramethylpiperidinyl-1 -oxy. (6. 45) POLYMER CHEMISTRY
D. Leaving free radical polymerization : Atom transfer polymerization. a. Copper(I) bypyridyl(bpy) complex: (6. 42) (6. 43) (6. 44) b. TEMPO (18) : 2, 2, 6, 6 -tetramethylpiperidinyl-1 -oxy. (6. 45) POLYMER CHEMISTRY
 6. 4 Kinetic and Mechanism of polymerization. c. Synthesis of block copolymers like anionic polymerization. d. Monodisperse polymerization (PI=1. 05). E. Kinetics of Emulsion polymerization. a. N : the number of particles. b. POLYMER CHEMISTRY
6. 4 Kinetic and Mechanism of polymerization. c. Synthesis of block copolymers like anionic polymerization. d. Monodisperse polymerization (PI=1. 05). E. Kinetics of Emulsion polymerization. a. N : the number of particles. b. POLYMER CHEMISTRY
 6. 5 Stereochemistry of polymerization. A. General consideration. a. Stereoregular polymerization : Ionic and complex coordination polymerization. 1) Terminal ion pair : counter ion. 2) Terminal complex active site. 3) Low temperature. b. Stereo-irregular polymerization : Free-radical polymerization. 1) No stereoregulating radical terminal group. 2) Somewhat higher temperature. POLYMER CHEMISTRY
6. 5 Stereochemistry of polymerization. A. General consideration. a. Stereoregular polymerization : Ionic and complex coordination polymerization. 1) Terminal ion pair : counter ion. 2) Terminal complex active site. 3) Low temperature. b. Stereo-irregular polymerization : Free-radical polymerization. 1) No stereoregulating radical terminal group. 2) Somewhat higher temperature. POLYMER CHEMISTRY
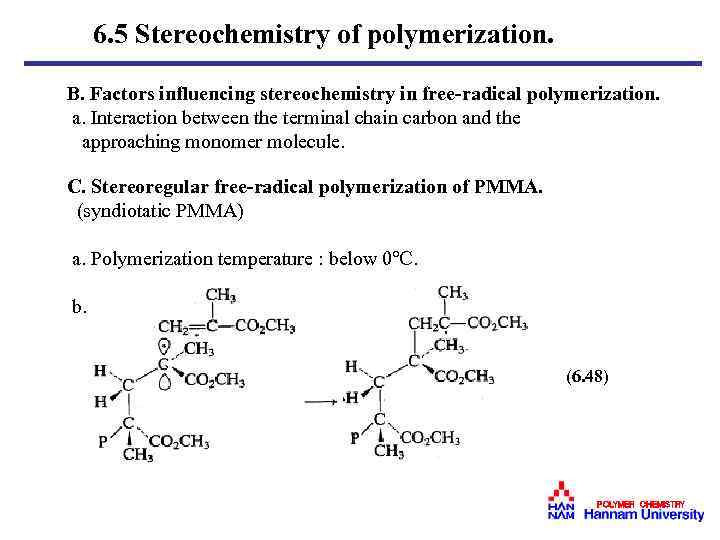 6. 5 Stereochemistry of polymerization. B. Factors influencing stereochemistry in free-radical polymerization. a. Interaction between the terminal chain carbon and the approaching monomer molecule. C. Stereoregular free-radical polymerization of PMMA. (syndiotatic PMMA) a. Polymerization temperature : below 0℃. b. (6. 48) POLYMER CHEMISTRY
6. 5 Stereochemistry of polymerization. B. Factors influencing stereochemistry in free-radical polymerization. a. Interaction between the terminal chain carbon and the approaching monomer molecule. C. Stereoregular free-radical polymerization of PMMA. (syndiotatic PMMA) a. Polymerization temperature : below 0℃. b. (6. 48) POLYMER CHEMISTRY
