896b0fc298fe820ac410d4e83151a8c2.ppt
- Количество слайдов: 30

Advances in Nano Drugs for Cancer Chemotherapy: Biopharmaceutical Trends and Perspectives and Case Studies Panayiotis P. Constantinides, Ph. D Biopharmaceutical & Drug Delivery Consulting, LLC Gurnee, Illinois, USA ppconstantinides@bpddc. com Keynote Forum 3 rd International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems April 8 -10, Northbrook, Illinois

OUTLINE • Biopharmaceutical Aspects of Anticancer Nano Drugs – – Image-guided drug delivery and multifunctional nanoparticles Nanoparticle targeting principles Marketed drug products and in development Formulation development, manufacturing, Toxicity/PK/ADME • Case Studies • A. Parenteral Nano drugs – Nanoemulsions : paclitaxel – Liposomes and polymeric micelles : phospho-Ibuprofen (NME) • B. Oral Nano drugs – SNEDDS : Tamoxifen – Reverse Micelles : Leuprolide • Conclusions and Future Perspectives 4/8/2013 BPDDC LLC www. bpddc. com 2

BIOPHARMACEUTICAL ASPECTS OF ANTICANCER NANODRUGS 4/8/2013 BPDDC LLC www. bpddc. com 3
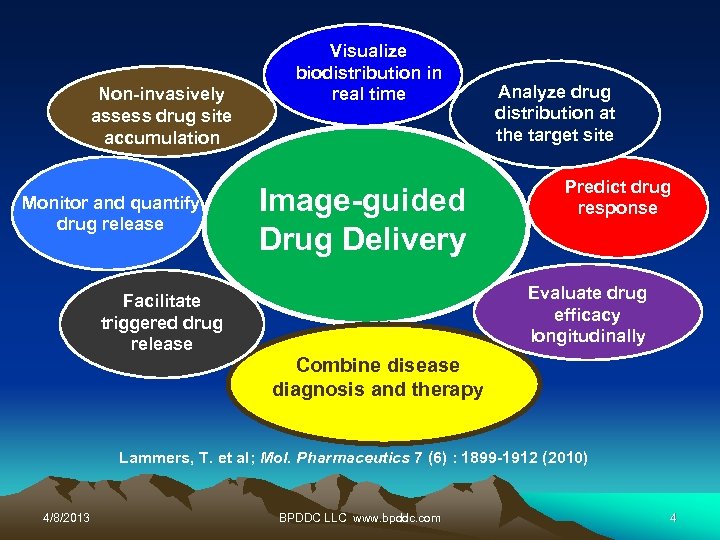
Non-invasively assess drug site accumulation Monitor and quantify drug release Visualize biodistribution in real time Image-guided Drug Delivery Analyze drug distribution at the target site Predict drug response Evaluate drug efficacy longitudinally Facilitate triggered drug release Combine disease diagnosis and therapy Lammers, T. et al; Mol. Pharmaceutics 7 (6) : 1899 -1912 (2010) 4/8/2013 BPDDC LLC www. bpddc. com 4
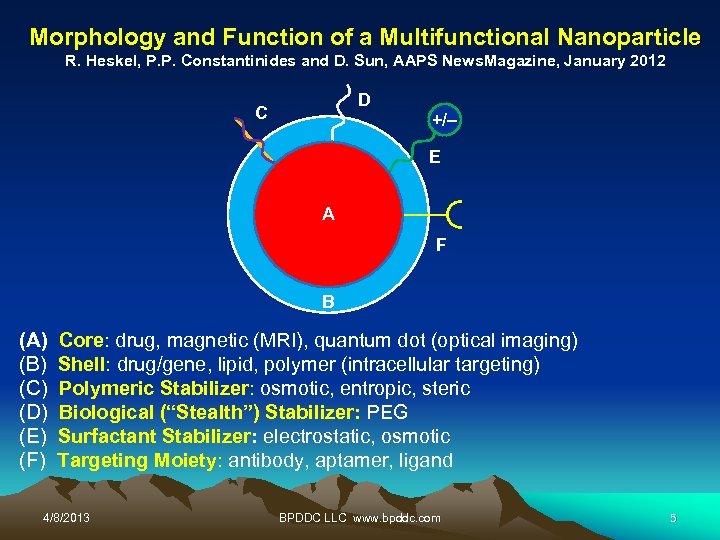
Morphology and Function of a Multifunctional Nanoparticle R. Heskel, P. P. Constantinides and D. Sun, AAPS News. Magazine, January 2012 D C +/– E A F B (A) Core: drug, magnetic (MRI), quantum dot (optical imaging) (B) Shell: drug/gene, lipid, polymer (intracellular targeting) (C) Polymeric Stabilizer: osmotic, entropic, steric (D) Biological (“Stealth”) Stabilizer: PEG (E) Surfactant Stabilizer: electrostatic, osmotic (F) Targeting Moiety: antibody, aptamer, ligand 4/8/2013 BPDDC LLC www. bpddc. com 5