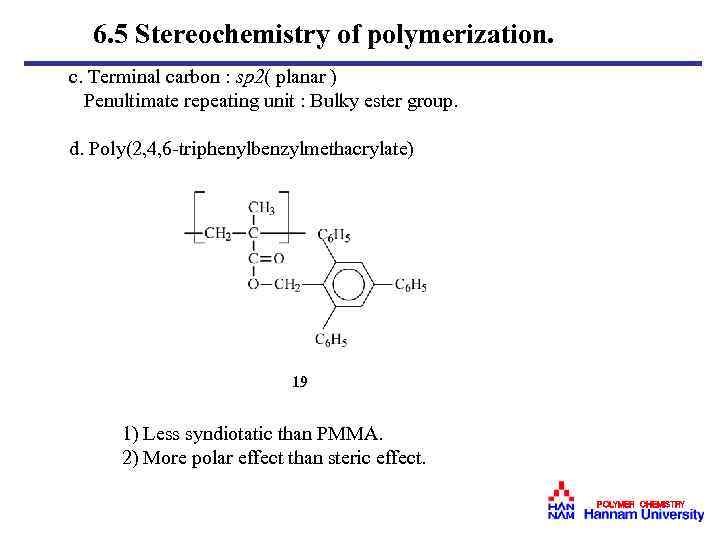 6. 5 Stereochemistry of polymerization. c. Terminal carbon : sp 2( planar ) Penultimate repeating unit : Bulky ester group. d. Poly(2, 4, 6 -triphenylbenzylmethacrylate) 19 1) Less syndiotatic than PMMA. 2) More polar effect than steric effect. POLYMER CHEMISTRY
6. 5 Stereochemistry of polymerization. c. Terminal carbon : sp 2( planar ) Penultimate repeating unit : Bulky ester group. d. Poly(2, 4, 6 -triphenylbenzylmethacrylate) 19 1) Less syndiotatic than PMMA. 2) More polar effect than steric effect. POLYMER CHEMISTRY
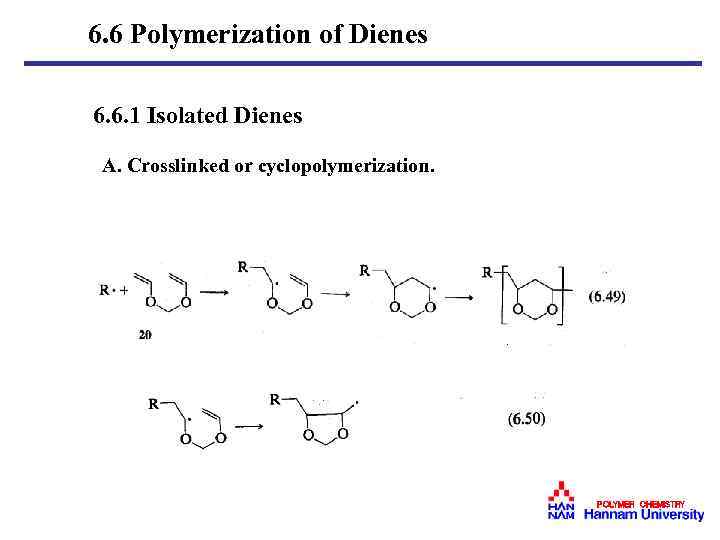 6. 6 Polymerization of Dienes 6. 6. 1 Isolated Dienes A. Crosslinked or cyclopolymerization. POLYMER CHEMISTRY
6. 6 Polymerization of Dienes 6. 6. 1 Isolated Dienes A. Crosslinked or cyclopolymerization. POLYMER CHEMISTRY
 6. 6. 2 Conjugated Dienes. A. Structure of conjugated Diene monoer. Isoprene 23 B. a. 1, 2 -Addition : Pendent vinyl group. 25 b. Stereochemistry : isotactic, syndiotactic, atactic. POLYMER CHEMISTRY
6. 6. 2 Conjugated Dienes. A. Structure of conjugated Diene monoer. Isoprene 23 B. a. 1, 2 -Addition : Pendent vinyl group. 25 b. Stereochemistry : isotactic, syndiotactic, atactic. POLYMER CHEMISTRY
 6. 6. 2 Conjugated Dienes. C. 1, 4 -Addition : Delocalized double bond a. 24 26 27 D. 3, 4 -Addition 29 E. Polymerization reaction and temperature. POLYMER CHEMISTRY
6. 6. 2 Conjugated Dienes. C. 1, 4 -Addition : Delocalized double bond a. 24 26 27 D. 3, 4 -Addition 29 E. Polymerization reaction and temperature. POLYMER CHEMISTRY
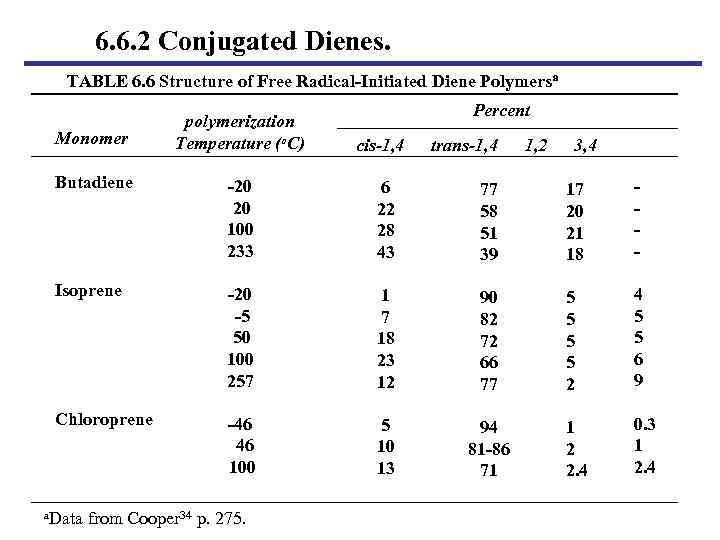 6. 6. 2 Conjugated Dienes. TABLE 6. 6 Structure of Free Radical-Initiated Diene Polymersa Monomer polymerization Temperature (o. C) Percent cis-1, 4 trans-1, 4 1, 2 3, 4 Butadiene -20 20 100 233 6 22 28 43 77 58 51 39 17 20 21 18 - Isoprene -20 -5 50 100 257 1 7 18 23 12 90 82 72 66 77 5 5 2 4 5 5 6 9 Chloroprene -46 46 100 5 10 13 94 81 -86 71 1 2 2. 4 0. 3 1 2. 4 a. Data from Cooper 34 p. 275.
6. 6. 2 Conjugated Dienes. TABLE 6. 6 Structure of Free Radical-Initiated Diene Polymersa Monomer polymerization Temperature (o. C) Percent cis-1, 4 trans-1, 4 1, 2 3, 4 Butadiene -20 20 100 233 6 22 28 43 77 58 51 39 17 20 21 18 - Isoprene -20 -5 50 100 257 1 7 18 23 12 90 82 72 66 77 5 5 2 4 5 5 6 9 Chloroprene -46 46 100 5 10 13 94 81 -86 71 1 2 2. 4 0. 3 1 2. 4 a. Data from Cooper 34 p. 275.
 6. 6. 2 Conjugated Dienes. F. s-cis and s-trans POLYMER CHEMISTRY
6. 6. 2 Conjugated Dienes. F. s-cis and s-trans POLYMER CHEMISTRY
 6. 7 Monomer Reactivity A. Thermodynamic feasibility. a. ΔGp = ΔHp - TΔSp ΔGp : Gibbs free energy change of polymerization. ΔHp : Enthalpy change of polymerization. ΔSp : Entropy change of polymerization. ΔGp < 0 : favorable free energy of polymerization. b. Values of ΔH and ΔS for several monomers. c. Polypropylene and isobutylene : ΔG < 0 → unfavorable polymerization. because of kinetic feasibility POLYMER CHEMISTRY
6. 7 Monomer Reactivity A. Thermodynamic feasibility. a. ΔGp = ΔHp - TΔSp ΔGp : Gibbs free energy change of polymerization. ΔHp : Enthalpy change of polymerization. ΔSp : Entropy change of polymerization. ΔGp < 0 : favorable free energy of polymerization. b. Values of ΔH and ΔS for several monomers. c. Polypropylene and isobutylene : ΔG < 0 → unfavorable polymerization. because of kinetic feasibility POLYMER CHEMISTRY
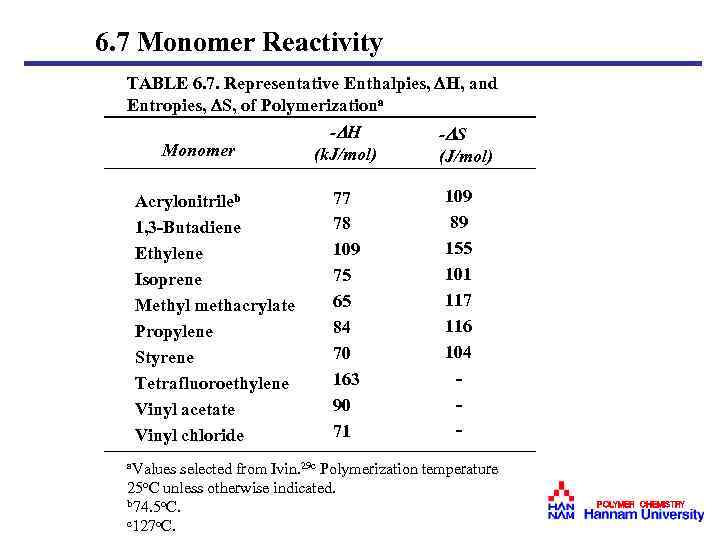 6. 7 Monomer Reactivity TABLE 6. 7. Representative Enthalpies, H, and Entropies, S, of Polymerizationa Monomer Acrylonitrileb 1, 3 -Butadiene Ethylene Isoprene Methyl methacrylate Propylene Styrene Tetrafluoroethylene Vinyl acetate Vinyl chloride - H (k. J/mol) 77 78 109 75 65 84 70 163 90 71 - S (J/mol) 109 89 155 101 117 116 104 - a. Values selected from Ivin. 29 c Polymerization temperature 25 o. C unless otherwise indicated. b 74. 5 o. C. c 127 o. C. POLYMER CHEMISTRY
6. 7 Monomer Reactivity TABLE 6. 7. Representative Enthalpies, H, and Entropies, S, of Polymerizationa Monomer Acrylonitrileb 1, 3 -Butadiene Ethylene Isoprene Methyl methacrylate Propylene Styrene Tetrafluoroethylene Vinyl acetate Vinyl chloride - H (k. J/mol) 77 78 109 75 65 84 70 163 90 71 - S (J/mol) 109 89 155 101 117 116 104 - a. Values selected from Ivin. 29 c Polymerization temperature 25 o. C unless otherwise indicated. b 74. 5 o. C. c 127 o. C. POLYMER CHEMISTRY