Nanodimensions of Drug Delivery Nanoparticles Mattheolabakis, G. , Rigas B. , and Constantinides, P. P. Nanomedicine (2012) 7: 1577 -1590 The true nanorange is narrowly defined as the 1 -100 nm particles. Marketed injectable liposomal (Dauno. Xome®, Doxil®) and albumin -bound nanoparticles (Abraxane®) anticancer drug products, as well as the oral Nano. Crystal® drug products (Rapamune®, EMEND®, Tri. Cor® 145, Megace® ES and INVEGA® SUSTENNA®) are within the submicron range (100 – 1000 nm). 4/8/2013 BPDDC LLC www. bpddc. com 6

Drug Targeting Active Passive • Liver and spleen • Receptor- mediated – Immunotargeting • Antigenic sites on pathogens • Infected cells expressing antigenic structures • Tumor-associated antigens – Targeting Ligands • EGF, Transferrin • Folate, RGD • VIP 4/8/2013 – Macrophage uptake • Lysosomal enzyme deficiencies • Size-mediated – EPR effect • p. H-mediated – p. H-responsive drug carriers • Temperature-mediated – Thermo-responsive drug carriers BPDDC LLC www. bpddc. com 7

Diverse Targeted Nanoparticles in Cancer in Preclinical and Clinical Development • • Carbon nanotubes Quantum Dots Nanoparticle-aptamer bioconjugates Lipid-based nanocarriers – Liposomes, nanoemulsions, micelles, solid lipid nanoparticles and nanosuspensions • Anti-angiogenic molecules in NPs • Brain-targeted NPs • Polymeric micelles • Polymer-drug conjugates and immunoconjugates • Combination of NPs with other physical and diagnostic methods – radiotherapy, photodynamic therapy and ultrasound – nanoshells and paramagnetic NPs 4/8/2013 BPDDC LLC www. bpddc. com 8
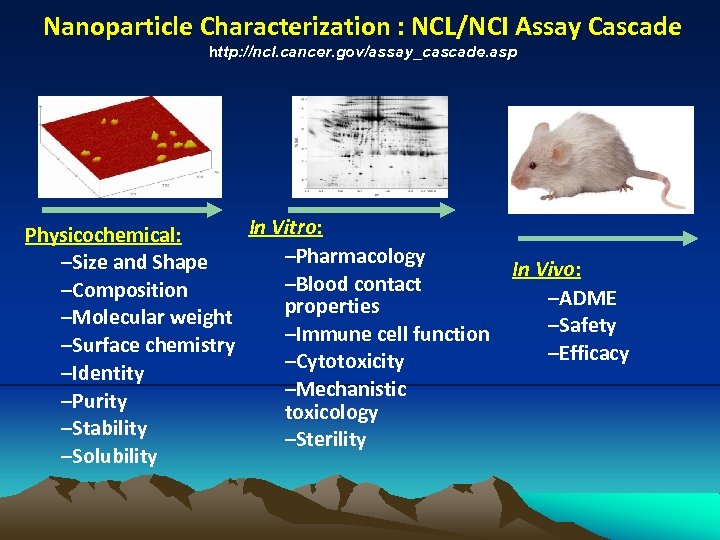
Nanoparticle Characterization : NCL/NCI Assay Cascade http: //ncl. cancer. gov/assay_cascade. asp In Vitro: Physicochemical: –Pharmacology –Size and Shape –Blood contact –Composition properties –Molecular weight –Immune cell function –Surface chemistry –Cytotoxicity –Identity –Mechanistic –Purity toxicology –Stability –Sterility –Solubility In Vivo: –ADME –Safety –Efficacy

Formulation and Process Considerations • Lessons learned from the marketed nanoparticle drug products (100 - 1000 nm) that can be applied to the 1 -100 nm particles - what is truly new knowledge? – New processing equipment and characterization methods – No reference standards and specifications are available • Need to develop and validate suitable methods and set meaningful controls and drug product specifications – establish reference standards for 1 -100 nm nanoparticles • Scale up and manufacturing challenges with acceptable shelf -life of complex multifunctional nanoparticulate systems 4/8/2013 BPDDC LLC www. bpddc. com 10
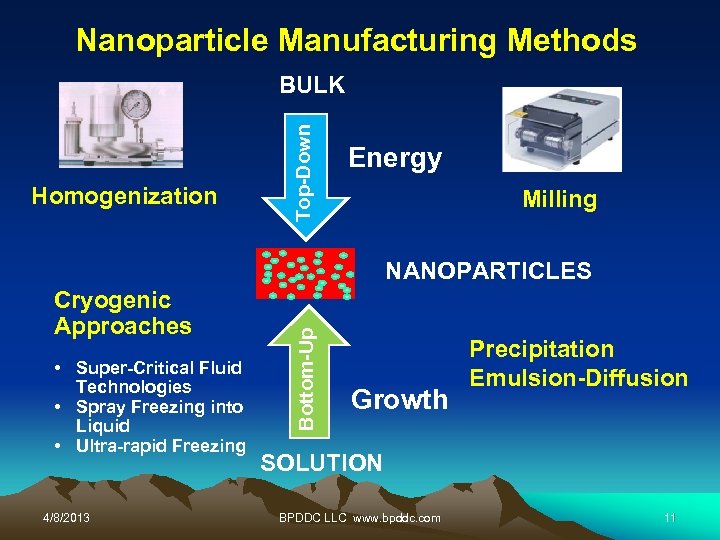
Nanoparticle Manufacturing Methods Homogenization Top-Down BULK Energy Milling Cryogenic Approaches • Super-Critical Fluid Technologies • Spray Freezing into Liquid • Ultra-rapid Freezing 4/8/2013 Bottom-Up NANOPARTICLES Growth Precipitation Emulsion-Diffusion SOLUTION BPDDC LLC www. bpddc. com 11

Nanotoxicity : Why the Concern? • Unusual physicochemical properties attributable to : – Small size (surface area, size distribution) • Particle Toxicity Rank (in general): (+) > (-) > (0) (net charge) – Chemical composition (e. g. , purity, crystallinity, electronic properties) – Surface structure (e. g. , surface reactivity, surface groups, inorganic or organic coatings) – Solubility, shape and aggregation • Opportunities for increased uptake and interaction with biological tissues relative to bulk materials • Need to establish nanotoxicity guidelines • No specific regulations at the present time – June 9, 2011 FDA Draft Guidance on regulated products involve Nanotech applications 4/8/2013 BPDDC LLC www. bpddc. com 12
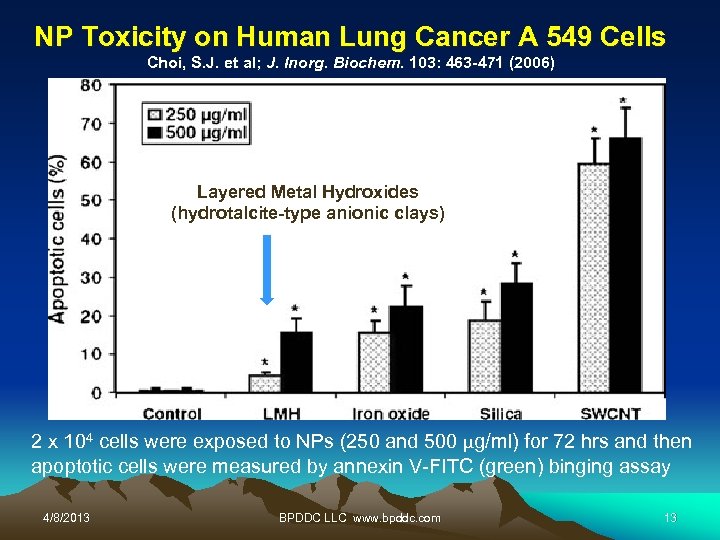
NP Toxicity on Human Lung Cancer A 549 Cells Choi, S. J. et al; J. Inorg. Biochem. 103: 463 -471 (2006) Layered Metal Hydroxides (hydrotalcite-type anionic clays) 2 x 104 cells were exposed to NPs (250 and 500 µg/ml) for 72 hrs and then apoptotic cells were measured by annexin V-FITC (green) binging assay 4/8/2013 BPDDC LLC www. bpddc. com 13