 6. 7 Monomer Reactivity B. Factors of monomer reactivity in free radical polymerization. a. The stability of the monomer toward addition of a free radical. b. The stability of the monomer radicals. c. Order of monomer reactivity. Acrylonitrile > Styrene > Vinyl acetate. d. Order of benzoyloxy radical initiation. Syrene > Vinyl acetate > Acrylonitrile Benzoyloxy radical : Ph 14 CO 2․ POLYMER CHEMISTRY
6. 7 Monomer Reactivity B. Factors of monomer reactivity in free radical polymerization. a. The stability of the monomer toward addition of a free radical. b. The stability of the monomer radicals. c. Order of monomer reactivity. Acrylonitrile > Styrene > Vinyl acetate. d. Order of benzoyloxy radical initiation. Syrene > Vinyl acetate > Acrylonitrile Benzoyloxy radical : Ph 14 CO 2․ POLYMER CHEMISTRY
 6. 7 Monomer Reactivity C. The inverse relationship between monomer stability and polymerization rate. a. Vinyl acetate: not Stable monomer but high rate constant. b. Steric and polar effects: Not clear-cut generalization. Lower rate constant of MMA than MA. c. 1, 2 disubstituted monomer difficult to polymerize in free radical. Exception: Tetrafluoroethylene. POLYMER CHEMISTRY
6. 7 Monomer Reactivity C. The inverse relationship between monomer stability and polymerization rate. a. Vinyl acetate: not Stable monomer but high rate constant. b. Steric and polar effects: Not clear-cut generalization. Lower rate constant of MMA than MA. c. 1, 2 disubstituted monomer difficult to polymerize in free radical. Exception: Tetrafluoroethylene. POLYMER CHEMISTRY
 6. 7 Monomer Reactivity D. Ceiling temperature (Tc) a. b. Definition of ceiling temperature. ΔGp = 0 : equal forward and backword reactions. c. High Tc : favorable polymerization. Low Tc : unfavorable polymerization. Exception : α-methylstyrene (Tc=66℃). POLYMER CHEMISTRY
6. 7 Monomer Reactivity D. Ceiling temperature (Tc) a. b. Definition of ceiling temperature. ΔGp = 0 : equal forward and backword reactions. c. High Tc : favorable polymerization. Low Tc : unfavorable polymerization. Exception : α-methylstyrene (Tc=66℃). POLYMER CHEMISTRY
 6. 8 Copolymerization. A. Mechanism of copolymerization. POLYMER CHEMISTRY
6. 8 Copolymerization. A. Mechanism of copolymerization. POLYMER CHEMISTRY
 B. Kinetics of copolymerization. a. b. c. let, (reactivity ratio) and steady state assumption. d. solving : Copolymer equation or copolymer composition equation. d[M 1]/d[M 2] : the molar ratio of the two monomers in the copolymer [M 1], [M 2] : the initial molar concentration of monomers in the reaction mixture