Nanoparticle Pharmacokinetics • Fate of particles upon administration – Are all nanoparticles taken up by cells and cross anatomical barriers ? – What is their fate after cell uptake? • Drug absorption, distribution, metabolism and elimination (ADME): – How is affected by size, surface composition and charge of the particles? • Biopharmaceutical and pharmacokinetic data: – How to use it to optimize quality and performance of nanoparticles? 4/8/2013 BPDDC LLC www. bpddc. com 14

Nanomaterial and Nanoparticle ADME Findings Riviere, J. E. Wiley Wires/Nanomed, Vol 1. 26 -34 (2009) • Most nanomaterials (NMs) accumulate in the liver – Depending on size and charge, can also accumulate in kidney and other tissues • Nanoparticles (NPs) with hydrodynamic radii < 5 - 6 nm may be eliminated from the kidney • All classes of NPs have extensive tissue retention – Carbon based materials and quantum dots - toxicological implications • State of NMs once deposited in tissue largely unknown • Comparisons within and across complex NMs difficult 4/8/2013 BPDDC LLC www. bpddc. com 15

PARENTERAL NANO DRUGS CASE STUDIES 4/8/2013 BPDDC LLC www. bpddc. com 16
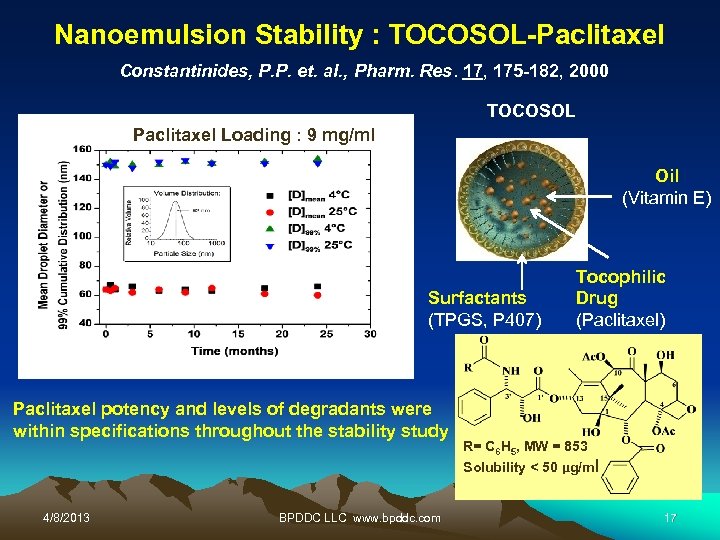
Nanoemulsion Stability : TOCOSOL-Paclitaxel Constantinides, P. P. et. al. , Pharm. Res. 17, 175 -182, 2000 TOCOSOL Paclitaxel Loading : 9 mg/ml Oil (Vitamin E) Surfactants (TPGS, P 407) Paclitaxel potency and levels of degradants were within specifications throughout the stability study 4/8/2013 BPDDC LLC www. bpddc. com Tocophilic Drug (Paclitaxel) R= C 6 H 5, MW = 853 Solubility < 50 µg/ml 17
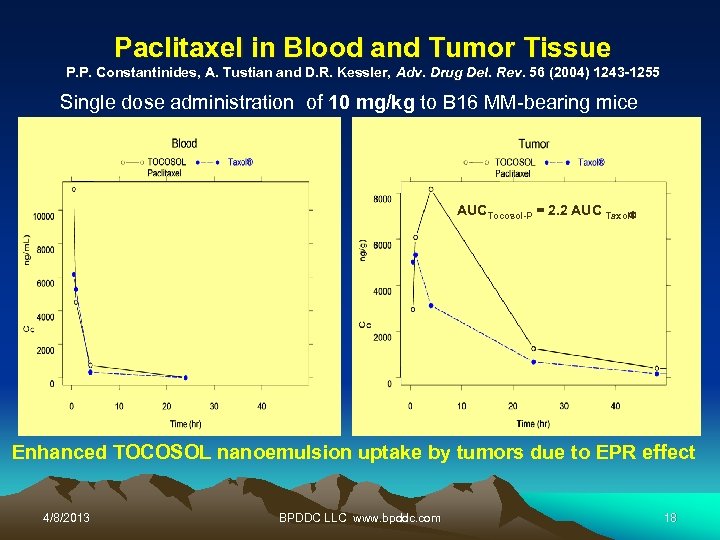
Paclitaxel in Blood and Tumor Tissue P. P. Constantinides, A. Tustian and D. R. Kessler, Adv. Drug Del. Rev. 56 (2004) 1243 -1255 Single dose administration of 10 mg/kg to B 16 MM-bearing mice AUCTocosol-P = 2. 2 AUC Taxol Enhanced TOCOSOL nanoemulsion uptake by tumors due to EPR effect 4/8/2013 BPDDC LLC www. bpddc. com 18

Ibuprofen vs Phospho-Ibuprofen (MDC-917) : Physical Properties and Cytotoxicity (Nie, T. et al; (2011) British J. Pharmacol. ) MDC : Medicon Pharmaceuticals PEO-b-PLA diblock copolymer Soy PC + DSPE-PEG 209 ± 16 nm P-I -15. 5 ± 2. 6 m. V 78 ± 8 nm ; -6. 2 ± 0. 6 m. V Ibuprofen Partition coefficient (log P) IC 50, µM, range (HT 29, HCT 116, SW 480 colon cancer cell lines) Cell uptake, nmol/mg protein 4/8/2013 Phosphoibuprofen 3. 75 28 -104 0. 1 MICELLES 5. 43 748 -1, 554 LIPOSOMES 18 BPDDC LLC www. bpddc. com 200 nm 50 nm 19
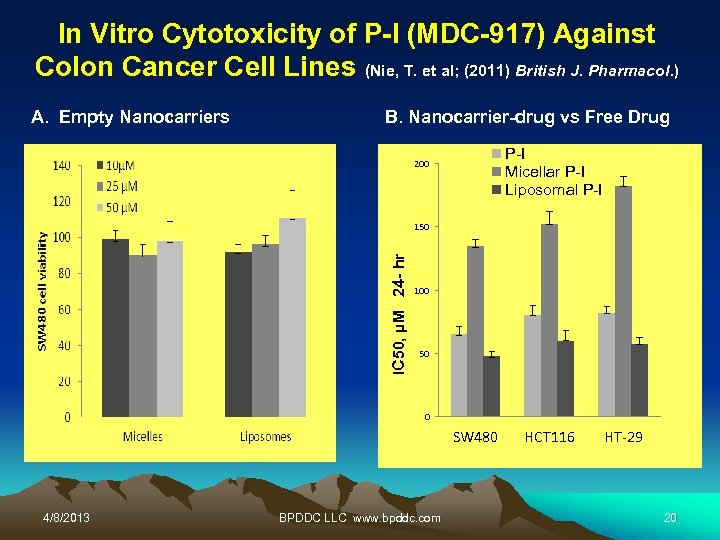
In Vitro Cytotoxicity of P-I (MDC-917) Against Colon Cancer Cell Lines (Nie, T. et al; (2011) British J. Pharmacol. ) A. Empty Nanocarriers B. Nanocarrier-drug vs Free Drug P-I Micellar P-I Liposomal P-I 200 B IC 50, µM 24 - hr 150 100 50 0 SW 480 4/8/2013 BPDDC LLC www. bpddc. com HCT 116 HT-29 20
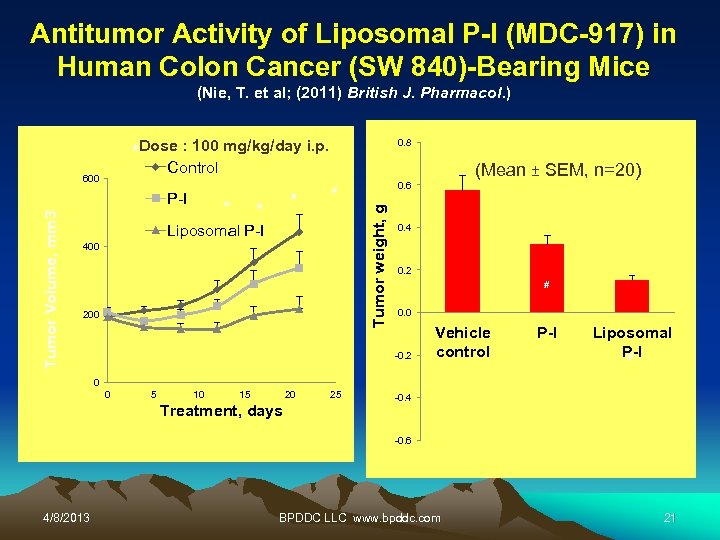
Antitumor Activity of Liposomal P-I (MDC-917) in Human Colon Cancer (SW 840)-Bearing Mice (Nie, T. et al; (2011) British J. Pharmacol. ) Dose : 100 mg/kg/day i. p. Control 0. 8 # Tumor Volume, mm 3 P-I * # # # Liposomal P-I 400 200 (Mean ± SEM, n=20) 0. 6 Tumor weight, g 600 0. 4 0. 2 # 0. 0 -0. 2 Vehicle control P-I Liposomal P-I # 0 0 5 10 15 20 Treatment, days 25 -0. 4 -0. 6 4/8/2013 BPDDC LLC www. bpddc. com 21