B. Kinetics of copolymerization. a. b. c. let, (reactivity ratio) and steady state assumption. d. solving : Copolymer equation or copolymer composition equation. d[M 1]/d[M 2] : the molar ratio of the two monomers in the copolymer [M 1], [M 2] : the initial molar concentration of monomers in the reaction mixture
 C. Significance of reactivity ratio (r 1, r 2). a. r 1 = r 2 = ∞ : Homopolymer. b. r 1 = r 2 = 0 : Alternating polymer. c. r 1 = r 2 = 1 : Copolymer composition depending on feeding monomers in the reaction temperature. d. r 1 × r 2 = 1 : Ideal copolymerization like ideal liquid vaporization. e. r 1 × r 2 > 1 : Azotropic copolymerization (polymer composition not depending on feeding). f. Determination of r 1, r 2 : Measure copolymer composition by NMR or other method at low conversion ( <10% ) POLYMER CHEMISTRY
C. Significance of reactivity ratio (r 1, r 2). a. r 1 = r 2 = ∞ : Homopolymer. b. r 1 = r 2 = 0 : Alternating polymer. c. r 1 = r 2 = 1 : Copolymer composition depending on feeding monomers in the reaction temperature. d. r 1 × r 2 = 1 : Ideal copolymerization like ideal liquid vaporization. e. r 1 × r 2 > 1 : Azotropic copolymerization (polymer composition not depending on feeding). f. Determination of r 1, r 2 : Measure copolymer composition by NMR or other method at low conversion ( <10% ) POLYMER CHEMISTRY
 D. Alfrey-price Q-e scheme. a. b. c. For styrene Q=1. 0 , e=-0. 8 d. Q : resonance stabilization. e : less negative values equal more electron attracting. POLYMER CHEMISTRY
D. Alfrey-price Q-e scheme. a. b. c. For styrene Q=1. 0 , e=-0. 8 d. Q : resonance stabilization. e : less negative values equal more electron attracting. POLYMER CHEMISTRY
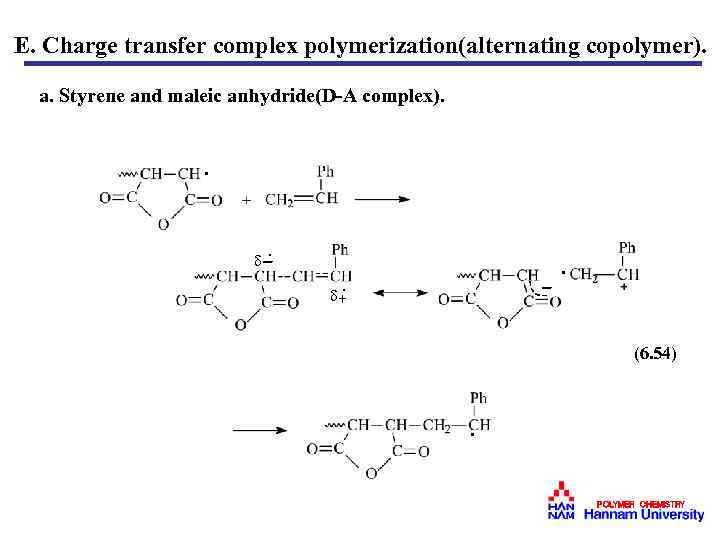 E. Charge transfer complex polymerization(alternating copolymer). a. Styrene and maleic anhydride(D-A complex). · · - · + · (6. 54) · POLYMER CHEMISTRY
E. Charge transfer complex polymerization(alternating copolymer). a. Styrene and maleic anhydride(D-A complex). · · - · + · (6. 54) · POLYMER CHEMISTRY
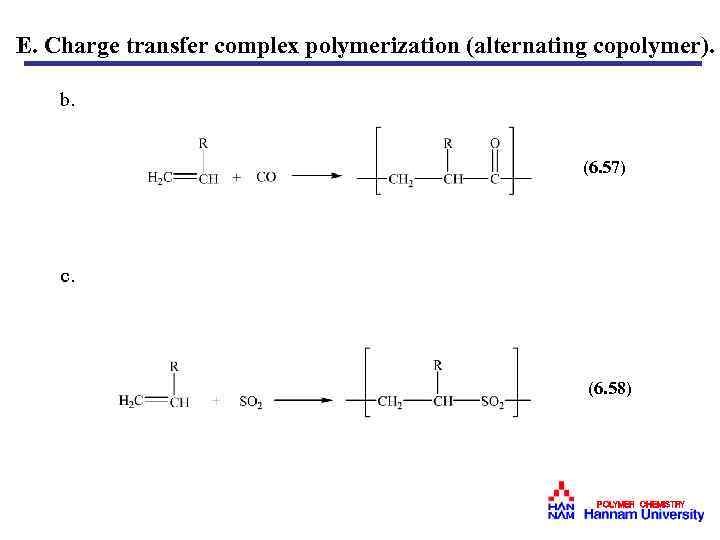 E. Charge transfer complex polymerization (alternating copolymer). b. (6. 57) c. (6. 58) POLYMER CHEMISTRY
E. Charge transfer complex polymerization (alternating copolymer). b. (6. 57) c. (6. 58) POLYMER CHEMISTRY