ORAL NANO DRUGS CASE STUDIES 4/8/2013 BPDDC LLC www. bpddc. com 22

Tamoxifen Citrate VAN Life Sciences Pvt Ltd, www. van. in Log PO/W = 3. 7, p. H 7. 0 BCS IV Molecule • Oral anti-estrogen for breast cancer treatment • Available as a Tablet and Oral Solution in a daily dose 10 -20 mg; chronic therapy (3 -5 yrs) • Hepatotoxicity is a major toxicity with TMX-Citrate therapy • Poor oral bioavailability (20 -30%); large inter-subject variability • Intestinal P-gp substrate; First-pass metabolism (CYP 34 A) • Use of CYP 34 A inhibitors improves bioavailability • Use of lipid-based systems (SNEDDS, SLN/NLC) to improve the oral BA of TMX 4/8/2013 BPDDC LLC www. bpddc. com 23
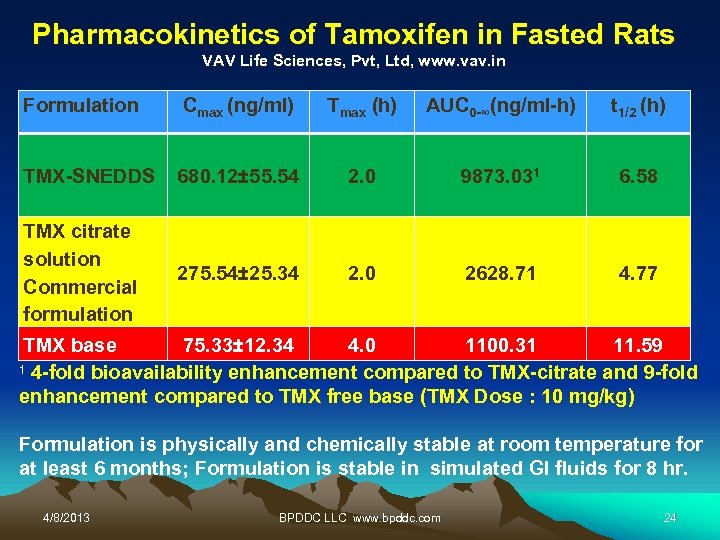
Pharmacokinetics of Tamoxifen in Fasted Rats VAV Life Sciences, Pvt, Ltd, www. vav. in Formulation Cmax (ng/ml) TMX-SNEDDS TMX citrate solution Commercial formulation 680. 12± 55. 54 275. 54± 25. 34 (h) Tmax AUC 0 -∞(ng/ml-h) t 1/2 (h) 2. 0 9873. 031 6. 58 2. 0 2628. 71 4. 77 TMX base 75. 33± 12. 34 4. 0 1100. 31 11. 59 1 4 -fold bioavailability enhancement compared to TMX-citrate and 9 -fold enhancement compared to TMX free base (TMX Dose : 10 mg/kg) Formulation is physically and chemically stable at room temperature for at least 6 months; Formulation is stable in simulated GI fluids for 8 hr. 4/8/2013 BPDDC LLC www. bpddc. com 24
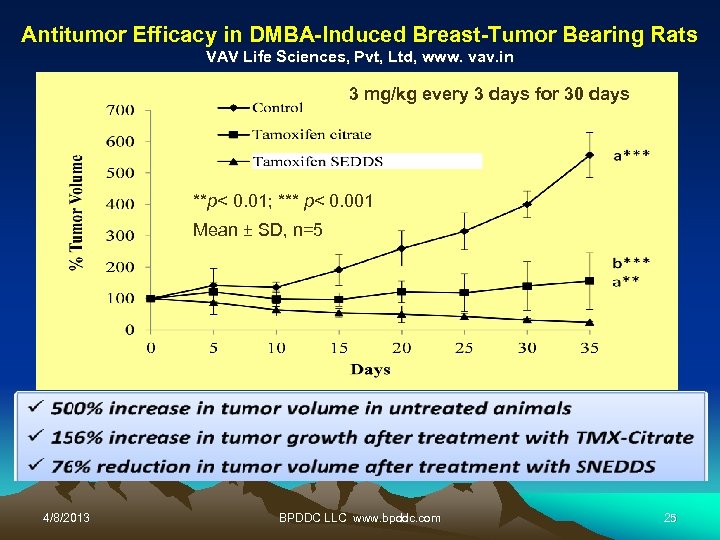
Antitumor Efficacy in DMBA-Induced Breast-Tumor Bearing Rats VAV Life Sciences, Pvt, Ltd, www. vav. in 3 mg/kg every 3 days for 30 days **p< 0. 01; *** p< 0. 001 Mean ± SD, n=5 4/8/2013 BPDDC LLC www. bpddc. com 25
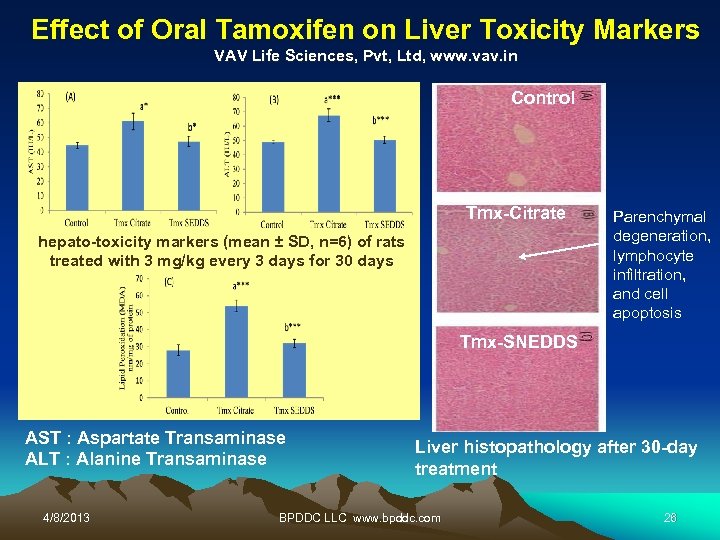
Effect of Oral Tamoxifen on Liver Toxicity Markers VAV Life Sciences, Pvt, Ltd, www. vav. in Control Tmx-Citrate hepato-toxicity markers (mean ± SD, n=6) of rats treated with 3 mg/kg every 3 days for 30 days Parenchymal degeneration, lymphocyte infiltration, and cell apoptosis Tmx-SNEDDS AST : Aspartate Transaminase ALT : Alanine Transaminase 4/8/2013 Liver histopathology after 30 -day treatment BPDDC LLC www. bpddc. com 26

Peroral Absorption of Leuprolide in Dogs in EC HGCs Constantinides P. P. et al. 2002 AAPS Meeting and Exposition, Toronto, Canada Formulation n Dose mg/k g LPM EC (2 X) 5 2. 09 31277. 2 ± 9413 199. 2 ± 78. 5 11. 6 ± 3. 7 LPM EC (4 X) 6 1. 04 16158. 7 ± 3093 117. 9 ± 20. 3 11. 4 ± 2. 2 LPM EC (2 X) 6 0. 35 8563 ± 1535 81. 5 ± 16. 8 16. 6 ± 3. 4 LPM EC (2 X) 3 0. 35 6208 ± 2311 68. 3 ± 24. 4 13. 8 ± 5. 1 LPM EC (4 X) 3 0. 35 7488. 3 ± 2609 39. 2 ± 15. 0 16. 6 ± 5. 8 AUC (min*ng/ml) Cmax (ng/ml) Bioavailability (mean SEM) EC : Enteric Coated; X is the number of layers of coating applied: HGC : Hard Gelatin Capsule (Li. Caps®) Oral (i. d. ) BA of Leurpolide from solution = 2. 2 ± 0. 2 (n=4) Reverse Micelle Solubility in 0. 1 M acetate buffer p. H 5. 1 > 30 mg/ml Intestinal permeability: very low Leuprolide (LHRH analogue, MW = 1209) p-Glu-His-Trp-Ser-Tyr -D-Leu-Arg-Pro. NHC 2 H 5 Prostate Cancer • BPDDC LLC www. bpddc. com Endometriosis • 4/8/2013 • Precocious Puberty 27

Conclusions and Future Perspectives • There have been significant and promising advances in drug delivery NPs in cancer drug delivery and targeting. • The most promising systems/approaches are those that combine disease diagnosis with therapy (Nanotheranostics). – Formulation development and manufacturing challenges • Better understanding of the ADMET and PK of NPs is critical to their progress from bench to clinic and commercialization • Toxicity is dependent on the nature and composition of NPs – In general, inorganic NPs are more toxic than organic ones – Need to establish nanotoxicity guidelines • Expanded use of parenteral and oral lipidic and/or polymeric nanoparticles in cancer drug delivery for both poorly soluble and water-soluble molecules/macromolecules. 4/8/2013 BPDDC LLC www. bpddc. com 28

Acknowledgements • Nanoemulsions : TOCOSOL-Paclitaxel – Karl Lambert, Alex Tustian, Dean Kessler, former R&D associates at SONUS Pharmaceuticals • Liposomes/Polymeric Micelles : Phospho-Ibuprofen – Collaboration with Dr. Basil Rigas and laboratory staff, Stony Brook University, Medical School, Division of Cancer Prevention and Medicon Pharmaceuticals • LPM™ (Reverse Micelles) : Leuprolide (DOR Biopharma) – Andy Jang, Likan Liang, Dave Fast, Liangxiu He, former R&D associates at DOR Biopharma • Tamoxifen-SNEDDS – Arun Kedia, General Manager of VAV Life Sciences Pvt, Ltd, Mumbai, INDIA, a partnering company 4/8/2013 BPDDC LLC www. bpddc. com 29
ENABLED ANTICANCER DRUG PRODUCTS THANK YOU ! LIPIDS/POLYMERS ORGANIC/INORGANIC MATERIALS 4/8/2013 BPDDC LLC www. bpddc. com FUNCTIONAL NANOPARTICLES 30
896b0fc298fe820ac410d4e83151a8c2.ppt